Introduction
With the continuous development of global economy
and the accelerated rhythms of life and work, people's daily
dietary patterns have changed. As a result, the incidence rate of
hypertension is on the increase, and increasingly younger patients
are affected by this disease (1).
Drug therapies are used as the main way of treatment for
hypertension, which consist of β-receptor blocker, diuretics,
calcium channel blocker, angiotensin-converting enzyme inhibitor,
angiotensin II receptor blocker and other drugs. Although the
therapies can effectively lower the blood pressure and control it
at a certain level, they bring a series of side effects to the body
at the same time, including abnormal lipid metabolism, evocation of
diabetes, increase in heart rate, gastrointestinal dysfunction,
abnormal liver function and impairment of renal function.
Therefore, it is necessary and important to explore prevention and
treatment measures for hypertension (2,3). There
are huge numbers of microorganisms in human body, which mainly
concentrate in the gastrointestinal tract. Currently the intestinal
floras that have been detected are Bacteroides, Firmicutes,
Fusobacterium, Actinomycetes, Verrucomicrobia,
Proteobacteria, Spirochaetes, Cyanobacteria, VadinBE97 and
Methanobrevibacter (4). The
intestinal floras are maintained in a relatively balanced state in
the human body under normal conditions, which can regulate and
nurture the intestines, influence the immune system of the body,
effectively prevent the invasion of pathogenic bacteria and have a
close association with hypertension (5). Bacteroides thetaiotaomicron is
one of the largest microflora in the intestinal tract.
Eubacterium rectale is a conditional pathogen.
Bifidobacterium is a probiotic bacterium that has a variety
of functions in the intestine. The selection of these three
bacteria can completely reflect the basic situation of the whole
intestinal flora of human body. The treatment principle of
hypertension is that the blood pressure is decreased slowly and
steadily. Τherefore, dietary intervention and other
non-pharmacologic treatments have a positive effect on the body's
metabolism; they have important impacts on the intestinal flora
(6). In this study, the content of
intestinal flora in hypertension patients was studied, and its
correlation with blood pressure was analyzed.
Patients and methods
General patient data
Ninety-four patients with hypertension who were
admitted and treated in No. 215 Hospital of Shaanxi Nuclear
Industry (Xianyang, China) from May 2016 to April 2017 were
selected as the observation group. Inclusion criteria: i) Patients
whose blood pressure was measured 3 times for 2 consecutive days,
with systolic blood pressure (SBP) ≥140 mmHg and (or) diastolic
blood pressure (DBP) ≥90 mmHg; ii) patients who had clear
consciousness and could communicate with others normally; and iii)
patients who signed the informed consent. Exclusion criteria: i)
Patients with congenital heart disease, severe infectious disease,
malignant tumor or serious liver disease; ii) patients with a
history of diarrhea or other gastrointestinal diseases in the
previous 1 month; iii) patients who took steroidal drugs,
antibiotics and probiotics in the prior 4 weeks; and iv) patients
with secondary hypertension. Ninety-four healthy people from the
physical examination center of No. 215 Hospital of Shaanxi Nuclear
Industry in the same time period were selected as the control
group. This study was checked and approved by the Ethics Committee
of No. 215 Hospital of Shaanxi Nuclear Industry (Xianyang, China),
and the differences in the general information between the two
groups of participants were not statistically significant
(P>0.05) (Table I) Signed
informed consents were obtained from the patients or guardians.
 | Table I.General data of research objects. |
Table I.
