Introduction
As a common noncommunicable chronic disease,
diabetes mellitus (DM) has become a global health issue. It is
estimated that the morbidity of DM is expected to rise to 552
million by 2030 (1). Subjected to a
relative or absolute deficiency of insulin secretion, patients with
DM present numerous complications due to the long-term conditions
of hyperglycemia, such as diabetic cardiomyopathy. DM may also
cause abnormality in the heart metabolic signaling pathway. These
mediators can result in diastolic dysfunction at the early stage of
DM and damaged systolic function at the late stage of DM (2).
A number of studies have been performed to explore
potential critical signaling pathways in DM (3–5). For
example, in the pathogenesis of diabetic cardiomyopathy, the state
of chronic hyperglycemia increases the formation of diacylglycerol
(DAG), activates protein kinase C (PKC) and accelerates
non-enzymatic formation of advanced glycated end products (6). These intracellular changes eventually
cause structural and functional alteration of cardiomyocytes.
The PKC family comprises of more than ten different
isozymes and was found to be associated with cardiovascular
diseases such as cardiac hypertrophy and ischemia-reperfusion
injury (7,8). It was reported that PKC inhibition
could partly reverse both structural abnormalities and cardiac
dysfunction in cardiac hypertrophy (9). PKC-α and PKC-β2 are two
highly expressed PKC isoforms in the heart. Previous studies showed
that high glucose stimulation could activate PKC-α and
PKC-β2 at the early stage of diabetic cardiomyopathy
(10). Such changes of those two
isoforms suggested they may act as an important mediator in the
pathological progress of diabetic cardiomyopathy (11). However, the exact role of the PKC
family and its downstream signaling pathway in diabetic
cardiomyopathy remains to be completely elucidated.
β-receptor blockers (β-blockers) are one of the most
commonly used medicines for the treatment of cardiovascular
diseases, including cardiac hypertrophy, heart failure, arrhythmia
and angina (12,13). Use of β-blockers could effectively
improve cardiac function, reverse the remodeling of left-ventricle
and enhance the ability of physical exercise-related capacity
(14). Although the prognostic
benefits of β-blockers for patients with cardiovascular disease are
commonly recognized, they are yet to be elucidated in patients with
diabetes, as beta-blockers could delay the development of diabetes
by counteracting hypoglycemia symptoms (15). However, β-blockers can also disrupt
glycemic control and lipid metabolism of diabetic patients. In a
Glycemic Effects in Diabetes Mellitus: Carvedilil-Metoprolol
Comparison in Hypertensives study, patients with hypertension and
type 2 DM (T2DM) treated with metaprolol showed a significant
increase in hemoglobin A1c levels compared with baseline
levels (16). In another study,
insulin-stimulated endothelial function in diabetic patients was
significantly decreased following treatment with metaprolol
(17). However, diabetic patients
who have developed systolic heart failure (SHF) may still benefit
from appropriate β-blocker therapy. A meta-analysis of large-scale
clinical trials has verified that β-blockers could reduce mortality
rates in patients with SHF with DM (18).
Metoprolol and bisoprolol, both β-antagonists,
improve the prognosis of heart failure and prevent the progression
of left ventricle remodeling (19).
In ischemic or non-ischemic heart failure, both metoprolol and
bisoprolol have been reported to reduce cardiac events, attenuate
cardiac dysfunction, and reduce mortality (20,21).
Therapy with beta-blockers, such as bisoprolol, has a favorable
effect in clinical outcomes of reducing mortality and morbidity of
heart failure patients with new-onset DM, either with heart failure
with preserved or reduced ejection fraction (22). However, how metoprolol and bisoprolol
attenuate left ventricle remodeling or dysfunction associated with
diabetic cardiomyopathy remains to be determined.
In the present study, PKC and its downstream
PKC/NF-κB/c-fos signaling pathway was hypothesized to be critical
for the development of cardiac hypertrophy related to diabetic
cardiomyopathy. Moreover, the current study also aimed to determine
whether metoprolol and bisoprolol could attenuate cardiac
hypertrophy induced by hyperglycemia and elucidate the underlying
mechanism of this process.
Materials and methods
Experimental animals
Cardiomyocytes of each experiment were derived from
1 to 3-day-old neonatal Sprague-Dawley rats (total number of rats,
30; weight, 4–6 g; male rats, 15; female rats, 15), provided by the
Laboratory Animal Center of Zhejiang University. Animals were
housed in a temperature of 22–26°C and a humidity of 50–65%
controlled environment with a 12-h light-dark cycle. The rats had
free access to water and food. Procedures in the current study were
all conducted according to the guidelines for animal care (23) and approved by the Ethics Committee of
Zhejiang University.
Reagents, chemicals and drugs
DMEM, FBS and 0.25% trypsin were obtained from
Gibco; Thermo Fisher Scientific, Inc.; type II collagenase, glucose
and [3H]-leucine were provided by Sigma-Aldrich; Merck
KGaA. Ro-31-8220 and BAY11-7082 were obtained from Selleck
Chemcicals; metoprolol and bisoprolol were obtained from
AstraZeneca. Primary antibodies for detection of PKC-α (cat. no.
2056), p-PKC-β2 (cat. no. 9371), NF-κB p65 (cat. no.
8242), p-NF-κB (cat. no. 3033), Histone H3 (cat. no. 9717) and
c-fos (cat. no. 2250) were purchased from Cell Signaling
Technology. PKC-β2 (cat. no. sc-210) and p-PKC-α (cat.
no. sc-12356) were purchased from Santa Cruz Biotechnology. β-actin
primary antibody (cat. no. 70-ab010-100) and horseradish
peroxidase-linked goat anti-rabbit secondary antibodies (cat. no.
70-GAM0072) were purchased from MultiSciences Biotech Co., Ltd.
Primers were designed according to our previous study (11) and synthesized by TSINGKE. TRIzon
reagent (cat. no. CW0580) for extracting total RNA and UltraSYBR
Mixture (cat. no. CW0957) for reverse transcription-quantitative
PCR (RT-qPCR) assays were purchased from CWBIO Biosciences.
PrimeScript RT-PCR kit was purchased from Takara Bio, Inc.
Isolation and culture of neonatal rat
cardiomyocytes
Cardiomyocytes were obtained and cultured as
follows. Neonatal Sprague-Dawley rats were sacrificed and hearts
were dissected. A diluted solution made up of 0.125% trypsin and
0.1% type II collagenase was used to digest the ventricular tissues
for 8 min at 37°C. Following each digestion, the supernatant was
collected and added to DMEM with 10% FBS. The step was repeated six
to eight times until the ventricles were completely digested. The
collected mixture was then centrifuged at 55.9 × g for 5 min at
room temperature. The supernatant was discarded and cells were
resuspended in ACK Lysis Buffer (Beijing Solarbio Science and
Technology Co., Ltd.) for 2 min. The cells were centrifuged as
described above once again, and the collected cells were
resuspended in DMEM with 10% FBS. Differential adhesion method was
used for separating cardiomyocytes from myofibroblasts.
Cardiomyocytes were diluted to (3–5)×105/ml in DMEM with 10% FBS
and cultured in six-well plates in a humidified atmosphere with 5%
CO2 at 37°C. Culture medium was replaced with serum-free
DMEM after 48 h.
Treatment groups
After the first 48 h, cardiomyocytes were cultured
in serum-free DMEM with different concentrations of glucose and
drugs for another 48, or 72 h for the [3H]-leucine
incorporation measurements at 37°C with humidified 5%
CO2 as follows: Low glucose (LG; 5.5 mM), high glucose
(HG; 25.5 mM), HG + metoprolol (0.5 or 10 µM), HG + bisoprolol (50
or 200 nM), HG + PKC inhibitor Ro-31-8220 (50 nM) and HG + NF-κB
inhibitor BAY11-7082 (5 µM). A higher dose of metoprolol or
bisoprolol was used in combination with PKC inhibitor (Ro-31-8220)
or NF-κB inhibitor (BAY11-7082) to determine whether the
hypertrophic response could be attenuated to a lower extent
compared with metoprolol or bisoprolol treatment alone. Cells were
cultured in serum-free DMEM in the following conditions: HG +
metoprolol (10 µmol/l) + Ro-31-8220 (50 nmol/l), HG + metoprolol
(10 µmol/l) + BAY11-7082 (5 µmol/l), HG + bisoprolol (200 nmol/l) +
Ro-31-8220 (50 nmol/l) and HG + bisoprolol (200 nmol/l) +
BAY11-7082 (5 µmol/l).
