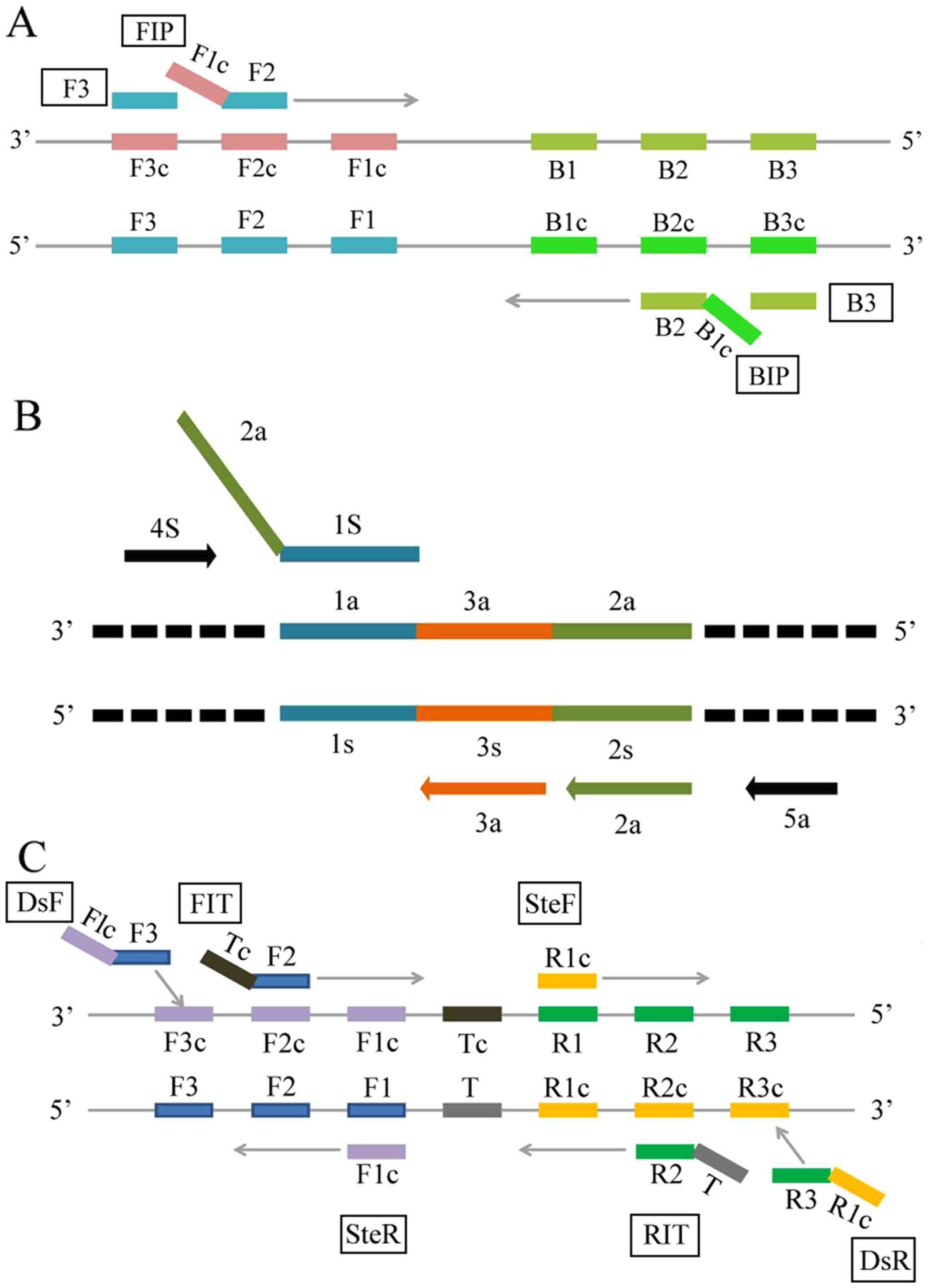
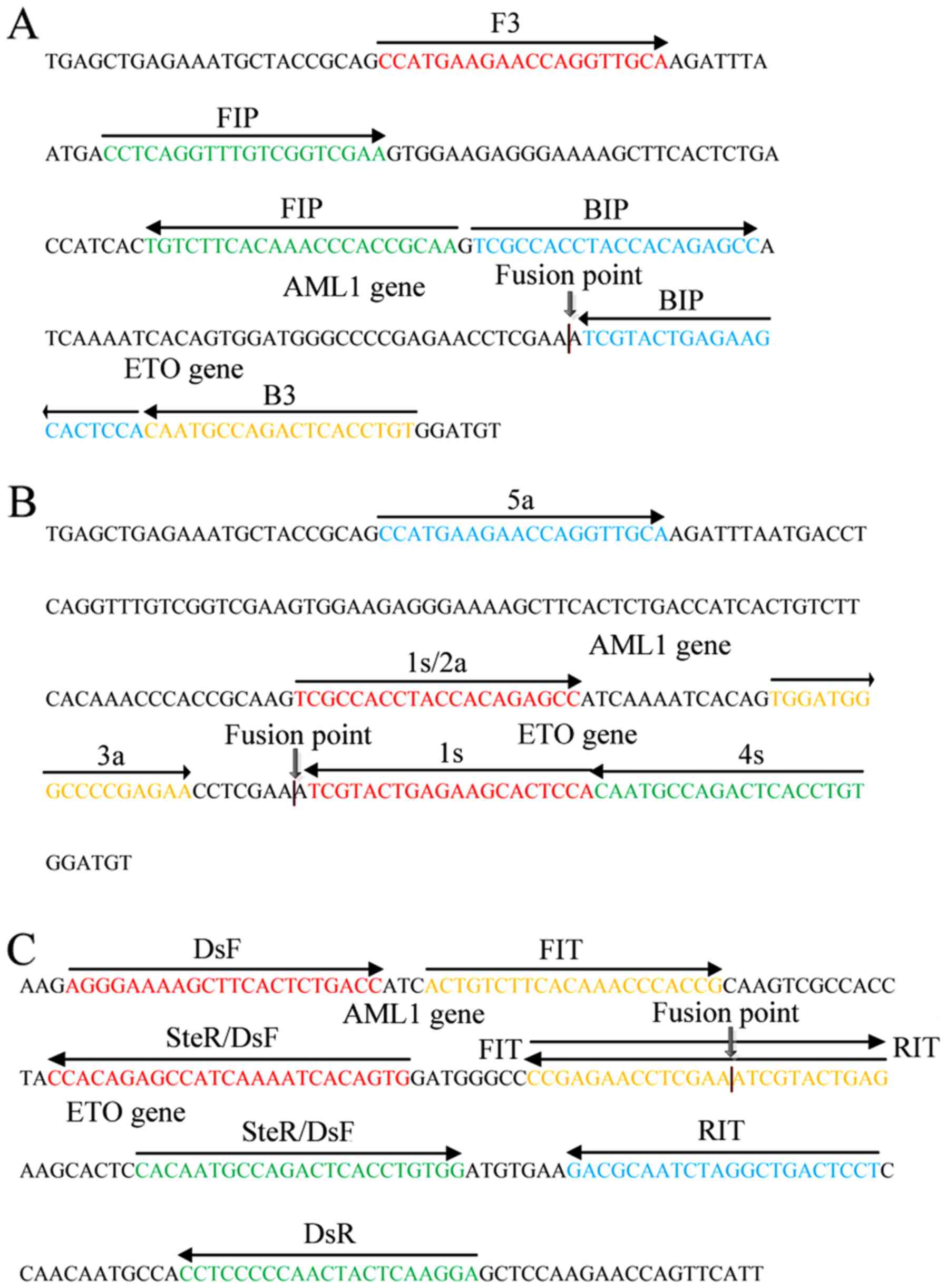
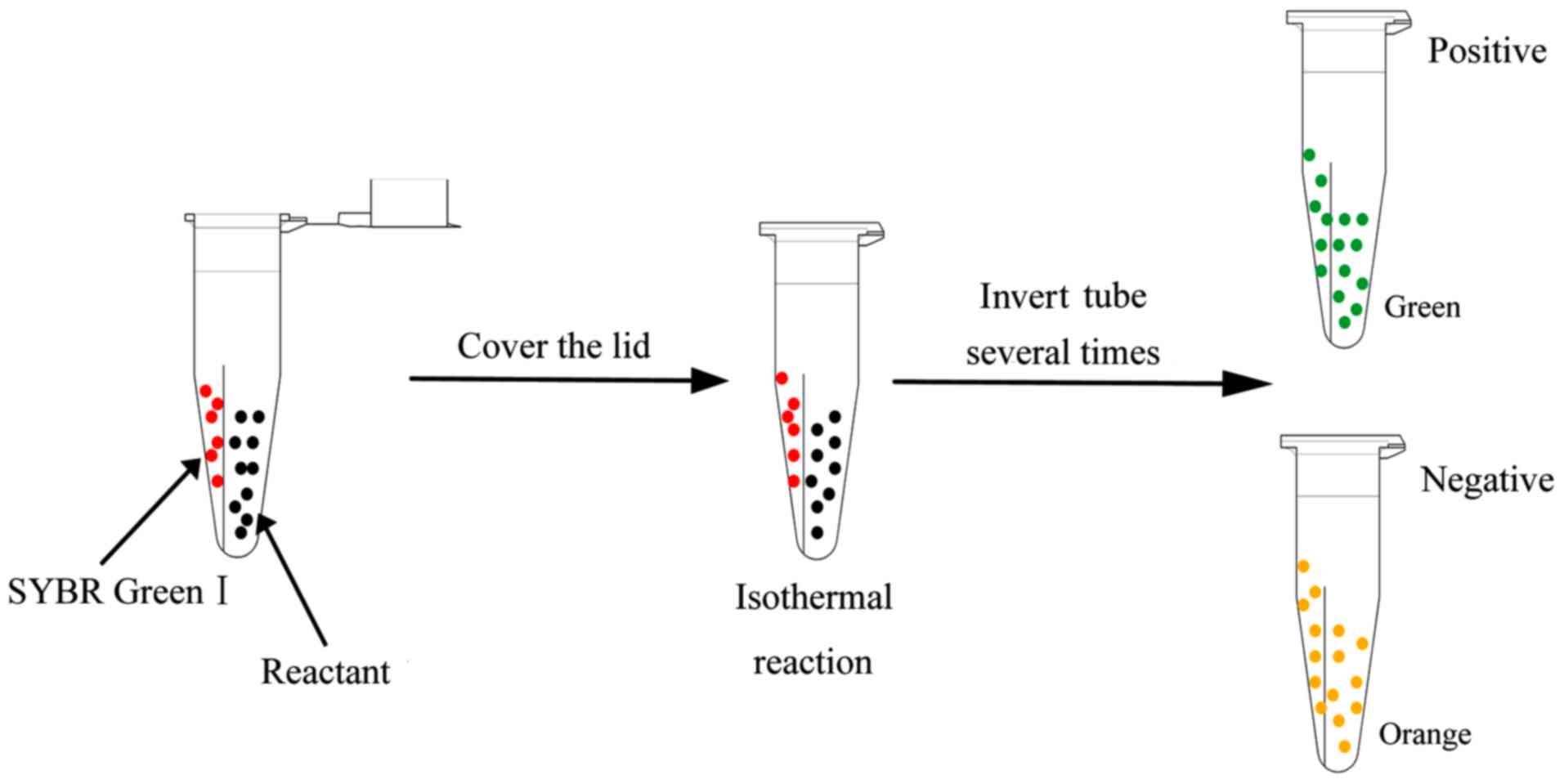
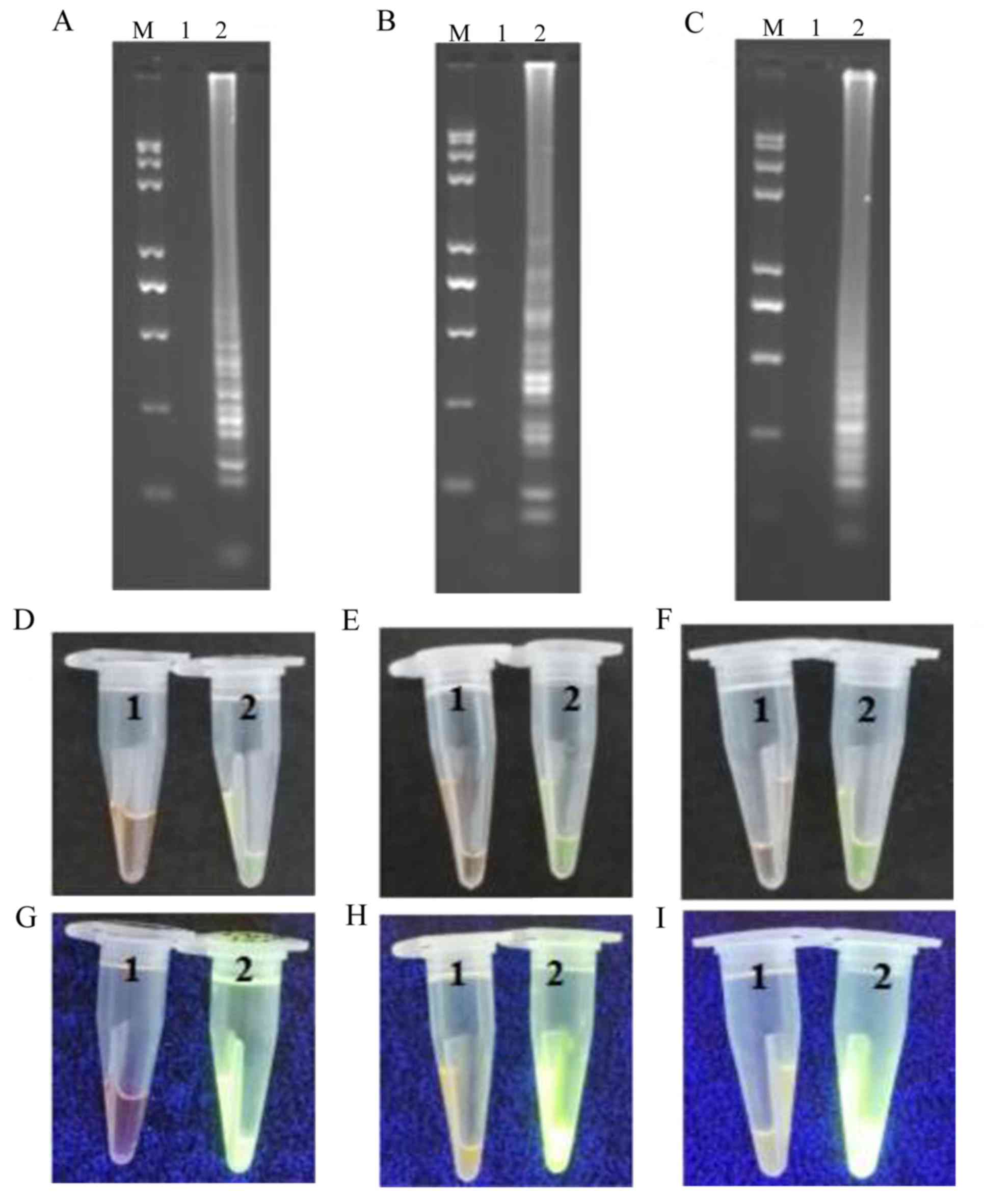
|
1
|
Salmon JM, Bots M, Vidacs E, Stanley KL,
Atadja P, Zuber J and Johnstone RW: Combining the differentiating
effect of panobinostat with the apoptotic effect of arsenic
trioxide leads to significant survival benefit in a model of
t(8;21) acute myeloid leukemia. Clin Epigenetics. 