Introduction
The incidence of diabetes mellitus (DM), a common
chronic disease in clinical practice that affected over 100 million
people in China, is increasing each year (1). It has been reported that DM is a
persistent metabolic disorder, which can be induced by hypoglycemia
and impaired glucose tolerance due to insulin deficiency and
resistance (2,3). Type 1 DM is caused by the reduction of
insulin due to the destruction of islet cells and accounts for a
minority of patients with DM (4,5). Type 2
DM (T2DM) is the more common type of DM, accounting for > 90% of
all cases. Cardiovascular complications are a major cause of
mortality in patients with DM (6).
Vascular lesions induced by DM often cause pathological changes to
the tissues and organs (6). Blood
vessels transport blood, secreting bioactive substances, regulating
blood pressure and maintaining tissue and organ perfusion (7). It has been demonstrated that
microvascular lesions cause diabetic ophthalmopathy and nephropathy
and that DM-induced vascular injuries are a key factor in the
development of microvascular lesions (8). Vascular endothelial cells serve
important roles in maintaining the integrity of vascular structure
and function (9,10). Patients with DM often exhibit damages
to vascular endothelial cells (11).
However, the underlying molecular mechanism remains unclear.
MicroRNA (miRNA or miR) is a class of non-coding
small RNA molecules (18–22 nucleotides) that bind to the
3′-untranslated region of target gene mRNA to regulate their
expression (12,13). As miRNA molecules serve diverse
biological roles with multiple targets, they have become novel
candidates for the diagnosis and treatment of diseases (14). MiRNAs are involved in a majority of
pathophysiological processes and they are associated to vascular
endothelial cell damages in two aspects (15,16). The
first aspect describes a decreased ability to repair endothelial
cell damage and is characterized by inhibited proliferation and
migration of cells (15). The second
aspect describes endothelial dysfunction, characterized by abnormal
secretion of vascular endothelial cell factors (16). Studies have demonstrated that a
variety of miRNA molecules are associated with endothelial cell
damage (17–22). MiR-200b serves a role in the
regulation of vascular endothelial injury induced by acute ischemia
by targeting Kruppel like factor 2 gene (17). Furthermore, miR-142 regulates
angiokinesis by upregulating endothelial nitric oxide synthase
(eNOS) expression in vascular endothelial progenitor cells
(18). MiR-199a-3p is a highly
conserved miRNA molecule that serves important functions in the
occurrence and development of various tumors (19,20). A
previous study demonstrated that miR-199a-3p is associated with the
injury and protection of cardiac myocytes and is downregulated in
the peripheral blood of patients with myocardial ischemia
reperfusion injury (21). The
specific expression of miR-199a-3p in pancreatic tissues may also
be associated with T2DM (22).
However, it remains unclear whether miR-199a-3p is associated with
DM-induced endothelial cell injury. In the present study, reverse
transcription-quantitative polymerase chain reaction (RT-qPCR),
western blotting, cell counting kit (CCK)-8 assays and flow
cytometry were performed to examine the function and mechanism of
action of miR-199a-3p in endothelial cell injuries induced by T2DM
at the clinical and cellular levels.
Patients and methods
Patients
A total of 36 patients with T2DM (26 males and 10
females; mean age, 52.5±7.0 years) who received treatments at the
Affiliated Hospital of Taishan Medical University (Taian, China)
between January 2016 and January 2017 were included in the
experimental group. In addition, 20 healthy subjects (10 males and
10 females; mean age, 55.6±4.5 years) who undertook physical
examinations in the same time period were included in the control
group. Fasting peripheral blood (5 ml) was obtained from all
participants in the morning. According to the diagnostic standards
for DM published by World Health Organization in 1999 (23) and the results of oral glucose
tolerance tests, patients with T2DM were divided into subgroup 1
(17 cases; T2DM with no complications), subgroup 2 (13 cases; T2DM
combined with macroangiopathy) and subgroup 3 (6 cases; T2DM
combined with macrovascular and microvascular lesions). All
patients were diagnosed for the first time and had no history of
long-term medication, tumors or chronic diseases. All procedures
were approved by the Ethics Committee of Taishan Medical
University. Written informed consent was obtained from all patients
or their families.
RT-qPCR analysis
Serum (250 µl) was separated from peripheral blood
by centrifugation at 2,000 × g for 10 min at 4°C and mixed with 750
µl TRIzol reagent (Thermo Fisher Scientific, Inc., Waltham, MA,
USA) for lysis following the manufacturer's protocol. Following
lysis, total RNA was extracted using the phenol chloroform method.
The purity of RNA was determined by A260/A280 using ultraviolet
spectrophotometry (Nanodrop ND2000; Thermo Fisher Scientific, Inc.,
Pittsburgh, PA, USA). cDNA was obtained by RT at 37°C for 1 h using
miScript II RT kit (Qiagen GmbH, Hilden, Germany) from 0.5 µg RNA
according to the manufacturer's protocol and samples were stored at
−20°C.
qPCR was performed using miScript
SYBR®-Green PCR kit (Qiagen GmbH). The reaction mixture
comprised 10 µl RT-qPCR-mix, 0.5 µl upstream primer
(5′-ACAGTAGUCTGCACATTGGTTA-3′), 0.5 µl downstream primer (universal
primer provided by the kit), 2 µl cDNA and 7 µl ddH2O.
Thermocycling conditions were as follows: Initial denaturation at
95°C for 10 min followed by 40 cycles of 95°C for 1 min and 60°C
for 30 sec. The 2−ΔΔCq method (24) was used to calculate the relative
expression of miR-199a-3p against U6 (forward primer,
5′-GCGCGTCGTGAAGCGTTC-3′ and reverse primer,
5′-GTGCAGGGTCCGAGGT-3′). Each sample was tested in triplicate.
Cells
Human umbilical vein endothelial cells (HUVECs; Type
Culture Collection of the Chinese Academy of Sciences, Shanghai,
China) were seeded at a density of 1×105 cells/well in
24-well plates containing RPMI-1640 medium supplemented with 10%
fetal bovine serum (FBS; Thermo Fisher Scientific, Inc.) and
cultured at 37°C in an atmosphere containing 5% CO2.
Cells were divided into negative control (NC) and miR-199a-3p
mimics groups. When cells reached 70% confluence, 1.25 µl miR-NC
(universal sequence; Sangon Biotech Co., Ltd., Shanghai, China) or
miR-199a-3p mimics (5′-ACAGTAGTCTGCACATTGGTTA-3′; Sangon Biotech
Co., Ltd.) and 2 µl Lipofectamine® 3000 were added to
individual vials containing 50 µl Opti Mem medium (both Thermo
Fisher Scientific, Inc.) at room temperature for 5 min. Vials were
mixed and incubated at room temperature for 15 min. Mixtures were
added to the cells and incubated at 37°C for 6 h, following which
the medium was replaced with RPMI-1640 medium supplemented with 10%
FBS. Cells were cultured at 37°C in an atmosphere containing 5%
CO2 for 48 h prior to experiments.