Determination of cellular pulsatile
frequency
Individual cellular pulsatile frequency of
cardiomyocytes was determined using an inverted light microscope.
The inverted microscope and 24-well plates were briefly placed in a
homoiothermic air convection assembly, which was fully covered by
convection air at 37°C. Five random fields from each group were
selected and 20 individual cardiomyocytes of each field were
counted for the pulsatile frequency at ×400 magnification.
Measurement of cellular diameter
Cardiomyocytes on glass coverslips were washed with
warm PBS twice, fixed with 4% paraformaldehyde for 10 min at room
temperature and permeabilized with 0.5% Triton-100 for 5 min at
room temperature. After washing, cells were incubated with 100 nM
rhodamine-phalloidin (Beijing Solarbio Science and Technology Co.,
Ltd.) for 30 min at room temperature. Cells were then stained with
DAPI (Beijing Solarbio Science and Technology Co., Ltd.) for 30 sec
at room temperature. Images of five randomly selected fields of
view were captured for each group and used to measure the cellular
diameters of cardiomyocytes using a fluorescent microscope at ×400
magnification.
Determination of cell protein
content
Cardiomyocytes were washed with Hanks' balanced salt
solution (HBSS) three times before being lysed with 1% SDS. Protein
content measurement was then carried out in accordance with the
Lowry method (24). A standard
protein solution was utilized to prepare a standard curve for the
final estimation of protein content. The absorbance of the final
solution was then measured at a wavelength of 750 nm.
Incorporation of
[3H]-leucine
To measure newly synthesized protein of
cardiomyocytes (25),
[3H]-leucine incorporation method was conducted as
previously described by Luo et al (26). Firstly, 1 µCi [3H]-leucine
was added to cell culture medium with corresponding levels of
glucose for cardiomyocytes treatment. β-blockers and inhibitors
were then co-incubated with the cells for 72 h. Following
incubation, cells were quickly washed with cold HBSS three times. A
total of 1 ml 1% SDS was added to each well to lyse cells and the
lysates were collected. Subsequently, 1 ml 5% trichloroacetic acid
was added to the lysates at 4°C for 1 h before the lysates were
precipitated and transferred to fiberglass filters. Finally,
lysates were washed with HBSS before the precipitates were dried
and moved to scintillation fluid. Radioactivity was detected and
expressed as cpm/well by liquid scintillation counting.
Nucleus extraction
To detect the translocation of NF-κB and p-NF-κB in
the nucleus by the glucose stimulation, nucleus extraction was
conducted using Nuclear Extraction kit (cat. no. SN0020) according
to the manufacture's protocol (Beijing Solarbio Life Science &
Technology Co., Ltd.). Cells in the cultured plates with a density
of 5×105/ml were firstly digested with EDTA buffer and
washed with PBS. Those cells were centrifuged at the speed of 800 ×
g for 5 min at 4°C and resuspended with 1 ml cold lysis buffer with
PMSF and 50 µl Reagent A provided by the kit. Then, the 1.05 ml
cell suspension was transferred to a small glass homogenizer, and
the cells were grinded 20–30 times in an ice bath. Then, the cell
homogenates were centrifuged at the speed of 700 × g at 4°C for 5
min to collect the sediments. After resuspension with 0.5 ml cold
lysis buffer, the same amount of medium buffer were mixed and
centrifuged at the speed of 700 × g at 4°C for 5 min. The nucleus
was resuspended with lysis buffer and centrifuged at the speed of
1,000 × g at 4°C for 10 min. The final sediments were resuspended
by the store buffer provided by the kit.
RT-qPCR
Measurement of atrial natriuretic peptide (ANP),
α-myosin heavy chain (α-MHC) and β-myosin heavy chain (β-MHC) and
tumor necrosis factor-α (TNF-α) mRNA transcripts was performed
using RT-qPCR. Total RNA from cardiomyocytes was extracted using
TRIzon reagent and reverse transcribed into cDNA using a
PrimeScript RT-PCR kit (Takara Bio, Inc.) according to the
manufacturer's instructions. mRNA quantification was conducted
using Nanodrop 2000 (Applied Biosystems; Thermo Fisher Scientific,
Inc.) (27). The thermcycling
condition used were as follows: Initial denaturation at 95°C for 10
min; followed by 40 cycles, each cycle included denaturation at
95°C for 15 sec and an extension at 60°C for 1 min. RT-qPCR was
performed with UltraSYBR Mixture on the Viia 7 system (Applied
Biosystems; Thermo Fisher Scientific, Inc.) following the
manufacturer's instructions. The primers used in the current study
are listed in Table I.
 | Table I.List of primers used for reverse
transcription-quantitative PCR. |
Table I.
List of primers used for reverse
transcription-quantitative PCR.
|
| Primer sequence
5′-3′ |
|---|
|
|
|
|---|
| Gene | Forward | Reverse |
|---|
| ANP |
GCTCGAGCAGATCGCAAAAG |
CACCACCTCTCAGTGGCAAT |
| α-MHC |
GCCGAGTCCCAGGTCAACA |
TATTGGCCACAGCGAGGGTCT |
| β-MHC |
CACCCGCGAGTACAACCTTC |
CCCATACCCACCATCACACC |
| TNF-α |
GAACAACCCTACGAGCACCT |
GGGTAGTTTGGCTGGGATAA |
| GAPDH |
ACCCACTTCTCCACCTTTGAC |
TGTTGCTGTAGCCAAATTCG |
RT-qPCR and Southern blot
The mRNA expresssion level of TNF-α was measured by
RT-qPCR and Southern blot. RT-qPCR was performed to convert the
mRNA into cDNA as described above, followed by PCR using the
primers listed in Table I to amplify
the target fragments. The resulting products were then separated on
1.2% agarose gel and stained with 1 µg/ml ethidium bromide for 30
min at room temperature. The bands were then exposed and quantified
using Gel DOC XR image system (Bio-Rad Laboratories, Inc.). The
intensity of the TNF-α band was normalized by GAPDH. Densitometry
was analyzed using ImageJ software (version 2.1.4.7; National
Institutes of Health).
Western blot analysis of cultured
cardiomyocytes
Cells were washed with cold PBS twice and lysed with
RIPA buffer and PMSF (Beijing Solarbio Science and Technology Co.,
Ltd.; 100:1) for 15 min. Lysates were centrifuged at 8,049.6 × g
for 15 min at 4°C. After quantification by bicinchoninic acid
assay, the collected supernatant was added to 5X loading buffer
(4:1) and incubated at 100°C for 5 min. Protein separation was
conducted using 10% SDS-PAGE gels. A total of 20 µg of protein from
each group and 5 µl of marker were loaded into each lane. The
separated protein was then transferred to PVDF membranes and
blocked with 5% non-fat milk for 1 h at room temperature. The
membranes were incubated with primary antibodies and 5% BSA
(1:1,000) overnight at 4°C. Following primary antibody incubation,
membranes were incubated with secondary antibodies (1:5,000) for 2
h at room temperature. Protein bands were visualized using the
HRP-ECL kit (Bio-Rad Laboratores, Inc.), and optical density was
measured using the Amersham Imager 600 System (GE Healthcare Life
Sciences). The intensity of bands were normalized to β-actin.
Densitometry of western blot bands was analyzed using ImageJ
software (version 2.1.4.7; National Institutes of Health).
Statistical analysis
All experimental data are presented as the mean ± SD
for at least three individual experiments. Statistical analysis of
multiple comparisons was performed by one-way ANOVA followed by a
Tukey's post-hoc test using GraphPad Prism software (Version 8.0;
GraphPad Software, Inc.). P<0.05 indicated statistically
significant differences.
Results
Metoprolol and bisoprolol decrease the
pulsatile frequency, cellular diameter and cell surface area of
HG-treated cardiomyocytes
Cardiomyocytes treated with HG showed increased
pulsatile frequency and cellular diameter compared with LG-treated
samples (69.42±1.66 vs. 57.63±1.82 bpm, P<0.05; and 24.81±0.78
vs. 18.50±0.67 µm, P<0.05). The cell surface area of HG-treated
cardiomyocytes was also increased by 2.6-fold compared with the
LG-treated cardiomyocytes. After adding metoprolol, bisoprolol,
Ro-31-8220 and BAY11-7082, the pulsatile frequency, cellular
diameter and cell surface area of cardiomyocytes decreased
significantly compared with the HG group, as shown in Table II and Fig. 1A.
