Introduction
Panniculitis is a group of heterogeneous
inflammatory disorders of the subcutaneous adipose tissue. The
diagnosis of panniculitis requires histopathologic examination.
Clinically, patients with panniculitis frequently present with
multiple palpable nodules on the trunk or lower extremities.
According to the predominant location of the microscopic
inflammation determined by histopathology, panniculitis can be
divided into two subtypes: Septal or lobular (1,2).
However, panniculitis of breast tissue as the first manifestation
has rarely been reported and is often misdiagnosed as mastitis, or
even breast cancer during clinical examination. Without proper and
timely treatment, breast panniculitis can cause significant
morbidity. First, at acute inflammatory phase, breast becomes
painful. Later the skin develops ulceration and infection. At
chronic phase, the inflammation is resolved, but breast can be left
with deep atrophic scars, cosmetic disfigurement, which may lead to
psychiatric sequelae for some patients (3). So, early diagnosis and treatment are
very important for the prognosis of breast panniculitis. Hereby we
reported a patient suffered from the liquefactive fat necrosis of
breast tissue, a rare case of panniculitis who first presented with
irregular breast nodules, followed by erythema nodosum of both
lower extremities, to help understand the early presentation of
breast panculitis for future diagnosis.
Case report
A 37-year-old woman without a relevant medical
history presented with fever and recently occurred painful
subcutaneous nodules predominantly in the upper quadrants of his
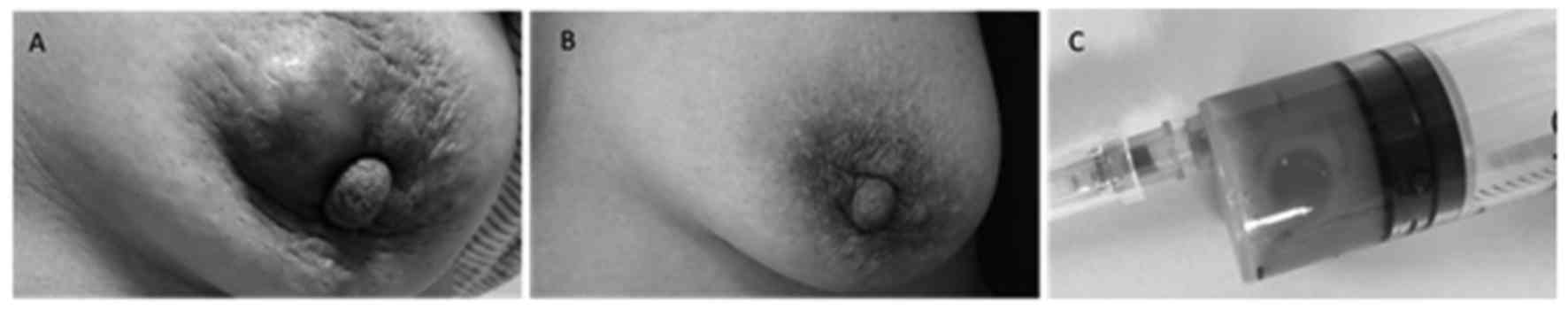
left breast. Physical exam identified an area of poorly-defined,
marked induration and tenderness on her breast, with inflammatory
changes on the skin but without nipple discharge or retraction
(Fig. 1). The right breast appeared
normal. The highest body temperature was 38.5°C.
The patient was initially seen in the Department of
Surgeon. At the first visit, physical examination showed tenderness
and irregular subcutaneous nodules at the upper inner quadrant of
her left breast, accompanied by mild local skin inflammation,
without nipple discharge and swelling of axillary lymph nodes. The
patient lacks any history of breast trauma, tumor and connective
tissue diseases. X-ray of her left breast showed no clear
abnormalities of the skin, nipple and subcutaneous adipose tissue.
