Introduction
Rhabdomyosarcoma is the most common childhood
sarcoma. It is a highly aggressive malignant tumor arising in the
mesenchymal cells and developing in the striated muscle (1). This particular tumor may appear in any
organ or tissue of the body, with the exception of the bone.
Approximately 35% of rhabdomyosarcomas occur in the head and neck
region, with ~50% of them arising in the parameningeal sites,
including the paranasal sinuses and nasal cavity (2).
Due to the rarity and largely unknown biological
behavior of rhabdomyosarcomas, the management of pediatric
sinonasal rhabdomyosarcoma (PSR) remains challenging. The cure rate
for localized disease has increased over the past 30 years due to
the use of multimodal therapy, which includes surgery and
multiagent chemotherapy with or without radiotherapy (1,3).
However, the majority of the patients have a poor prognosis, since
the tumor has a tendency towards early and extensive local and
metastatic dissemination (4).
The present study reported the case of a 2-year old
girl with rapid progressive clinical course of PSR, despite
multimodal therapy, including primary and salvage surgeries and
chemotherapy.
Case report
The present study was conducted in accordance with
the Declaration of Helsinki and was approved by the Ethics
Committee of the Akdeniz University School of Medicine (Antalya,
Turkey). Written informed consent was obtained from the patient's
family.
A 2-year old girl was referred to the Akdeniz
University School of Medicine in August 2013 with a 3-month history
of unilateral nasal obstruction and purulent rhinorrhea. Multiple
medications, including amoxicillin clavulanate, clarithromycin,
hypertonic nasal irrigation solutions and nasal decongestants,
prescribed by several different specialists due to diagnosis of
rhinosinusitis failed to improve the symptoms. Physical examination
showed a hemorrhagic, fragile and polypoid tumoral mass within the
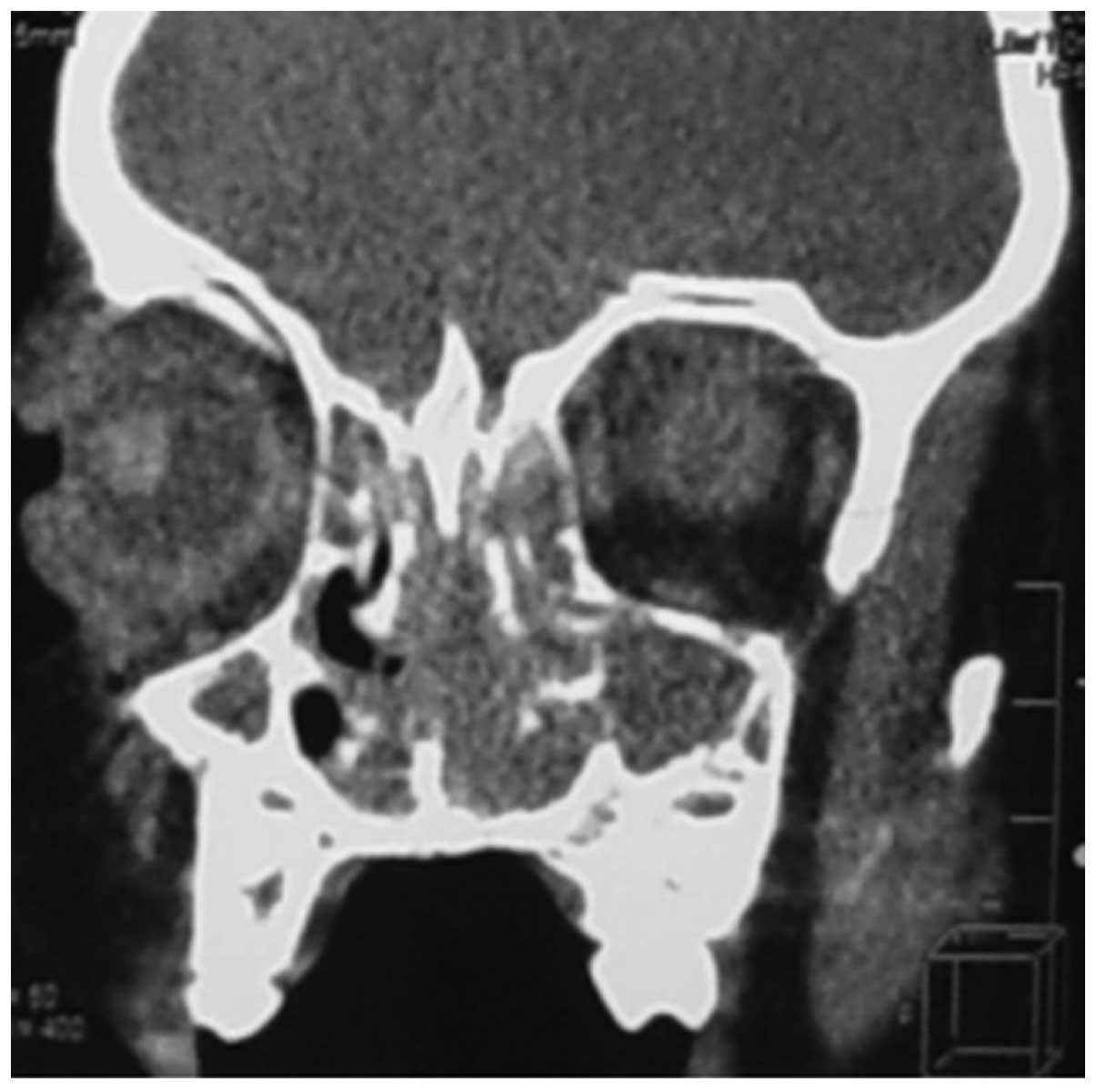
left nasal cavity. A paranasal computed tomography (CT) scan
revealed an irregular mass filling the entire left maxillary sinus,
ethmoidal sinus and nasopharynx (Fig.
1). Biopsy samples were obtained for definitive
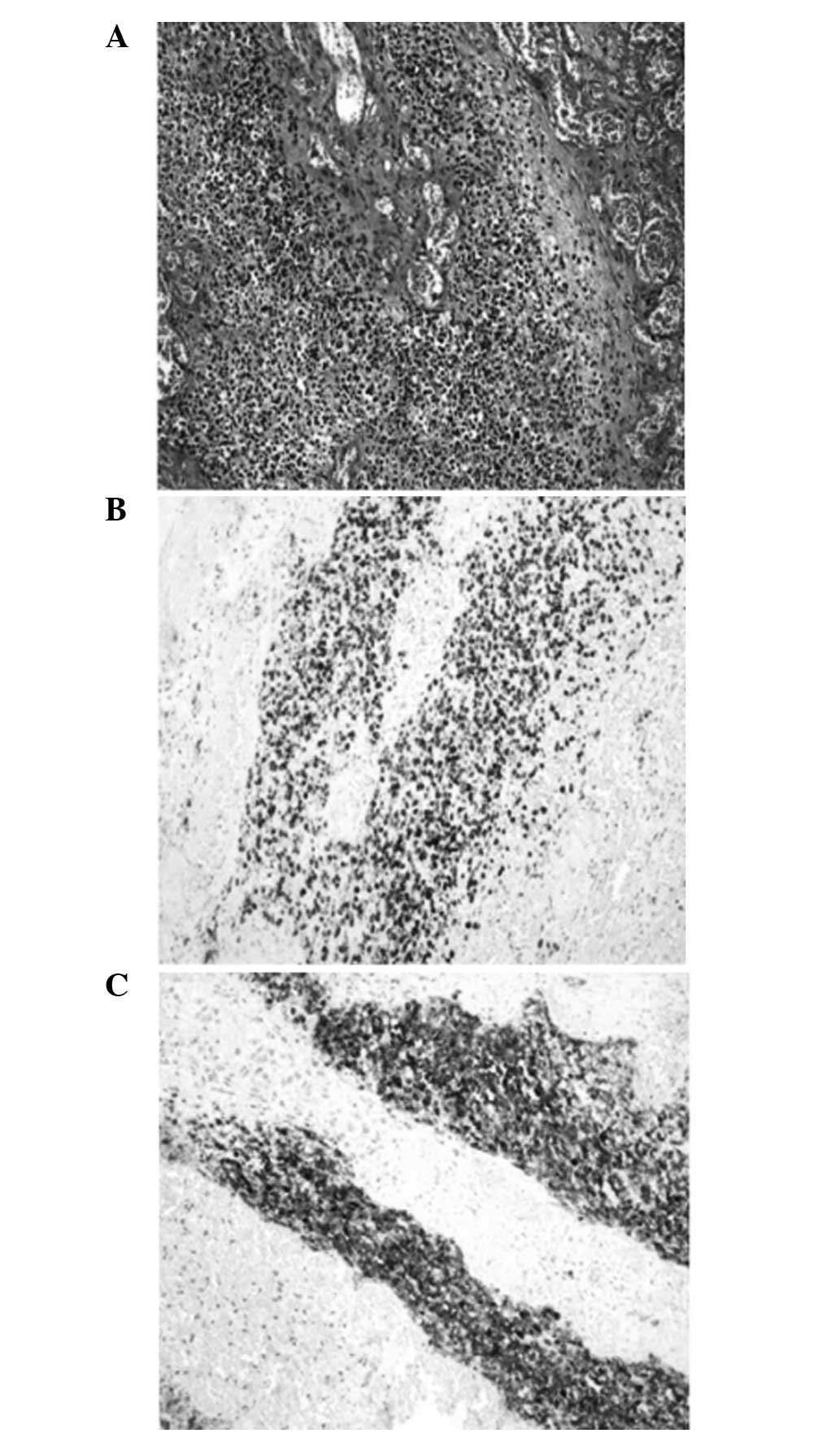
histopathological analysis. Mitotically active, small-round tumor
cells with a high nucleocytoplasmic ratio were detected upon
microscopic examination (Fig. 2A).
Immunohistochemical staining showed that the tumor exhibited strong
immunoreactivity for CD56 and desmin (Fig. 2B–C); however, no staining was
observed for myogenin, CD99 and CD20. These histological and
immunohistochemical findings were consistent with a diagnosis of
embryonal rhabdomyosarcoma.
The tumor was completely resected using an
endoscopic approach and the surgical margins were found to be