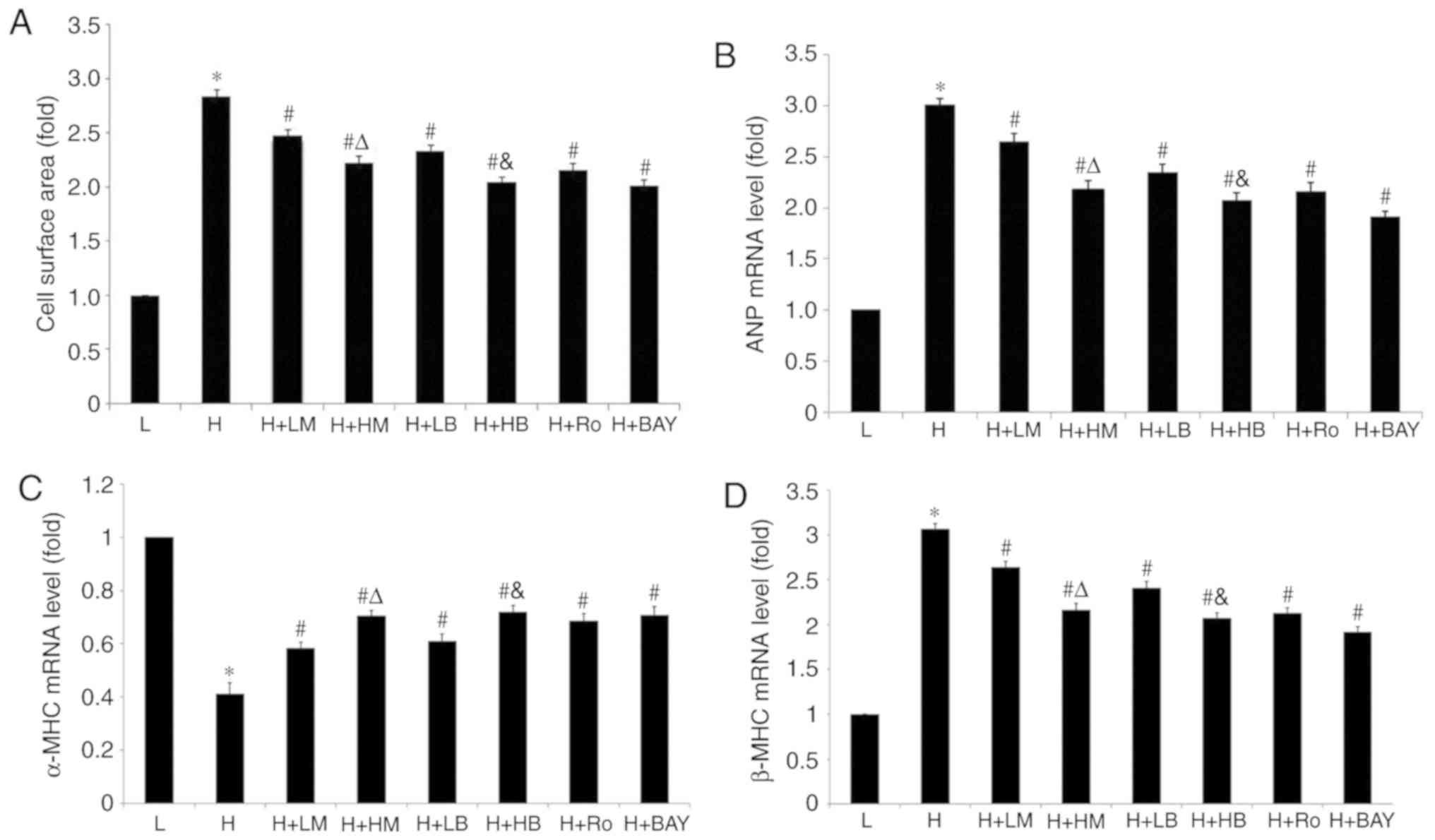 | Figure 1.Metoprolol and bisoprolol decrease
cell surface area and ANP and β-MHC mRNA levels and increase α-MHC
mRNA levels of cardiomyocytes cultured in high glucose. (A) Cell
surface area. mRNA expression levels of (B) ANP, (C) α-MHC and (D)
β-MHC. n=100 cardiomyocytes for measurement of each group.
*P<0.05 vs. L group, #P<0.05 vs. H group,
ΔP<0.05 vs. H + LM group, &P<0.05
vs. H + LB group. L, low glucose; H, high glucose; LM, low
metoprolol dose; HM, high metoprolol dose; LB, low bisoprolol dose;
HB, high bisoprolol dose; Ro, Ro-31-8220; BAY, BAY11-7082; ANP,
atrial natriuretic peptide; MHC, myosin heavy chain. |
 | Table II.Effects of metoprolol, bisoprolol,
Ro-31-8220 and BAY11-7082 on pulsatile frequency and cellular
diameter of cardiomyocytes cultured in high glucose. |
Table II.
Effects of metoprolol, bisoprolol,
Ro-31-8220 and BAY11-7082 on pulsatile frequency and cellular
diameter of cardiomyocytes cultured in high glucose.
| Treatment | Pulsatile frequency
(bpm) | Diameter (µm) |
|---|
| Low glucose (5.5
mmol/l) | 57.63±1.82 | 18.50±0.67 |
| High glucose (25.5
mmol/l) |
69.42±1.66a |
24.81±0.78a |
| High glucose +
metoprolol (0.5 µmol/l) |
64.25±1.64b |
21.82±0.60b |
| High glucose +
metoprolol (10 µmol/l) |
61.74±2.11b,c |
20.12±0.57b,c |
| High glucose +
bisoprolol (50 nmol/l) |
63.83±1.65b |
21.54±0.48b |
| High glucose +
bisoprolol (200 nmol/l) |
61.12±1.88b,d |
19.76±0.6b,d |
| High glucose +
Ro-31-8220 (50 nmol/l) |
63.90±1.38b |
19.98±0.55b |
| High glucose +
BAY11-7082 (5 µmol/l) |
62.31±1.70b |
19.70±0.62b |
Metoprolol and bisoprolol decrease the
total protein content and [3H]-leucine incorporation of
HG-treated cardiomyocytes
Cardiomyocytes treated with HG presented increased
total protein content and [3H]-leucine incorporation
compared with LG-treated cardiomyocytes (57.21±5.29 vs. 31.22±2.30
µg/well, P<0.05; and 1,510.64±82.31 vs. 1,033.21±60.33 cpm/well,
P<0.05). Following addition of metoprolol, bisoprolol,
Ro-31-8220 and BAY11-7082, total protein content and
[3H]-leucine incorporation of cardiomyocytes was
significantly reduced compared with the HG group (P<0.05), as
shown in Table III.
 | Table III.Effects of metoprolol, bisoprolol,
Ro-31-8220 and BAY11-7082 on total protein content and
[3H]-leucine incorporation of cardiomyocytes cultured in
high glucose. |
Table III.
Effects of metoprolol, bisoprolol,
Ro-31-8220 and BAY11-7082 on total protein content and
[3H]-leucine incorporation of cardiomyocytes cultured in
high glucose.
| Treatment | Protein
(µg/well) |
[3H]-leucine incorporation
(cpm/well) |
|---|
| Low glucose (5.5
mmol/l) | 31.22±2.30 | 1,033.21±60.33 |
| High glucose (25.5
mmol/l) |
57.21±5.29a |
1,510.64±82.31a |
| High glucose +
metoprolol (0.5 µmol/l) |
45.73±4.97b |
1,318.15±63.91b |
| High glucose +
metoprolol (10 µmol/l) |
39.94±5.20b,c |
1,152.82±87.84b,c |
| High glucose +
bisoprolol (50 nmol/l) |
42.14±4.89b |
1,140.53±76.26b |
| High glucose +
bisoprolol (200 nmol/l) |
41.76±3.71b |
1,130.29±73.05b |
| High glucose +
Ro-31-8220 (50 nmol/l) |
47.88±4.76b |
1,330.27±74.84b |
| High glucose +
BAY11-7082 (5 µmol/l) |
40.60±3.66b |
1,158.76±87.43b |
Metoprolol and bisoprolol regulate
ANP, α-MHC and β-MHC mRNA levels of HG-treated cardiomyocytes
Cardiomyocytes treated with HG showed increased mRNA
levels of ANP and β-MHC and decreased mRNA levels of α-MHC compared
with LG-treated cardiomyocytes (all P<0.05). Following addition
of metoprolol, bisoprolol, Ro-31-8220 and BAY11-7082, mRNA
expression levels of ANP and β-MHC in cardiomyocytes were
significantly reduced compared with HG-treated samples (P<0.05).