Several patchy hyperintense signals, linear streaky shadows and
fuzzy nodules were seen at the left breast. All signals were evenly
dispersed and scattered calcifications were also visible. There was
no localized soft tissue mass and clustered micro-calcification in
the left breast tissue (Fig. 2). The
patient was first treated with cefaclor (Sustained Release Tablets,
0.375 g orally twice daily) and topical drugs lactate ethacridine
solution, twice daily for 3 months. However, the treatment was not
effective. Subsequently, she underwent breast ultrasonography. The
pattern of left breast tissue was clear, the echo of the glands was
uneven, mixed with strong and weak signals, and there was no
expansion of the breast duct. A reduced echo of subcutaneous soft
tissue was shown at the upper inner quadrant of her left breast.
The area was approximately 4.4×1.5 cm in size, without a clear
margin, and the echo was not uniform, indicating abundant blood
flow but a lack of clear boundary with the rear glands. The rear
glands also showed a reduced and uneven echo without evident mass,
and significant local tenderness (Fig.
2B).
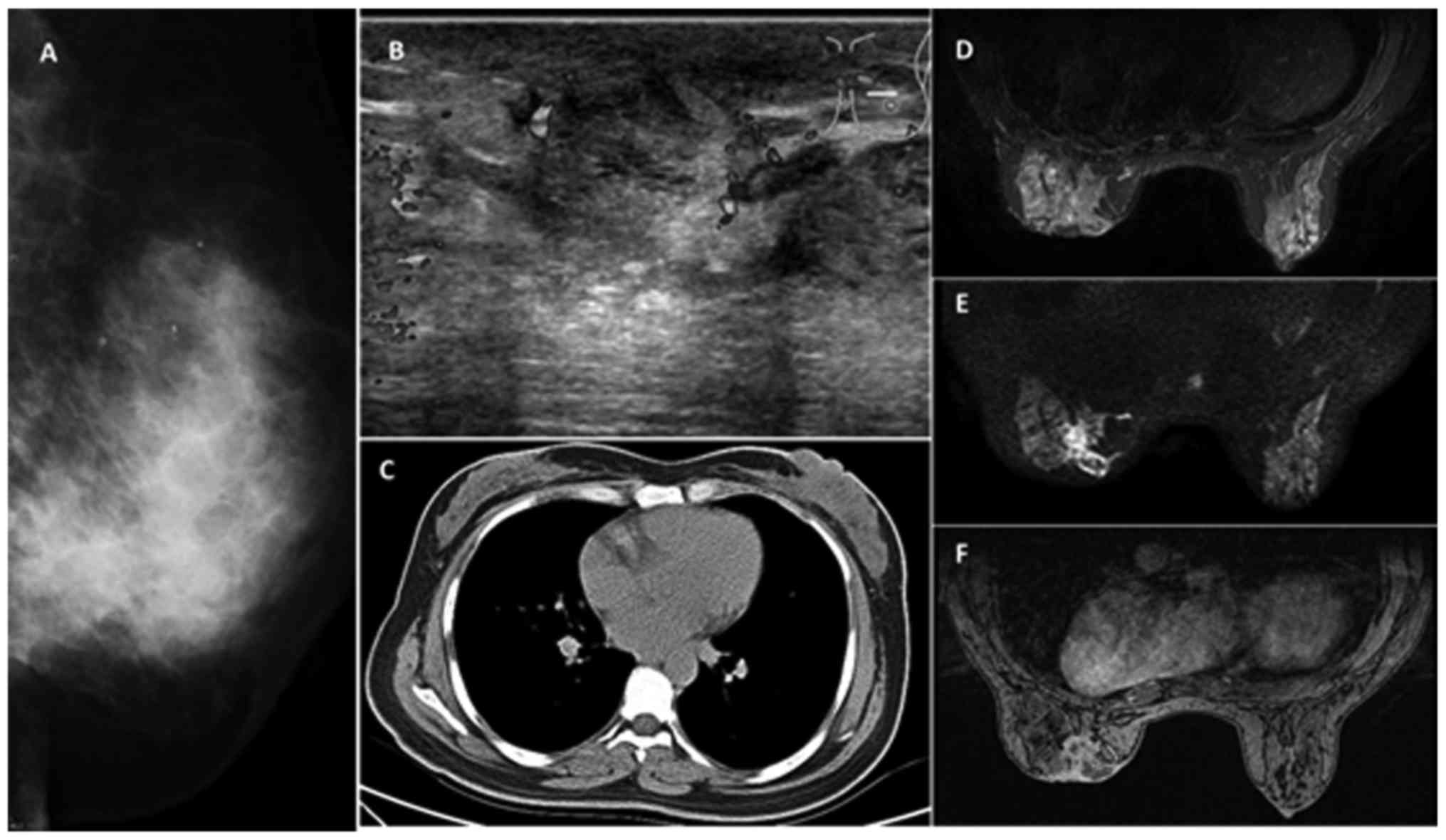 | Figure 2.(A) Mammography, multiple patchy
hyperintense signals, linear shadows and fuzzy nodules at the left
breast. All signals were evenly dispersed. (B) Breast ultrasound,
gland echo is not homogeneous, mixed with strong and weak signals,
no expansion of the breast duct, a reduced echo of subcutaneous
soft tissue at the upper inner quadrant. The area was approximately
