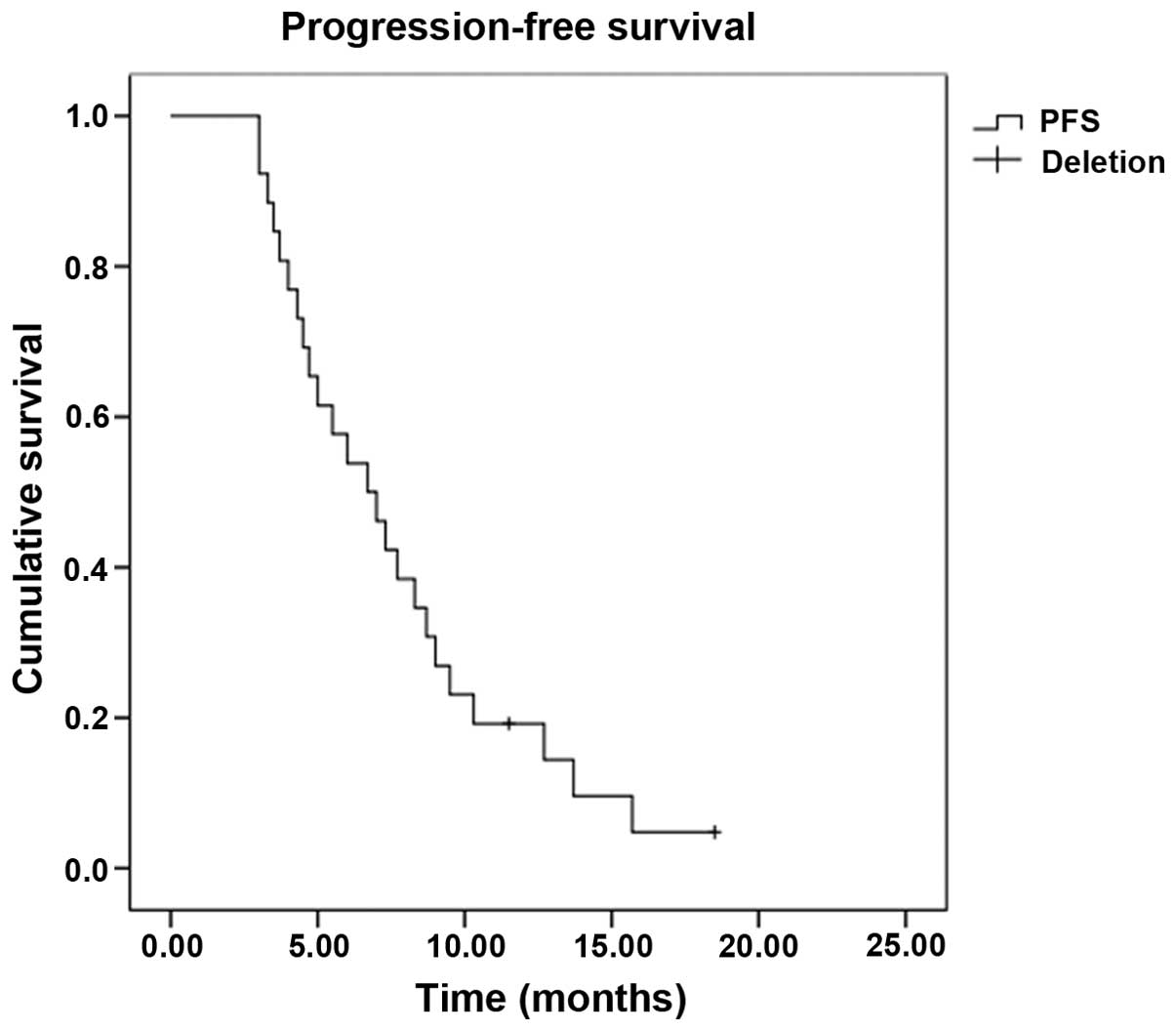
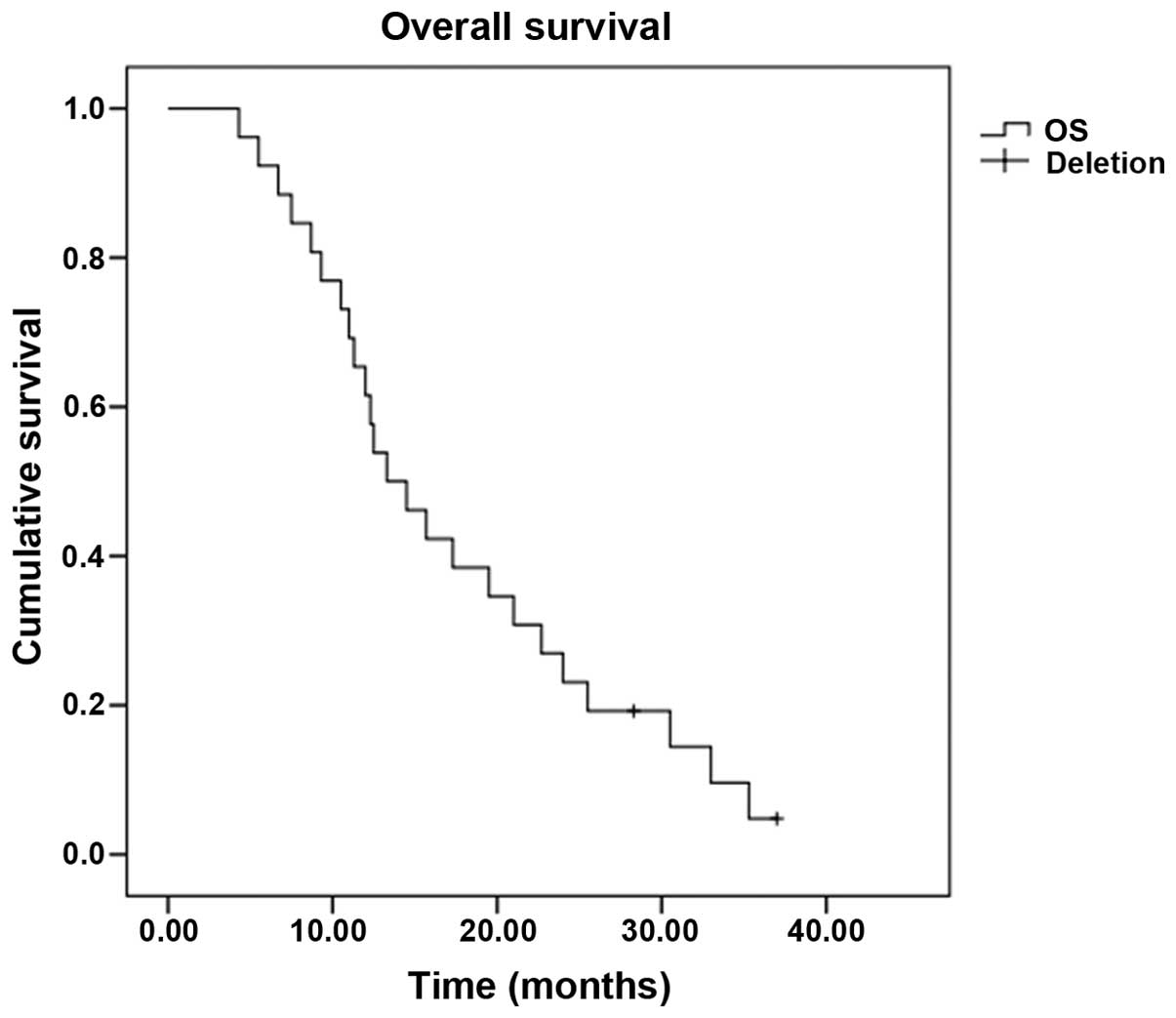
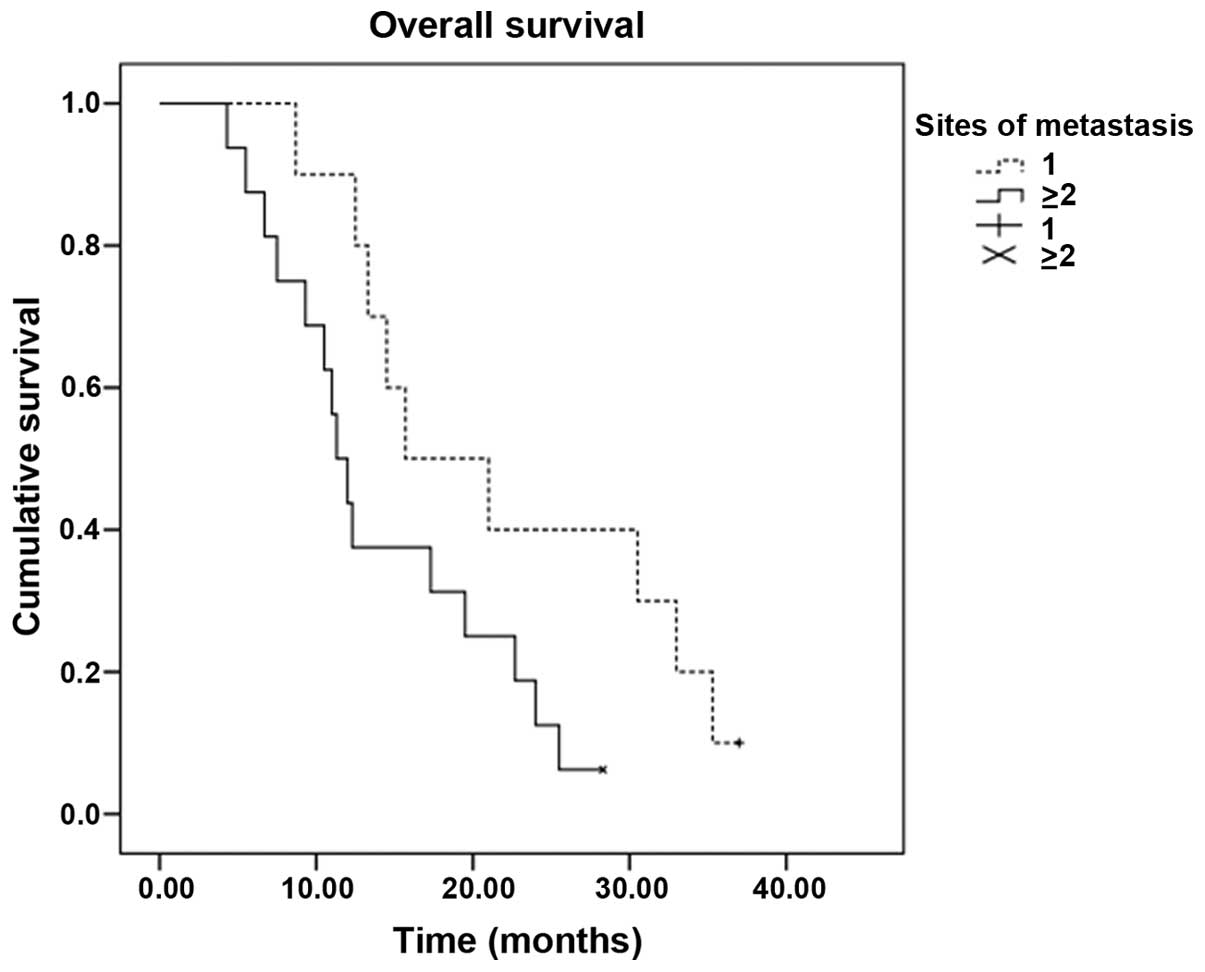
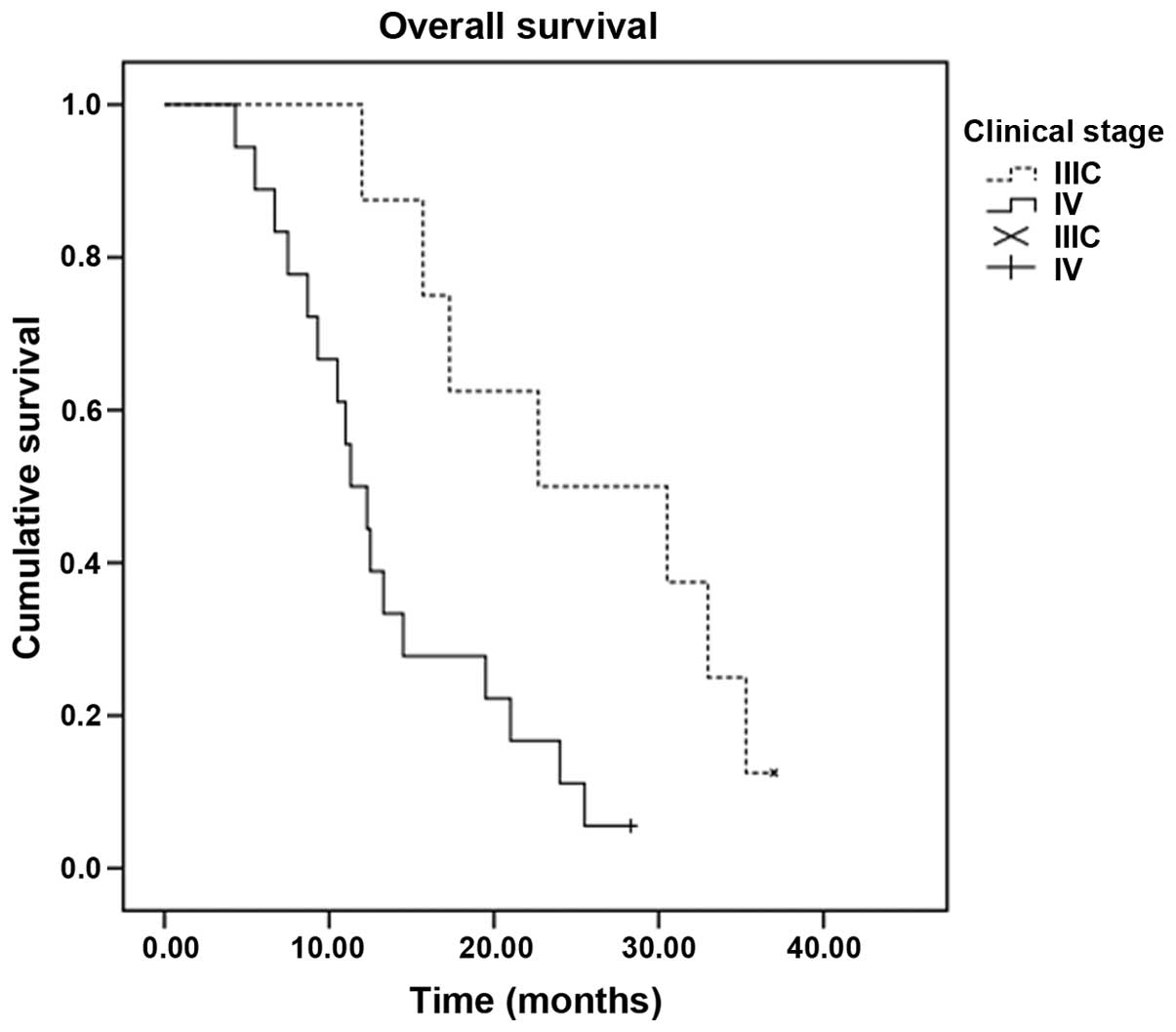
|
1
|
Vrdoljak E, Miše BP, Lukić B, et al:
Long-lasting control of triple-negative metastatic breast cancer
with the novel drug combination ixabepilone and capecitabine - case
report. Onkologie. 33:53–56. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rakha EA and Ellis IO:
Triple-negative/basal-like breast cancer: Review. Pathology.
41:40–47. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fei F, Chen C, Xue J, et al: Efficacy and
safety of docetaxel combined with oxaliplatin as a neoadjuvant
chemotherapy regimen for Chinese triple-negative local advanced
breast cancer patients. A prospective, open, and unicentric Phase
II clinical trial. Am J Clin Oncol. 36:545–551. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Oakman C, Viale G and Di Leo A: Management
of triple negative breast cancer. Breast. 19:312–321. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tan DS, Marchió C, Jones RL, et al: Triple
negative breast cancer: Molecular profiling and prognostic impact
in adjuvant anthracycline-treated patients. Breast Cancer Res
Treat. 111:27–44. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu M, Mo QG, Wei CY, et al:
Platinum-based chemotherapy in triple-negative breast cancer: A
meta-analysis. Oncol Lett. 5:983–991. 2013.PubMed/NCBI
|
|
7
|
Sumpter K, Harper-Wynne C, Cunningham D,
et al: Report of two protocol planned interim analyses in a
randomised multicentre phase III study comparing capecitabine with
fluorouracil and oxaliplatin with cisplatin in patients with
advanced oesophagogastric cancer receiving ECF. Br J Cancer.