Meanwhile, α-MHC mRNA levels of cardiomyocytes following treatment
with of metoprolol, bisoprolol, Ro-31-8220 and BAY11-7082 were
upregulated compared with HG-treated samples (all P<0.05), as
shown in Fig. 1B-D.
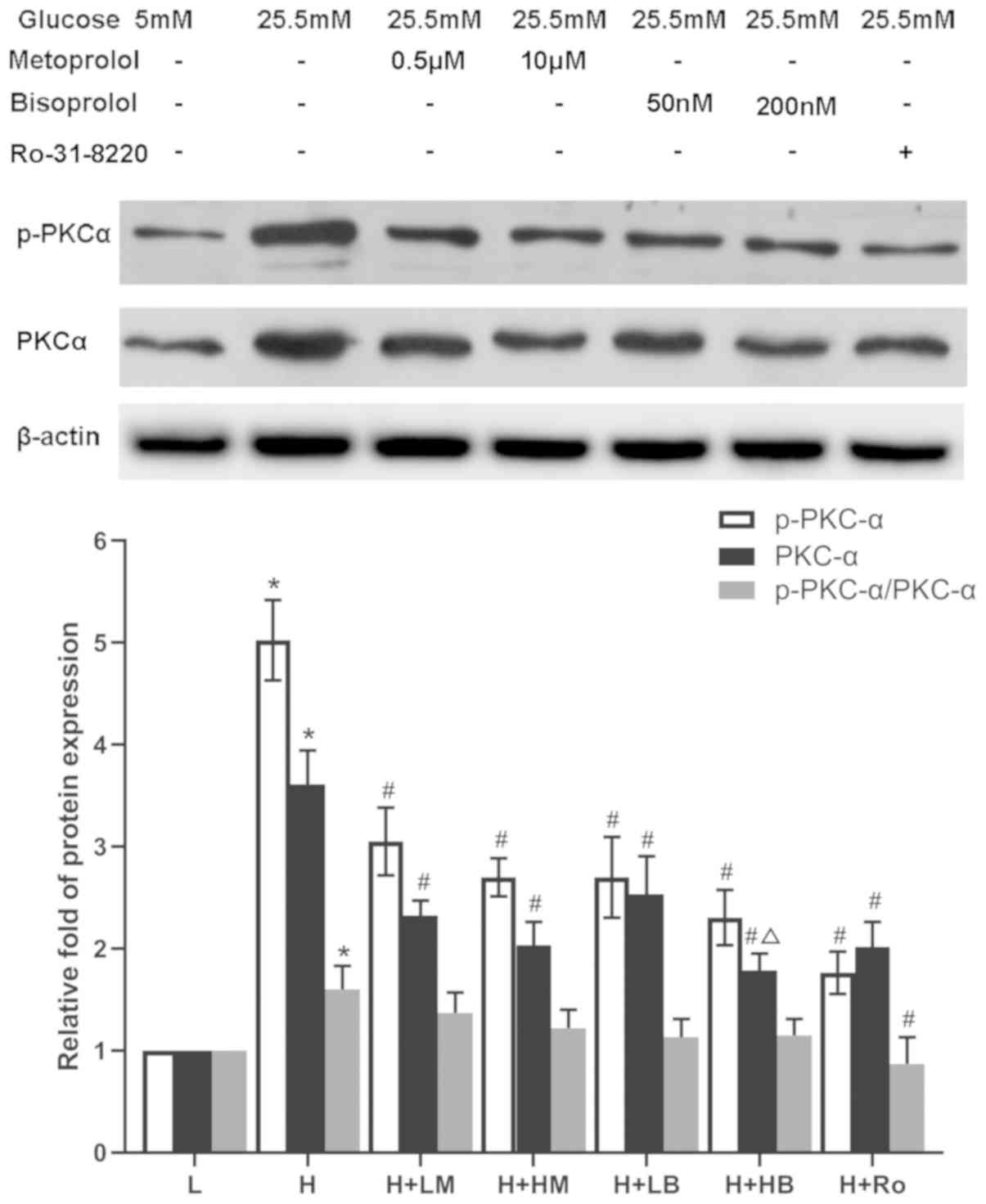
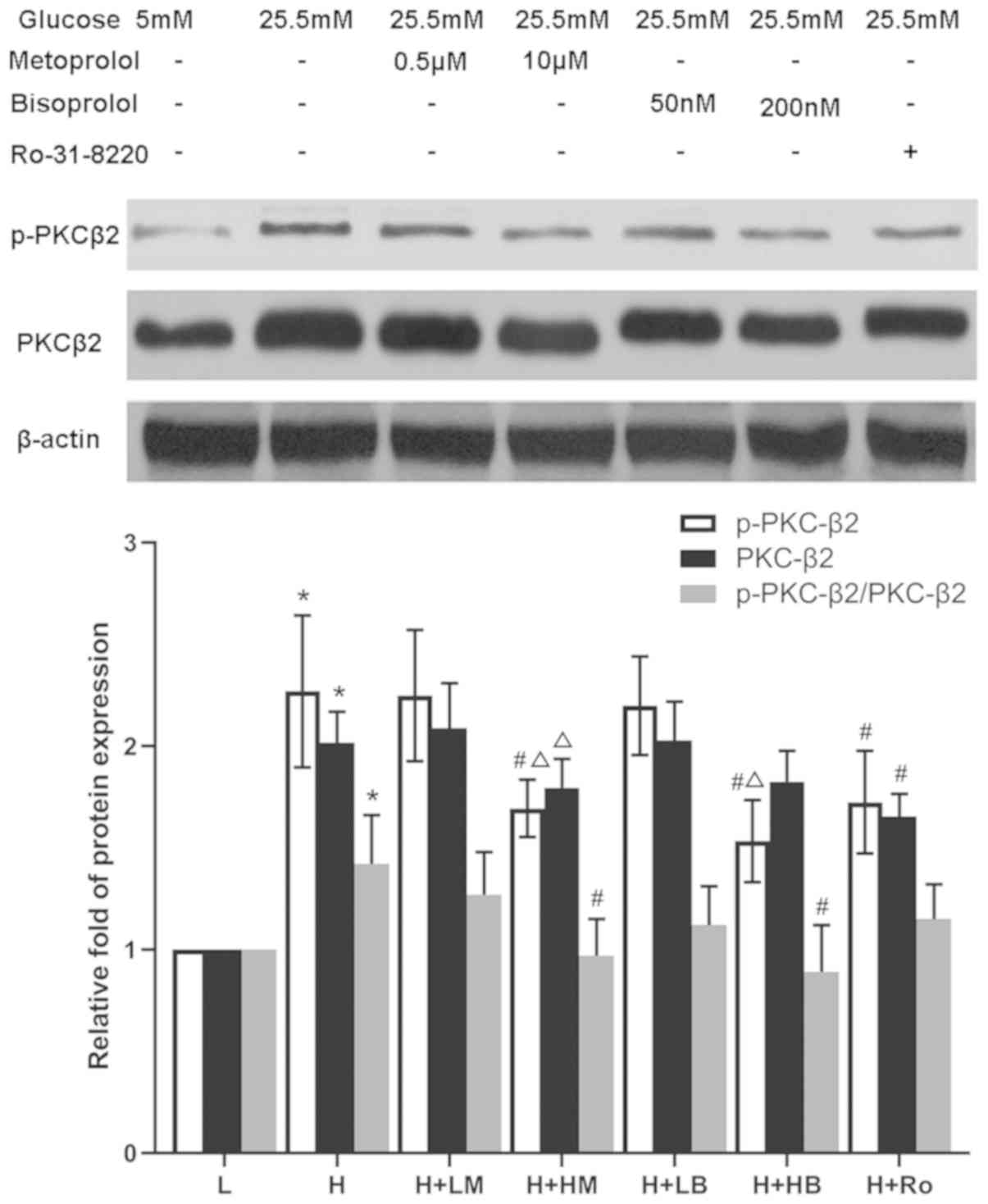
Metoprolol and bisoprolol attenuate
the expression and activity of PKC-α and PKC-β2 in
HG-treated cardiomyocytes
Cardiomyocytes treated with HG showed increased
expression and activity of PKC-α and PKCβ2, reflected by
the increased expression of PKC-α, p-PKC-α, PKC-β2,
p-PKC-β2, as well as the ratios of p-PKC-α/PKC-α and
p-PKC-β2/PKC-β2, compared with LG-treated
samples (P<0.05). Following addition of Ro-31-8220 or either
dose of metoprolol and bisoprolol, the expression levels of PKC-α
and p-PKC-α were significantly decreased compared with HG-treated
samples. In addition, the ratio of p-PKC-α/PKC-α was decreased in
the Ro-31-8220 group. However, only high doses of metoprolol and
bisoprolol could significantly attenute the increased
PKC-β2 and p-PKC-β2 protein levels, and the
ratio of p-PKC-β2/PKC-β2, except for the
PKC-β2 level treated by high dose of bisoprolol,
compared with HG-treated samples (P<0.05; Figs. 2 and 3).