4.4×1.5 cm in size, without a clear margin, and the echo was not
uniform. (C) Breast computed tomography, left breast skin
thickening, uneven density of breast tissues and destruction of
normal gland structure. (D) Breast MRI, T2WI imaging, focal,
irregular sized abscess shadow. (E) Breast MRI diffusion-weighted
sequence, focal, patchy and circular signal. (F) Breast MRI dynamic
enhancement imaging, flake-like and circular lesion signals, and
large irregular abscess walls. MRI, magnetic resonance imaging;
T2WI, T2-weighted image. |
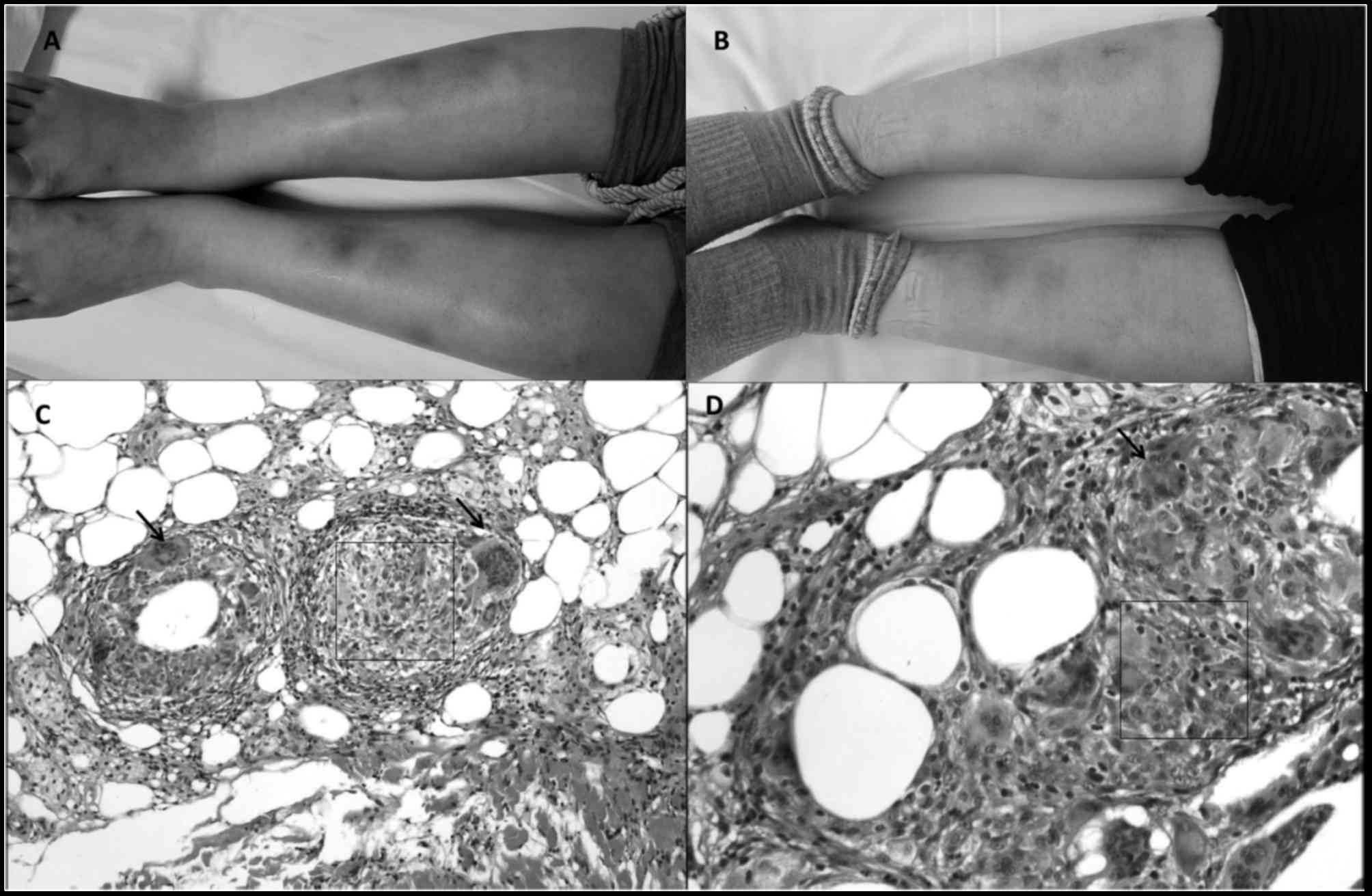
After three months of antibacterial and topical
treatment, dark reddish plaques and tender skin lesions were
identified over both legs, 3–4 cm in size (Fig. 3). The patient was then referred to
the Department of Rheumatology and Immunology. At the time of
visit, the patient was depressed and admitted for further
evaluation. Upon admission, initial laboratory results revealed
normal blood count, increased erythrocyte sedimentation rate (27
mm/h) and C-reactive protein (1.05 mg/ml). Pro-calcitonin,
β-D-glucan, PPD-test, T-SPOT were all negative. Breast puncture
fluid was purulent (Fig. 1C). The
puncture fluid routine test showed white blood cell of 40/HPF, and
negative on bacteria fungus mycobacterium tuberculosis infection
from the puncture fluid culture. Histopathological findings of
lower limb skin and subcutaneous tissue showed no obvious
atypicality of epithelial cells, mild proliferation of
subepithelial fibrous tissue, the distribution of granuloma