CCK-8 assay
At 48 h following transfection, HUVECs were
trypsinized, collected by centrifugation at 500 × g for 5 min at
room temperature and inoculated into 96-well plates containing 200
µl RPMI-1640 medium at a density of 2,000 cells/well. At 0, 24, 48
and 72 h, 20 µl CCK-8 (5 g/l; Beyotime Institute of Biotechnology,
Haimen, China) was added to the cells. Following incubation at 37°C
for 2 h, the absorbance (490 nm) of each well was determined and
cell proliferation curves were plotted. Each group was tested in
triplicate and the values were averaged.
Transwell assay
Matrigel chambers (Corning Incorporated, Corning,
NY, USA) were used to determine the migration ability of cells.
Matrigel was diluted with serum-free RPMI-1640 medium at a ratio of
1:2. A total of 50 µl diluted Matrigel was added to the upper
chamber and incubated at 37°C for 1 h, following which
2×105 HUVECs and 200 µl serum-free RPMI-1640 medium were
added. In the lower chamber, 500 µl RPMI-1640 medium supplemented
with 10% FBS was added. Following incubation at 37°C and 5%
CO2 for 24 h, cells in upper chamber were removed using
a cotton swab. The chamber was fixed using 4% formaldehyde for 10
min at room temperature and subjected to 5% Giemsa's staining at
room temperature for 1 min. Following washing for 3 times, migrated
cells were counted using a light microscope (5 fields;
magnification, ×200).
Flow cytometry
To simulate the high glucose environment in DM,
HUVECs were cultured in RPMI-1640 medium supplemented with 40
mmol/l glucose following transfection in an atmosphere containing
5% CO2 at 37°C for 6 h. HUVECs were washed twice with
PBS, trypsinized, collected by centrifugation at 500 × g for 5 min
at room temperature and adjusted to a density of 1×106
cells/100 µl. Apoptosis was assessed using flow cytometry with an
ANXN V FITC Apoptosis DTEC kit I (BD Biosciences, Franklin Lakes,
NJ, USA) according to the manufacturer's protocol. Using a flow
cytometer and FlowJo software (version 7.6.1; BD Biosciences),
cells with Annexin V-positive values were determined to be in early
apoptosis, those with propidium iodide-positive values were
necrotic and those with double positive values were in late
apoptosis.
Laser-scanning confocal
microscopy
At 24 h following transfection, HUVECs were seeded
onto culture plates at a density of 1×105 cells/well.
When cells reached 70% confluence, they were infected following the
manufacturer's instructions with Ad-GFP-LC3B adenovirus (Hanbio
Biotechnology Co., Ltd., Shanghai, China) at a multiplicity of
infection of 20 for 48 h at 37°C. Without fixing, cells were
observed under a laser confocal microscope (SP8; Leica Microsystems
GmbH, Wetzlar, Germany). Green vesicles represent autophagosomes.
Autophagosome numbers in HUVECs were counted in five fields using a
confocal microscope and averaged to evaluate autophagy
activity.
Western blotting
HUVECs were trypsinized and collected by
centrifugation at 500 × g for 5 min at room temperature. Cold
radioimmunoprecipitation assay lysis buffer (500 µl; Beyotime
Institute of Biotechnology) was mixed with the samples for 30 min
on ice, followed by centrifugation at 12,000 × g at 4°C for 10 min.
Extraction of nuclear proteins was performed using Cell nuclear
protein and cytoplasmic protein extraction kit (P0027; Beyotime
Institute of Biotechnology) A bicinchoninic acid protein
concentration determination kit [RTP7102, Real-Times (Beijing)
Biotechnology Co., Ltd., Beijing, China] was used to determine the
protein concentration in the supernatant. Protein samples (10 µg)
were mixed with 5X SDS loading buffer and the mixture was denatured
by boiling in a water bath for 10 min. Proteins were separated by
10% SDS-PAGE and transferred to polyvinylidene difluoride
membranes, which were subsequently blocked with 5% skimmed milk at
room temperature for 1 h. Membranes were incubated with
phosphatidylinositol 3-kinase (PI3K) catalytic subunit p110α
(1:1,000; cat. no. 4249; Cell Signaling Technology, Inc., Danvers,
MA, USA), PI3K regulatory subunit p85 (1:1,000; cat. no. AF1729;
Beyotime Institute of Biotechnology) LC3BII (1:1,000; cat. no.
AL221; Beyotime Institute of Biotechnology) and mouse anti-human
GAPDH (1:5,000; cat. no. AF0006; Beyotime Institute of
Biotechnology) primary antibodies at 4°C overnight. For nuclear
proteins, membranes were incubated with rabbit anti-human
polyclonal nuclear factor (NF)-κB (1:1,000; cat. no. AF0246;
Beyotime Institute of Biotechnology), protein kinase B (AKT;
1:1,000; cat. no. AA326; Beyotime Institute of Biotechnology),
phosphorylated-(p)AKT (1:1,000; cat. no. AA329; Beyotime Institute
of Biotechnology) and histone H (internal reference for nuclear
proteins; 1:5,000; cat. no. AF0009; Beyotime Institute of
Biotechnology) primary antibodies at 4°C overnight. Membranes were
washed with PBS with Tween-20 (PBST) five times for 5 min and
incubated with goat anti-mouse (1:4,000; cat. no. A0216; Beyotime
Institute of Biotechnology) and goat anti-rabbit (1:4,000; cat. no.
A0208; Beyotime Institute of Biotechnology) horseradish
peroxidase-conjugated secondary antibodies at room temperature for
1 h. Subsequently, membranes were washed five times with PBST for 5
min and developed using an enhanced chemiluminescence detection kit
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). Image lab v3.0
software (Bio-Rad Laboratories, Inc., Hercules, CA, USA) was used
to analyze imaging data. The relative expression of target proteins
was normalized to GAPDH.
Statistical analysis
Data were analyzed using SPSS 17.0 statistical
software (SPSS, Inc., Chicago, IL, USA). Data are expressed as the
mean ± standard deviation. Data were tested for normality and
multigroup measurement data were analyzed using one-way analysis of
variance. Least Significant Difference and Student-Newman-Keuls
post hoc tests were used for homogeneous data, while Tamhane's T2
or Dunnett's T3 tests were performed for heterogeneous data.
Comparisons between two groups were made using Student's t-tests.
P<0.05 was considered to indicate a statistically significant
difference.
