Introduction
B-lineage acute lymphoblastic leukemia is the most
common type of acute leukemia and remains a major life-threatening
disease in children (1). Compared
with conventional chemotherapy, targeted monoclonal antibody (mAb)
therapy is more effective and has fewer side-effects due to greater
selectivity and specificity (2–6).
mAbs directed against CD molecules on the surface of leukemic cells
have shown promising results for targeted therapy (7). CD19 is a specific surface marker of B
cells and is consistently and steadily expressed in almost all the
differentiation stages of B-lineage cells (8). Therefore, CD19 may be an ideal target
for the development of antibody-based treatments for B-lineage
malignancies (9–11).
Currently, the majority of high-affinity mAbs
available are of mouse origin. The clinical application of mouse
antibodies has been greatly restricted due to the occurrence of the
human anti-mouse antibody response, which decreases the efficacy of
therapy and may cause severe anaphylaxis (12,13).
Thus, it is necessary to reduce the immunogenicity of mouse
antibodies, but maintain maximal antigen-recognizing activity. A
successful model of a chimeric antibody used in targeted therapy in
combination with chemotherapy is rituximab (human-mouse chimeric
anti-human-CD20 antibody). Treatment with rituximab has improved
the efficacy of B-lineage non-Hodgkin lymphoma therapy by 40%
(3). However, the majority of
B-lineage leukemic cells are immature and lack expression of CD20
on the cell surface, restricting the application of rituximab in
leukemia treatment. By contrast, CD19 is expressed at various
differentiation stages of B lymphocytes, from stem cells to mature
B cells, throughout B-lineage leukemia. Therefore, CD19 is an
improved target molecule for B-lineage leukemia therapy (8).
The baculovirus expression system is widely applied
as an important eukaryotic expression system due to a number of
advantages, including the ease of inserting desired genes, high
yield, the presence of post-translational modifications similar to
those found in human cells, ease of use and suitable biological
material safety. The baculovirus expression vector pAc-κ-CH3 was
designed particularly for the expression of chimeric antibodies; it
comprises IgG expression cassette elements, including authentic
IgGκ and heavy chain signal sequences, as well as light chain κ and
the heavy chain constant region, that are integrated into a single
vector and controlled by p10 and polyhedrin promoters, respectively
(14). IgG yields have been
reported to be between 6 and 18 mg/l and the antigen-binding
function has been observed to be well-preserved (15). Therefore, this system was selected
for the expression of recombinant protein in the present study.
However, modifying IgM to IgG is likely to be a difficult procedure
since changing the pentameric IgM unit to a dimeric IgG unit may
result in the loss of binding activity (16,17).
Zhejiang Children’s Hospital (ZCH)-4-2E8 or simply
2E8, is an antibody belonging to the murine IgM subtype that was
generated in the Department of Hematology and Oncology, School of
Medicine, Children’s Hospital of Zhejiang University (Hangzhou,
China) (18). 2E8 was assigned to
the CD19 category by the 6th International Workshop and Conference
on Human Leukocyte Differentiation Antigens (HLDA6) in 1996. The
aim of the present study was to insert 2E8 Fv genes into the
pAc-κ-CH3 baculovirus shuttle vector and express the novel protein
in the insect Sf9 cell line, using the baculoviral expression
system. The characteristics and physiological activity of the
chimeric antibody were also examined.
Materials and methods
Cell culture
Experimental procedures were approved by the Medical
Ethics Committee of the Children’s Hospital of Zhejiang
University.
The Sf9 insect cell line was provided by Professor
Mifang Liang from the Chinese Center for Disease Control
Prevention, Institute for Viral Disease Control and Prevention
(Beijing, China) (15). Sf9 cells
were cultured in serum-free medium (SFM; SF900 II; Invitrogen Life
Technologies, Shanghai, China) and fetal bovine serum (Gibco-BRL,
Gaithersburg, MD, USA), but were gradually adapted to SFM prior to
the expression studies. NALM-6, a B-lineage leukemic cell line, was
cultured in RPMI-1640 medium (Gibco-BRL) and fetal bovine
serum.