composed of epithelioid cells and multinucleated giant cells along
the leaflet interval. Immunohistochemical staining: acid-fast
staining, PAS and PASM were all negative.
The pathological results indicated septal
panniculitis, known as erythema nodosum (Fig. 3C). ANA titer was 1:320, but the
results of anti-dsDNA, anti-CCP antibody, antineutrophil
cytoplasmic antibody (ANCA), anti-phospholipid antibody,
immunoglobulin G, complement factors C3 and C4 were all within the
normal range. Chest computed tomography (CT; Fig. 2C) showed thickening of the left
breast skin, rugged local skin, increased density of subcutaneous
adipose tissue, CT value of 10. The density of left breast tissue
was not uniform, and the normal gland structure also disappeared.
Left breast magnetic resonance imaging (MRI) also showed the
disappearance of normal gland structure. T2-weighted image (T2WI)
imaging revealed small focal lesions, large areas of irregular
abscess-like changes, most significant within the inner upper
quadrant, outer lower quadrant and behind the areola. The lesions
at the inner upper quadrant also involved the subcutaneous fat and
skin, showing local skin thickening with rugged surface and abscess
formation connected with the glands, as well as the retraction of
left nipple (Fig. 2D). DWI imaging
showed dotted, patchy and annular high signals (Fig. 2E). Dynamic enhancement imaging
revealed the flake-like and circular lesions, with large irregular
abscess walls that were shown as mild to moderate early
enhancement, and strengthened late continuous enhancement (Fig. 2F).
