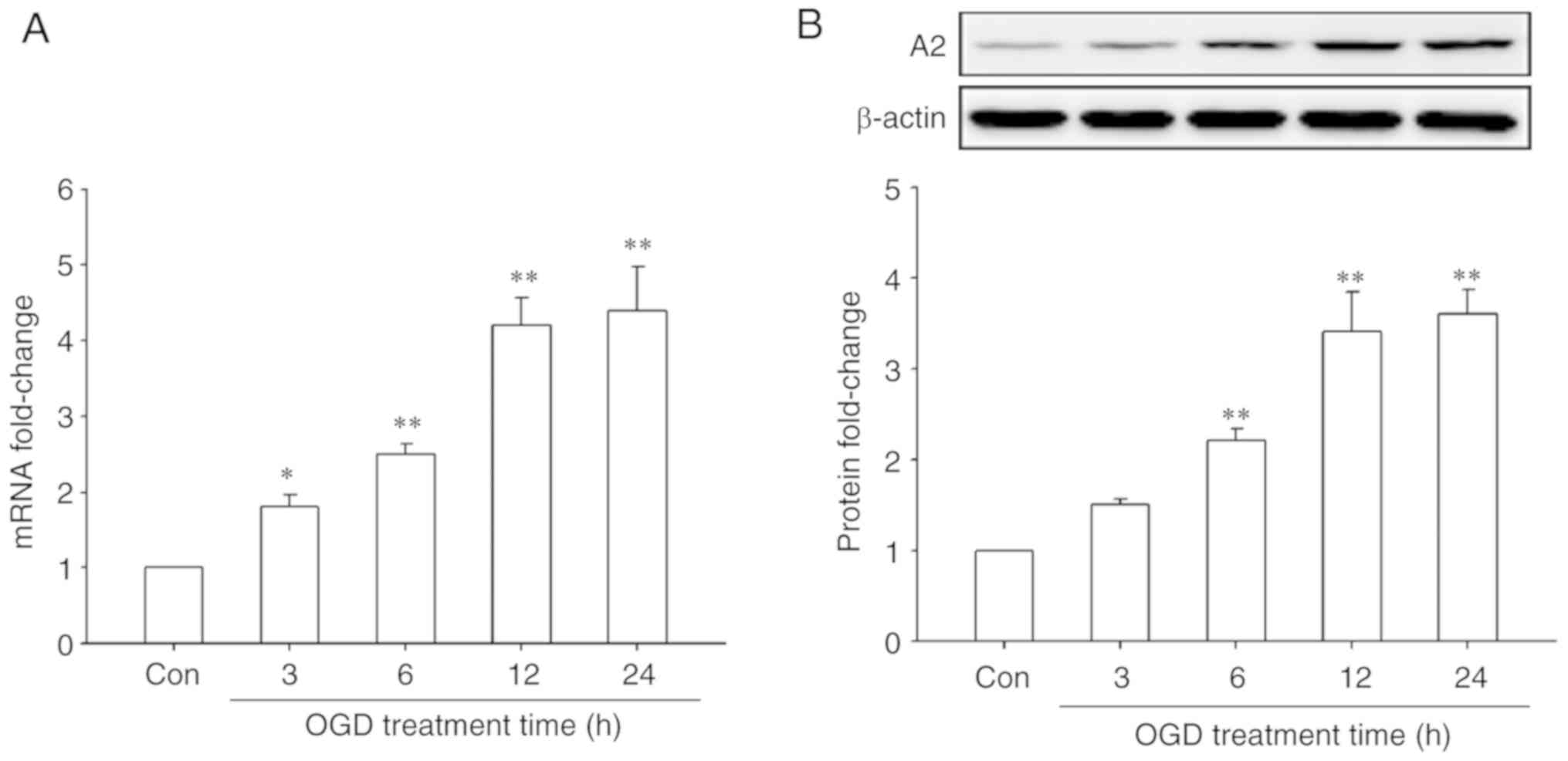
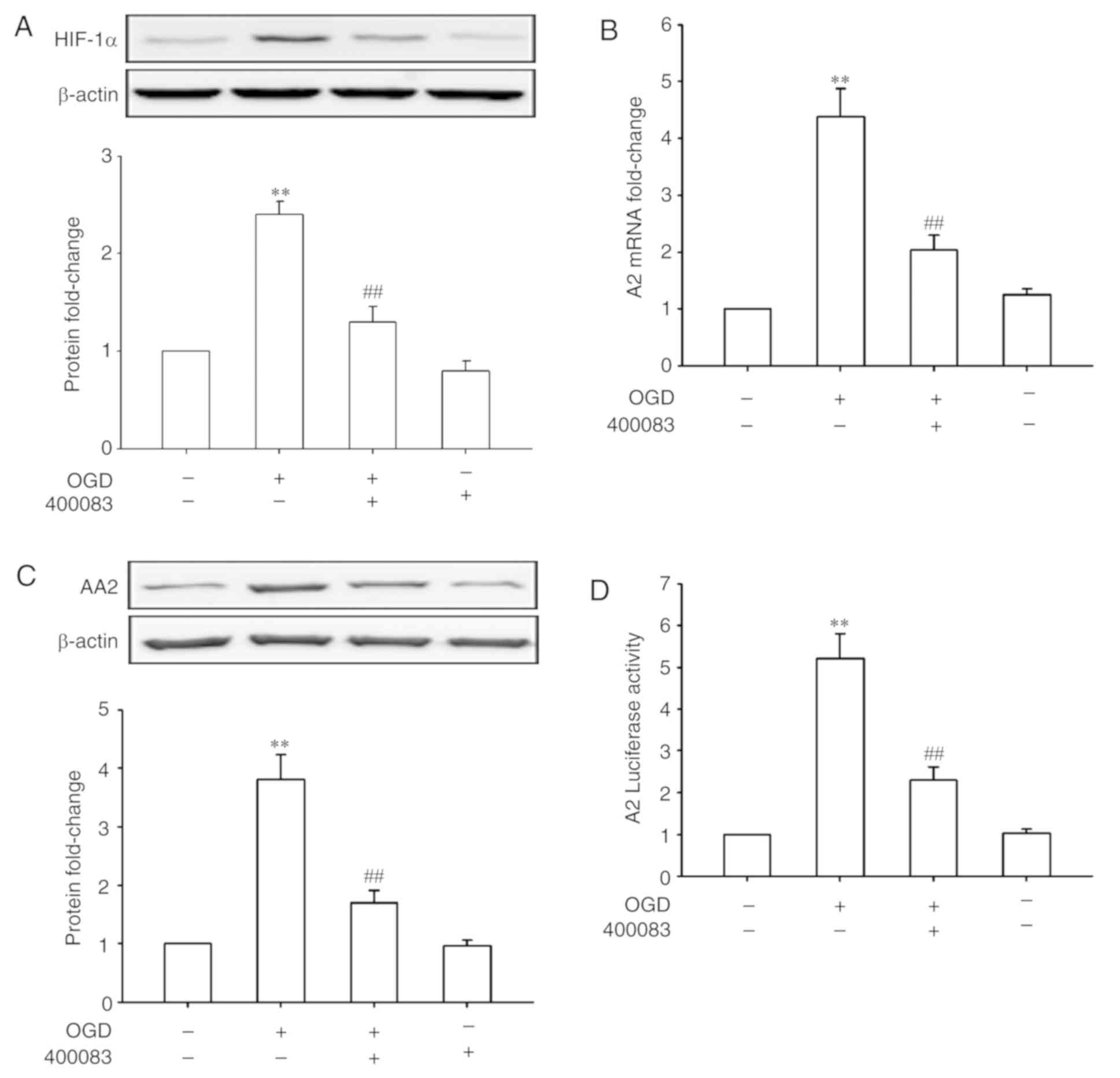
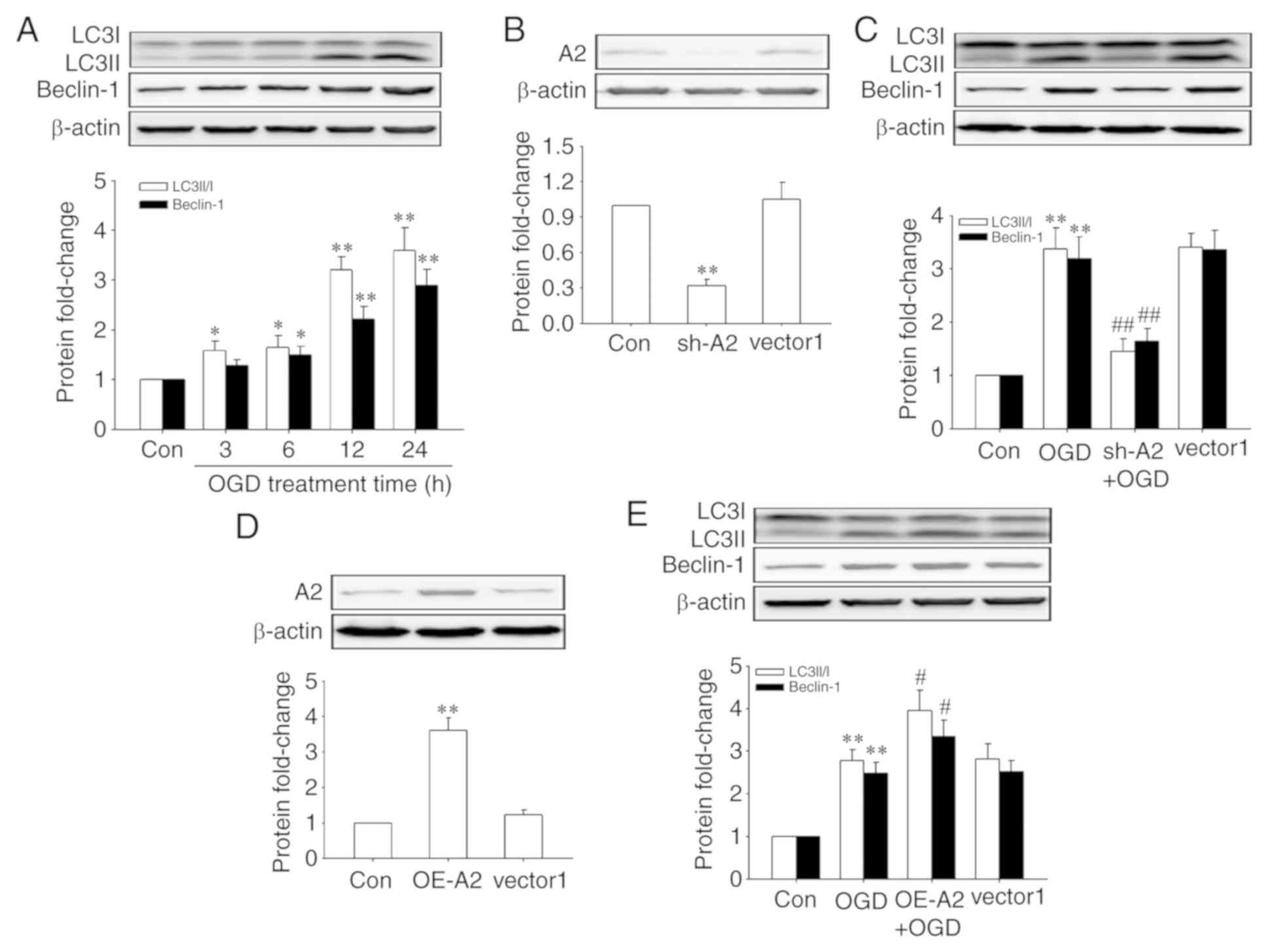
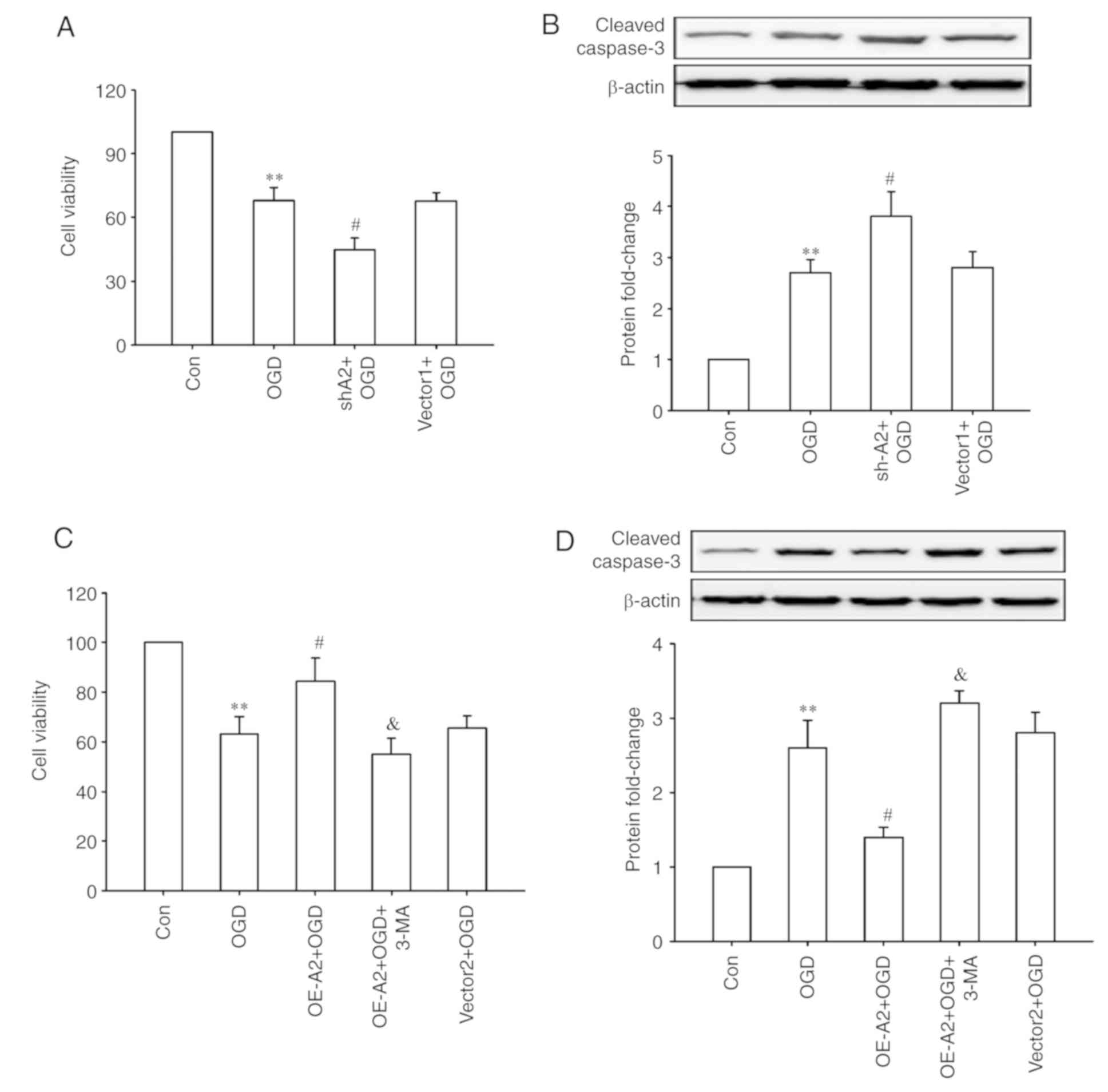
|
1
|
Zhang SX, Ma JH, Bhatta M, Fliesler SJ and
Wang JJ: The unfolded protein response in retinal vascular
diseases: Implications and therapeutic potential beyond protein
folding. Prog Retin Eye Res. 45:111–131. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mao XB, You ZP, Wu C and Huang J:
Potential suppression of the high glucose and insulin-induced
retinal neovascularization by Sirtuin 3 in the human retinal
endothelial cells. Biochem Biophys Res Commun. 482:341–345. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hajjar KA: The biology of Annexin A2: From
vascular fibrinolysis to innate immunity. Trans Am Clin Climatol
Assoc. 126:144–155. 2015.PubMed/NCBI
|
|
4
|
Smith LE, Wesolowski E, McLellan A, Kostyk
SK, D'Amato R, Sullivan R and D'Amore PA: Oxygen-induced
retinopathy in the mouse. Invest Ophthalmol Vis Sci. 35:101–111.
