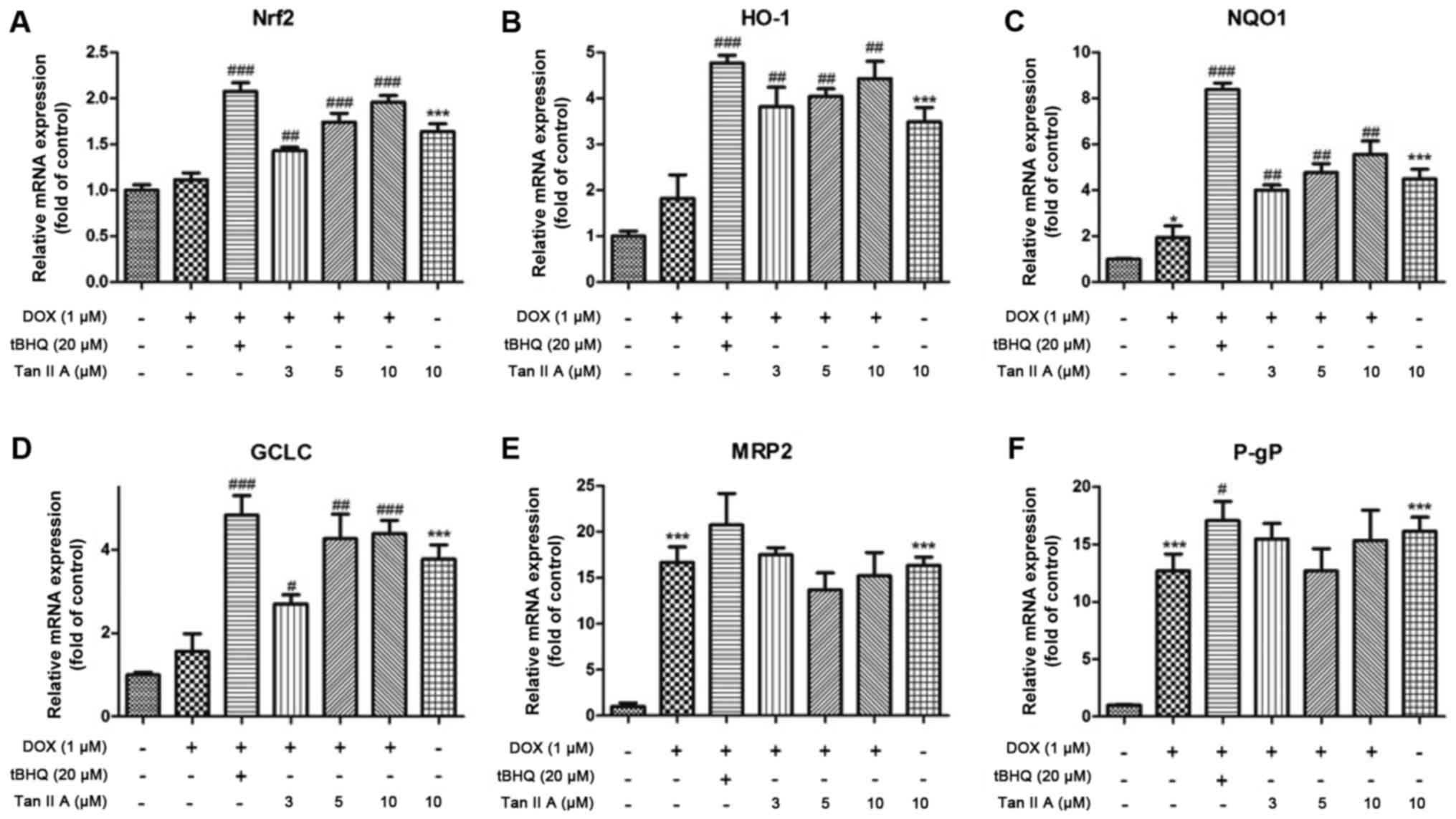
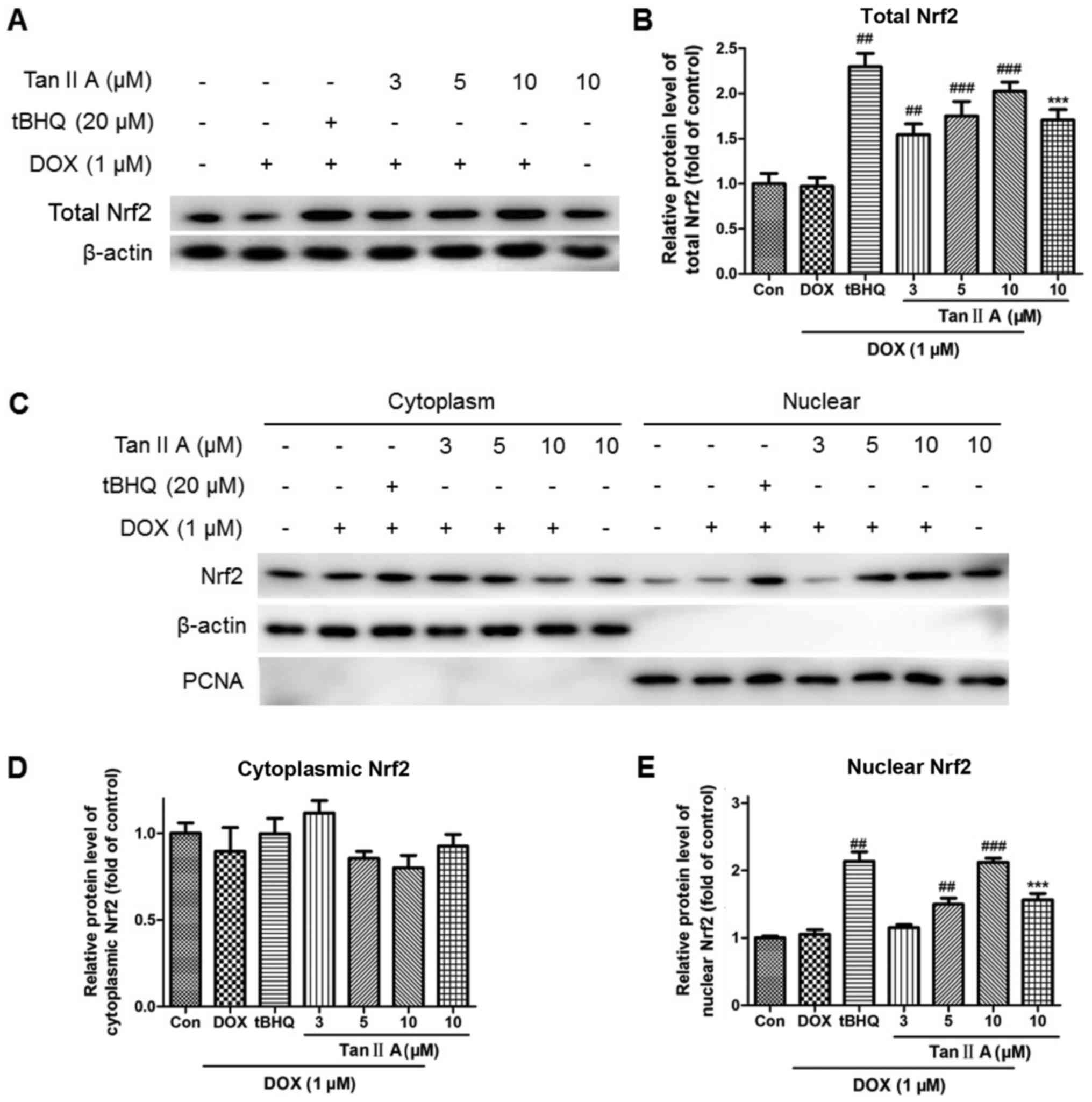
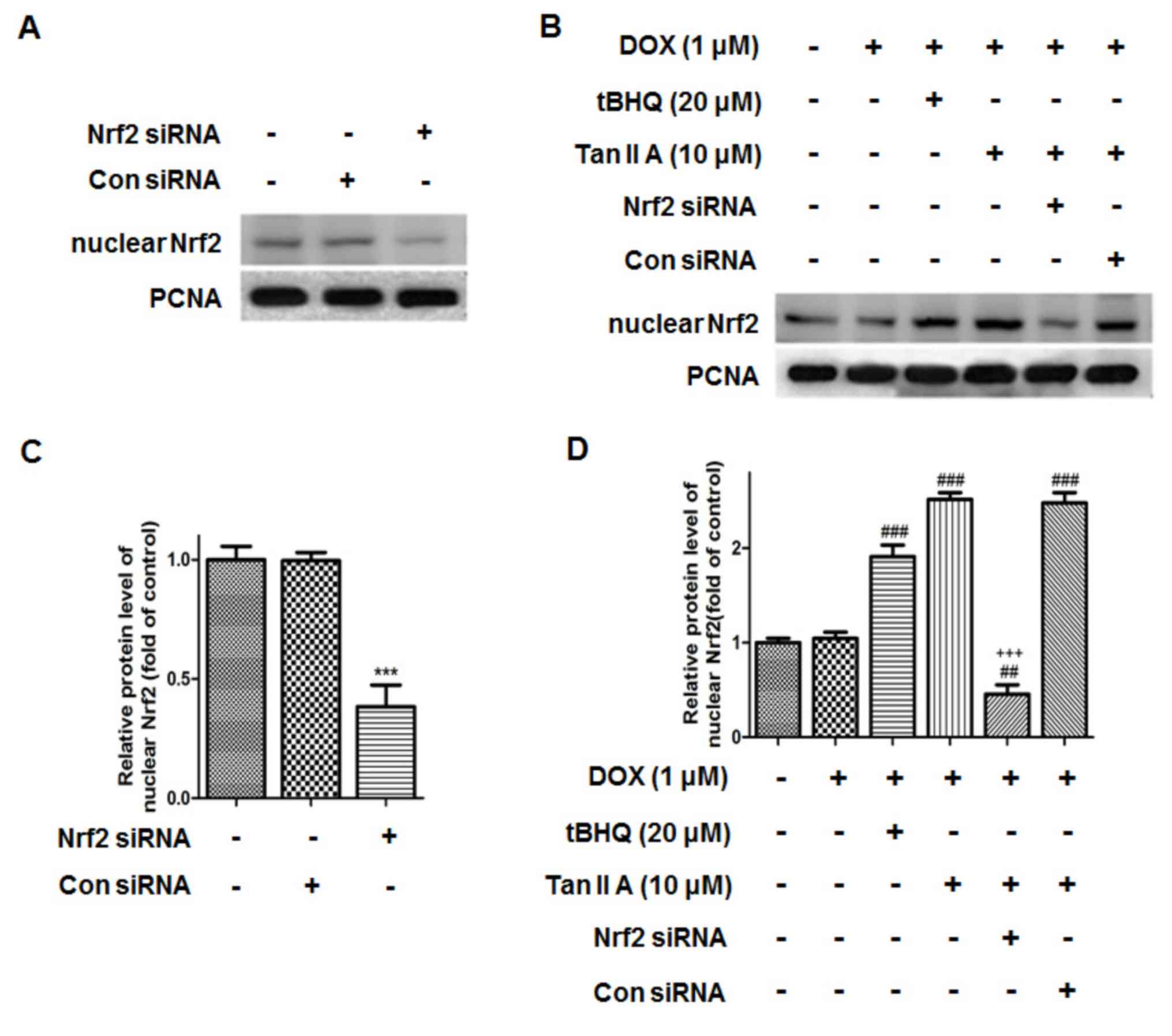
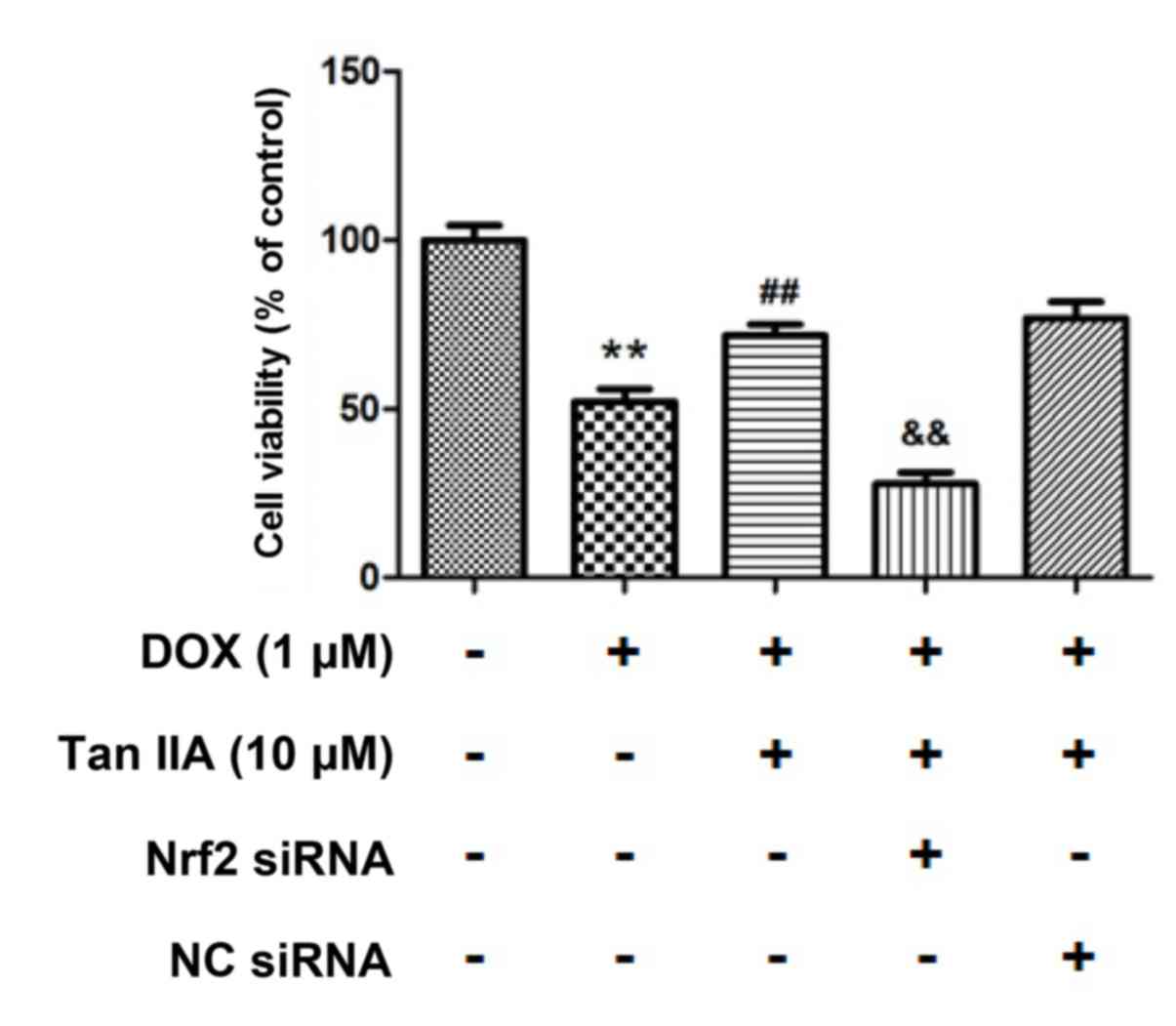
|
1
|
Gorini S, De Angelis A, Berrino L, Malara
N, Rosano G and Ferraro E: Chemotherapeutic drugs and mitochondrial
dysfunction: Focus on doxorubicin, trastuzumab, and sunitinib. Oxid
Med Cell Longev. 2018:75827302018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Vejpongsa P and Yeh ET: Prevention of