According to the clinical presentation, laboratory,
imaging and pathological results, the patient was diagnosed as
breast panniculitis with liquefactive fat necrosis. The patient
received systemic corticosteroids, intravenous methylprednisolone
80 mg daily for 3 days, following oral prednisone 60 mg daily,
combined with methotrexate (12.5 mg/W) and thalidomide (50
mg/day).
After 3 days of treatment, the patient's body
temperature returned to normal, nodular erythema of lower
extremities, and breast tenderness all significantly subsided.
After 1 month of treatment, glucocorticoids were tapered off. After
2 months of treatment, mastoid nodules and lower extremity nodules
erythema disappeared (Figs. 1B and
3B). The breast appeared to be
normal without scar formation. The patient's anxiety and depression
were also improved. The patient provided written informed consent
for the publication of their data.
Discussion
Panculitis is a rare inflammatory disorder of
subcutaneous adipose tissue. The characteristic painful nodules are
observed primarily in the area of the lower extremities and the
trunk (4). However, the breast can
also be seldom involved. For mammary glanditis the imaging study
and systematic treatment are rarely reported.
Breast panniculitis is characterized by subcutaneous
painful mass of the breast, accompanied by fever and fatigue and
other systemic inflammation, as well as depression especially in
women. This paper reported a case of papillitis with mammary gland
involvement as the early presentation, and provided a detailed
description of ultrasonography, X-ray, CT, MRI and other imaging
findings of breast panniculitis. The treatment of breast
panniculitis and follow-up were also recorded in detail. Together
this case report provided a good reference for the future diagnosis
and treatment of breast panniculitis. In clinical practice when the
patient presents with breast pain, in addition to the commonly seen
suppurative mastitis and tumor, papillitis should also be taken
into consideration.
Breast nodule within a few centimeters in size may
be visualized by ultrasound. The imaging findings of
ultrasonography have been shown to be associated with the stage of
disease and the degree of fibrosis (5,6). In the
acute inflammatory stage, adipocytes are destroyed or necrotized,
accompanied by the infiltration of inflammatory cells. The
ultrasound imaging features high echo nodules with internal
hypoechoic cyst or no echo capsule, and unevenly echoed tissue
shadow. In the recovery and chronic phase, along with tissue
fibrosis, ultrasound imaging shows diffused high echo mass, and
lesions with margins that can be clear, fuzzy or needle-shaped
depending on the degree of fibrosis (7).
Breast malignancy typically features: i) Irregular
shaped tumor with spicule sign and lobular sign; clustered or sandy
like micro-calcifications; invasion of local skin, subcutaneous fat
layer and chest wall, and axillary lymph node enlargement. ii)
Blood flow increase in and around the nodule (8). The breast lesion of our patient did not
present these features. Rather, the sonography report suggested
inflammatory lesions. This result suggested the breast lesion was
inflammation.
It has also been reported that for the diagnosis of
breast panniculitis, mammography is superior to ultrasonography,
especially for evaluating early micro calcification. Early
calcifications frequently resemble malignancy and might present in
a ductal distribution. In our case, breast X-ray imaging showed
local mass shadow that could be penetrated by the X-ray, there was
no apparent calcification, and fat necrosis is also shown as the
penetrable cavity. The variable mammography features can be
attributed to the degree of fibrosis in different stages of
panculitis (6,9). Characteristic presentation of breast
malignancy include: local invasion, spicule sign, clustered
calcifications; local skin thickened, nipple retraction, etc. The
mammography of our patient showed patchy hyper densities, to which
the radiologist suggested further evaluation.
We noted the breast lesion on the CT. The lesion was
an irregular shaped mass. However, non-contrast chest CT is
insufficient to differentiate between benign and malignant breast
lesions (10). Contrast-enhanced
breast CT might clearly display the lesion size, location, density,
appearance, blood vessel around the lesion, and provide better
differential diagnosis for early breast cancer. Unfortunately, our
patient only underwent high-resolution CT scans.