negative for tumor infiltration, while metastatic work-up was
negative for systemic dissemination. Thus, the patient was
diagnosed with stage IB disease, according to the Intergroup
Rhabdomyosarcoma Study (IRS) staging system (5). A total of six cycles of adjuvant
chemotherapy with vincristine (1.5 mg/m2, weekly) and
actinomycin-D (1.5 mg/m2, three times weekly) were
administered with no severe adverse events. On the 10th month of
follow-up (June 2014), a paranasal CT scan revealed a
local-regional recurrence within the left nasal cavity. Salvage
surgery was performed in order to remove the recurrent tumor using
an endoscopic approach. The recurrent tumor was completely resected
and histopathological analysis indicated a diagnosis of recurrent
rhabdomyosarcoma. The same chemotherapy regimen was resumed,
following salvage surgery. The pediatric patient developed a fungal
bronchopneumonia subsequent to the first cycle of chemotherapy and
was further treated with intravenous antifungal antibiotics.
However, medical treatment failed to improve the clinical condition
of the patient, and she was admitted to the Intensive Care Unit.
The results of blood gas test and chest radiography, which revealed
diffuse pulmonary infiltrates, were compatible with acute
respiratory distress syndrome (ARDS). The patient succumbed due to
disease progression, ARDS and septic shock 12 months after
diagnosis (August 2014).
