Introduction
Over the past decade, the emergence of multi-and
even pan-drug-resistant Acinetobacter baumannii (PDR-Ab) has
brought a tremendous challenge to hospital infection control and
clinical treatment. As an opportunistic pathogen widely distributed
in the hospital and the natural environment, Acinetobacter
baumannii is normally colonized in the respiratory, digestive,
urinary and reproductive systems. It causes nosocomial infections
such as sepsis, ventilator-associated pneumonia and urinary tract
infections under certain conditions (1). In recent years, the extensive use and
misuse of antibiotics have led to the increasing resistance of
Acinetobacter baumannii, which has attracted significant
attention from relevant personnel.
Drugs that are potentially effective for the
treatment of multi-resistant Acinetobacter baumannii include
carbapenems, tetracyclines and enzyme inhibitors, and polymyxins
(1). Polymyxins are regarded as
the last line of defense. However, a single medication is typically
ineffective in clinical treatment (1). Therefore, while it remains necessary
to develop new drugs, at present, the only method for the treatment
of Acinetobacter baumannii infection is the novel
utilization of traditional drugs or the combined use of multiple
drugs (2).
In the current study, we examined the effects of
combinations of fosfomycin (3–5),
minocycline (6) and polymyxin B
(7) in the treatment of PDR-Ab.
The three drugs were chosen as they show considerable effectiveness
and are commonly used in combination in the treatment of
multi-resistant bacteria. Our results suggest that the effects of
multiple drugs used in combination are synergistic and additive,
particularly for the combined use of polymyxin B and
minocycline.
Materials and methods
Experimental strains
A total of 25 strains of PDR-Ab were collected from
the Qilu Hospital of Shandong University (Jinan, China). They were
identified to be resistant to multiple drugs, including
carbapenems, quinolones, cephalosporins, aminoglycosides and
sulfonamides, by the Kirby-Bauer (K-B) method. Among them, 24
strains were from sputum specimens and one was from wound
secretions. The strains were stored at −80°C. Escherichia
coli ATCC25922 was used as a quality control strain.
Materials and instruments
Minocycline and fosfomycin were purchased from the
National Institute for the Control of Pharmaceutical and Biological
Products (Beijing, China). Polymyxin B in this study was
INALCO1758-9325 (Baierdi Biotechnology Company, Beijing, China). MH
agar and MH broth were purchased from Boshang Biotechnology Company
(Jinan, China). The VITEK32 microbial analysis instrument was
purchased from Boshang Biotechnology Company.
Broth microdilution method
Bacterial suspensions were prepared by inoculation
of colonies from a freshly cultured plate, followed by culturing
for 4–6 h at 35°C. The turbidity of the cultures was then
calibrated to 0.5 McFarland (1.5×108 CFU/ml) using a
spectrophotometer. Stock solutions of antibiotics were prepared and
stored at −60°C.
For the determination of the minimum inhibitory
concentration (MIC) value of each drug, various concentrations of
the drugs were added to a 96-well plate. Bacterial suspensions were
added to each well at a final concentration of 1.5×105
CFU/ml and incubated at 35±2°C for 18–24 h. The MIC value was
determined as the drug concentration at which bacterial growth was
completely inhibited.
For the joint drug susceptibility test, the fold
drug dilutions were determined according to the MIC values of each
drug. Each combination of two drugs at various concentrations was
mixed with a bacterial suspension at a final concentration of
1.5×105 CFU/ml, and incubated at 35±2°C for 18–24 h. The
MIC values of each drug were recorded. The fractional inhibitory
concentration index (FICI) was calculated as follows: FICI =
MICA2/MICA1 +
MICB2/MICB1, where MICA2, the MIC
value of drug A in combinative use; MICA1, the MIC value
of drug A used alone; MICB2, the MIC value of drug B in
combinative use; and MICB1, the MIC value of drug B used
alone. FICI values of ≤0.5, 0.5–1.0, 1.0–4.0 and >4.0 were
considered to indicate a synergistic effect, additive effect,
independent effect and antagonistic effect, respectively (8).
Statistical analysis
SPSS 17.0 software (SPSS, Inc., Chicago, IL, USA)
was used to perform the statistical analysis, using a paired t-test
and the geometric mean. P<0.05 was considered to indicate a
statistically significant result.