The hybrid cell line 2E8, which secreted the
parental 2E8 anti-human CD19 mAb, was cultured in RPMI-1640 medium
and fetal bovine serum.
Plasmids and reagents
The Psectag2A/Scfv2E8 plasmid, which contained heavy
and light chains of the 2E8 mAb, was established in the Department
of Hematology and Oncology, School of Medicine, Children’s Hospital
of Zhejiang University. Psectag2A, Escherichia coli (E.
coli) DH5α strain cells, high-fidelity platinum Taq
polymerase, Taq DNA polymerase, RQ1
5-bromo-4-chloro-indolyl-β-D-galactopyranoside,
isopropylthio-β-galactoside, RNasin and RNase-free DNase were
purchased from Invitrogen Life Technologies. The pAc-κ-CH3
baculovirus expression vector, which contained authentic IgGκ,
heavy chain signal sequences and constant regions, was provided by
Professor Mifang Liang from the Chinese Center for Disease Control
Prevention, Institute for Viral Disease Control and Prevention
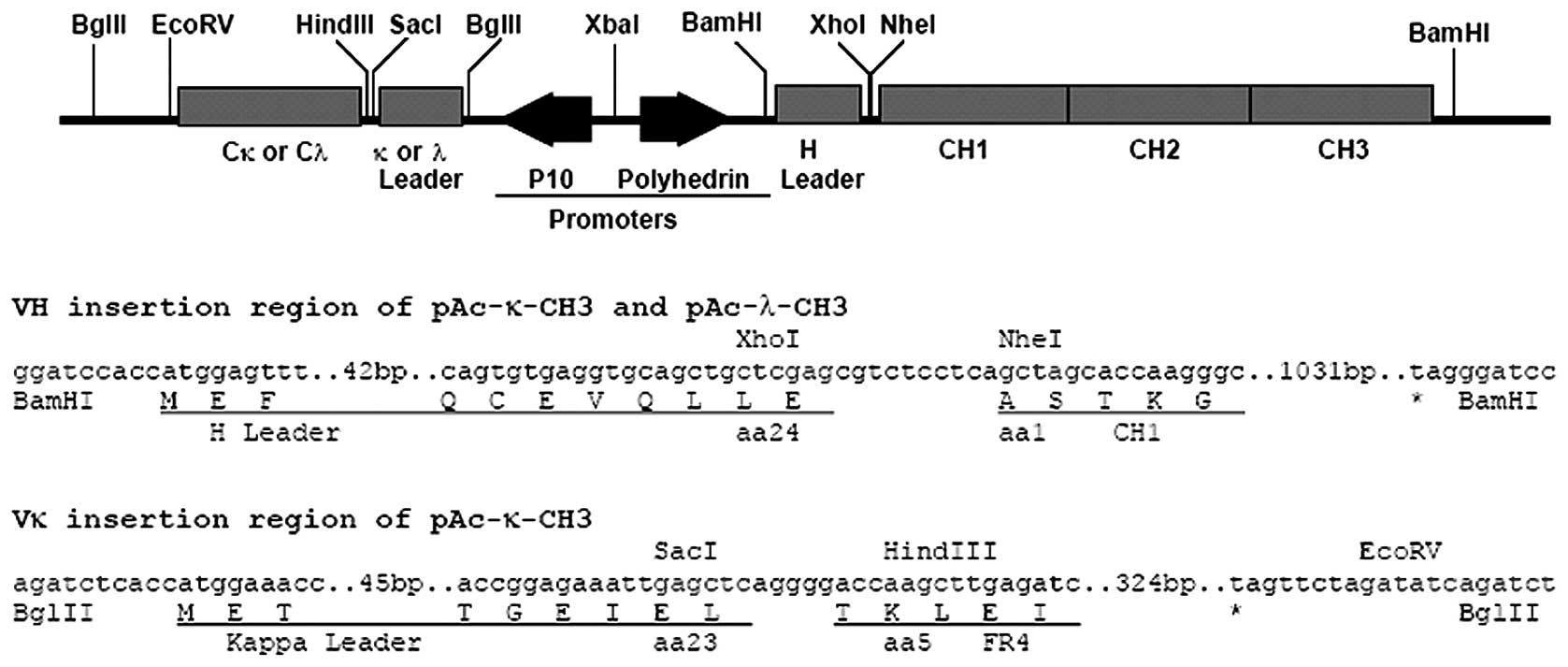
(15). The structure of this
plasmid is shown in Fig. 1
(15).
