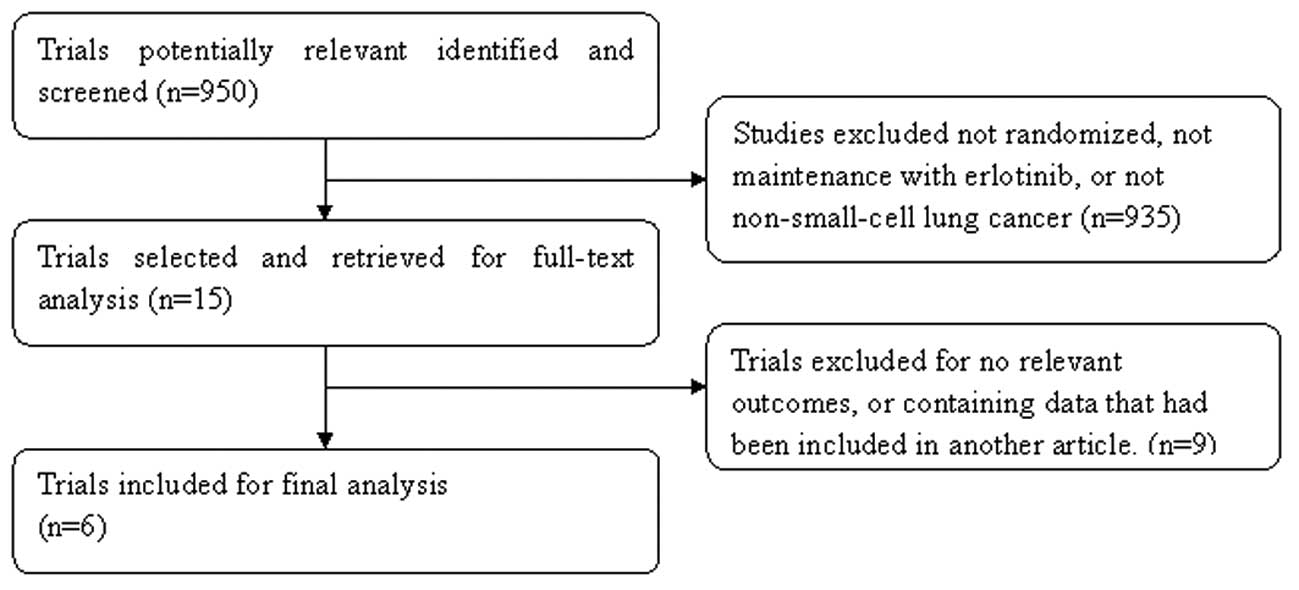
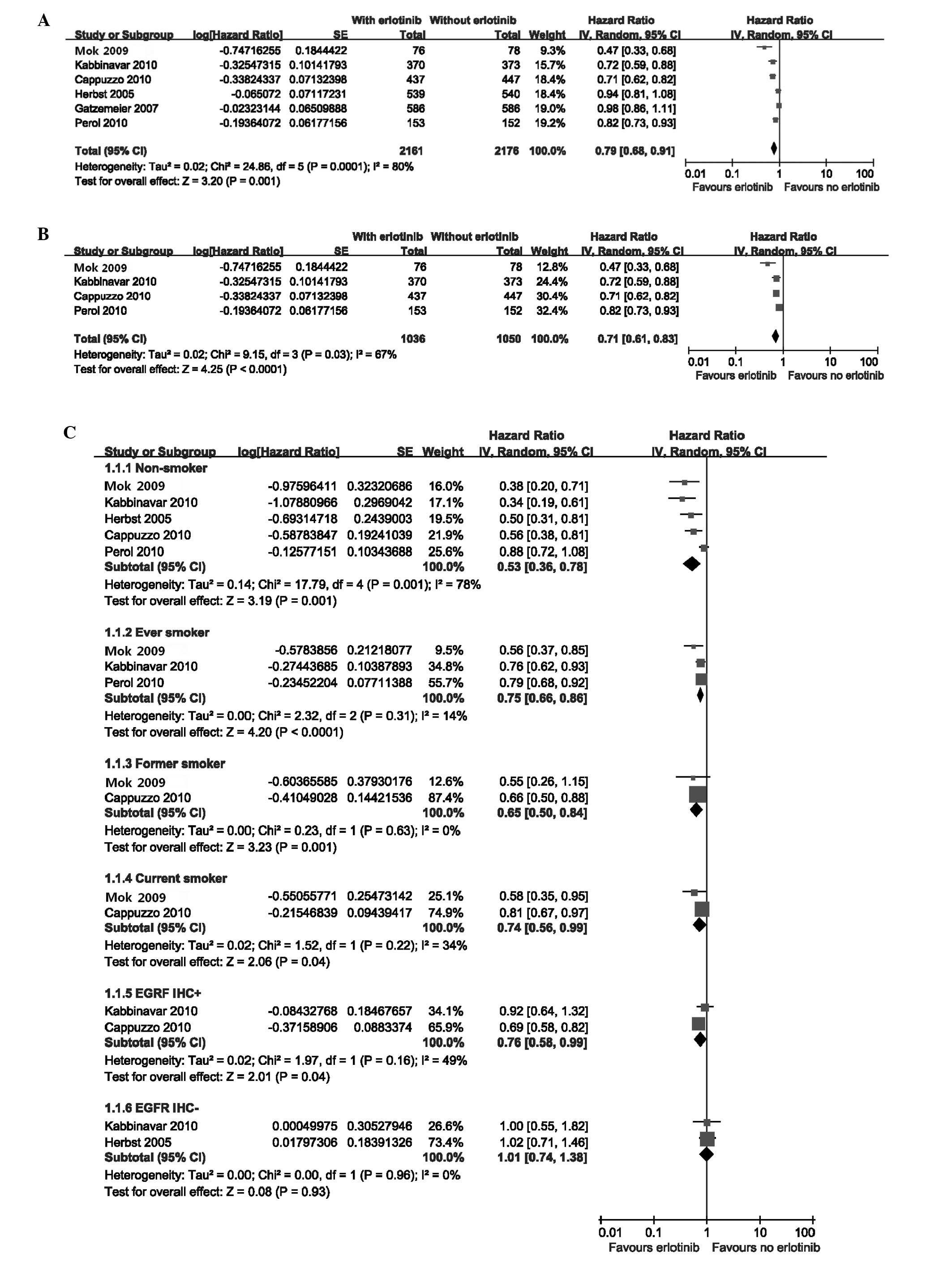
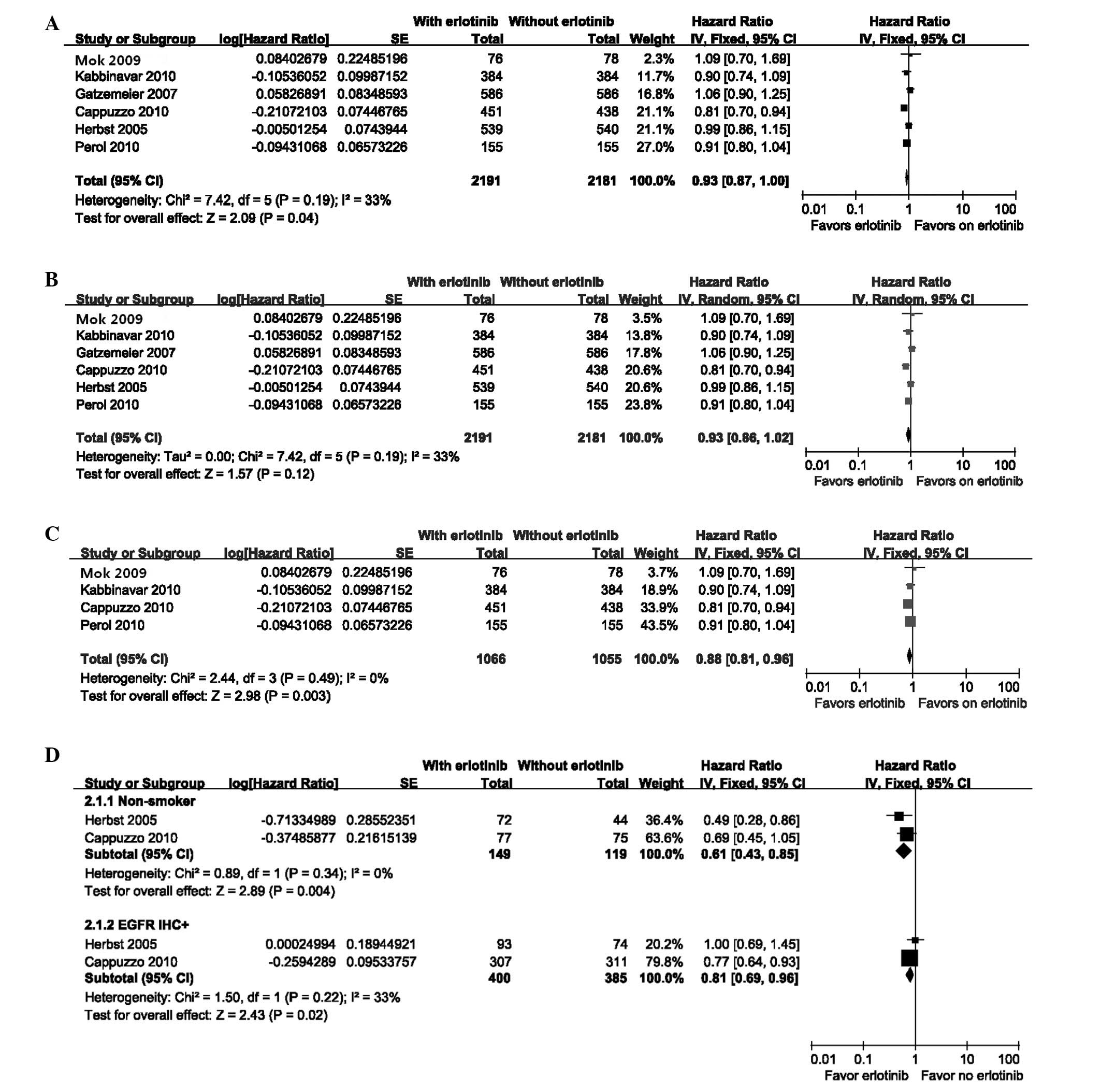
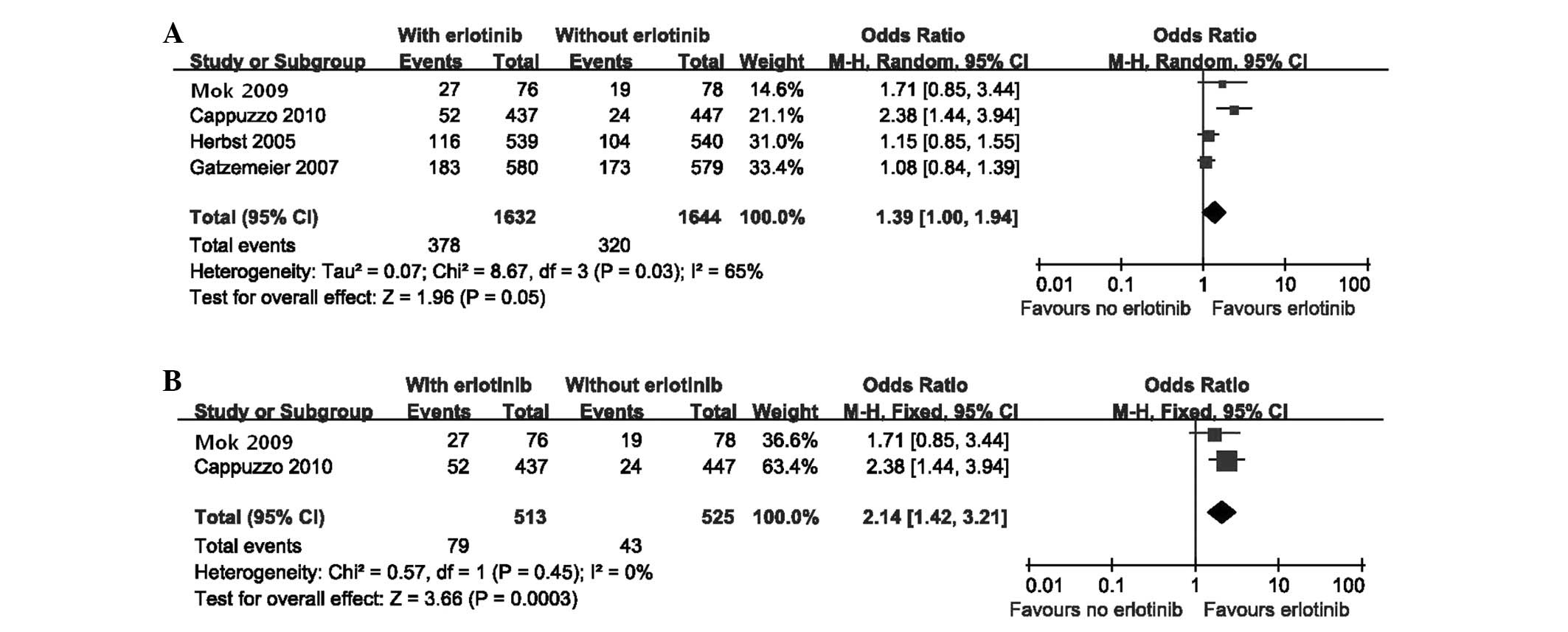
|
1
|
Jemal A, Siegel R, Ward E, et al: Cancer
statistics. CA Cancer J Clin. 58:71–96. 2008.
|
|
2
|
Govindan R, Page N, Morgensztern D, et al:
Changing epidemiology of small-cell lung cancer in the United
States over the last 30 years: analysis of the surveillance,
epidemiologic, and end results database. J Clin Oncol.