Results
MIC values are significantly reduced when
fosfomycin is used in combination with minocycline or polymyxin
B
As shown in Tables
I and II, the MIC values of
fosfomycin, minocycline and polymyxin B when used in combination
were significantly reduced when compared with those when used
alone. Notably, while fosfomycin alone showed no significant
antibacterial effects on Acinetobacter baumannii, its MIC
value was significantly reduced when used in combination with
minocycline or polymyxin B (Tables
I and II). We further analyzed
antibacterial effects for combinations of minocycline and polymyxin
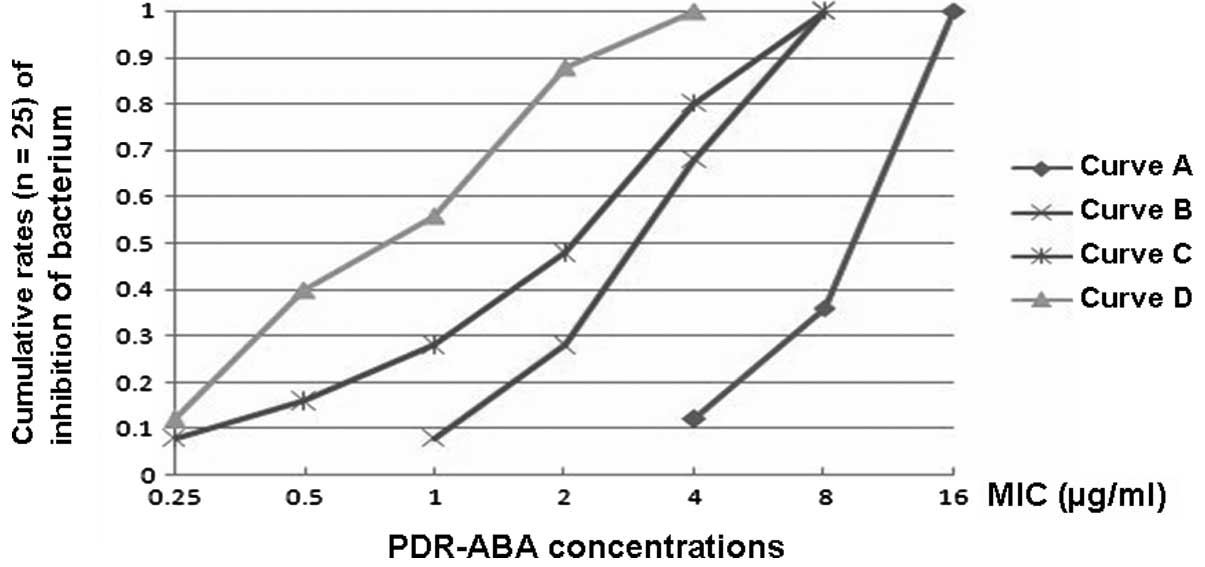
B. The results in Fig. 1 indicate
that the MIC values of minocycline and polymyxin B when used in
combination were significantly reduced when compared with those
when used alone (Fig. 1).
 | Table IAntimicrobial MICG,
MIC50, and MIC90 values of fosfomycin,
minocycline and polymyxin B (μg/ml). |
Table I
Antimicrobial MICG,
MIC50, and MIC90 values of fosfomycin,
minocycline and polymyxin B (μg/ml).
| Antimicrobial
drugs |
MICrange | MICG | MIC50 | MIC90 |
|---|
| Polymyxin B | 1–8 | 3.8906 | 4 | 8 |
| Minocycline | 4–16 | 11.4716 | 16 | 16 |
| Fosfomycin | >512 | >512 | - | - |
 | Table IIMIC values of fosfomycin, minocycline
and polymyxin B in combined use (μg/ml). |
Table II
MIC values of fosfomycin, minocycline
and polymyxin B in combined use (μg/ml).
| Combined
medication |
MICrange | MICG | MIC50 | MIC90 |
|---|
| Polymyxin B and
minocycline | 0.25–4 | 1.0281 | 1 | 2 |
| 0.25–8 | 2.2974 | 2 | 8 |
| Polymyxin B and
fosfomycin | 0.25–4 | 1.3195 | 2 | 4 |
| 64–1024 | 484.381 | 512 | 1024 |
| Minocycline and
fosfomycin | 0.5–4 | 2.1140 | 2 | 4 |
| 64–1024 | 526.39 | 512 | 1024 |
Effects of combinations of drugs
As demonstrated in Table III, the FICI values for
combinations of minocycline and polymyxin B were generally ≤0.5 or
>0.5–1.0, suggesting that the two drugs have a synergistic or
additive effect. However, the FICI values for fosfomycin and
polymyxin B were mostly within the ranges 0.5–1.0 and 1.0–4.0,
suggesting that the effects of fosfomycin and polymyxin B were
additive or independent. Similar results were also observed for
fosfomycin and minocycline. No antagonistic effects for the drug
combinations were observed. Based on these findings, the drugs when
used in combination have synergistic, additive or independent
effects, rather than antagonistic effects. The synergistic and
additive effects were the most prominent, particularly for the
combination of minocycline and polymyxin B.