1994.PubMed/NCBI
|
|
5
|
Ling Q, Jacovina AT, Deora A, Febbraio M,
Simantov R, Silverstein RL, Hempstead B, Mark WH and Hajjar KA:
Annexin II regulates fibrin homeostasis and neoangiogenesis in
vivo. J Clin Invest. 113:38–48. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gerke V, Creutz CE and Moss SE: Annexins:
Linking Ca2+ signalling to membrane dynamics. Nat Rev Mol Cell
Biol. 6:449–461. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Grindheim AK, Saraste J and Vedeler A:
Protein phosphorylation and its role in the regulation of Annexin
A2 function. Biochim Biophys Acta Gen Subj. 1861:2515–2529. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Moss SE and Morgan RO: The annexins.
Genome Biol. 5:2192004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Luo M and Hajjar KA: Annexin A2 system in
human biology: Cell surface and beyond. Semin Thromb Hemost.
39:338–346. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Song H, Pan D, Sun W, Gu C, Zhang Y, Zhao
P, Qi Z and Zhao S: SiRNA directed against annexin II receptor
inhibits angiogenesis via suppressing MMP2 and MMP9 expression.
Cell Physiol Biochem. 35:875–884. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jiang SL, Pan DY, Gu C, Qin HF and Zhao
SH: Annexin A2 silencing enhances apoptosis of human umbilical vein
endothelial cells in vitro. Asian Pac J Trop Med. 8:952–957. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huang G, Su J, Zhang M, Jin Y, Wang Y,
Zhou P and Lu J: RhoB regulates the function of macrophages in the
hypoxia-induced inflammatory response. Cell Mol Immunol.
14:265–275. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li R, Wang LZ, Du JH, Zhao L and Yao Y:
Autophagy activation and the mechanism of retinal microvascular
endothelial cells in hypoxia. Int J Ophthalmol. 11:567–574.
2018.PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wu D, Luo N, Wang L, Zhao Z, Bu H, Xu G,
Yan Y, Che X, Jiao Z, Zhao T, et al: Hydrogen sulfide ameliorates
chronic renal failure in rats by inhibiting apoptosis and
inflammation through ROS/MAPK and NF-κB signaling pathways. Sci
Rep. 7:4552017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
You P, Wu H, Deng M, Peng J, Li F and Yang
Y: Brevilin A induces apoptosis and autophagy of colon
adenocarcinoma cell CT26 via mitochondrial pathway and
PI3K/AKT/mTOR inactivation. Biomed Pharmacother. 98:619–625. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Semenza GL: Targeting HIF-1 for cancer
therapy. Nat Rev Cancer. 3:721–732. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Riboldi E, Porta C, Morlacchi S, Viola A,
Mantovani A and Sica A: Hypoxia-mediated regulation of macrophage
functions in pathophysiology. Int Immunol. 25:67–75. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Klionsky DJ, Abdalla FC, Abeliovich H,
Abraham RT, Acevedo-Arozena A, Adeli K, Agholme L, Agnello M,
Agostinis P, Aguirre-Ghiso JA, et al: Guidelines for the use and
interpretation of assays for monitoring autophagy. Autophagy.