Magnetic resonance imaging has used high spatial
resolution techniques for soft tissue evaluations, and has unique
contributions for the diagnosis of breast disease as a
radiation-free and no-invasive method. To our knowledge, the MRI of
breast panniculitis has rarely been reported in the literature.
Pinho et al found that on MRI imaging, breast panculitis was
shown as a large irregular-shaped, heterogeneous shadow with high
signal intensity on T2-weighted images, medium signal intensity on
T1-weighted images, and regional enhancement. These features are
nonspecific and can be attributed to inflammatory process and edema
(9). Kinetic analysis also showed
continuous enhancement, suggesting a benign lesion. For the early
stage of panculitis accompanied by focal edema, MRI shows
hyperintense signal on T2-weighted images. The stage of fat
necrosis consists of oil cysts, which can be shown as circular,
well-circumcised hyperintense areas on T1-weighted images (11). Our patient presented nodular, patchy,
small annular and large irregular cavities and hyperintense signal
on T2-weighted images, and circular, focal, patchy hyperintense
signal on DWI. These features suggested inflammatory and benign
nature of the lesions. But at the final stage of the disease,
panculitis could present with indistinct or speculated margins and
structural distortion on MRI, which may be indistinguishable from
breast malignancy.
Histopathology can also be helpful to ascertain the
type of breast panniculitis, distinguish it from other lesions, and
guide the further therapy. The diagnosis of breast panniculitis is
different. However, upon careful review of histopathology, a
specific diagnosis can be appropriately made. First, we should know
that breast panniculitides are mixed panniculitis because of
inflammatory infiltration involving both the septa and the lobules
of the subcutaneous adipose tissue. Second, the pathological
examination is to assess whether vasculitis is present. Third, it
is necessary to identify the nature of the inflammatory
infiltration and additional histopathologic features of the
lesions, in order to draw a specific diagnosis of the disease that
involves the subcutaneous fat (12,13). Our
patient mostly presented fat necrosis of left breast. According to
the characteristics of adipocytes necrosis, there are five
histopathologic types, including lipophagic necrosis, liquefactive
fat necrosis, hyalinizing fat necrosis, membranous fat necrosis,
and ischemic fat necrosis (14).
With regard to our patient, the extracted fatty tissue was
liquefactive, but etiological examination was negative. Therefore,
the lesion was considered mostly lobular panniculitis with
liquefactive fat necrosis. The presence of vasculitis and nature of
infiltrated cells were not assessed.
At present, there is no established therapeutic
strategies for breast panniculitis. Many patients underwent
surgical treatment. Therefore, the early diagnosis and timely
treatment is very important, to avoid surgery and irreversible
damage of the breast. Firstly, lobular panniculitis is generally
resulted from systematic diseases, such as systemic lupus
erythematosus, pancreatic diseases, α1-antitrypsin deficiency,
infections, trauma, etc. So, the treatment of root causes is
critical. When the etiology cannot be determined, such as in this
case, panniculitis is termed as idiopathic. According to
pathological characteristics, a large number of inflammatory cells
were present in the lesion. Idiopathic panniculitis refers to a
condition of non-infective fat tissue inflammation. Rotaru et
al reported a case of non-suppurative nodular breast
panniculitis with systemic symptoms. After treated with systemic
corticosteroids, the condition improved (15). Yuan reported a case of isolated
breast panniculitis without evidence of systemic inflammation, who
underwent resection without systemic corticosteroid or
immunosuppressive therapy (16). The
patient did not suffer a relapse. Others also reported the isolated
breast panniculitis cases that had an excellent prognosis with
resection only (17,18). Our patient was complicated by fever,
nodular erythema of both lower extremities, and ESR elevation.
Although the lesion was punctured and drained, the patient's
overall symptoms had not improved. Therefore, we gave the patient
experimental glucocorticoid therapy, and follow-up with
immunosuppressive therapy. Her general condition, breast lesions,
lower extremity erythema all markedly improved as a result. So far,
the patient has not had a relapse.