pGEM®-T Easy Vector (TA cloning) and the
restriction endonucleases, EcoRI, SacI,
HindIII, XhoI, NheI, BamHI and
BglI, were purchased from Promega Corporation (Beijing,
China). The BaculoGold transfection kit, mouse anti-human (MAH)
γ1-fluorescein isothiocyanate (FITC) and dialysis solution were
purchased from Becton Dickinson (Franklin Lakes, NJ, USA).
Horseradish peroxidase (HRP)-conjugated MAH-Fc-HRP antibodies were
purchased from Sigma-Aldrich (Shanghai, China). Goat anti-mouse
(GAM)-Fab-rhodamine, HRP-conjugated GAM-μ-HRP and GAM-Fab-HRP
antibodies were purchased from Rockland Immunochemicals, Inc.
(Gilbertsville, PA, USA). MAH-Fc-FITC, GAM-Fab-(κ)-FITC and
SuperSignal West Durab Extended Duration Substrate were purchased
from Pierce Biotechnology, Inc. (Rockford, IL, USA). T4 DNA ligase
and Triton X-100 were purchased from Gibco-BRL and a DL2000 marker
was purchased from Takara Bio, Inc. (Dalian, China). A QIAquick gel
extraction kit was purchased from Qiagen (Valencia, CA, USA) and
the SuperFect transfection reagent was purchased from Roche
Diagnostics (Shanghai, China). Oligo(dT)12–18 primers
and polyvinylidene fluoride (PVDF) membranes were purchased from
Bio-Rad Laboratories, Inc. (Shanghai, China). A prestained protein
molecular weight marker was purchased from Fermentas, (Shenzhen,
China) and HRP-labeled GAM IgG (heavy and light chains) was
purchased from Beijing Zhongshan Golden Bridge Biotechnology Co.,
Ltd. (Beijing, China).
Construction of the
pAc-κ-CH3-VH2E8-VL2E8 baculovirus shuttle
vector
VH2E8 and VL2E8 genes were
cloned from pSectag2A/ScFv2E8, which had been
successfully established previously by polymerase chain reaction
amplification using the primers listed in Table I. Specific endonuclease sites were
located within the primer pairs and the amplified fragments were
inserted using TA cloning techniques. This was followed by
transformation into E. coli DH5α cells. Recombinants were
selected and amplification and sequencing of the inserted sequences
were performed. Target sequences were confirmed by comparison with
the previously cloned VH2E8 and VL2E8 gene
sequences to enable further study.
 | Table IPrimers used to clone VH2E8
and VL2E8 genes for insertion into pAc-κ-CH3. |
Table I
Primers used to clone VH2E8
and VL2E8 genes for insertion into pAc-κ-CH3.
| Gene | Up/down | Sequence |
|---|
| VH2E8 | Forward |
CTCGAGGAGGTGAAGCTGGTGGAGT
(XhoI) |
| VH2E8 | Reverse |
GCTAGCCTCTGAGGAGACGGTGACT
(NheI) |
| VL2E8 | Forward |
GAGCTCGATATCCAGATGACACAGACTTC
(SacI) |
| VL2E8 | Reverse |
AAGCTTTTTGATTTCCAGCTTGGTGCCT
(HindIII) |
VH2E8 and VL2E8 gene fragments
were cleaved with corresponding endonucleases (VH2E8,
XhoI and NheI; VL2E8, SacI and
HindIII) and inserted sequentially into the secretive
pAc-κ-CH3 baculovirus expression shuttle vector. Following
transformation into E. coli DH5α cells, recombinants were
selected, plasmid DNA was purified and the insertions were
amplified and sequenced using the method described by Liang et
al (14). The sequences were
then compared with the previously identified VH2E8 and
VL2E8 gene sequences to confirm that the insertions were
correct. DNA manipulation and bacterial transformation procedures
were conducted as previously described by Filpula et al
(16).