 | Table IIIFractional inhibitory concentration
index (FICI) values of fosfomycin, minocycline and polymyxin B upon
combined use. |
Table III
Fractional inhibitory concentration
index (FICI) values of fosfomycin, minocycline and polymyxin B upon
combined use.
| Combined
medication | FICI (≤0.5) | FICI (0.5–1) | FICI (1–4) | FICI (>4) |
|---|
| Polymyxin B and
minocycline | 44% | 48% | 8% | 0 |
| Polymyxin B and
fosfomycin | 16% | 44% | 40% | 0 |
| Minocycline and
fosfomycin | 12% | 56% | 32% | 0 |
Discussion
In the treatment of infections caused by
pan-drug-resistant bacteria, a single medication is typically
ineffective. Therefore, the combined use of multiple drugs is
recommended. The medication regimen is usually designed by
referring to the joint drug susceptibility test in vitro.
For the evaluation of the combined effect of antimicrobial agents,
the FICI value is an important parameter (9). It is considered that the combined use
of drugs with a synergistic or additive effect as determined by the
FICI is likely to be effective in clinical treatments.
In the current study, we examined the effects of
combinations of fosfomycin, minocycline and polymyxin B in the
treatment of PDR-Ab. Our results revealed that the MIC values of
the drugs were reduced when they were used in combination,
suggesting a synergistic or additive effect. This effect was
further demonstrated by determining the FICI values.
Based on these findings, the combined use of
multiple drugs is effective, which provides a basis for the use of
drug combinations in the clinical treatment of PDR-Ab. Furthermore,
it also has other advantages over the traditional use of a single
medication. By using combinations of multiple drugs, bacterial
resistance is likely to be reduced. The incidence of adverse drug
responses is also likely to be reduced due to the use of lower drug
doses.
Acknowledgements
This study was supported by the 2012
Innovation Fund (Free Exploration Grant type I, grant no.
26010172611152) of Shandong University.
References
|
1.
|
Shi Y and Liu DW: Exploration of treatment
of multi-drug resistant Acinetobacter. Prescription Drugs.
9:11–16. 2006.(In Chinese).
|
|
2.
|
Liang W, Liu XF, Huang J, Zhu DM, Li J and
Zhang J: Activities of colistin-and minocycline-based combinations
against extensive drug resistant Acinetobacter baumannii
isolates from intensive care unit patients. BMC Infect Dis.
11:1092011. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Lian JJ and Tang QY: Synergy and the
clinical applications of fosfomycin as well as other antimicrobial
agents. Foreign Medicine (Antibiotics). 1:44–50. 2003.(In
Chinese).
|
|
4.
|
Pang XJ, Chen Y and Lu MD: Time difference
attack therapy producing ESBLs G - 22 cases of bacillus infection.
Chinese Journal of New Drugs. 3:351–352. 2005.(In Chinese).
|
|
5.
|
Pang XJ, Zhou HW and Wei H: Clinical
studies of the treatment of Pan-drug resistant Acinetobacter
baumannii infection. Chinese Journal of Pharmaceutical
Sciences. 5:397–398. 2009.(In Chinese).
|
|
6.
|
Huang ZG, Huang C and Ma DY: Antibacterial
activity of minocycline on Acinetobacter baumannii.
International Journal of Laboratory Medicine. 10:1155–1157.
2010.(In Chinese).
|
|
7.
|
Shi YB and Dong Y: In vitro studies of
tigecycline and polymyxin B treatments on pan-drug resistant
Acinetobacter baumannii strains resistant to cefoperazone.
Zhongguo weishengtaixue zazhi. 9:825–827. 2010.(In Chinese).
|
|
8.
|
Sopirala MM, Mangino JE, Gebreyes WA, et
al: Synergy testing by Etest, microdilution checkerboard, and
time-kill methods for pan-drug-resistant Acinetobacter
baumannii. Antimicrob Agents Chemother. 54:4678–4683. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Lee YS, Kang OH, Choi JG, et al:
Synergistic effect of emodin in combination with ampicillin or
oxacillin against methicillin-resistant Staphylococcus
aureus. Pharm Biol. 48:1285–1290. 2010. View Article : Google Scholar : PubMed/NCBI
|