anthracycline-induced cardiotoxicity: Challenges and opportunities.
J Am Coll Cardiol. 64:938–945. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
van Dalen EC, van der Pal HJ, Caron HN and
Kremer LC: Different dosage schedules for reducing cardiotoxicity
in cancer patients receiving anthracycline chemotherapy. Cochrane
Database Syst Rev: CD005008. 2006. View Article : Google Scholar
|
|
4
|
Pugazhendhi A, Edison TNJI, Velmurugan BK,
Jacob JA and Karuppusamy I: Toxicity of doxorubicin (Dox) to
different experimental organ systems. Life Sci. 200:26–30. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Carvalho FS, Burgeiro A, Garcia R, Moreno
AJ, Carvalho RA and Oliveira PJ: Doxorubicin-induced
cardiotoxicity: From bioenergetic failure and cell death to
cardiomyopathy. Med Res Rev. 34:106–135. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen CT, Wang ZH, Hsu CC, Lin HH and Chen
JH: In vivo protective effects of diosgenin against
doxorubicin-induced cardiotoxicity. Nutrients. 7:4938–4954. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li B, Kim DS, Yadav RK, Kim HR and Chae
HJ: Sulforaphane prevents doxorubicin-induced oxidative stress and
cell death in rat H9c2 cells. Int J Mol Med. 36:53–64. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Howden R: Nrf2 and cardiovascular defense.