8:445–544. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Guo T, Song H, Zhao Z, Qi Z and Zhao S:
Overexpression of Annexin A2 receptor inhibits neovascularization
via the promotion of krüppel-like transcription factor 2. Cell
Physiol Biochem. 46:1617–1627. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zagórska A and Dulak J: HIF-1: The knowns
and unknowns of hypoxia sensing. Acta Biochim Pol. 51:563–585.
2004.PubMed/NCBI
|
|
22
|
Huang B, Deora AB, He KL, Chen K, Sui G,
Jacovina AT, Almeida D, Hong P, Burgman P and Hajjar KA:
Hypoxia-inducible factor-1 drives annexin A2 system-mediated
perivascular fibrin clearance in oxygen-induced retinopathy in
mice. Blood. 118:2918–2929. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li R, Tan S, Yu M, Jundt MC, Zhang S and
Wu M: Annexin A2 regulates autophagy in pseudomonas aeruginosa
infection through the Akt1-mTOR-ULK1/2 signaling pathway. J
Immunol. 195:3901–3911. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Moreau K, Ghislat G, Hochfeld W, Renna M,
Zavodszky E, Runwal G, Puri C, Lee S, Siddiqi F, Menzies FM, et al:
Transcriptional regulation of Annexin A2 promotes
starvation-induced autophagy. Nat Commun. 6:80452015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang Y, Song H, Guo T, Zhu Y, Tang H, Qi
Z, Zhao P and Zhao S: Overexpression of Annexin II receptor-induced
autophagy protects against apoptosis in uveal melanoma cells.
Cancer Biother Radiopharm. 31:145–151. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Choi Y, Bowman JW and Jung JU: Autophagy
during viral infection-a double-edged sword. Nat Rev Microbiol.
16:341–354. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Huang Y, Jin Y, Yan CH, Yu Y, Bai J, Chen
F, Zhao YZ and Fu SB: Involvement of Annexin A2 in p53 induced
apoptosis in lung cancer. Mol Cell Biochem. 309:117–123. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang YX, Lv H, Li ZX, Li C and Wu XY:
Effect of shRNA mediated down-regulation of Annexin A2 on
biological behavior of human lung adencarcinoma cells A549. Pathol
Oncol Res. 18:183–190. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sharathchandra A, Lal R, Khan D and Das S:
Annexin A2 and PSF proteins interact with p53 IRES and regulate
translation of p53 mRNA. RNA Biol. 9:1429–1439. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang Y, Yang C, Gu Q, Sims M, Gu W,
Pfeffer LM and Yue J: KLF4 promotes angiogenesis by activating VEGF
signaling in human retinal microvascular endothelial cells. PLoS
One. 10:e01303412015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jo DH, Bae J, Chae S and Kim JH, Han JH,
Hwang D, Lee SW and Kim JH: Quantitative proteomics reveals β2
integrin-mediated cytoskeletal rearrangement in vascular
endothelial growth factor (VEGF)-induced retinal vascular
hyperpermeability. Mol Cell Proteomics. 15:1681–1691. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Domigan CK, Warren CM, Antanesian V,
Happel K, Ziyad S, Lee S, Krall A, Duan L, Torres-Collado AX,
Castellani LW, et al: Autocrine VEGF maintains endothelial survival
through regulation of metabolism and autophagy. J Cell Sci.
128:2236–2248. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhao S, Huang L, Wu J, Zhang Y, Pan D and
Liu X: Vascular endothelial growth factor upregulates expression of
annexin A2 in vitro and in a mouse model of ischemic retinopathy.
Mol Vis. 15:1231–1242. 2009.PubMed/NCBI
|
|
34
|
Genetos DC, Wong A, Watari S and Yellowley
CE: Hypoxia increases Annexin A2 expression in osteoblastic cells
via VEGF and ERK. Bone. 47:1013–1019. 2010. View Article : Google Scholar : PubMed/NCBI
|