Results
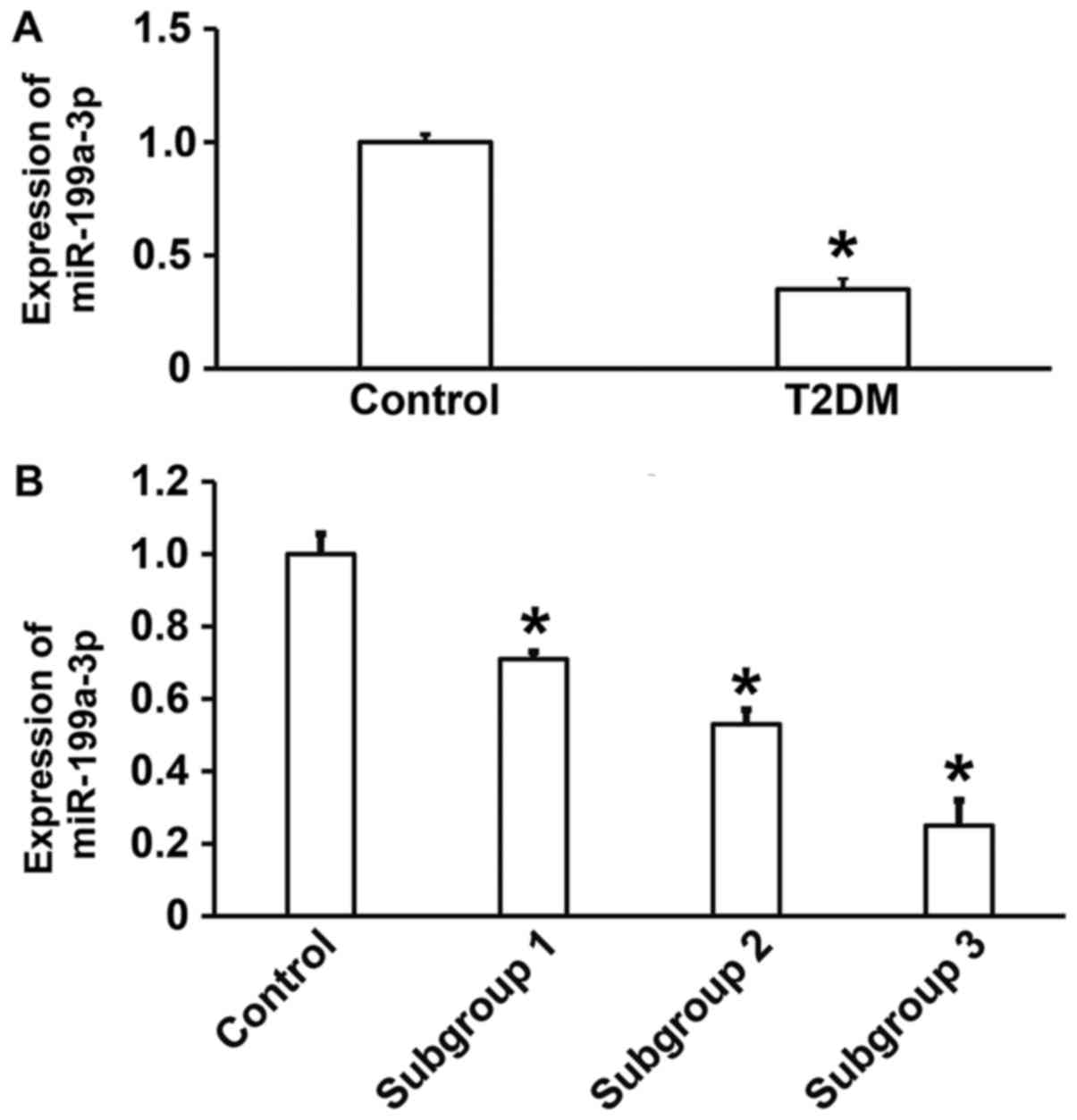
Reduced miR-199a-3p expression in T2DM
may be associated with vascular lesions
To measure the expression of miR-199a-3p in the
peripheral blood of patients with T2DM, RT-qPCR was performed. The
data illustrated that miR-199a-3p expression was significantly
lower in patients with T2DM compared with healthy subjects
(P<0.05; Fig. 1A). In addition,
significant differences in miR-199a-3p expression were observed
between healthy subjects and subgroups 1, 2 and 3 (P<0.05), with
lower levels in patients with T2DM combined with macroangiopathy
and patients with T2DM combined with macrovascular and
microvascular lesions compared with patients with T2DM without
complications (Fig. 1B). These
results suggest that reduced miR-199a-3p expression in T2DM may be
associated with vascular lesions.
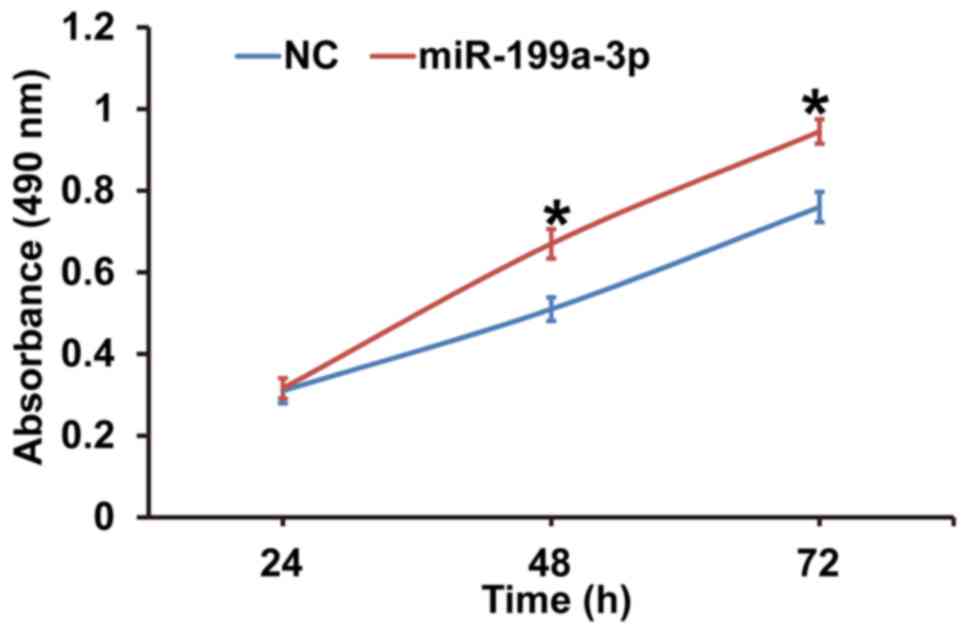
MiR-199a-3p overexpression promotes
the proliferation of HUVECs
To assess the effect of miR-199a-3p overexpression
on HUVEC proliferation, cells were transfected with miR-NC or
miR-199a-3p mimics and a CCK-8 assay was performed. The data
revealed that the absorbance of HUVECs transfected with miR-199a-3p
was significantly higher compared with the NC group at 48 h and 72
h (P<0.05; Fig. 2). The results
indicate that miR-199a-3p overexpression may promote the
proliferation of HUVECs.