Transfection of Sf9 cells with the
reconstructed baculovirus shuttle vector and the formation of the
pAc-κ-CH3-VH2E8-VL2E8 complete virion
(CV)
Recombinant baculoviruses were prepared by
homologous recombination using the BaculoGold transfection kit
(Becton Dickinson, Franklin Lakes, NJ, USA), according to the
manufacturer’s instructions. Sf9 cells were cotransfected with the
pAc-κ-CH3-VH2E8-VL2E8 reconstructed shuttle
vector and linearized DNA of the Autographa california
nuclear polyhedrosis virus (AcNPV). pXyIE and AcNPV linearized
DNA-transfected Sf9 cells and uninfected Sf9 cells were set as
positive and negative controls, respectively, as recommended by the
manufacturer’s instructions. Morphological changes in the cells
were observed every day following transfection using an inverted
microscope. Positive control cells expressing recombinant XyIE
turned yellow in the presence of catechol at day 4 following
transfection. The supernatants of the
pAc-κ-CH3-VH2E8-VL2E8-transfected Sf9 cells
were harvested as primary recombinant CVs, to produce
pAc-κ-CH3-VH2E8-VL2E8 CV (P0) for further
amplification. Transfected Sf9 cells were collected for detection
on day 7.
Through three passages of amplification, large viral
stocks were prepared by infecting Sf9 cells at a multiplicity of
infection (number of virions/number of cells being infected) of
<1. The supernatant was harvested at day 4 or 5 following
infection. Three passages were amplified and the virus stock was
saved for application in the expression studies.
For protein expression, Sf9 cells were cultured in
SFM. The supernatant was collected for detection at day 6 following
infection when ~30% of living cells remained.
Identification of the recombinant protein
by flow cytometry (FCM)
To analyze the activity levels of the recombinant
antibody in the supernatant and cell lysates, a 1×106
cells/tube suspension of fresh NALM-6 cells was prepared in six
tubes. Next, 100 μl concentrated expression supernatant or infected
Sf9 cell lysate was added to the cell suspension in two of the
tubes and the same volume of concentrated regular medium (each in
duplicates) was added to the other four tubes as negative controls.
After 30 min, the cells were washed twice with phosphate-buffered
saline (PBS). MAH-Fc-FITC and GAM-κ-FITC were added separately and
the reactions were incubated for 30 min, which was followed by two
washes with PBS. FCM analysis was utilized to observe whether the
chimeric antibody in the supernatant or infected Sf9 cell lysate
was able to bind to the CD19 antigen on the NALM-6 cell
surface.
Identification of the recombinant protein
by western blot analysis
Sf9 cells (2×107 cells) were placed in 1
ml lysis refolding solution [50 mmol/l Tris-HCl (pH 7.5), 50 mM
NaCl, 5 mM oxidized glutathione, 0.5 mM reduced glutathione and 1 M
urea] with 100 mM phenylmethylsulfonyl chloride, 1 μg/ml aprotinin
and 1 μg/ml leupeptin to prevent protein degradation. Cells were
sonicated to obtain the cell lysate and
pAc-κ-CH3-VH2E8-VL2E8 CV P3-infected Sf9
cells were set as the experimental group and the uninfected Sf9
cells lysates were used as the negative control. Lysates were
dialyzed in PBS (containing 0.02% sodium azide) for 48 h (used PBS
was replaced with fresh PBS six times in this duration), following
which PBS substitution ultrafiltration was performed twice. Protein
concentration was determined using the DC protein assay kit
(Bio-Rad Laboratories, Inc.). Total protein extracts (0.5 μg/well)
were separated by sodium dodecyl sulfate-polyacrylamide gel
electrophoresis (SDS-PAGE), according to the method described by
Sambrook et al (16), using
12% separation polyacrylamide gels and 5% condensed gels. Gels were
stained with Coomassie brilliant blue R250 (16). For western blot analysis, the
chimeric antibody was transferred onto PVDF membranes and blocked
with 5% skimmed milk. The transferred membrane was incubated with
MAH-Fc-HRP (1:20,000) for 2 h at 37°C and washed three times.