7:22015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Okuda T, Cai Z, Yang S, Lenny N, Lyu CJ,
van Deursen JM, Harada H and Downing JR: Expression of a knocked-in
AML1-ETO leukemia gene inhibits the establishment of normal
definitive hematopoiesis and directly generates dysplastic
hematopoietic progenitors. Blood. 91:3134–3143. 1998.PubMed/NCBI
|
|
3
|
Saia M, Termanini A, Rizzi N, Mazza M,
Barbieri E, Valli D, Ciana P, Gruszka AM and Alcalay M: AML1/ETO
accelerates cell migration and impairs cell-to-cell adhesion and
homing of hematopoietic stem/progenitor cells. Sci Rep.
6:349572016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Peterson LF and Zhang DE: The 8;21
translocation in leukemogenesis. Oncogene. 23:4255–4262. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kumari P, Lingappa Kavitha B, Obula Reddy
C, Mangalagowri M, Madhumathi DS, Mahadeva Prasad M, Raghavendra
HV, Premalata CS, Lakshmaiah KC and Mir Mazloumi SH: A rare
cytogenetic presentation of acute myeloid leukemia (AML-M2). Acta
Med Iran. 50:827–830. 2012.PubMed/NCBI
|
|
6
|
Gabert J, Beillard E, van der Velden VH,
Bi W, Grimwade D, Pallisgaard N, Barbany G, Cazzaniga G, Cayuela
JM, Cavé H, et al: Standardization and quality control studies of
‘real-time’ quantitative reverse transcriptase polymerase chain
reaction of fusion gene transcripts for residual disease detection
in leukemia-a Europe against cancer program. Leukemia.