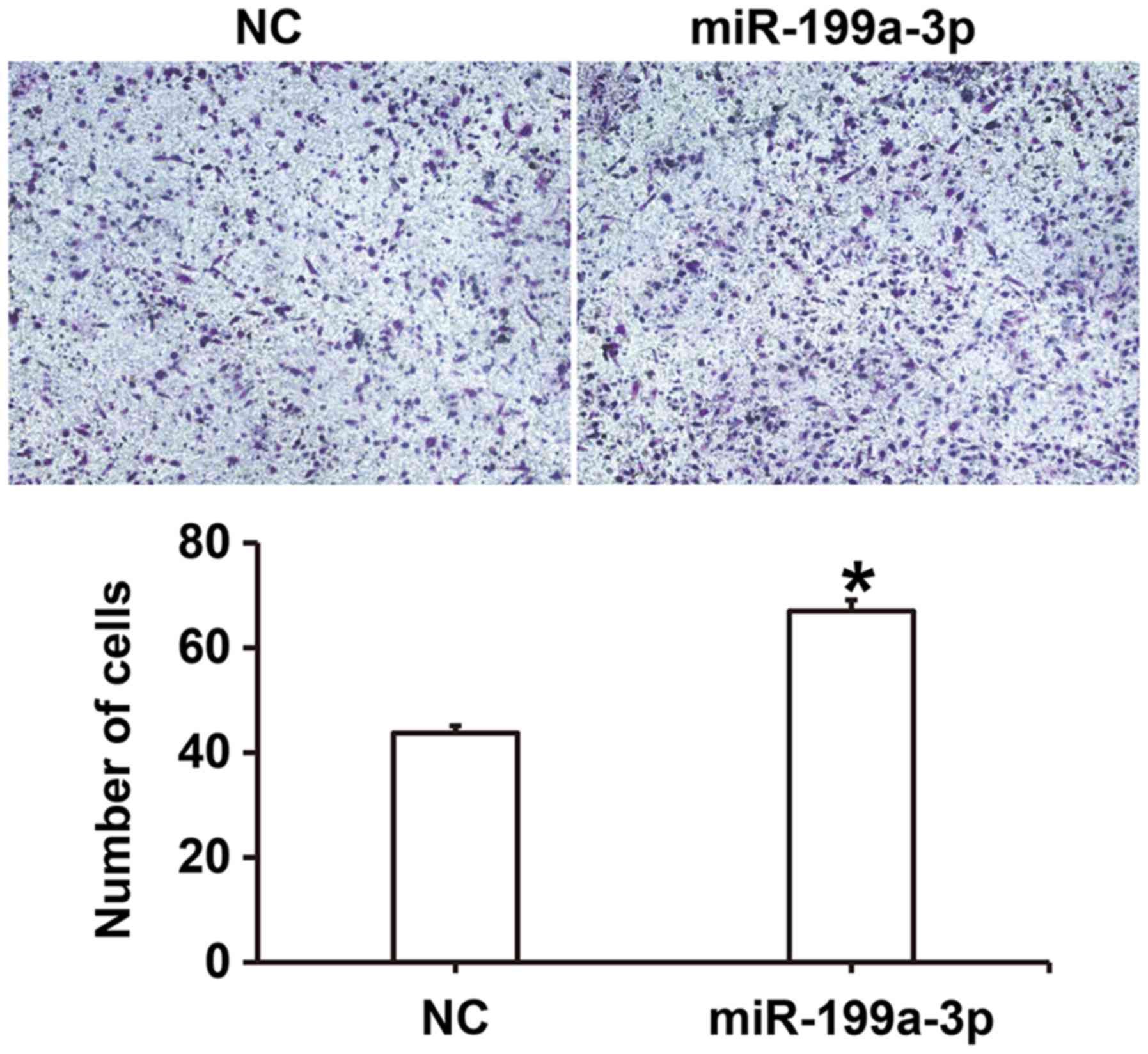
miR-199a-3p overexpression enhances
the migration ability of HUVECs
To examine the effect of miR-199a-3p on HUVEC
migration, a Transwell assay was performed. The number of migrated
cells was significantly higher in the miR-199a-3p group compared
with the NC group (P<0.05; Fig.
3). These results suggest that miR-199a-3p overexpression may
enhance the migration ability of HUVECs.
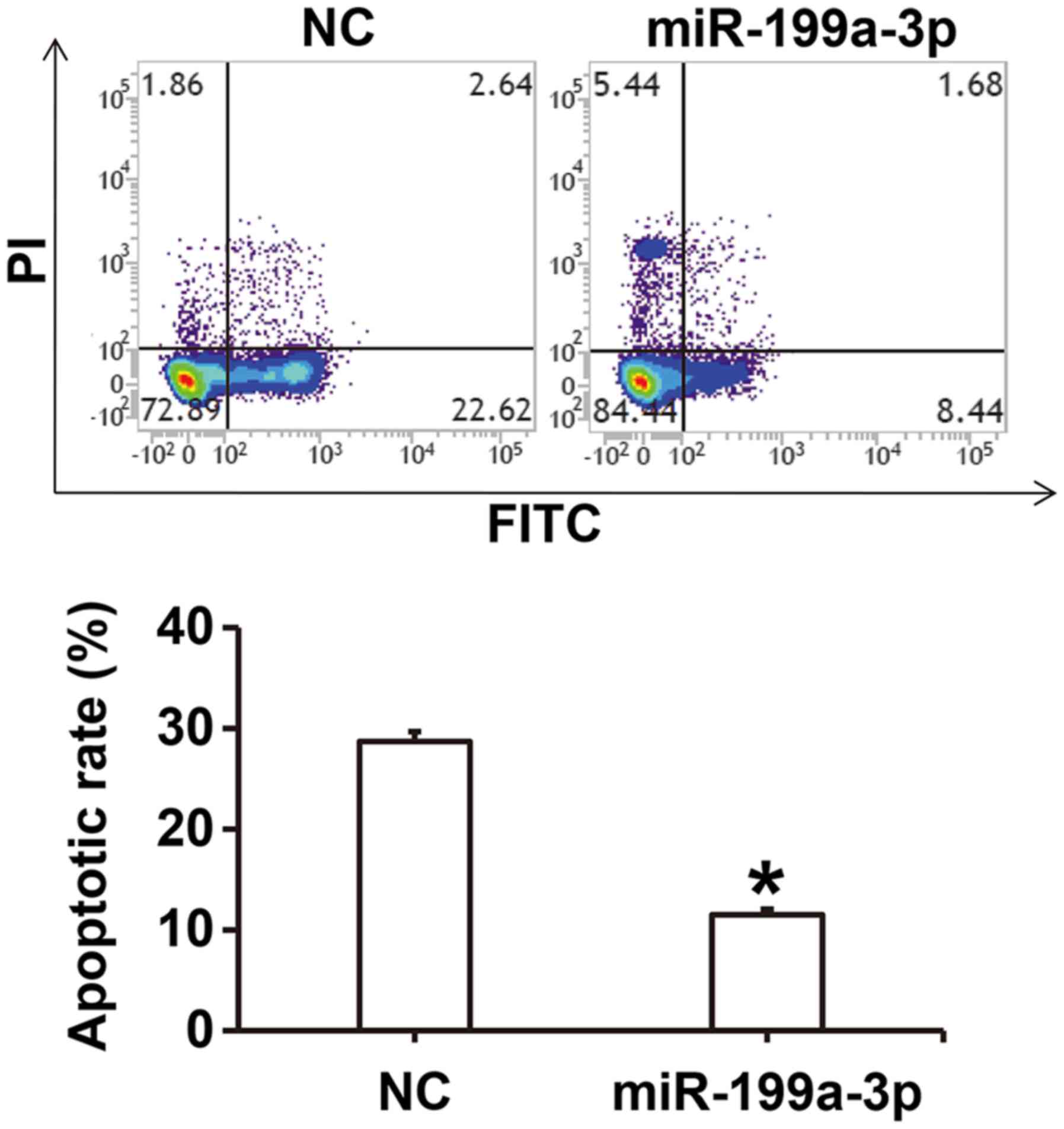
MiR-199a-3p overexpression inhibits
the apoptosis of HUVECs
To investigate how miR-199a-3p overexpression
affects apoptosis in HUVECs cultured under high glucose conditions,
flow cytometry was performed. The results revealed that the
apoptotic rate of HUVECs in the miR-199a-3p group was significantly
lower compared with the NC group (P<0.05; Fig. 4).