General data of research objects.
|
| Groups |
|
|
|---|
|
|
|
|
|
|---|
| Item | Observation
(n=94) | Control (n=94) | t/χ2 | P-value |
|---|
| Age (years) | 30–78 | 30–75 |
|
|
| Sex
(male/female) | 49/45 | 46/48 | 0.085 | 0.771 |
| Average age
(years) | 57.76±7.42 | 58.15±7.73 | 0.353 | 0.724 |
| Body mass index (BMI)
(kg/m2) | 21.78±2.54 | 22.07±2.23 | 0.832 | 0.406 |
| Educational level (n,
%) |
| Junior
high school and below | 17 (18.08) | 16 (17.02) |
|
|
| Senior
high school and special secondary school | 46 (48.94) | 49 (52.13) | 0.192 | 0.909 |
| College
and above | 31 (32.98) | 29 (30.85) |
|
|
| Smoking (n, %) | 26 (27.66) | 23 (24.47) | 0.110 | 0.739 |
| Drinking (n, %) | 37 (39.36) | 39 (41.49) | 0.022 | 0.882 |
Methods
Data collection
The background information (demographic
characteristics) and disease information (duration of hypertension)
of the 94 hypertension patients were collected.
Detection of blood pressure
All the research objects had a rest for 10 min under
appropriate conditions, and then a mercury sphygmomanometer was
used to measure the blood pressure of the brachial artery. Τhe
blood pressure was measured 3 consecutive times, and the mean value
was calculated.
Measurement of intestinal flora
i) Collection of fecal samples: After 2 g or more
fecal sample of each research object was collected and sent to the
laboratory, the sample was separated in an Eppendorf (EP) tube and
labeled under sterile conditions, and then it was stored at −80°C.
ii) Collection of cells in the feces: Fecal sample (0.5 g) was
taken and mixed with 10 ml phosphate-buffered saline (PBS) (Beijing
Dingguo Changsheng Biotechnology Co., Ltd., Beijing, China); then
the sample was centrifuged at 2,750 × g for 5 min at 4°C, the
precipitate was discarded and the supernatant was taken; after the
procedure was repeated another two times, the supernatant was taken
and marked as supernatant I. Supernatant I (1 ml) was added into
the EP tube and centrifuged at 10,500 × g for 10 min at 4°C. After
the supernatant was discarded, another 1 ml supernatant I was added
and centrifuged at 10,500 × g for 10 min at 4°C, and then the
supernatant was discarded. After the procedure was repeated twice,
the precipitate was taken.iii) Extraction of total DNA of
intestinal flora: The total DNAs of the bacteria in the precipitate
were extracted strictly according to the instructions of the
extraction kit of bacterial genome DNA (Beijing Dingguo Changsheng
Biotechnology Co., Ltd.), and ultraviolet spectrophotometric assay
was utilized to detect the concentration and purity of the total
DNA, so as to ensure that the concentration ratio was 1.6–1.9. iv)
Synthesis of primers for reverse transcription-quantitative
polymerase chain reaction (RT-qPCR): All the primers were
synthesized by Shanghai Sangong Pharmaceutical Co., Ltd. (Shanghai,
China). The primers for Eubacterium rectale were: Forward,
CATAAAATCATAGGTTCGCATGAA and reverse, TTTTACCCTACCAACCAGCTAATC. The
primers for Bacteroides thetaiotaomicron multiforme were:
Forward, TGTCGTAACAACGATTTCATAGGT and reverse, TGTTA
GACTCGGCTTCTTTCTTTT. The primers for Bifidobacterium were:
Forward, TCGCCTCCGGGTGAGAGTGG and reverse, CGAAGCCATGGTGGGCCGTT; v)
RT-qPCR: The routine PCR was performed with the genome DNA
extracted from the feces in the control group as a template
(relevant kits were provided by Beijing Zhongshan Golden Bridge
Biotechnology Co., Ltd. (OriGene Technologies, Inc., Rockville, MD,
USA). Reaction conditions: Denaturation at 94°C for 3 min and at
94°C for 30 sec, annealing at 57°C for 30 sec and at 72°C for 60
sec for 35 cycles, and final elongation at 72°C for 5 min. After
the PCR underwent electrophoresis, efficient agarose gel DNA
recovery kit (centrifugal column type) (Beijing Dingguo Changsheng
Biotechnology Co., Ltd.) was used for purification and recovery of
DNA which was taken as a standard substance. LightCycler 480 data
analysis software (Roche Diagnostics, Basel, Switzerland) was
applied to draw the standard curves of Eubacterium rectale,
Bacteroides thetaiotaomicron and Bifidobacterium; the
RT-qPCR was simultaneously performed on blank controls, diluted
standard substances and bacterial DNA samples from the feces to be
detected [relevant kits were provided by Beijing Zhongshan Golden
Bridge Biotechnology Co., Ltd.; OriGene Technologies, Inc.)].