Metoprolol and bisoprolol reduce the
expression of phospho-NF-κB, NF-κB, TNF-α and c-fos in HG-treated
cardiomyocytes
The relative expression of p-NF-κB, NF-κB, TNF-α and
c-fos significantly increased in cardiomyocytes treated with HG
compared with LG-treated samples (P<0.05). In addition, the
ratio of p-NF-κB/NF-κB was also increased. Following addition of
metoprolol, bisoprolol, Ro-31-8220 and BAY11-7082, the relative
expression of NF-κB was significantly reduced in those groups
compared with HG-treated samples. However, only the high metoprolol
dose group, Ro-31-8220 group and BAY11-7082 group decreased the
expression level of p-NF-κB. The ratio of p-NF-κB/NF-κB was not
significantly decreased following these treatments. Treatments of
metoprolol, bisoprolol, Ro-31-8220 and BAY11-7082 reduced the mRNA
expression levels of TNF-α and protein expression levels of c-fos
compared with HG-treated samples (P<0.05; Figs. 4–6).
In addition, high dose of bisoprolol further suppressed the mRNA
expression levels of TNF-α and protein expression levels of c-fos
compared with the low bisoprolol dose group. A similar
dose-dependent suppressive effect was also found in high metoprolol
dose group on the expression of c-fos protein levels.
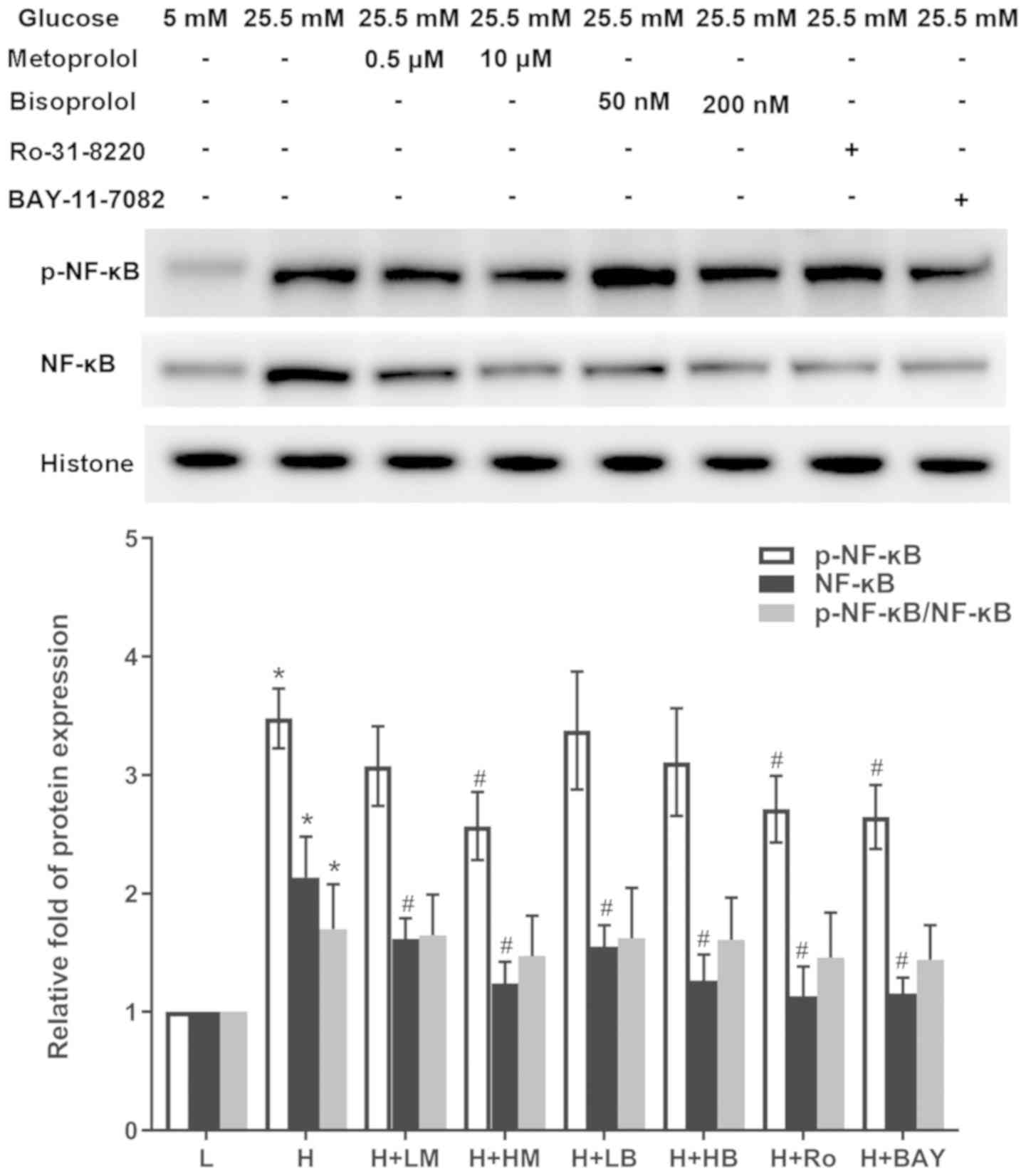 | Figure 4.Effects of metoprolol, bisoprolol,
PKC inhibitor Ro-31-8220 and NF-κB inhibitor BAY11-7082 on the
expression and activity of NF-κB in cardiomyocytes cultured in high
glucose. Cardiomyocytes cultured in HG showed higher expression and
activity of NF-κB compared with LG-cultured cardiomyocytes. n=4–5.