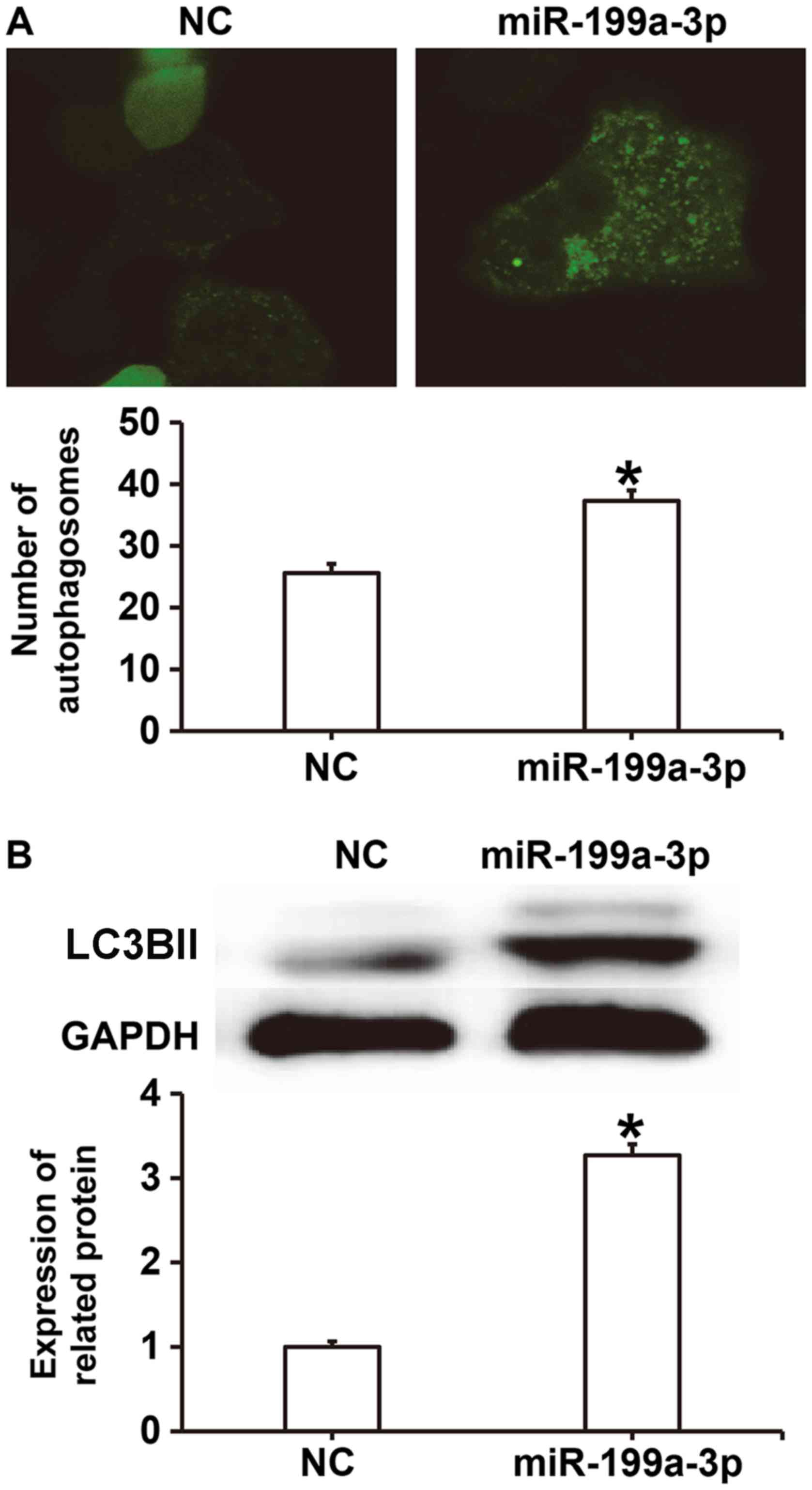
MiR-199a-3p overexpression facilitates
HUVEC autophagy via targeting autophagy-associated signaling
pathways
To investigate the effect of miR-199a-3p on
autophagy, laser-scanning confocal microscopy and western blotting
were performed. The results revealed that the number of
autophagosomes in the miR-199a-3p group was significantly higher
compared with the NC group (P<0.05; Fig. 5A). Western blotting demonstrated that
LC3BII protein expression was significantly increased in the
miR-199a-3p group compared with the NC group (P<0.05; Fig. 5B). These results suggest that
miR-199a-3p overexpression may facilitate HUVEC autophagy by
affecting autophagy-associated signaling pathways.