Reaction conditions: 94°C for 5 min, 94°C for 30 sec, 60°C for 30
sec, 72°C for 1 min for 35 cycles, and 72°C for 5 min. The
amplification curves were drawn automatically using the instrument
for RT-qPCR (PerkinElmer, Inc., Waltham, MA, USA). At the end of
the reaction, the copy number of the bacterial genes to be measured
was automatically analyzed utilizing the built-in software of the
instrument for PCR, which was used as the quantitative result.
Evaluation indexes
Parameters and indexes of blood
pressure
All the patients had a rest for 10 min, and then a
mercury sphygmomanometer was used to measure the blood pressure of
the upper limbs. The SBP and DBP were measured 3 times,
respectively, and the mean values were calculated.
More than 2 g fecal sample of every research object
was collected on the same day, which was separated in an EP tube
and labeled, and then it was stored at −80°C. RT-qPCR was applied
to detect the quantities of Eubacterium rectale, Bacteroides
thetaiotaomicron and Bifidobacterium in the
intestines.
Statistical analysis
Statistical Product and Service Solutions (SPSS)
19.0 software (IBM Corp., Armonk, NY, USA) was used for data
processing. The measurement data were presented as mean ± standard
deviation, and t-test was performed. The enumeration data were
presented as ratio and χ2 was used. The Pearson's
correlation coefficient was utilized to analyze the correlation.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Comparisons of the blood pressure and
intestinal flora between the two groups
The comparisons of the blood pressures and
intestinal floras between the two groups showed that the content of
Eubacterium rectale in the observation group was
significantly higher than that in the control group, the
differences were significantly significant(p<0.05). And the
levels of SBP and DBP in the observation group were significantly
higher than those in the control group, the differences were
significantly significant(p<0.05). While the contents of
Bacteroides thetaiotaomicron and Bifidobacterium in
the observation group was obviously lower than that in the control
group, the differences were significantly significant (p<0.05)
(Table II).
 | Table II.Comparison of blood pressure and
intestinal flora between the two groups. |
Table II.
Comparison of blood pressure and
intestinal flora between the two groups.
| Groups | Case | Eubacterium
rectale (×103) | Bacteroides
thetaiotaomicron (×103) |
Bifidobacterium
(×106) | SBP (mmHg) | DBP (mmHg) |