24:4539–4544. 2006.PubMed/NCBI
|
|
3
|
Stinchcombe TE, Bogart J, Wigle DA and
Govindan R: Annual review of advances in lung cancer clinical
research: a report for the year 2009. J Thorac Oncol. 5:935–939.
2010.PubMed/NCBI
|
|
4
|
Pfister DG, Johnson DH, Azzoli CG, et al:
American Society of Clinical Oncology treatment of unresectable
non-small cell lung cancer guideline: update 2003. J Clin Oncol.
22:330–353. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Park JO, Kim SW, Ahn JS, et al: Phase III
trial of two versus four additional cycles in patients who are
nonprogressive after two cycles of platinum-based chemotherapy in
non small-cell lung cancer. J Clin Oncol. 25:5233–5239. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Socinski MA, Schell MJ, Peterman A, et al:
Phase III trial comparing a defined duration of therapy versus
continuous therapy followed by second-line therapy in
advanced-stage IIIB/IV non-small cell lung cancer. J Clin Oncol.
20:1335–1343. 2002. View Article : Google Scholar
|
|
7
|
von Plessen C, Bergman B, Andresen O, et
al: Palliative chemotherapy beyond three courses conveys no
survival or consistent quality-of-life benefits in advanced
non-small cell lung cancer. Br J Cancer. 95:966–973.
2006.PubMed/NCBI
|
|
8
|
Ciuleanu T, Brodowicz T, Zielinski C, et
al: Maintenance pemetrexed plus best supportive care versus placebo
plus best supportive care for non-small cell lung cancer: a
randomised, double-blind, phase 3 study. Lancet. 374:1432–1440.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fidias PM, Dakhil SR, Lyss AP, et al:
Phase III study of immediate compared with delayed docetaxel after
front-line therapy with gemcitabine plus carboplatin in advanced
non-small cell lung cancer. J Clin Oncol. 27:591–598. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Belani CP, Barstis J, Perry MC, et al:
Multicenter, randomized trial for stage IIIB or IV non-small-cell
lung cancer using weekly paclitaxel and carboplatin followed by
maintenance weekly paclitaxel or observation. J Clin Oncol.
21:2933–2939. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pirker R, Pereira JR, Szczesna A, et al:
Cetuximab plus chemotherapy in patients with advanced
non-small-cell lung cancer (FLEX): an open-label randomised phase
III trial. Lancet. 373:1525–1531. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Scagliotti GV, Parikh P, von Pawel J, et
al: Phase III study comparing cisplatin plus gemcitabine with
cisplatin plus pemetrexed in chemotherapy-naive patients with
advanced-stage non-small cell lung cancer. J Clin Oncol.
26:3543–3551. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Stinchcombe TE and Socinski MA: Treatment
paradigms for advanced stage non-small cell lung cancer in the era
of multiple lines of therapy. J Thorac Oncol. 4:243–250. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gridelli C, Maione P, Rossi A, et al:
Potential treatment options after first-line chemotherapy for
advanced NSCLC: maintenance treatment or early second-line?