Breast panniculitis is a rare condition, can be
mistaken for cancer at clinical examination, frequently associated
with systemic symptoms and occasional visceral involvement. Through
histopathology and imaging examinations, it can be diagnosed and
treated early, to avoid further visceral involvement and breast
disfiguration. From the experience of our patient, glucocorticoids
combined with immunosuppressive therapy could prevent the
relapse.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Requena L and Yus Sanchez E: Panniculitis.
Part II. Mostly lobular panniculitis. J Am Acad Dermatol.
45:325–364. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Requena L and Yus ES: Panniculitis. Part
I. Mostly septal panniculitis. J Am Acad Dermatol. 45:163–186.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Grossberg E, Scherschun L and Fivenson DP:
Lupus profundus: Not a benign disease. Lupus. 10:514–516. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chowaniec M, Starba A and Wiland P:
Erythema nodosum-review of the literature. Reumatologia. 54:79–82.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Taboada JL, Stephens TW, Krishnamurthy S,
Brandt KR and Whitman GJ: The many faces of fat necrosis in the
breast. AJR Am J Roentgenol. 192:815–825. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tan PH, Lai LM, Carrington EV, Opaluwa AS,
Ravikumar KH, Chetty N, Kaplan V, Kelley CJ and Babu ED: Fat
necrosis of the breast-a review. Breast. 15:313–318. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ng AM, Dissanayake D, Metcalf C and Wylie
E: Clinical and imaging features of male breast disease, with
pathological correlation: A pictorial essay. J Med Imaging Radiat
Oncol. 58:189–198. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tan TJ, Leong LC and Sim LS: Clinics in
diagnostic imaging (147). Male breast carcinoma. Singapore Med J.
54:347–352. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pinho MC, Souza F, Endo E, Chala LF,
Carvalho FM and de Barros N: Nonnecrotizing systemic granulomatous
panniculitis involving the breast: Imaging correlation of a breast
cancer mimicker. AJR Am J Roentgenol. 188:1573–1576. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bach AG, Abbas J, Jasaabuu C, Schramm D,
Wienke A and Surov A: Comparison between incidental malignant and
benign breast lesions detected by computed tomography: A systematic
review. J Med Imaging Radiation Oncol. 57:529–533. 2013. View Article : Google Scholar
|
|
11
|
Kuhl CK, Mielcareck P, Klaschik S, Leutner
C, Wardelmann E, Gieseke J and Schild HH: Dynamic breast MR
imaging: Are signal intensity time course data useful for
differential diagnosis of enhancing lesions? Radiology.
211:101–110. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ferrara G, Stefanato CM, Gianotti R, Kubba
A and Annessi G: Panniculitis with vasculitis. G Ital Dermatol
Venereol. 148:387–394. 2013.PubMed/NCBI
|
|
13
|
Wick MR: Panniculitis: A summary. Semin
Diagn Pathol. 34:261–272. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
White WL, Wieselthier JS and Hitchcock MG:
Panniculitis: Recent developments and observations. Semin Cutan Med
Surg. 15:278–299. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rotaru N, Punga J, Codreanu I,
Gavrilasenco I, Manea D and Cujba N: Nonsuppurative nodular
panniculitis of the breast. Clin Breast Cancer. 15:e219–e221. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yuan WH, Li AF, Hsu HC and Chou YH:
Isolated panniculitis with vasculitis of the male breast suspicious
for malignancy on CT and ultrasound: A case report and literature
review. Springerplus. 3:6422014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hernandez-Rodriguez J, Tan CD, Molloy ES,
Khasnis A, Rodriguez ER and Hoffman GS: Vasculitis involving the
breast: A clinical and histopathologic analysis of 34 patients.
Medicine (Baitimore). 87:61–69. 2008. View Article : Google Scholar
|
|
18
|
Kafantari E, Sotiropoulou M, Sfikakis P,
Dimitrakakis K, Zagouri F, Mandrekas K, Dimopoulos S, Dimopoulos MA
and Papadimitriou CA: Giant cell arteritis of the breast and breast
cancer: Paraneoplastic manifestation or concomitant disease? A case
report. Onkologie. 31:685–688. 2008.PubMed/NCBI
|