|---|
| Observation | 94 | 3.95±1.61 | 2.92±1.67 | 2.56±1.38 | 145.89±3.72 | 94.65±3.63 |
| Control | 94 | 2.49±1.56 | 4.13±1.86 | 9.87±2.07 | 129.74±3.26 | 85.32±3.14 |
| t value |
| 6.314 | 4.693 | 28.488 | 31.656 | 18.847 |
| P-value |
| <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
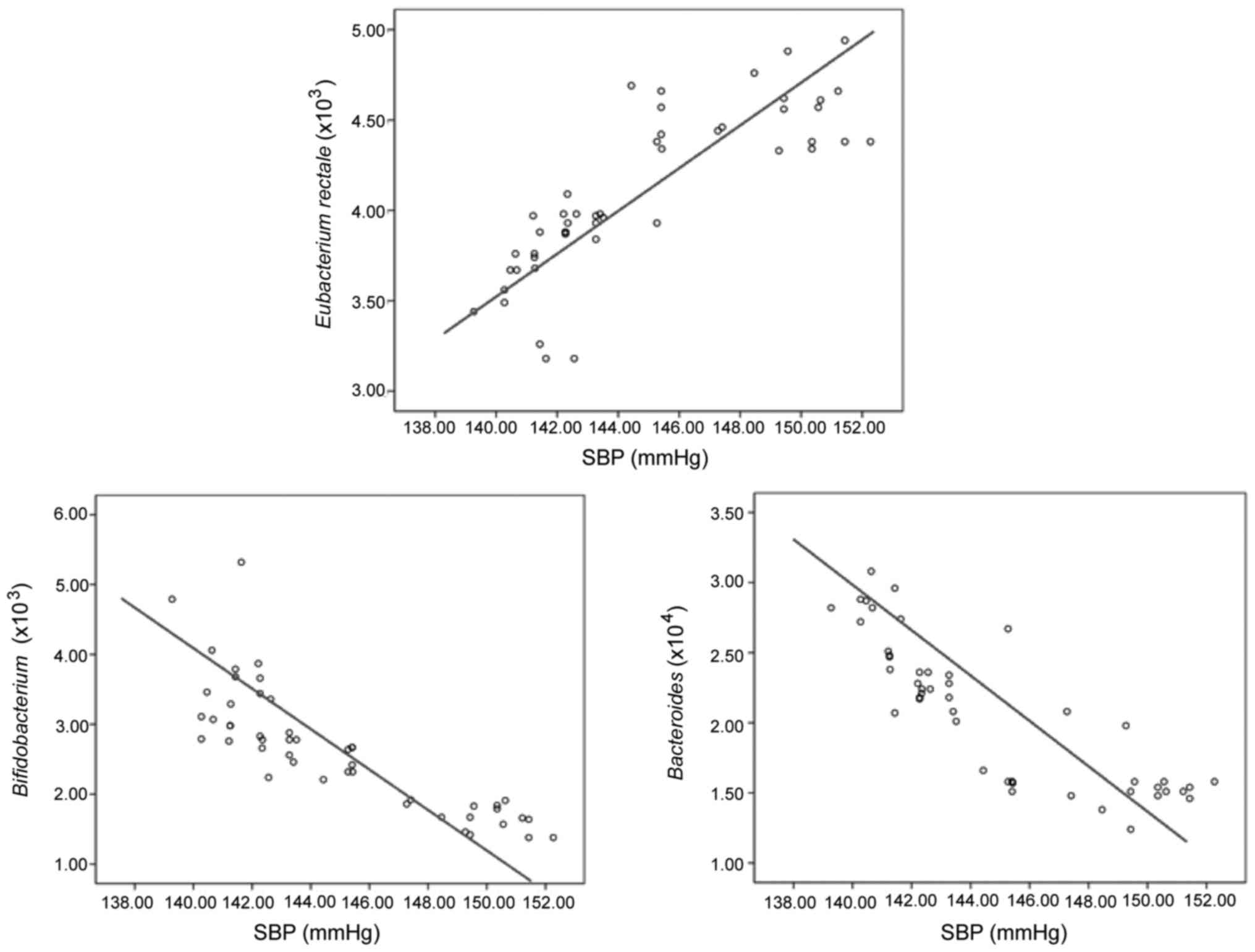
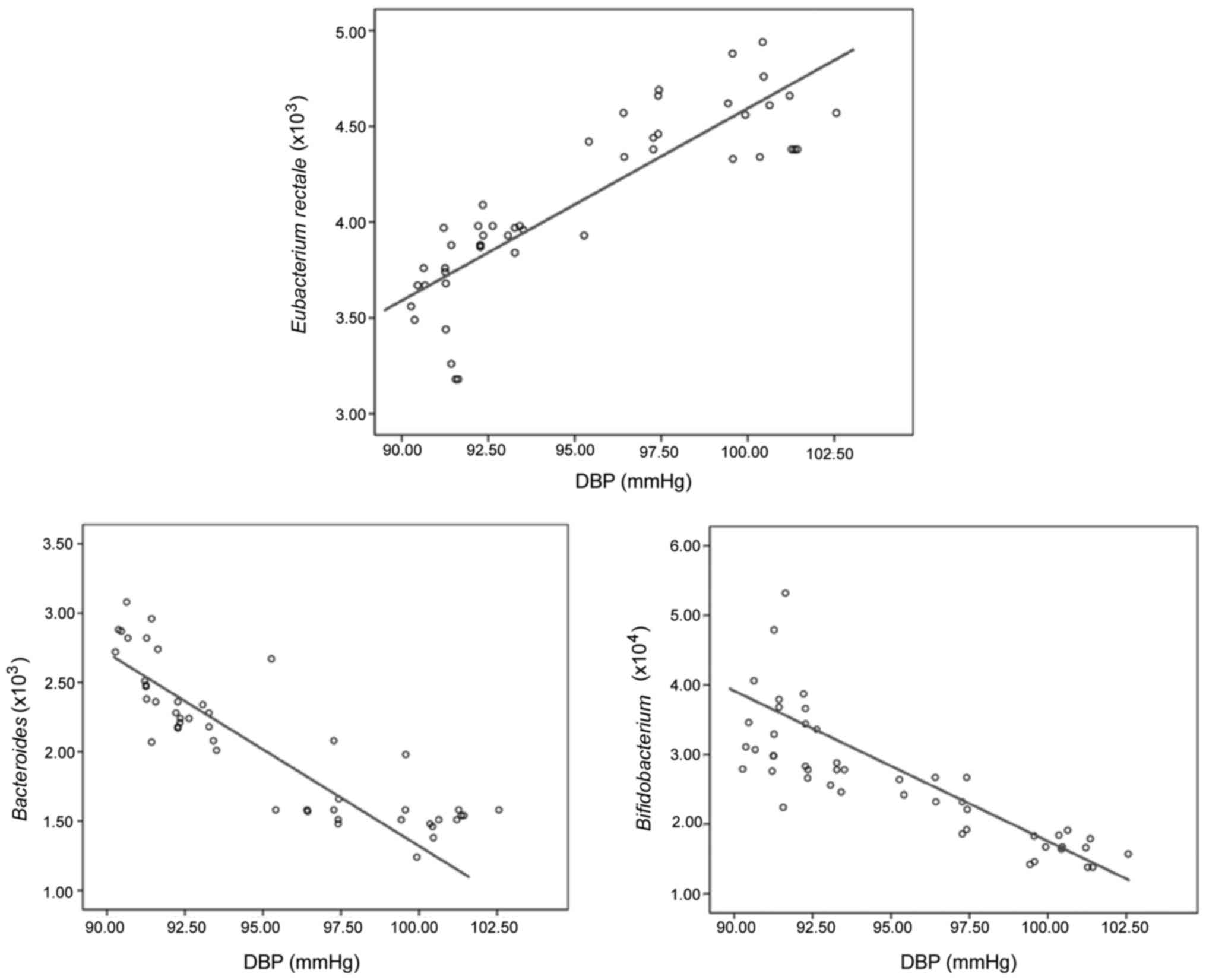
Analyses on correlation of intestinal
flora with SBP and DBP
The analyses via Pearson's correlation coefficients
demonstrated that the content of Eubacterium rectale was
positively correlated with the levels of SBP and DBP, the
differences were significantly significant(p<0.05), and the
correlation of the content of Eubacterium rectale with the
level of DBP was better than that with the level of SBP. While the
content of Bacteroides thetaiotaomicron and
Bifidobacterium had a negative correlation with the levels
of SBP and DBP, the differences were significantly significant
(p<0.05), and the correlation of the content of Bacteroides
multiforme and Bifidobacterium with the level of DBP was
better than that with the level of SBP. (Figs 1 and 2;
Table III).
 | Table III.Analyses on correlation of SBP and
DBP with indexes of intestinal floras. |
Table III.