Discussion
PSR, particularly at its earliest stages, produces
symptoms that may be incorrectly attributed to allergy and
infection. Nasal obstruction, epistaxis and drainage are often
exhibited prior to the presentation of more severe symptoms, such
as cranial neuropathy (4). As
observed in the present case, these indistinct symptoms may impede
the early diagnosis and treatment of the disease. All patients
suffering from these symptoms should therefore undergo an initial
complete clinical evaluation of their medical history, physical
examination and nasopharyngoscopy. Endoscopic evaluation usually
reveals a unilateral, polypoid mass (6), and a biopsy should be performed to rule
out the possibility of an invasive tumor.
Although pediatric rhabdomyosarcoma is a
morphologically and genetically heterogeneous tumor, two cell types
are common, the embryonal and alveolar types, each characterized by
specific genetic alterations (7).
The embryonal type is the most common histological type of head and
neck rhabdomyosarcoma (2), and has
been linked to the loss of homozygosity on chromosome 11p15, the
location of insulin-like growth factor II. By contrast, alveolar
tumors often have a chromosomal translocation [t(2;13)(q35;q14)]
that fuses the PAX3 gene with the FKHR gene,
contributing to tumorigenesis (7).
In contrast to other mucosa-associated head and neck
rhabdomyosarcomas, PSR predominantly shows an alveolar histology,
particularly the solid alveolar type (8). Using immunohistochemical staining, PSR
shows positive immunoreactivity for desmin and myogenin, and
negative immunoreactivity for cytokeratin, epithelial membrane
antigen, CD45, CD99 and S-100 protein (9). The majority of alveolar tumors are
strongly positive for myogenin, whereas embryonal tumors often
exhibit positivity to a lesser degree (9,10), which
is in agreement with the immunohistochemistry results of the
present case.
The embryonal and alveolar subtypes of pediatric
rhabdomyosarcoma have diverse prognostic factors and outcomes, with
5-year failure-free survival rates ranging between 31 and 87%
according to the patient age, stage, histological type and tumor
site (11). The overall outcomes of
patients with the alveolar histology are worse than those of
patients with the embryonal histology. In addition, due to its
parameningeal location, PSR has a potential for cerebrospinal fluid
dissemination, which contributes to a poor prognosis (12). Thereby, early diagnosis and optimal
management are important in reducing morbidity and mortality.
Comprehensive imaging studies, including paranasal CT or magnetic
resonance imaging scans, should be performed in order to
anatomically localize the tumor site and extent of the disease, and
to determine the correct disease stage.
The treatment of pediatric rhabdomyosarcoma often
requires a multimodal therapy, including surgery, chemotherapy and
radiotherapy (3); however, it is not
currently possible to determine the best treatment strategy on the
basis of high-level scientific evidence, since previous IRS trials
comprise a heterogeneous population and do not evaluate the
treatment outcomes specifically for PSR (11). The surgical goal is the complete
excision of the primary tumor, whenever possible, without causing
major functional or cosmetic deficits. Incompletely resected tumors
or tumors with a positive resection margin are generally treated
with radiotherapy (13). Novel
techniques of implementing radiotherapy, including
intensity-modulated, proton beam radiotherapy and brachytherapy may
reduce long-term sequelae, as compared with three-dimensional
conformal radiotherapy (14). In
addition, all patients with rhabdomyosarcoma require treatment with
chemotherapy (15). The most
commonly used regimens include vincristine and actinomycin-D in
combination with an alkylating agent, such as cyclophosphamide or
ifosfamide. Compared with tumors in other regions, PSRs have a
decreased response to treatment, a relapse rate of 30–40% and a
greater potential for early metastasis (16). The majority of rhabdomyosarcoma
recurrences involve the primary site or adjacent region. Although
salvage surgery is advocated for resectable residual or recurrent
PSR, the evidence for its efficacy is considerably limited
(15,16).