*P<0.05 vs. L group, #P<0.05 vs. H group, L, low
glucose; H, high glucose; LM, low metoprolol dose; HM, high
metoprolol dose; LB, low bisoprolol dose; HB, high bisoprolol dose;
Ro, Ro-31-8220; BAY, BAY11-7082. |
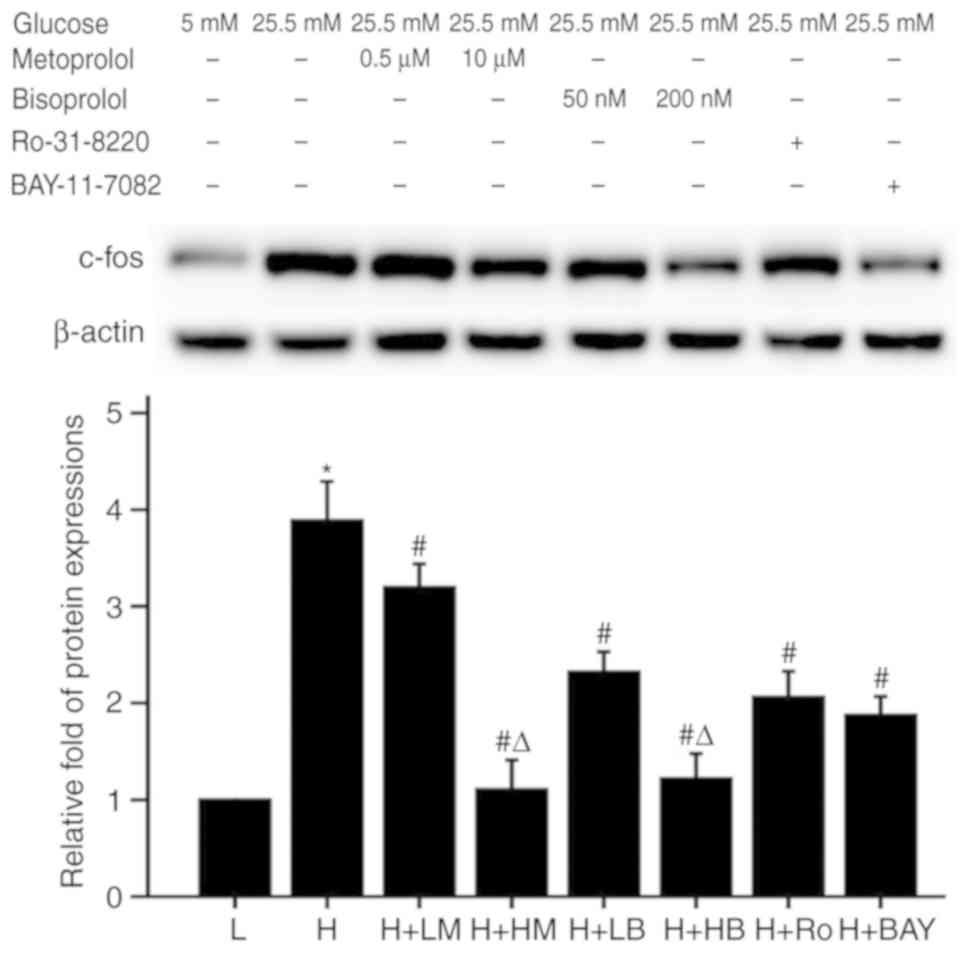 | Figure 6.Effects of metoprolol, bisoprolol,
PKC inhibitor Ro-31-8220 and NF-κB inhibitor BAY11-7082 on c-fos
expression in cardiomyocytes cultured in HG. n=4–5. *P<0.05 vs.
L group, #P<0.05 vs. H group, ΔP<0.05
vs. corresponding low dose group of metoprolol or bisoprolol. L,
low glucose; H, high glucose; LM, low metoprolol dose; HM, high
metoprolol dose; LB, low bisoprolol dose; HB, high bisoprolol dose;
Ro, Ro-31-8220; BAY, BAY11-7082. |
Combined use of PKC inhibitor, NF-κB
inhibitor with metoprolol or bisoprolol further decreases the
pulsatile frequency, cellular diameter, cell surface area and
regulates ANP, α-MHC and β-MHC mRNA levels of HG-treated
cardiomyocytes
Metoprolol reduced the pulsatile frequency, cellular
diameter and cell surface area of cardiomyocytes treated with HG
(Table IV; Fig. 7A). The mRNA levels of ANP and β-MHC
of cardiomyocytes were significantly reduced, while α-MHC mRNA
levels of cardiomyocytes showed a significant increase compared
with HG-treated samples (Fig. 7B-D).
Combined use of Ro-31-8220 or BAY11-7082 with metoprolol further
decreased the pulsatile frequency, cellular diameter and cell
surface area of cardiomyocytes treated with HG (Table IV; Figs.
7A and 8), as well as ANP and
β-MHC mRNA expression levels (Fig. 7B
and D) compared with the HG-treated samples. In addition,
increased α-MHC mRNA expression levels were also observed in these
treatment groups compared with the HG-treated samples. Similar
results were observed when bisoprolol was used in combination with
Ro-31-8220 or BAY11-7082 (P<0.05; Table IV; Fig.
7).
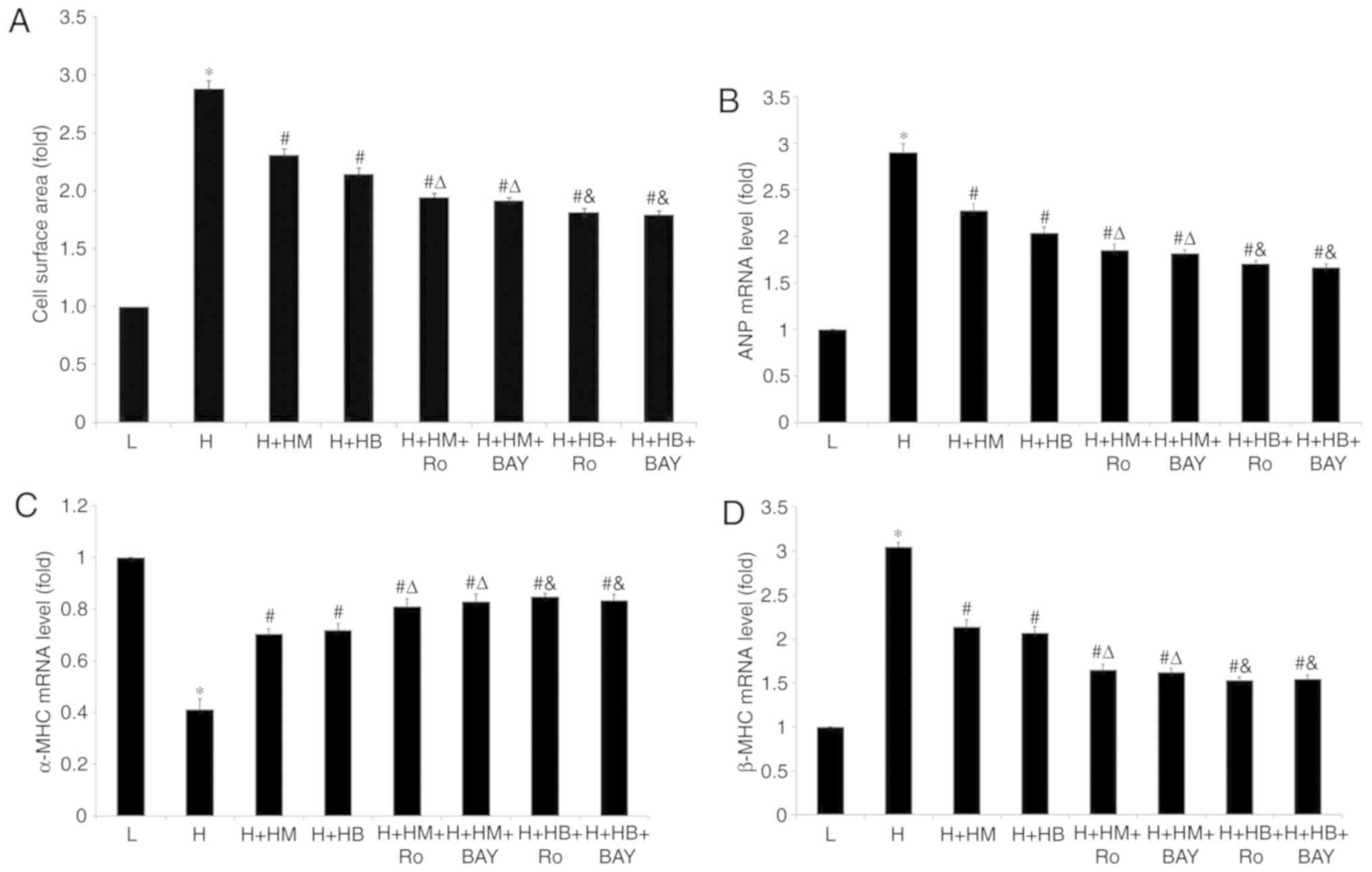 | Figure 7.Combined use of PKC inhibitor
Ro-31-8220 or NF-κB inhibitor BAY11-7082 with metoprolol or
bisoprolol further decreases cell surface area, ANP and β-MHC mRNA
levels and increases α-MHC mRNA levels of cardiomyocytes cultured
in high glucose. (A) Cell surface area. mRNA expression levels of
(B) ANP, (C) α-MHC and (D) β-MHC. n=100 cardiomyocytes for
measurement of each group. *P<0.05 vs. L group,
#P<0.05 vs. H group, ΔP<0.05 vs. HG +
HM group, &P<0.05 vs. HG + HB group. L, low
glucose; H, high glucose; LM, low metoprolol dose; HM, high
metoprolol dose; LB, low bisoprolol dose; HB, high bisoprolol dose;
Ro, Ro-31-8220; BAY, BAY11-7082; ANP, atrial natriuretic peptide;
MHC, myosin heavy chain; PKC, protein kinase C. |
 | Table IV.Combined use of protein kinase C
inhibitor Ro-31-8220 or NF-κB inhibitor BAY11-7082 with metoprolol
or bisoprolol further decreased the pulsatile frequency and
cellular diameter of cardiomyocytes cultured in high glucose. |
Table IV.