Oxid Med Cell Longev. 2013:1043082013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ma Q and He X: Molecular basis of
electrophilic and oxidative defense: Promises and perils of Nrf2.
Pharmacol Rev. 64:1055–1081. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Han X, Pan J, Ren D, Cheng Y, Fan P and
Lou H: Naringenin-7-O-glucoside protects against
doxorubicin-induced toxicity in H9c2 cardiomyocytes by induction of
endogenous antioxidant enzymes. Food Chem Toxicol. 46:3140–3146.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yu X, Cui L, Zhang Z, Zhao Q and Li S:
α-Linolenic acid attenuates doxorubicin-induced cardiotoxicity in
rats through suppression of oxidative stress and apoptosis. Acta
Biochim Biophys Sin (Shanghai). 45:817–826. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu S and Liu P: Tanshinone II-A: New
perspectives for old remedies. Expert Opin Ther Pat. 23:149–153.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gao S, Liu Z, Li H, Little PJ, Liu P and
Xu S: Cardiovascular actions and therapeutic potential of
tanshinone IIA. Atherosclerosis. 220:3–10. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jiang B, Zhang L, Wang Y, Li M, Wu W, Guan
S, Liu X, Yang M, Wang J and Guo DA: Tanshinone IIA sodium
sulfonate protects against cardiotoxicity induced by doxorubicin in
vitro and in vivo. Food Chem Toxicol. 47:1538–1544. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang W, Guan C, Sun X, Zhao Z, Li J, Fu X,
Qiu Y, Huang M, Jin J and Huang Z: Tanshinone IIA protects against
acetaminophen-induced hepatotoxicity via activating the Nrf2
pathway. Phytomedicine. 23:589–596. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang L, Zhang C, Guo Y, Su ZY, Yang Y, Shu
L and Kong AN: Blocking of JB6 cell transformation by tanshinone
IIA: Epigenetic reactivation of Nrf2 antioxidative stress pathway.
AAPS J. 16:1214–1225. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Alam MF, Khan G, Safhi MM, Alshahrani S,
Siddiqui R, Sivagurunathan Moni S and Anwer T: Thymoquinone
ameliorates doxorubicin-induced cardiotoxicity in swiss albino mice
by modulating oxidative damage and cellular inflammation. Cardiol
Res Pract. 2018:14830412018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sies H, Berndt C and Jones DP: Oxidative
stress. Annu Rev Biochem. 86:715–748. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bellezza I, Giambanco I, Minelli A and
Donato R: Nrf2-Keap1 signaling in oxidative and reductive stress.
Biochim Biophys Acta. 1865:721–733. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Silva-Islas CA and Maldonado PD: Canonical
and non-canonical mechanisms of Nrf2 activation. Pharmacol Res.
134:92–99. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jiang S, Yang Y, Li T, Ma Z, Hu W, Deng C,
Fan C, Lv J, Sun Y and Yi W: An overview of the mechanisms and
novel roles of Nrf2 in cardiovascular diseases. Expert Opin Ther
Targets. 20:1413–1424. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ramprasath T, Vasudevan V, Sasikumar S,
Puhari SS, Saso L and Selvam GS: Regression of oxidative stress by
targeting eNOS and Nrf2/ARE signaling: A guided drug target for
cardiovascular diseases. Curr Top Med Chem. 15:857–871. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tacar O, Sriamornsak P and Dass CR:
Doxorubicin: An update on anticancer molecular action, toxicity and
novel drug delivery systems. J Pharm Pharmacol. 65:157–170. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li S, Wang W, Niu T, Wang H, Li B, Shao L,
Lai Y, Li H, Janicki JS, Wang XL, et al: Nrf2 deficiency
exaggerates doxorubicin-induced cardiotoxicity and cardiac
dysfunction. Oxid Med Cell Longev. 2014:7485242014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang LF, Su SW, Wang L, Zhang GQ, Zhang R,
Niu YJ, Guo YS, Li CY, Jiang WB, Liu Y and Guo HC:
Tert-butylhydroquinone ameliorates doxorubicin-induced
cardiotoxicity by activating Nrf2 and inducing the expression of
its target genes. Am J Transl Res. 7:1724–1735. 2015.PubMed/NCBI
|
|
27
|
Liu Z, Wang J, Huang E, Gao S, Li H, Lu J,
Tian K, Little PJ, Shen X, Xu S and Liu P: Tanshinone IIA
suppresses cholesterol accumulation in human macrophages: Role of
heme oxygenase-1. J Lipid Res. 55:201–213. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sarkar S, Mukherjee S, Chattopadhyay A and
Bhattacharya S: Low dose of arsenic trioxide triggers oxidative
stress in zebrafish brain: Expression of antioxidant genes.