In conclusion, PSR is a rare aggressive malignancy
associated with poor outcomes. Only a limited number of PSR cases
have previously been reported (2,4,6,8,11,13,14). The
present report emphasized the prognostic significance of the tumor
site and the age upon diagnosis of rhabdomyosarcoma, as well as the
necessity of a multidisciplinary management strategy in clinical
centers with adequate experience.
References
|
1
|
McDowell HP: Update on childhood
rhabdomyosarcoma. Arch Dis Child. 88:354–357. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hicks J and Flaitz C: Rhabdomyosarcoma of
the head and neck in children. Oral Oncol. 38:450–459. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Walterhouse D and Watson A: Optimal
management strategies for rhabdomyosarcoma in children. Paediatr
Drugs. 9:391–400. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Herrmann BW, Sotelo-Avila C and Eisenbeis
JF: Pediatric sinonasal rhabdomyosarcoma: Three cases and a review
of the literature. Am J Otolaryngol. 24:174–180. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lawrence W Jr, Anderson JR, Gehan EA and
Maurer H: Pretreatment TNM staging of childhood rhabdomyosarcoma: A
report of the Intergroup Rhabdomyosarcoma Study Group. Children's
Cancer Study Group. Pediatric Oncology Group. Cancer. 80:1165–1170.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fyrmpas G, Wurm J, Athanassiadou F,
Papageorgiou T, Beck JD, Iro H and Constantinidis J: Management of
paediatric sinonasal rhabdomyosarcoma. J Laryngol Otol.
123:990–996. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pappo AS, Shapiro DN, Crist WM and Maurer
HM: Biology and therapy of pediatric rhabdomyosarcoma. J Clin
Oncol. 13:2123–2139. 1995.PubMed/NCBI
|
|
8
|
Ahmed AA and Tsokos M: Sinonasal
rhabdomyosarcoma in children and young adults. Int J Surg Pathol.
15:160–165. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kumar S, Perlman E, Harris CA, Raffeld M
and Tsokos M: Myogenin is a specific marker for rhabdomyosarcoma:
An immunohistochemical study in paraffin-embedded tissues. Mod
Pathol. 13:988–993. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cessna MH, Zhou H, Perkins SL, Tripp SR,
Layfield L, Daines C and Coffin CM: Are myogenin and MyoD1
expression specific for rhabdomyosarcoma? A study of 150 cases,
with emphasis on spindle cell mimics. Am J Surg Pathol.
25:1150–1157. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Meza JL, Anderson J, Pappo AS and Meyer
WH: Children's Oncology Group: Analysis of prognostic factors in
patients with nonmetastatic rhabdomyosarcoma treated on intergroup
rhabdomyosarcoma studies III and IV: The Children's Oncology Group.
J Clin Oncol. 24:3844–3851. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sturgis EM and Potter BO: Sarcomas of the
head and neck region. Curr Opin Oncol. 15:239–252. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Daya H, Chan HS, Sirkin W and Forte V:
Pediatric rhabdomyosarcoma of the head and neck: Is there a place
for surgical management? Arch Otolaryngol Head Neck Surg.
126:468–472. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gosiengfiao Y, Reichek J and Walterhouse
D: What is new in rhabdomyosarcoma management in children? Paediatr
Drugs. 14:389–400. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Buwalda J, Blank LE, Schouwenburg PF,
Copper MP, Strackee SD, Voûte PA, Merks JH and Caron HN: The AMORE
protocol as salvage treatment for non-orbital head and neck
rhabdomyosarcoma in children. Eur J Surg Oncol. 30:884–892. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Paulino AC, Bauman N, Simon JH, Nguyen TX,
Ritchie JM and Tannous R: Local control of parameningeal
rhabdomyosarcoma: Outcomes in non-complete responders to
chemoradiation. Med Pediatr Oncol. 41:118–122. 2003. View Article : Google Scholar : PubMed/NCBI
|