Combined use of protein kinase C
inhibitor Ro-31-8220 or NF-κB inhibitor BAY11-7082 with metoprolol
or bisoprolol further decreased the pulsatile frequency and
cellular diameter of cardiomyocytes cultured in high glucose.
| Treatment | Pulsatile frequency
(bpm) | Diameter (µm) |
|---|
| Low glucose (5.5
mmol/l) | 57.22±1.69 | 18.16±0.78 |
| High glucose (25.5
mmol/l) |
69.85±1.87a |
25.02±0.19a |
| High glucose +
metoprolol (10 µmol/l) |
62.23±1.58b |
20.71±0.17b |
| High glucose +
bisoprolol (200 nmol/l) |
61.75±1.63b |
20.44±0.36b |
| High glucose +
metoprolol+Ro-31-8220 |
60.29±1.16b,c |
19.03±0.55b,c |
| High glucose +
metoprolol+BAY11-7082 |
60.13±1.79b,c |
19.14±0.41b,c |
| High glucose +
bisoprolol+Ro-31-8220 |
59.68±1.82b,d |
18.76±0.63b,d |
| High glucose +
bisoprolol+BAY11-7082 |
59.56±1.67b,d |
18.62±0.38b,d |
Combined use of PKC inhibitor and
NF-κB inhibitor with metoprolol or bisoprolol further decreases
total protein content and [3H]-leucine incorporation of
HG-treated cardiomyocytes
In the HG-treated cardiomyocytes, the protein
content and [3H]-leucine incorporation were
significantly upregulated compared with LG-treated samples. The
present results suggested that reatments with metoprolol or
bisoprolol could inhibit this upregulation in HG-treated samples.
Moreover, when combining the use of the PKC inhibitor and NF-κB
inhibitor with metoprolol or bisoprolol, the protein content and
[3H]-leucine incorporation could be further
downregulated compared with metoprolol or bisoprolol treatment
alone (P<0.05; Table V).
 | Table V.Combined use of protein kinase C
inhibitor Ro-31-8220 or NF-κB inhibitor BAY11-7082 with metoprolol
or bisoprolol further decreased the total protein content and
[3H]-leucine incorporation of cardiomyocytes cultured in
high glucose. |
Table V.
Combined use of protein kinase C
inhibitor Ro-31-8220 or NF-κB inhibitor BAY11-7082 with metoprolol
or bisoprolol further decreased the total protein content and
[3H]-leucine incorporation of cardiomyocytes cultured in
high glucose.
| Treatment | Protein
(µg/well) |
[3H]-leucine incorporation
(cpm/well) |
|---|
| Low glucose (5.5
mmol/l) | 30.74±3.25 | 984.52±74.48 |
| High glucose (25.5
mmol/l) |
58.54±4.87a |
1,578.93±69.23a |
| High glucose +
metoprolol (10 µmol/l) |
40.35±4.66b |
1,201.67±76.53b |
| High glucose +
bisoprolol (200 nmol/l) |
40.16±3.32b |
1,189.38±68.39b |
| High glucose +
metoprolol+Ro-31-8220 |
37.61±3.91b,c |
1,065.36±54.64b,c |
| High glucose +
metoprolol+BAY11-7082 |
36.82±3.58b,c |
1,052.81±62.71b,c |
| High glucose +
bisoprolol+Ro-31-8220 |
37.45±3.34b,d |
1,071.27±66.42b,d |
| High glucose +
bisoprolol+BAY11-7082 |
36.78±3.79b,d |
1,045.65±59.16b,d |
Discussion
Patients with diabetic cardiomyopathy generally
display progressive development of impaired systolic and diastolic
dysfunction and heart failure (2).
However, the exact underlying pathological mechanism remains to be
further elucidated. The DAG-PKC signaling pathway plays an
important role in the initiation and development of diabetic
cardiomyopathy. A number of studies investigated the impact of HG
on the structural and functional alterations in cardiac myocytes.
The classical phenotype in diabetic cardiomyopathy is characterized
by increased pulsatile frequency, myocardial size and protein
content and synthesis (28). The
present study mainly explored the protective effects of two types
of beta-blockers, metoprolol and bisoprolol, on such hypertrophic
changes and the associated signaling pathway.
Previous studies found that diabetic rats showed a
significantly increased probability of developing cardiac
hypertrophy compared with normal rats (28,29),
which was regulated by the PKC/NF-κB/c-fos signaling pathway. In
addition, another previous study found that overexpression of PKC
activated NF-κB and promoted the expression of c-fos, resulting in
cardiac hypertrophy and decreased contractility of cardiomyocytes,
consequently leading to heart failure (30). However, upon treatment with
pharmacological PKC inhibitors, such hypertrophic alteration in
cardiomyocytes were reversed. In addition, cardiac hypertrophy in
ischemic and heart failure models usually resulted in the
activation of NF-κB and TNF-α (31,32).
Hence, these factors may play a role in the pathological
development of diabetic cardiomyopathy.
In the present study, the expression levels of
PKC-α, PKC-β2, NF-κB, TNF-α and c-fos of cardiomyocytes
cultured in HG were all upregulated compared with LG. The increased
pulsatile frequency and hypertrophic changes, including cellular
diameter, cell surface area, protein content and synthesis and mRNA
levels of ANP, α-MHC, β-MHC of cardiomyocytes treated with HG were
reversed upon treatment with the PKC inhibitor Ro-31-8220. The
expression and activity of PKC-α and PKC-β2
significantly decreased, alongside reduced expression of NF-κB,
TNF-α, and c-fos levels. Although it was reported that Ro-31-8220
inhibited glycogen synthase kinase-3 (33), there was no doubt that PKC inhibition
was its principal effect as RO-31-8220 is widely accepted as a PKC
inhibitor (34). The present results
suggested that the pricinple effect of Ro-31-8220 may be mediated
by PKC inhibition rather than GSK-3 inhibition, as shown by the
suppression of the total and phosphrylated PKC-α and
PKCβ2. Likewise, following treatment with the NF-κB
inhibitor BAY11-7082, the HG-induced increase in pulsatile
frequency and cardiac hypertrophy was reversed, and a significant
decrease in expression of NF-κB, TNF-α, and c-fos levels was
observed.
NF-κB is a key transcription factor regulating
inflammatory responses and the expression of hyperglycemic stress
related immediate early genes (35).
Previous studies showed that NF-κB regulated several signal
transduction pathways in cardiomyocytes under stimulation with HG
(36,37). Hence, blocking NF-κB might attenuate
HG-induced cardiac hypertrophy (38)
in cardiac hypertrophy models. TNF-α is a cytokine associated with
NF-κB. As a contributor of cardiac dysfunction, elevated TNF-α
level could trigger NF-κB translocation to the nucleus, allowing
NF-κB to promote transcription of TNF-α (39,40).
c-fos is a proto-oncogene, whose activation could
induce the enlargement of cardiomyocytes and increased protein
content. TNF-α stimulates the expression of c-fos as an adaptive
response to cardiac dysfunction, such as cardiac hypertrophy and
ischemic heart disease (41,42). Moreover, previous studies
demonstrated that c-fos is also regulated by PKC to participate in
inendothelin-1-induced proliferation of neonatal cardiomyocytes
(43,44). A previous study also revealed that
the PKC/c-fos pathway was involved in HG-induced cardiac
hypertrophy (11).
Beta-blockers are well studied and play a
cardioprotective role in ameliorating cardiac dysfunction in rats
with diabetic cardiomyopathy (45–47).
Since numerous types of beta-blockers with different effects on
glucose control are available at present, it would be beneficial
for DM patients to select the appropriate beta-blocker to prevent
or delay cardiovascular complication (48). According to evidence-based medicine,
metoprolol and bisoprolol are two types of highly selective
beta-blockers widely used in clinical practice (49). Autoantibodies against the β1 receptor
could be a predictor for left ventricular hypertrophy for T2DM
patients with hypertension (50,51).
Treatment with metoprolol could inhibit intrinsic damage to the
heart during diabetic cardiomyopathy (52). Several studies indicated that use of
beta-blockers, such as bisoprolol, did not aggravate glycemic
control, lipid profile or albuminuria status in T2DM patients with
SHF (53,54).