Detection was performed using an enhanced chemiluminescence
substrate and exposure to X-ray film. The anti-human CD20
mouse-human chimeric antibody, rituximab (Roche/Genentech, San
Francisco, CA, USA), was used as a positive control.
Results
Construction of the
pAc-κ-CH3-VH2E8-VL2E8 recombinant baculovirus
shuttle vector
The 380-bp VH2E8 and 330-bp
VL2E8 gene fragments were amplified using the primers
listed in Table I and inserted
into the TA-cloning vector. Endonuclease digestion, sequencing and
sequence analysis were then performed. The correct sequences were
named TA-VH2E8 and TA-VL2E8.
TA-VH2E8 and pAc-κ-CH3 vectors were
digested with XhoI and NheI and ligated with T4 DNA
ligase. The recombinant was treated with BamHI endonuclease
and sequenced to confirm the correct insertion and orientation of
recombinant pAc-κ-CH3-VH2E8. The VL2E8
fragment was inserted into pAc-κ-CH3-VH2E8, which was
confirmed by sequencing.
Transfection, virion amplification and
expression of the recombinant antibody
Sf9 cells were transfected with the
pAc-κ-CH3-VH2E8-VL2E8 baculovirus expression
vector and transfected cells were identified to be bigger and of
irregular shape (Fig. 2A) when
compared with the negative control cells (Fig. 2B). The positive control cells
turned yellow in the presence of catechol when harvested at day 4
following transfection (Fig. 3A),
indicating that the transfection procedure was successful.
Following three passages of amplification,
high-titer virus stocks were ready for use in the expression
studies.
Determination of recombinant protein
activity levels by FCM
Antibody activity was detected using FCM in the cell
lysates from infected Sf9 cells, but not in the supernatant.
NALM-6 cells, incubated with the cell lysates from
infected Sf9 cells, were 14.35% positive (vs. 2.97% in the negative
control) when labeled with GAM-Fab-FITC. The percentage of positive
cells (28.67 vs. 2.76% in the negative control) was even higher
when labeled with MAH-Fc-FITC, which indicated the existence of
functional antibody in the infected Sf9 cell lysates (Fig. 4).
Identification of the recombinant protein
by immunofluorescence and western blot analysis
Since Sf9 cells exhibited green autofluorescence,
immunofluorescence in the transfected Sf9 cells was monitored by
incubation with GAM-Fab-rhodamine instead of FITC. It was found
that 80% of the cells were positive for red fluorescence in the
cytoplasm (Fig. 5A), indicating
the expression of novel protein in the cytoplasm of infected Sf9
cells. However, no fluorescence was observed in the uninfected Sf9
cells (Fig. 5B).
No specific protein bands corresponding to the heavy
and light chains were observed in the supernatant from the infected
cells. However, specific bands were observed in the lysates of the
infected Sf9 cells, which was consistent with the FCM results.
Specific bands corresponding to the heavy and light chains of
rituximab were observed by western blot analysis in the infected
Sf9 cell lysate, while the uninfected Sf9 cell lysates did not
exhibit any positive activity (Fig.
3B).
Discussion
There are five main classes of Ig: IgG, IgA, IgD,
IgE and IgM, divided according to the various genes encoding the
constant regions of the heavy chain. IgG and IgA classes are in
turn subdivided into six isotypes: IgG1, IgG2, IgG3, IgG4, IgA1 and
IgA2. When reconstructing expression vectors for antibodies, it is
generally accepted that IgGs, including IgG1, IgG2 and IgG4
subtypes, may be genetically manipulated with relative ease whilst
maintaining antibody function. In total, >20 recombinant IgG
antibody-based therapeutic drugs are now licensed for the treatment
of a variety of diseases, the majority of which belong to the IgG1
subclass. In addition, there are hundreds of new drugs currently
under development (9). In the
present study, a vector with the IgG1 form of the final product was
selected to express a chimeric antibody. It was previously reported
that difficulties may arise when transforming IgM into IgG
(17). Thus, the outcome of the
CD19 antibody in the current study was not clear. Genetic
manipulation was performed on ZCH-4-2E8, an IgM-type antibody
developed previously and identified to be a novel CD19 mAb by HLDA6
in 1996. Gene cloning of the variable regions of the heavy and
light chains of this antibody was successfully performed and a
eukaryotic expression system was constructed. Following the
insertion of the genes encoding the chimeric antibody into the
pAc-κ-CH3 baculovirus vector and transfection into the insect host
Sf9 cell line, the antibody was shown to be expressed in the
cytoplasm by immunostaining with a rhodamine-labeled GAM-Fab
antibody. This was further confirmed by SDS-PAGE and western blot
analysis. The results obtained indicate that genes encoding the IgM
variable region may be reconstructed with an IgG backbone and
retain part of the antibody function. In addition, the results
demonstrate that specific IgM antibodies may also be amenable to
genetic manipulation and are likely to retain part of the capacity
to recognize antigens.