Oncologist. 14:137–147. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Butts C, Murray N, Maksymiuk A, et al:
Randomized phase IIB trial of BLP25 liposome vaccine in stage IIIB
and IV non-small cell lung cancer. J Clin Oncol. 23:6674–6681.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cappuzzo F, Ciuleanu T, Stelmakh L, et al
SATURN investigators: Erlotinib as maintenance treatment in
advanced non-small cell lung cancer: a multicentre, randomised,
placebo-controlled phase 3 study. Lancet Oncol. 11:521–529. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sandler A, Gray R, Perry MC, et al:
Paclitaxel-carboplatin alone or with bevacizumab for non-small cell
lung cancer. N Engl J Med. 355:2542–2550. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shepherd FA, Rodrigues Pereira J, Ciuleanu
T, et al National Cancer Institute of Canada Clinical Trials Group:
Erlotinib in previously treated non-small cell lung cancer. N Engl
J Med. 353:123–132. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Reck M, van Zandwijk N, Gridelli C, et al:
Erlotinib in advanced non-small cell lung cancer: efficacy and
safety findings of the global phase IV tarceva lung cancer survival
treatment study. J Thorac Oncol. 5:1616–1622. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gatzemeier U, Pluzanska A, Szczesna A, et
al: Phase III study of erlotinib in combination with cisplatin and
gemcitabine in advanced non-small-cell lung cancer: the Tarceva
Lung Cancer Investigation Trial. J Clin Oncol. 25:1545–1552. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Herbst RS, Prager D, Hermann R, et al
TRIBUTE Investigator Group: TRIBUTE: a phase III trial of erlotinib
hydrochloride (OSI-774) combined with carboplatin and paclitaxel
chemotherapy in advanced non-small cell lung cancer. J Clin Oncol.
23:5892–5899. 2005. View Article : Google Scholar
|
|
22
|
NHS Centre for Reviews and Dissemination:
Undertaking systematic reviews of research on effectiveness: CRD’s
guidance for carrying out or commissioning reviews. CRD Report 4.
2nd edition. University of York; York: 2001
|
|
23
|
Parmar MK, Torri V and Stewart L:
Extracting summary statistics to perform meta-analyses of the
published literature for survival endpoints. Stat Med.
17:2815–2834. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Higgins JP, Thompson SG, Deeks JJ and
Altman DG: Measuring inconsistency in meta-analyses. BMJ.
327:557–560. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Egger M, Davey Smith G, Schneider M and
Minder C: Bias in meta-analysis detected by a simple, graphical
test. BMJ. 315:629–634. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Altman DG and Deeks JJ: Meta-analysis,
Simpson’s paradox, and the number needed to treat. BMC Med Res
Methodol. 2:32002.
|
|
27
|
McQuay HJ and Moore RA: Using numerical
results from systematic reviews in clinical practice. Ann Intern
Med. 126:712–720. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Smeeth L, Haines A and Ebrahim S: Numbers
needed to treat derived from meta-analyses–sometimes informative,
usually misleading. BMJ. 318:1548–1551. 1999.
|
|
29
|
Moher D, Cook DJ, Eastwood S, Olkin I,
Rennie D and Stroup DF: Improving the quality of reports of
meta-analyses of randomised controlled trials: the QUOROM
statement. Quality of Reporting of Meta-analyses. Lancet.
354:1896–1900. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mok TS, Wu YL, Yu CJ, et al: Randomized,
placebo-controlled, phase II study of sequential erlotinib and
chemotherapy as first-line treatment for advanced non-small-cell
lung cancer. J Clin Oncol. 27:5080–5087. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kabbinavar FF, Miller VA, Johnson BE,
O’Connor PG and Soh C; ATLAS Investigators: Overall survival (OS)
in ATLAS, a phase IIIb trial comparing bevacizumab (B) therapy with
or without erlotinib (E) after completion of chemotherapy (chemo)
with B for first-line treatment of locally advanced, recurrent, or
metastatic non-small cell lung cancer (NSCLC). J Clin Oncol
(meeting abstracts). 28:75262010.
|
|
32
|
Perol M, Chouaid C, Milleron BJ, et al:
Maintenance with either gemcitabine or erlotinib versus observation
with predefined second-line treatment after cisplatin-gemcitabine
induction chemotherapy in advanced NSCLC: IFCT-GFPC 0502 phase III
study. J Clin Oncol (meeting abstracts). 28:75072010.
|
|
33
|
Davies AM, Ho C, Lara PN Jr, Mack P,
Gumerlock PH and Gandara DR: Pharmacodynamic separation of
epidermal growth factor receptor tyrosine kinase inhibitors and
chemotherapy in non-small cell lung cancer. Clin Lung Cancer.
7:385–388. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Piperdi B, Ling YH and Perez-Soler R:
Schedule-dependent interaction between the proteosome inhibitor
bortezomib and the EGFR-TK inhibitor erlotinib in human
non-small-cell lung cancer cell lines. J Thorac Oncol. 2:715–721.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Higgins B, Kolinsky K, Smith M, et al:
Antitumor activity of erlotinib (OSI-774, Tarceva) alone or in
combination in human non-small cell lung cancer tumor xenograft
models. Anticancer Drugs. 15:503–512. 2004. View Article : Google Scholar : PubMed/NCBI
|