Ecotoxicol Environ Saf. 107:1–8. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cheraghi M, Namdari M, Daraee H and
Negahdari B: Cardioprotective effect of magnetic hydrogel
nanocomposite loaded N,α-L-rhamnopyranosyl vincosamide isolated
from Moringa oleifera leaves against doxorubicin-induced cardiac
toxicity in rats: In vitro and in vivo studies. J Microencapsul.
34:335–341. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhu H, Long MH, Wu J, Wang MM, Li XY, Shen
H, Xu JD, Zhou L, Fang ZJ, Luo Y and Li SL: Ginseng alleviates
cyclophosphamide-induced hepatotoxicity via reversing disordered
homeostasis of glutathione and bile acid. Sci Rep. 5:175362015.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kawase A, Norikane S, Okada A, Adachi M,
Kato Y and Iwaki M: Distinct alterations in ATP-binding cassette
transporter expression in liver, kidney, small intestine, and brain
in adjuvant-induced arthritic rats. J Pharm Sci. 103:2556–2564.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Park J, Park E, Ahn BH, Kim HJ, Park JH,
Koo SY, Kwak HS, Park HS, Kim DW, Song M, et al: NecroX-7 prevents
oxidative stress-induced cardiomyopathy by inhibition of NADPH
oxidase activity in rats. Toxicol Appl Pharmacol. 263:1–6. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hajra S, Patra AR, Basu A and Bhattacharya
S: Prevention of doxorubicin (DOX)-induced genotoxicity and
cardiotoxicity: Effect of plant derived small molecule
indole-3-carbinol (I3C) on oxidative stress and inflammation.
Biomed Pharmacother. 101:228–243. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Al-Harthi SE, Alarabi OM, Ramadan WS,
Alaama MN, Al-Kreathy HM, Damanhouri ZA, Khan LM and Osman AM:
Amelioration of doxorubicininduced cardiotoxicity by resveratrol.
Mol Med Rep. 10:1455–1460. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Thandavarayan RA, Giridharan VV, Arumugam
S, Suzuki K, Ko KM, Krishnamurthy P, Watanabe K and Konishi T:
Schisandrin B prevents doxorubicin induced cardiac dysfunction by
modulation of DNA damage, oxidative stress and inflammation through
inhibition of MAPK/p53 signaling. PLoS One. 10:e01192142015.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chen RC, Xu XD, Zhi Liu X, Sun GB, Zhu YD,
Dong X, Wang J, Zhang HJ, Zhang Q and Sun XB: Total flavonoids from
clinopodium chinense (Benth.) O. Ktze protect against
doxorubicin-induced cardiotoxicity in vitro and in vivo. Evid Based
Complement Alternat Med. 2015:4725652015.PubMed/NCBI
|
|
37
|
Chen Y, Tu JH, He YJ, Zhang W, Wang G, Tan
ZR, Zhou G, Fan L and Zhou HH: Effect of sodium tanshinone II A
sulfonate on the activity of CYP1A2 in healthy volunteers.
Xenobiotica. 39:508–513. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Munagala R, Aqil F, Jeyabalan J and Gupta
RC: Tanshinone IIA inhibits viral oncogene expression leading to
apoptosis and inhibition of cervical cancer. Cancer Lett.
356:536–546. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Su CC: Tanshinone IIA potentiates the
efficacy of 5-FU in Colo205 colon cancer cells in vivo through
downregulation of P-gp and LC3-II. Exp Ther Med. 3:555–559. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lin C, Wang L, Wang H, Yang L, Guo H and
Wang X: Tanshinone IIA inhibits breast cancer stem cells growth in
vitro and in vivo through attenuation of IL-6/STAT3/NF-kB signaling
pathways. J Cell Biochem. 114:2061–2070. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Branco AF, Sampaio SF, Moreira AC, Holy J,
Wallace KB, Baldeiras I, Oliveira PJ and Sardão VA:
Differentiation-dependent doxorubicin toxicity on H9c2
cardiomyoblasts. Cardiovasc Toxicol. 12:326–340. 2012. View Article : Google Scholar : PubMed/NCBI
|