FCM was applied to detect the activity levels of the
recombinant antibody. Activity was detected in the lysates,
however, the levels were reduced compared with those of the
parental antibody. In addition, activity was not detected in the
supernatants. These results indicate that the antibody was produced
inside the cells, but was unable to be secreted outside the cells.
The results indicate that it was possible to modify IgM into an IgG
form and partially retain its activity. However, questions remain
with regard to promoting the secretion of the engineered antibody,
while retaining or improving its biological functions. Correct
antibody conformation is critical to biological function, including
the recognition of the relevant antigen. It remains to be
investigated whether transformation from IgM to IgG causes changes
in the spatial conformations of the chimeric antibody that prevent
secretion or whether the leader sequence of the IgG form is not
suitable for the secretion of the IgM variable region. Therefore,
further studies are required to engineer antibodies that are able
to be secreted from cells, to improve the binding activity of the
antibodies and to identify appropriate leader sequences that
promote efficient secretion, while retaining optimal biological
function via modulation of the gene sequences.
In the present study, the recombinant shuttle
vector, pAc-κ-CH3-VH2E8-VL2E8, was
successfully reconstructed and CVs with the capacity for natural
infection of insect cells were obtained. Recombinant antibody was
successfully expressed and functional antibody existed inside the
infected Sf9 cells but was not secreted. Therefore, the results
indicate that it is possible to reconstruct an IgG-form chimeric
antibody from a parental IgM antibody. However, modifications of
the procedure are required to obtain a secreted chimeric antibody
with appropriate biological activity.
Acknowledgements
The study was supported by grants from the Zhejiang
Provincial Natural Scientific Foundation (no. Z205166), the
National Natural Science Foundation of China (no. 81100338), the
Zhejiang Provincial Traditional Chinese Medicine Scientific and
Technological Innovation Platform for Severe Diseases (no.
2009ZDJB01-05) and the Science Technology Department of Zhejian
Province, Qianjiang Project (no. 2009R10037). The authors thank Ms.
Baiqin Qian and Mr. Hongqiang Shen for their technical support and
Xiaojun Xu and Sisi Li for their assistance during the study.
References
|
1
|
Salzer WL, Devidas M, Carroll WL, Winick
N, Pullen J, Hunger SP and Camitta BA: Long-term results of the
pediatric oncology group studies for childhood acute lymphoblastic
leukemia 1984–2001: a report from the children’s oncology group.
Leukemia. 24:355–370. 2010.
|
|
2
|
Portell CA and Advani AS: Antibody therapy
for acute lymphoblastic leukemia. Curr Hematol Malig Rep.
7:153–159. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shimoni A, Zwas ST, Oksman Y, Hardan I,
Shem-Tov N, Rand A, Yerushalmi R, Avigdor A, Ben-Bassat I and
Nagler A: Ibritumomab tiuxetan (Zevalin) combined with
reduced-intensity conditioning and allogeneic stem-cell
transplantation (SCT) in patients with chemorefractory
non-Hodgkin’s lymphoma. Bone Marrow Transplant. 41:355–361.