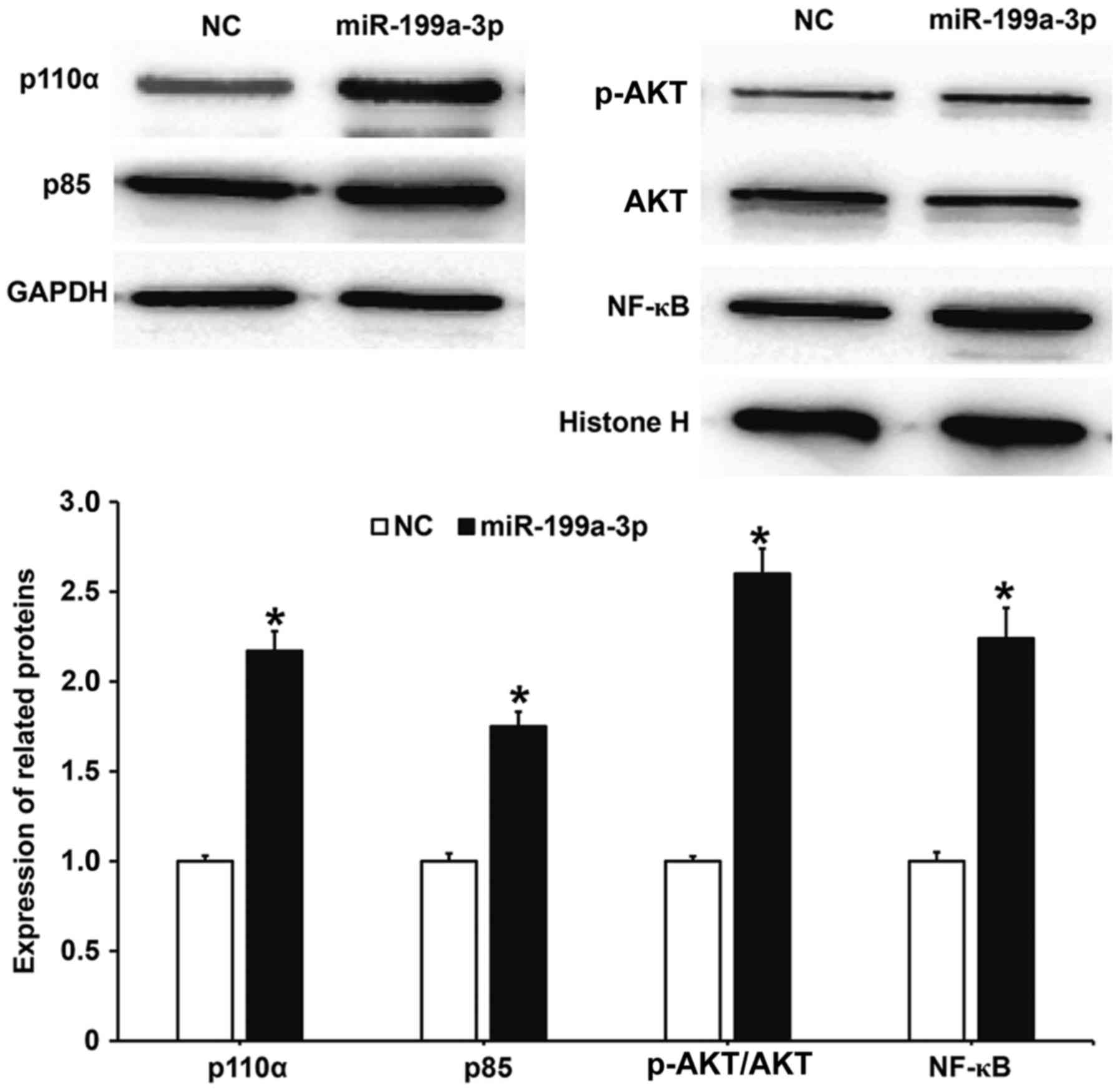
MiR-199a-3p overexpression may
regulate the biological functions of HUVECs via the PI3K/AKT/NF-κB
signaling pathway
To determine whether the biological functions of
miR-199a-3p were associated with activity changes in the
PI3K/AKT/NF-κB signaling pathway, western blotting was performed.
The results demonstrated that protein levels of the PI3K catalytic
subunit p110α and regulatory subunit p85 in HUVECs transfected with
miR-199a-3p were significantly increased compared with the NC group
(P<0.05; Fig. 6). Additionally,
the phosphorylation level of AKT (p-AKT/AKT) in HUVECs transfected
with miR-199a-3p was significantly increased compared with the NC
group (P<0.05). The nuclear expression of NF-κB in HUVECS
transfected with miR-199a-3p was significantly increased compared
with the NC group (P<0.05). These results suggest that
miR-199a-3p overexpression may regulate the biological functions of
HUVECs via the PI3K/AKT/NF-κB signaling pathway.
Discussion
Vascular injury is a basic pathological change in
patients with DM and it has an important influence on the
occurrence and development of DM. Sustained vascular injury
aggravates local inflammatory responses, stimulates the
proliferation and hypertrophy of smooth muscle cells, affects
systolic and diastolic functions and eventually leads to vascular
remodeling (25). In the present
study, it was discovered that the expression of miR-199a-3p in the
peripheral blood of patients with T2DM was significantly reduced
compared with healthy individuals. In vitro experiments
revealed that miR-199a-3p promoted the proliferation, migration and
autophagy of HUVECs potentially via the PI3K/AKT/NF-κB signaling
pathway, while apoptosis was inhibited.
Vascular endothelial cells cover the surface of
vascular intima and induce inflammatory signals, hormone levels,
shear stress or pressure in the blood environment, as well as
secreting a variety of vasoactive substances to regulate vascular
functions (26). Elevated blood
glucose in patients with DM results in repeated injury to vascular
endothelial cells, eventually leading to vascular remodeling and
further weakening blood vessels (27). It has been reported that miRNA serves
important roles in a number of biological functions, including the
proliferation, secretion and migration of vascular endothelial
cells (28). MiR-30b regulates
transforming growth factor-β2 expression to influence the ability
of HUVECS to form tubes in vitro (29). MiR-98 inhibits low-density
lipoprotein (LDL)-induced vascular endothelial cell injury by
targeting lectin-like oxidized LDL receptor-1 gene expression
(30), while miR-320a overexpression
promotes the proliferation of vascular endothelial cells (31). In the present study, it was
determined that miR-199a-3p downregulation in patients with T2DM
was associated with diabetic angiopathy. Furthermore, transfection
with miR-199a-3p promoted the proliferation and migration of
HUVECs, suggesting that miR-199a-3p may promote functional repair
of damaged vascular endothelial. Flow cytometry was used to
investigate HUVEC apoptosis and the results revealed that
miR-199a-3p overexpression inhibited the apoptosis of HUVECs
cultured under high-glucose conditions.
Autophagy is a process in which cells encapsulate
their own proteins or damaged organelles in vesicles that are then
fused with lysosomes to form autophagy lysosomes and the contents
are degraded (32). A study has
demonstrated that autophagy serves important roles in the survival
of vascular endothelial cells (33).