Analyses on correlation of SBP and
DBP with indexes of intestinal floras.
|
| Correlation with
SBP | Correlation with
DBP |
|---|
|
|
|
|
|---|
| Items | r | P-value | r | P-value |
|---|
| Eubacterium
rectale | 0.413 | 0.014 | 0.409 | 0.009 |
| Bacteroides
multiforme | −0.407 | 0.008 | −0.405 | 0.006 |
|
Bifidobacterium | −0.426 | 0.013 | −0.416 | 0.012 |
Discussion
Hypertension is a cardiovascular syndrome in a
continuous progression status, which can be divided into essential
and secondary hypertension (7). The
incidence rate of hypertension reaches as high as 20% or above in
some European and American countries. The disease may be triggered
by multiple causes, and can lead to functional and structural
changes of the heart and blood vessels (8). The pathogenic factors of hypertension
are influenced by the environmental and social factors jointly,
including toxin effect, infection, diet, medicine, psychological
pressure, socioeconomic status and urbanization (9). It is estimated that the intestinal
floras in the human body are composed of at least 1,014 bacteria
and archaebacteria, which play an important role in human health.
The coevolution and symbiosis of the human body and the intestinal
floras have formed many physiological functions of the human body,
and the intestinal floras are closely related to hypertension. For
instance, the blood pressure is generally elevated as the quantity
of Eubacterium rectale in the intestines is increased
(10). The possible mechanism of
intestinal floras affecting hypertension is that the intestinal
floras can produce short-chain fatty acids (SCFAs) which can
regulate the blood pressure through multiple approaches, and
control the rising and falling of the blood pressure via orphan G
protein-coupled receptor 41 (Gpr41) and olfactory receptor 78
(Olfr78). Among them, Olfr78 can affect the propionate and acetate
generated by the intestinal microbiota, thus inducing the release
of renin and increasing the blood pressure. Gpr41 can influence the
propionate, thus decreasing the blood pressure (11).
Bacteroides thetaiotaomicron is a major
representative of Bacteroides and a Gram-negative bacterium.
It is also one of the floras and microorganisms that have the
largest quantity in the intestines (12). Bacteroides thetaiotaomicron
can synthesize vitamins and proteins, so as to help the absorption
of food and maintain the balance of the intestinal microecology,
playing a dominant role in the intestines (13). Eubacterium rectale is a main
representative of Firmicutes and a kind of conditioned pathogen,
which can ferment the metabolic products of glucose (such as formic
acid, acetic acid and butyric acid) as well as proteins, thereby
inhibiting the proliferation of other beneficial bacteria in the
intestines and decreasing catabolic enzymes of glycan (14). Bifidobacterium is a class of
Gram-positive bacterium, which has multiple functions in the
intestines. Therefore, it is a probiotic of great significance for
human health (15). The results of
this investigation showed that the content of Eubacterium
rectale in the observation group was significantly higher than
that in the control group, while the contents of Bacteroides
thetaiotaomicron and Bifidobacterium were obviously
lower than those in the control group (P<0.05); it may be
related to imbalanced structure of intestinal floras, decreased
number of probiotics and increased quantity of spoilage bacteria in
patients with hypertension.
Some studies have proven (16) that dysbiosis of intestinal floras may
have a close correlation with hypertension. The results of this
research indicated that Eubacterium rectale was positively
correlated with SBP and DBP, while Bacteroides
thetaiotaomicron and Bifidobacterium had an inverse
correlation with SBP and DBP (P<0.05), these are consistent with
the results of relevant literature which suggest that
Eubacterium rectale may be closely related to hypertension.