92:1976–1983. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Huang J, Cao Y, Wu L, et al: S-1-based
therapy versus 5-FU-based therapy in advanced gastric cancer: A
meta-analysis. Med Oncol. 28:1004–1011. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hara F, Kiyoto S, Takahashi M, et al:
Efficacy and safety of S-1 in patients with metastatic breast
cancer: Retrospective review in a single institution. Oncology.
79:273–277. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Therasse P, Arbuck SG, Eisenhauer EA, et
al: New guidelines to evaluate the response to treatment in solid
tumors. European Organization for Research and Treatment of Cancer,
National Cancer Institute of the United States, National Cancer
Institute of Canada. J Natl Cancer Inst. 92:205–216. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Trotti A, Colevas AD, Setser A, et al:
CTCAE v3.0: Development of a comprehensive grading system for the
adverse effects of cancer treatment. Semin Radiat Oncol.
13:176–181. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Perou CM, Sørlie T, Eisen MB, et al:
Molecular portraits of human breast tumours. Nature. 406:747–752.
2000. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rastelli F, Biancanelli S, Falzetta A, et
al: Triple-negative breast cancer: Current state of the art.
Tumori. 96:875–888. 2010.PubMed/NCBI
|
|
14
|
Turner N, Tutt A and Ashworth A: Hallmarks
of ‘BRCAness’ in sporadic cancers. Nat Rev Cancer. 4:814–819. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fornier M and Fumoleau P: The paradox of
triple negative breast cancer: Novel approaches to treatment.
Breast J. 18:41–51. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
De Laurentiis M, Cianniello D, Caputo R,
et al: Treatment of triple negative breast cancer (TNBC): Current
options and future perspectives. Cancer Treat Rev. 36:(Suppl 3).
S80–S86. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rakha EA, El-Sayed ME, Green AR, et al:
Prognostic markers in triple-negative breast cancer. Cancer.
109:25–32. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rhee J, Han SW, Oh DY, et al: The
clinicopathologic characteristics and prognostic significance of
triple-negativity in node-negative breast cancer. BMC Cancer.
8:3072008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Geisler S, Lønning PE, Aas T, et al:
Influence of TP53 gene alterations and c-erbB-2 expression on the
response to treatment with doxorubicin in locally advanced breast
cancer. Cancer Res. 61:2505–2512. 2001.PubMed/NCBI
|
|
20
|
Gadhikar MA, Sciuto MR, Alves MV,
Pickering CR, Osman AA, Neskey DM, Zhao M, Fitzgerald AL, Myers JN
and Frederick MJ: Chk1/2 inhibition overcomes the cisplatin
resistance of head and neck cancer cells secondary to the loss of
functional p53. Mol Cancer Ther. 12:1860–1873. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dookeran KA, Dignam JJ, Ferrer K, et al:
p53 as a marker of prognosis in African-American women with breast
cancer. Ann Surg Oncol. 17:1398–1405. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chae BJ, Bae JS, Lee A, et al: p53 as a
specific prognostic factor in triple-negative breast cancer. Jpn J
Clin Oncol. 39:217–224. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hernandez-Aya LF, Chavez-Macgregor M, Lei
X, et al: Nodal status and clinical outcomes in a large cohort of
patients with triple-negative breast cancer. J Clin Oncol.
29:2628–2634. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lower EE, Glass E, Blau R and Harman S:
HER-2/neu expression in primary and metastatic breast cancer.
Breast Cancer Res Treat. 113:301–306. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lower EE, Glass EL, Bradley DA, et al:
Impact of metastatic estrogen receptor and progesterone receptor
status on survival. Breast Cancer Res Treat. 90:65–70. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lippman ME, Allegra JC, Thompson EB, et
al: The relation between estrogen receptors and response rate to
cytotoxic chemotherapy in metastatic breast cancer. N Engl J Med.