The MIF1 gene regulates the permeability of vascular endothelial
cells by inducing autophagy (34),
while the SIRT1 gene regulates the degradation of LDL in HUVECs via
the autophagy-lysosome pathway (35). In the present study, laser-scanning
confocal microscopy revealed that the number of autophagosomes was
increased in HUVECs transfected with miR-199a-3p compared with the
NC group. Western blotting demonstrated that miR-199a-3p promoted
the expression of LC3BII, suggesting that miR-199a-3p stimulates
the autophagy of HUVECs. The PI3K/AKT signaling pathway is
associated with the physiological and pathological processes of the
body and has important regulatory functions for cell survival,
apoptosis and the synthesis and secretion of inflammatory factors
(36). It has been reported that
PI3K/AKT and its downstream NF-κB signaling pathway promote
proliferation, migration and tube formation in endothelial cells
(37). Netrin-1 regulates high
glucose-induced vascular endothelial cell damage and angiogenesis
via the PI3K/AKT/eNOS signaling pathway (38), while phosphocreatine protects
vascular endothelial cells from oxidative stress-induced apoptosis
through the PI3K/AKT/eNOS and NF-κB signaling pathways (39). The results presented in the current
study suggest that the expression of p110α and p85 was upregulated
in cells transfected with miR-199a-3p compared with the NC group,
while AKT1 phosphorylation was also increased. Furthermore, NF-κB
protein aggregated in the nuclei of HUVECs transfected with
miR-199a-3p. These results suggest that the PI3K/AKT/NF-κB
signaling pathway was activated in HUVECs transfected with
miR-199a-3p and promoted cell proliferation and survival.
In conclusion, the present study demonstrates that
miR-199a-3p expression is downregulated in the peripheral blood of
patients with T2DM and is associated with disease progression.
Additionally, miR-199a-3p may activate the PI3K/AKT/NF-κB signaling
pathway, promote the proliferation, migration and autophagy of
vascular endothelial cells and suppress apoptosis, effectively
inhibiting vascular injury.
Acknowledgements
The authors would like to thank Dr Xiange Tang from
Endocrine Department of the Hospital.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HW and ZW collaborated to design the study. HW, ZW
and QT were responsible for experiments. HW and ZW analyzed the
data. All authors collaborated to interpret results and develop the
manuscript. The final version of the manuscript has been read and
approved by all authors, and each author believes that the
manuscript represents honest work.
Ethics approval and consent to
participate
All procedures performed in the current study were
approved by the Ethics Committee of Taishan Medical University.
Written informed consent was obtained from all patients or their
families.
Patient consent for publication
Written informed consents for publication of any
associated data and accompanying images were obtained from all
patients or their parents, guardians or next of kin.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Buraczynska M, Buraczynska K, Zukowski P
and Ksiazek A: Interleukin-4 gene intron 3 VNTR polymorphism in
type 2 diabetes patients with peripheral neuropathy. Immunol
Invest. 47:146–153. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Vernstrom L, Funck KL, Grove EL, Laugesen
E, Baier JM, Hvas AM and Poulsen PL: Antiplatelet effect of aspirin
during 24h in patients with type 2 diabetes without cardiovascular
disease. Thromb Res. 161:1–6. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhou ZW, Ju HX, Sun MZ, Chen HM, Fu QP and
Jiang DM: Serum fetuin-A levels in obese and non-obese subjects
with and without type 2 diabetes mellitus. Clin Chim Acta.
476:98–102. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Iciek R, Brazert M, Wender-Ozegowska E,
Pietryga M and Brazert J: Low placental visfatin expression is
related to impaired glycaemic control and fetal macrosmia in
pregnancies complicated by type 1 diabetes. J Physiol Pharmacol.
69:61–66. 2018.PubMed/NCBI
|
|
5
|
Plessas A, Robertson DP and Hodge PJ:
Radiographic bone loss in a Scottish non-smoking Type 1 Diabetes
mellitus population; a Bitewing Radiographic Study. J Periodontol.
May 15–2018.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cottam A, Cottam D, Zaveri H, Cottam S,
Surve A, Medlin W and Richards C: An analysis of mid-term
complications, weight loss, and type 2 diabetes resolution of
stomach intestinal pylorus-sparing surgery (SIPS) versus Roux-En-Y
gastric bypass (RYGB) with three-year follow-up. Obes Surg. May
22–2018.(Epub ahead of print). View Article : Google Scholar
|
|
7
|
Del Prato S and Chilton R: Practical
strategies for improving outcomes in T2DM: The potential role of
pioglitazone and DPP4 inhibitors. Diabetes Obes Metab. 20:786–799.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Suchkova OV, Gurfinkel YI and Sasonko ML:
Microcirculatory parameters in compensated and decompensated type 2
diabetes mellitus. Ter Arkh. 89:28–35. 2017.(In Russian; Abstract
available in Russian from the publisher). View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nakamura H, Kato M, Nakaya T, Kono M,
Tanimura S, Sato T, Fujieda Y, Oku K, Ohira H, Bohgaki T, et al:
Decreased haptoglobin levels inversely correlated with pulmonary
artery pressure in patients with pulmonary arterial hypertension: A
cross-sectional study. Medicine (Baltimore). 96:e83492017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Blanco PJ, Muller LO and Spence JD: Blood
pressure gradients in cerebral arteries: A clue to pathogenesis of
cerebral small vessel disease. Stroke Vasc Neurol. 2:108–117. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li X, Hou J, Du J, Feng J, Yang Y, Shen Y,
Chen S, Feng J, Yang D, Pei H, et al: Potential protective
mechanism in the cardiac microvascular injury. Hypertension.
72:116–127. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu X, Cao L, Zhang Y, Lian H, Sun Z and
Cui Y: MicroRNA-1246 inhibits cell invasion and epithelial
mesenchymal transition process by targeting CXCR4 in lung cancer
cells. Cancer Biomark. 21:251–260. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tesfaye D, Gebremedhn S, Salilew-Wondim D,
Hailay T, Hoelker M, Grosse-Brinkhaus C and Schellander K:
MicroRNAs: Tiny molecules with significant role in mammalian
follicular and oocyte development. Reproduction. 155:R121–R135.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu Y, He X, Li Y and Wang T:
Cerebrospinal fluid CD4+ T lymphocyte-derived miRNA-let-7b can
enhances the diagnostic performance of Alzheimer's disease
biomarkers. Biochem Biophys Res Commun. 495:1144–1150. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lv G, Zhu H, Li C, Wang J, Zhao D, Li S,
Ma L, Sun G, Li F, Zhao Y and Gao Y: Inhibition of IL-8-mediated
endothelial adhesion, VSMCs proliferation and migration by
siRNA-TMEM98 suggests TMEM98's emerging role in atherosclerosis.