In general, patients with hypertension have a habit of eating food
with high fat and protein content. As a result, the harmful
bacteria in the intestines are strengthened, especially the
reproduction of Eubacterium rectale, thus elevating the
blood pressure by promoting vasoconstriction (16). Moreover, a micro-inflammatory state
can reduce the diversity of the intestinal floras and accelerate
the ecological imbalance of the intestinal floras. Under normal
conditions, however, the intestinal floras can work as a defense
against the colonization of exotic pathogens, and in turn, the
disordered intestinal floras can lead to an overactive state of the
body's immune system, thus generating many kinds of inflammatory
factors [tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and
IL-8], aggravating inflammation and forming vicious cycles
(17). Various inflammatory factors
can trigger insulin resistance, thus becoming starting factors for
hypertension (18). Bacteroides
thetaiotaomicron is related to the substance and energy
metabolism in the body. When the number of Bacteroides
multiforme is decreased, metabolic disorders occur, bringing
negative impacts on the metabolic activities, forming metabolic
disturbance and inducing hypertension through the pro-inflammatory
mechanism (19).
Bifidobacterium can suppress the growth of spoilage bacteria
and has the actions of adjusting intestinal functions, exerting
anti-anaphylaxis and improving nutrition. Therefore, it can have a
positive effect on the prevention and treatment of a variety of
intestinal diseases (19).
Bifidobacterium can synthesize multiple vitamins, promote
the absorption of vitamin D and convert cholesterols into steroids,
thus lowering the concentration of cholesterols in the blood and
decreasing the blood pressure. When the quantity of
Bifidobacterium is reduced, the original balance of the
intestinal floras is broken, the release of renin is induced and
the blood pressure is elevated (20).
In conclusion, there are abnormal changes in the
content of intestinal floras in patients with hypertension. The
balance of the whole intestinal floras can be restored through
dietary intervention and intake of specific strains and bacterial
genera, so as to lower the blood pressure, thus providing new
thoughts for onset, prevention and treatment of hypertension. This
research adopts a small sample size. Therefore, it is inevitable
that there is a bias in the data, and larger sample sizes are
needed to further study and prove the results in the future.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JiL, NA, CM and XL conceived and designed the study.
JiL, JZ, WZ, YZ and JuL were responsible for the collection and
analysis of the patient data. NA, CM and JuL interpreted the data
and drafted the manuscript. JiL and NA revised the manuscript
critically for important intellectual content. All authors have
read and approved the final study.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
No. 215 Hospital of Shaanxi Nuclear Industry (Xianyang, China).
Signed informed consents were obtained from the patients or
guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Nkeh-Chungag BN, Sekokotla AM,
Sewani-Rusike C, Namugowa A and Iputo JE: Prevalence of
hypertension and pre-hypertension in 13–17 year old adolescents
living in Mthatha - South Africa: A cross-sectional study. Cent Eur
J Public Health. 23:59–64. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lin BM, Curhan SG, Wang M, Eavey R,
Stankovic KM and Curhan GC: Hypertension, diuretic use, and risk of
hearing loss. Am J Med. 129:416–422. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Matsumura K, Arima H, Tominaga M, Ohtsubo
T, Sasaguri T, Fujii K, Fukuhara M, Uezono K, Morinaga Y, Ohta Y,
et al COMFORT Investigators, : Effect of losartan on serum uric
acid in hypertension treated with a diuretic: The COMFORT study.
Clin Exp Hypertens. 37:192–196. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wu S, Xu W, Wang FR and Yang XW: Study of
the biotransformation of tongmai formula by human intestinal flora
and its intestinal permeability across the caco-2 cell monolayer.
Molecules. 20:18704–18716. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yao P, Cui M, Li Y, Deng Y and Wu H:
Effects of rhubarb on intestinal flora and toll-like receptors of
intestinal mucosa in rats with severe acute pancreatitis. Pancreas.
44:799–804. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bhatnagar D: Gut flora, diet and
intestinal metabolism on cardiovascular risk. Curr Opin Lipidol.
26:148–149. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pannarale G, Moroni C, Acconcia MC,
Pannitteri G, Truscelli G, Valente L, Gentile P, Lopreiato F,
Licitra R, Tancredi M, et al: The natural history of
prehypertension. A 20-year follow-up. Eur Rev Med Pharmacol Sci.