298:1223–1228. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Keam B, Im SA, Kim HJ, et al: Prognostic
impact of clinicopathologic parameters in stage II/III breast
cancer treated with neoadjuvant docetaxel and doxorubicin
chemotherapy: Paradoxical features of the triple negative breast
cancer. BMC Cancer. 7:2032007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Roy V, Pockaj BA, Allred JB, et al: A
Phase II trial of docetaxel and carboplatin administered every 2
weeks as preoperative therapy for stage II or III breast cancer:
NCCTG study N0338. Am J Clin Oncol. 36:540–544. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Villarreal-Garza C, Khalaf D, Bouganim N,
et al: Platinum-based chemotherapy in triple-negative advanced
breast cancer. Breast Cancer Res Treat. 146:567–572. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sirohi B, Arnedos M, Popat S, et al:
Platinum-based chemotherapy in triple-negative breast cancer. Ann
Oncol. 19:1847–1852. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Virag P, Perde-Schrepler M, Fischer-Fodor
E, et al: Superior cytotoxicity and DNA cross-link induction by
oxaliplatin versus cisplatin at lower cellular uptake in colorectal
cancer cell lines. Anticancer Drugs. 23:1032–1038. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dunn TA, Schmoll HJ, Grünwald V, Bokemeyer
C and Casper J: Comparative cytotoxicity of oxaliplatin and
cisplatin in non-seminomatous germ cell lines. Invest New Drugs.
15:109–114. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kang M, Jiang H, Wang S, et al: Efficacy
of docetaxel combined with oxaliplatin by different administration
routes in treatment of anthracycline-resistant metastatic breast
cancer. Zhong Guo Zhong Liu Lin Chuang Yu Kang Fu. 12:527–529.
2005.(In Chinese).
|
|
34
|
Thuss-Patience PC, von Minckwitz G,
Kretzschmar A, et al: Oxaliplatin and 5-fluorouracil for heavily
pretreated metastatic breast cancer: A preliminary phase II study.
Anticancer Drugs. 14:549–553. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Miura K, Kinouchi M, Ishida K, et al: 5-fu
metabolism in cancer and orally-administrable 5-fu drugs. Cancers
(Basel). 2:1717–1730. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lee JL, Kang YK, Kang HJ, et al: A
randomised multicentre phase II trial of capecitabine vs S-1 as
first-line treatment in elderly patients with metastatic or
recurrent unresectable gastric cancer. Br J Cancer. 99:584–590.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lv H, Yan M, Zhang M, et al: Efficacy of
capecitabine-based combination therapy and single-agent
capecitabine maintenance therapy in patients with metastatic breast
cancer. Chin J Cancer Res. 26:692–697. 2014.PubMed/NCBI
|
|
38
|
Gelmon K, Chan A and Harbeck N: The role
of capecitabine in first-line treatment for patients with
metastatic breast cancer. Oncologist. 11:(Suppl 1). 42–51. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shirasaka T, Tsukuda M, Inuyama Y and
Taguchi T: New oral anticancer drug, TS-1 (S-1) - from bench to
clinic. Gan To Kagaku Ryoho. 28:855–864. 2001.(In Japanese).
PubMed/NCBI
|
|
40
|
Koizumi W, Narahara H, Hara T, et al: S-1
plus cisplatin versus S-1 alone for first-line treatment of
advanced gastric cancer (SPIRITS trial): A phase III trial. Lancet
Oncol. 9:215–221. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Boku N: Gastrointestinal Oncology Study
Group of Japan Clinical Oncology Group: Chemotherapy for metastatic
disease: Review from JCOG trials. Int J Clin Oncol. 13:196–200.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shien T, Shimizu C, Akashi-Tanaka S, et
al: Clinical efficacy of S-1 in pretreated metastatic breast cancer
patients. Jpn J Clin Oncol. 38:172–175. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yamamoto D, Iwase S, Yoshida H, et al:
Efficacy of S-1 in patients with capecitabine-resistant breast
cancer-Japan Breast Cancer Research Network (JBCRN) 04-1 trial.
Anticancer Res. 30:3827–3831. 2010.PubMed/NCBI
|
|
44
|
Fujii K, Kosaka J, Mouri Y, et al:
Chemotherapy of a 2-week S-1 administration followed by 1-week rest
for advanced and metastatic breast cancer. Gan To Kagaku Ryoho.
38:1467–1470. 2011.(In Japanese). PubMed/NCBI
|
|
45
|
Saeki T: Clinical benefit of S-1 in
metastatic breast cancer. Gan To Kagaku Ryoho. 33:(Suppl 1).
202–206. 2006.(In Japanese). PubMed/NCBI
|