17:2318–2357. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang ZD, Qin YZ, Liu YR, Xu LP, Liu DH,
Liu KY and Huang XJ: Monitoring AML1-ETO mRNA levels by real-time
quantitative RT-PCR in t(8;21) acute myeloid leukemia patients
after hematopoietic stem cell transplantation. Zhonghua Xue Ye Xue
Za Zhi. 29:672–675. 2008.(In Chinese). PubMed/NCBI
|
|
8
|
Cui C, Shu W and Li P: Fluorescence in
situ hybridization: Cell-based genetic diagnostic and research
applications. Front Cell Dev Biol. 4:892016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Notomi T, Okayama H, Masubuchi H, Yonekawa
T, Watanabe K, Amino N and Hase T: Loop-mediated isothermal
amplification of DNA. Nucleic Acids Res. 28:E632000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xu G, Hu L, Zhong H, Wang H, Yusa S, Weiss
TC, Romaniuk PJ, Pickerill S and You Q: Cross priming
amplification: Mechanism and optimization for isothermal DNA
amplification. Sci Rep. 2:2462012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ding X, Nie K, Shi L, Zhang Y, Guan L,
Zhang D, Qi S and Ma X: Improved detection limit in rapid detection
of human enterovirus 71 and coxsackievirus A16 by a novel reverse
transcription-isothermal multiple-self-matching-initiated
amplification assay. J Clin Microbiol. 52:1862–1870. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ding X, Wu W, Zhu Q, Zhang T, Jin W and Mu
Y: Mixed-dye-based label-free and sensitive dual fluorescence for
the product detection of nucleic acid isothermal
multiple-self-matching-initiated amplification. Anal Chem.
87:10306–10314. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Viehmann S, Teigler-Schlegel A, Bruch J,
Langebrake C, Reinhardt D and Harbott J: Monitoring of minimal
residual disease (MRD) by real-time quantitative reverse
transcription PCR (RQ-RT-PCR) in childhood acute myeloid leukemia
with AML1/ETO rearrangement. Leukemia. 17:1130–1136. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Döhner H, Weisdorf DJ and Bloomfield CD:
Acute myeloid leukemia. N Engl J Med. 373:1136–1152. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Niessen L and Vogel RF: Detection of
Fusarium graminearum DNA using a loop-mediated isothermal
amplification (LAMP) assay. Int J Food Microbiol. 140:183–191.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yao X, Li P, Xu J, Zhang M, Ren R, Liu G
and Yang X: Rapid and sensitive detection of didymella bryoniae by
visual loop-mediated isothermal amplification assay. Front
Microbiol. 7:13722016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li F, Yan W, Long L, Qi X, Li C and Zhang
S: Development and application of loop-mediated isothermal
amplification assays for rapid visual detection of cry2Ab and cry3A
genes in genetically-modified crops. Int J Mol Sci. 15:15109–15121.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lu X, Li X, Mo Z, Jin F, Wang B, Zhao H,
Shan X and Shi L: Rapid identification of Chikungunya and Dengue
virus by a real-time reverse transcription-loop-mediated isothermal
amplification method. Am J Trop Med Hyg. 87:947–953. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ke Y, Wang Y, Wang Z, Du X, Huang L and
Chen Z: Sensitive and rapid detection of blaNDM-1 in clinical
samples by isothermal cross-priming amplification. J Microbiol
Methods. 95:215–217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wozniakowski G, Niczyporuk JS,
Samorek-Salamonowicz E and Gawel A: The development and evaluation
of cross-priming amplification for the detection of avian reovirus.
J Appl Microbiol. 118:528–536. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chandra A, Keizerweerd AT and Grisham MP:
Detection of puccinia kuehnii causing sugarcane orange rust with a
loop-mediated isothermal amplification-based assay. Mol Biotechnol.
58:188–196. 2016. View Article : Google Scholar : PubMed/NCBI
|