Oncotarget. 8:88043–88058. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang HH, Sun PF, Chen WK, Zhong J, Shi QQ,
Weng ML, Ma D and Miao CH: High glucose stimulates expression of
MFHAS1 to mitigate inflammation via Akt/HO-1 pathway in human
umbilical vein endothelial cells. Inflammation. 41:400–408. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bartoszewski R, Serocki M,
Janaszak-Jasiecka A, Bartoszewska S, Kochan-Jamrozy K, Piotrowski
A, Króliczewski J and Collawn JF: miR-200b downregulates Kruppel
Like Factor 2 (KLF2) during acute hypoxia in human endothelial
cells. Eur J Cell Biol. 96:758–766. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang HW, Li H, Yan H and Liu BL:
MicroRNA-142 promotes the expression of eNOS in human peripheral
blood-derived endothelial progenitor cells in vitro. Eur Rev Med
Pharmacol Sci. 20:4167–4175. 2016.PubMed/NCBI
|
|
19
|
Varshney A, Panda JJ, Singh AK, Yadav N,
Bihari C, Biswas S, Sarin SK and Chauhan VS: Targeted delivery of
microRNA-199a-3p using self-assembled dipeptide nanoparticles
efficiently reduces hepatocellular carcinoma in mice. Hepatology.
67:1392–1407. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Qu F, Zheng J, Gan W, Lian H, He H, Li W,
Yuan T, Yang Y, Li X, Ji C, et al: MiR-199a-3p suppresses
proliferation and invasion of prostate cancer cells by targeting
Smad1. Oncotarget. 8:52465–52473. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Park KM, Teoh JP, Wang Y, Broskova Z,
Bayoumi AS, Tang Y, Su H, Weintraub NL and Kim IM:
Carvedilol-responsive microRNAs, miR-199a-3p and −214 protect
cardiomyocytes from simulated ischemia-reperfusion injury. Am J
Physiol Heart Circ Physiol. 311:H371–H383. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhu H and Leung SW: Identification of
microRNA biomarkers in type 2 diabetes: A meta-analysis of
controlled profiling studies. Diabetologia. 58:900–911. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mathews E, Thomas E, Absetz P, D'Esposito
F, Aziz Z, Balachandran S, Daivadanam M, Thankappan KR and
Oldenburg B: Cultural adaptation of a peer-led lifestyle
intervention program for diabetes prevention in India: The Kerala
diabetes prevention program (K-DPP). BMC Public Health. 17:9742018.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Coelho SC, Berillo O, Caillon A, Ouerd S,
Fraulob-Aquino JC, Barhoumi T, Offermanns S, Paradis P and
Schiffrin EL: Three-month endothelial human endothelin-1
overexpression causes blood pressure elevation and vascular and
kidney injury. Hypertension. 71:208–216. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lai TS, Lindberg RA, Zhou HL, Haroon ZA,
Dewhirst MW, Hausladen A, Juang YL, Stamler JS and Greenberg CS:
Endothelial cell-surface tissue transglutaminase inhibits
neutrophil adhesion by binding and releasing nitric oxide. Sci Rep.
7:161632017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Pang LP, Li Y, Zou QY, Zhou C, Lei W,
Zheng J and Huang SA: ITE inhibits growth of human pulmonary artery
endothelial cells. Exp Lung Res. 43:283–292. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang X, He XQ, Li GD and Xu YQ:
AntagomiR-451 inhibits oxygen glucose deprivation (OGD)-induced
HUVEC necrosis via activating AMPK signaling. PLoS One.
12:e01755072017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Howe GA, Kazda K and Addison CL:
MicroRNA-30b controls endothelial cell capillary morphogenesis
through regulation of transforming growth factor beta 2. PLoS One.
12:e01856192017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chen Z, Wang M, He Q, Li Z, Zhao Y, Wang
W, Ma J, Li Y and Chang G: MicroRNA-98 rescues proliferation and
alleviates ox-LDL-induced apoptosis in HUVECs by targeting LOX-1.
Exp Ther Med. 13:1702–1710. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sun JY, Zhao ZW, Li WM, Yang G, Jing PY,
Li P, Dang HZ, Chen Z, Zhou YA and Li XF: Knockdown of MALAT1
expression inhibits HUVEC proliferation by upregulation of miR-320a
and downregulation of FOXM1 expression. Oncotarget. 8:61499–61509.
2017.PubMed/NCBI
|
|
32
|
Lin SY, Hsieh SY, Fan YT, Wei WC, Hsiao
PW, Tsai DH, Wu TS and Yang NS: Necroptosis promotes
autophagy-dependent upregulation of DAMP and results in
immunosurveillance. Autophagy. 14:778–795. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yuan Y, Li X and Li M: Overexpression of
miR-17-5p protects against high glucose-induced endothelial cell
injury by targeting E2F1-mediated suppression of autophagy and
promotion of apoptosis. Int J Mol Med. May 21–2018.(Epub ahead of
print). View Article : Google Scholar
|
|
34
|
Chao CH, Chen HR, Chuang YC and Yeh TM:
Macrophage migration inhibitory factor-induced autophagy
contributes to thrombin-triggered endothelial hyperpermeability in
sepsis. Shock. 50:103–111. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang Y, Sun J, Yu X, Shi L, Du W, Hu L,
Liu C and Cao Y: SIRT1 regulates accumulation of oxidized LDL in
HUVEC via the autophagy-lysosomal pathway. Prostaglandins Other
Lipid Mediat. 122:37–44. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Fan H, Ma X, Lin P, Kang Q, Zhao Z, Wang
L, Sun D, Cheng J and Li Y: Scutellarin prevents nonalcoholic fatty
liver disease (NAFLD) and hyperlipidemia via PI3K/AKT-dependent
activation of nuclear factor (Erythroid-Derived 2)-like 2 (Nrf2) in
rats. Med Sci Monit. 23:5599–5612. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liu LT, Liang L, Wang W, Yan CQ, Zhang J,
Xiao YC, Ye L, Zhao MX, Huang QS, Bian JJ, et al:
Isolariciresinol-9′-O-α-L-arabinofuranoside protects against
hydrogen peroxideinduced apoptosis of human umbilical vein
endothelial cells via a PI3K/Akt/Bad-dependent pathway. Mol Med
Rep. 17:488–494. 2018.PubMed/NCBI
|
|
38
|
Xing Y, Lai J, Liu X, Zhang N, Ming J, Liu
H and Zhang X: Netrin-1 restores cell injury and impaired
angiogenesis in vascular endothelial cells upon high glucose by
PI3K/AKT-eNOS. J Mol Endocrinol. 58:167–177. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chu P, Han G, Ahsan A, Sun Z, Liu S, Zhang
Z, Sun B, Song Y, Lin Y, Peng J and Tang Z: Phosphocreatine
protects endothelial cells from methylglyoxal induced oxidative
stress and apoptosis via the regulation of PI3K/Akt/eNOS and NF-κB
pathway. Vascul Pharmacol. 91:26–35. 2017. View Article : Google Scholar : PubMed/NCBI
|