21:1329–1334. 2017.PubMed/NCBI
|
|
8
|
Hao C, Kang C, Xue J, Shi K, Lv H and Li
Z: Effects of blood pressure and sex on heart-vessel coupling in
essential hypertension. Turk J Med Sci. 46:680–685. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ichihara A: Pathogenic mechanism of
primary hypertension. Nihon Rinsho. 73:1809–1814. 2015.(In
Japanese). PubMed/NCBI
|
|
10
|
Dobrijevic D, Abraham AL, Jamet A, Maguin
E and van de Guchte M: Functional comparison of bacteria from the
human gut and closely related Non-Gut bacteria reveals the
importance of conjugation and a paucity of motility and chemotaxis
functions in the gut environment. PLoS One. 11:e01590302016.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Goffredo M, Mass K, Parks EJ, Wagner DA,
McClure EA, Graf J, Savoye M, Pierpont B, Cline G and Santoro N:
Role of gut microbiota and short chain fatty acids in modulating
energy harvest and fat partitioning in youth. J Clin Endocrinol
Metab. 101:4367–4376. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kasai C, Sugimoto K, Moritani I, Tanaka J,
Oya Y, Inoue H, Tameda M, Shiraki K, Ito M, Takei Y, et al:
Comparison of human gut microbiota in control subjects and patients
with colorectal carcinoma in adenoma: Terminal restriction fragment
length polymorphism and next-generation sequencing analyses. Oncol
Rep. 35:325–333. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cuskin F, Lowe EC, Temple MJ, Zhu Y,
Cameron EA, Pudlo NA, Porter NT, Urs K, Thompson AJ, Cartmell A, et
al: Corrigendum: Human gut Bacteroidetes can utilize yeast mannan
through a selfish mechanism. Nature. 520:3882015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Rivière A, Gagnon M, Weckx S, Roy D and De
Vuyst L: Mutual cross-feeding interactions between Bifidobacterium
longum subsp. longum NCC2705 and Eubacterium rectale ATCC 33656
explain the bifidogenic and butyrogenic effects of arabinoxylan
oligosaccharides. Appl Environ Microbiol. 81:7767–7781. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Milani C, Lugli GA, Duranti S, Turroni F,
Mancabelli L, Ferrario C, Mangifesta M, Hevia A, Viappiani A,
Scholz M, et al: Bifidobacteria exhibit social behavior through
carbohydrate resource sharing in the gut. Sci Rep. 5:157822015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yang T, Santisteban MM, Rodriguez V, Li E,
Ahmari N, Carvajal JM, Zadeh M, Gong M, Qi Y, Zubcevic J, et al:
Gut dysbiosis is linked to hypertension. Hypertension.
65:1331–1340. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Peeters AC, Netea MG, Janssen MC, Kullberg
BJ, Van der Meer JW and Thien T: Pro-inflammatory cytokines in
patients with essential hypertension. Eur J Clin Invest. 31:31–36.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lai CL, Xing JP, Liu XH, Qi J, Zhao JQ, Ji
YR, Yang WX, Yan PJ, Luo CY and Ruan LF: Relationships of
inflammatory factors and risk factors with different target organ
damage in essential hypertension patients. Chin Med J (Engl).
130:1296–1302. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jiang F, Meng D, Weng M, Zhu W, Wu W,
Kasper D and Walker WA: The symbiotic bacterial surface factor
polysaccharide A on Bacteroides fragilis inhibits IL-1β-induced
inflammation in human fetal enterocytes via toll receptors 2 and 4.
PLoS One. 12:e01727382017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liao ZL, Zeng BH, Wang W, Li GH, Wu F,
Wang L, Zhong QP, Wei H and Fang X: Impact of the consumption of
tea polyphenols on early atherosclerotic lesion formation and
intestinal bifidobacteria in High-Fat-Fed ApoE-/- mice. Front Nutr.
3:422016. View Article : Google Scholar : PubMed/NCBI
|