2008.PubMed/NCBI
|
|
4
|
Pagel JM, Appelbaum FR, Eary JF, Rajendran
J, Fisher DR, Gooley T, Ruffner K, Nemecek E, Sickle E, Durack L,
Carreras J, Horowitz MM, Press OW, Gopal AK, Martin PJ, Bernstein
ID and Matthews DC: 131I-anti-CD45 antibody plus
busulfan and cyclophosphamide before allogeneic hematopoietic cell
transplantation for treatment of acute myeloid leukemia in first
remission. Blood. 107:2184–2191. 2006. View Article : Google Scholar
|
|
5
|
Herrera L, Stanciu-Herrera C, Morgan C,
Ghetie V and Vitetta ES: Anti-CD19 immunotoxin enhances the
activity of chemotherapy in severe combined immunodeficient mice
with human pre-B acute lymphoblastic leukemia. Leuk Lymphoma.
47:2380–2387. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Feldman EJ, Brandwein J, Stone R, Kalaycio
M, Moore J, O’Connor J, Wedel N, Roboz GJ, Miller C, Chopra R,
Jurcic JC, Brown R, Ehmann WC, Schulman P, Frankel SR, De Angelo D
and Scheinberg D: Phase III randomized multicenter study of a
humanized anti-CD33 monoclonal antibody, lintuzumab, in combination
with chemotherapy, versus chemotherapy alone in patients with
refractory or first-relapsed acute myeloid leukemia. J Clin Oncol.
23:4110–4116. 2005. View Article : Google Scholar
|
|
7
|
Dungarwalla M, Evans SO, Riley U, Catovsky
D, Dearden CE and Matutes E: High dose methylprednisolone and
rituximab is an effective therapy in advanced refractory chronic
lymphocytic leukemia resistant to fludarabine therapy.
Haematologica. 93:475–476. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen YH, Tang YM, Shen GQ, Song H, Yang
SL, Shi SW, Qian BQ, Xu WQ and Ning BT: The comparison of
expression patterns of CD19 and CD20 on 321 cases of acute
leukemia. Zhejiang Medical Journal. 25:461–463. 2003.(In
Chinese).
|
|
9
|
Jefferis R: Aglycosylated antibodies and
the methods of making and using them: WO2008030564. Expert Opin
Ther Pat. 19:101–105. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tsai LK, Pop LM, Liu X and Vitetta ES: A
comparison of the anti-tumor effects of a chimeric versus murine
anti-CD19 immunotoxins on human B cell lymphoma and Pre-B acute
lymphoblastic leukemia cell lines. Toxins (Basel). 3:409–419. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Davies JK, Singh H, Huls H, Yuk D, Lee DA,
Kebriaei P, Champlin RE, Nadler LM, Guinan EC and Cooper LJ:
Combining CD19 redirection and alloanergization to generate
tumor-specific human T cells for allogeneic cell therapy of B-cell
malignancies. Cancer Res. 70:3915–3924. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Michálek J, Collins RH and Vitetta ES:
Clinical-scale selective depletion of alloreactive T cells using an
anti-CD25 immunotoxin. Neoplasma. 50:296–299. 2003.PubMed/NCBI
|
|
13
|
Goto S, Goto H, Tanoshima R, Kato H,
Takahashi H, Sekiguchi O and Kai S: Serum sickness with an elevated
level of human anti-chimeric antibody following treatment with
rituximab in a child with chronic immune thrombocytopenic purpura.
Int J Hematol. 89:305–309. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liang M, Dubel S, Li D, Queitsch I, Li W
and Bautz EK: Baculovirus expression cassette vectors for rapid
production of complete human IgG from phage display selected
antibody fragments. J Immunol Methods. 247:119–130. 2001.
View Article : Google Scholar
|
|
15
|
van der Geld YM, Oost-Kort W, Limburg PC,
Specks U and Kallenberg CG: Recombinant proteinase 3 produced in
different expression systems: recognition by anti-PR3 antibodies. J
Immunol Methods. 244:117–131. 2000.PubMed/NCBI
|
|
16
|
Filpula D: Antibody engineering and
modification technologies. Biomol Eng. 24:201–215. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang Y, Feng J, Huang Y, Gu X, Sun Y, Li Y
and Shen B: The design, construction and function of a new chimeric
anti-CD20 antibody. J Biotechnol. 129:726–731. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tang Y and Guo S: Preparation,
identification and preliminary application of B lineage specific
monoclonal antibody ZCH-4-2E8. Journal of Zhejiang University
Science ABC. 23:246–249. 1994.
|