To explore whether the protective effects of
metoprolol and bisoprolol were associated with the PKC signal
transduction pathway, expression and activation of two PKC isoforms
(PKC-α and PKC-β2) were detected in cardiomyocytes
cultured with HG. Metoprolol and bisoprolol were found to decrease
cellular pulsatile frequency and improve cardiac hypertrophy, and
decrease expression and activity of PKC-α. These changes were more
significant when cultures were treated with a higher dose of
metoprolol and bisoprolol. Meanwhile, the expression of
p-PKC-β2 decreased when cells were treated with higher
doses of metoprolol and bisoprolol compared with cells treated with
HG. However, when treated with a lower dose of metoprolol and
bisoprolol, the expression of p-PKC-β2 in cardiomyocytes
resulted in no significant change when compared with HG-treated
samples, while the expression and activation of PKC-α of HG
cultured cardiomyocytes were significantly inhibited by treatment
with metoprolol or bisoprolol compared with those cultured with HG
alone. Therefore, the protective role of metoprolol and bisoprolol
on HG-induced hypertrophy was mainly conducted by inhibition of
PKC-α. Expression of NF-κB, TNF-α and c-fos also significantly
decreased when treated with metoprolol and bisoprolol. The present
results suggested that high dose of bisoprolol could further
inhibit the decrease in TNF-α expression leve in the low bisoprolol
dose group. In addition, higher doses of metoprolol and bisoprolol
showed a significant suppressive effect on c-fos expression
compared with low doses. Collectively, metoprolol and bisoprolol
provided a protective role against diabetic cardiomyopathy via the
PKC-induced NF-κB/TNF-α/c-fos signal transduction pathway.
In the current study, high dose of metoprolol and
bisoprolol inhibited c-fos expression to a lower degree compared
with PKC or NF-κB inhibitor, although the differences were not
significant. The present data suggested that beta-blockers may have
other targets other than PKC and NF-κB. One explanation for the
results is the involvement of another important cardiac
hypertrophic pathway in cultured cardiomyocytes, the PKC-δ/protein
kinase D (PKD)/histone deacetylases (HDAC) pathway. PKD, which can
be phosphorylated by PKC-δ, regulates the ability of cardiomyocyte
growth and contractility through phosphorylation of proteins such
as class IIa HDACs (55). Metoprolol
and bisoprolol may inhibit the expression of c-fos through
regulating the PKC-δ signaling pathways. Although there is little
evidence of the effects of signal transduction pathways of
beta-blockers on the expression of c-fos, one study showed that
bisoprolol, especially at high doses, could increase the survival
rate of hypertensive diastolic heart failure model of rats, at
least partly through the alleviation of inflammatory changes
(interleukin-1β, transforming growth factor-β1 and monocyte
chemoattractant protein-1) and oxidative stress (reactive oxygen
species production and nicotinamide adenine dinucleotide phosphate
oxidase activity) (52). Moreover,
isoproterenol (ISO)-induced heart failure could be inhibited by a
Rho-associated protein kinase (ROCK) inhibitor, fasudil, which
suppressed isoproterenol-induced JNK activation, translocation of
ERK to the nucleus, and increased expression of c-fos and c-jun
through RhoA/ROCK. Consequently, those changes could suppress
ISO-induced cardiac hypertrophy and ventricular remodeling in rat
models (56). Since ISO-induced
heart dysfunction was associated with RhoA/ROCK and its downstream
signaling pathways, beta-blockers may also have protective effects
on cardiomyocytes through similar signaling pathways, although this
warrants further studies.
Activation of PI3K-Akt and ERK pathways may also
contribute to the cardiac protective effects of metoprolol
(57). Therefore, beta-blockers may
have other targets than PKC and NF-κB, and ERK and inflammatory
cytokines could also cause cardiomyocyte hypertrophy. Fujioka et
al (58) proposed that the use
of another beta-blocker, propranolol, could promote post-hypoxic
contractile and metabolic recovery via non-beta-adrenoreceptors in
ischemia-reperfusion rat hearts. Hence, the role of such
non-beta-adrenorecepors activated by beta-blockers could be further
investigated in diabetic cardiomyopathy. In the present study,
combined use of PKC inhibitor Ro-31-8220 or NF-κB inhibitor
BAY11-7082 with metoprolol further decreased the cellular pulsatile
frequency, cellular diameter, cell surface area, total protein
content and [3H]-leucine incorporation of cardiomyocytes
cultured with HG. The same result was observed when bisoprolol was
combined with Ro-31-8220 or BAY11-7082. Combined use of
beta-blockers with PKC or NF-κB inhibitor attenuated cardiac
hypertrophy caused by HG. Beta-blockers may have targets other than
PKC and NF-κB that could also inhibit the expression of c-fos. This
hypothesis supports the observation that high doses of metoprolol
and bisoprolol may inhibit c-fos expression to a lower degree than
PKC or NF-κB inhibitors, although the differences were not
statistically significant in the present study.
In conclusion, the data presented depicted that
hyperglycemia could activate the PKC/NF-κB/TNF-α/c-fos signal
transduction pathway in diabetic cardiomyopathy. The protective
role of metoprolol and bisoprolol could significantly reverse
cardiac dysfunction and hypertrophy. Future studies can be
performed to identify other targets of beta-blockers and non-beta
receptor-mediated effects of beta-blockers in diabetic
cardiomyocytes.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural
Science Foundation of China (grant nos. 81500212 and 81800212) and
Zhejiang Natural Science Foundation (grant nos. LY18H020007 and
LQ16H020001).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
MW, GF and WZ designed the research. MW, QL, YW and
ZL performed the research. WZ contributed new reagents or analytic
tools. MW, LZ, YL and WZ analyzed the data. MW, QL and YL wrote the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All procedures were approved by the Ethics Committee
for the Use of Experimental Animals in Zhejiang University. All the
experiments followed the instructions for animal care and usage
provided by Zheijiang University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Whiting DR, Guariguata L, Weil C and Shaw
J: IDF diabetes atlas: Global estimates of the prevalence of
diabetes for 2011 and 2030. Diabetes Res Clin Pract. 94:311–321.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jia G, Whaley-Connell A and Sowers JR:
Diabetic cardiomyopathy: A hyperglycaemia- and
insulin-resistance-induced heart disease. Diabetologia. 61:21–28.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Suhara T, Baba Y, Shimada BK, Higa JK and
Matsui T: The mTOR signaling pathway in myocardial dysfunction in
type 2 diabetes mellitus. Curr Diab Rep. 17:382017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gao JR, Qin XJ, Fang ZH, Li-Sha n, Han LP,
Hui-Jian g, Guo MF and Jiang NN: To explore the pathogenesis of
vascular lesion of type 2 diabetes mellitus based on the PI3K/Akt
signaling pathway. J Diabetes Res. 2019:46509062019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang WK, Lu QH, Wang X, Wang B, Wang J,
Gong HP, Wang L, Li H and Du YM: Ulinastatin attenuates
diabetes-induced cardiac dysfunction by the inhibition of
inflammation and apoptosis. Exp Ther Med. 14:2497–2504. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brownlee M: The pathobiology of diabetic
complications: A unifying mechanism. Diabetes. 54:1615–1625. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ferreira JC, Brum PC and Mochly-Rosen D:
βIIPKC and εPKC isozymes as potential pharmacological targets in
cardiac hypertrophy and heart failure. J Mol Cell Cardiol.
51:479–484. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Churchill EN and Mochly-Rosen D: The roles
of PKCdelta and epsilon isoenzymes in the regulation of myocardial
ischaemia/reperfusion injury. Biochem Soc Trans. 35:1040–1042.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Weng LQ, Zhang WB, Ye Y, Yin PP, Yuan J,
Wang XX, Kang L, Jiang SS, You JY, Wu J, et al: Aliskiren
ameliorates pressure overload-induced heart hypertrophy and
fibrosis in mice. Acta Pharmacol Sin. 35:1005–1014. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Das Evcimen N and King GL: The role of
protein kinase C activation and the vascular complications of
diabetes. Pharmacol Res. 55:498–510. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Min W, Bin ZW, Quan ZB, Hui ZJ and Sheng
FG: The signal transduction pathway of PKC/NF-kappa B/c-fos may be
involved in the influence of high glucose on the cardiomyocytes of
neonatal rats. Cardiovasc Diabetol. 8:82009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Norozi K: The role of beta-blocker in
heart failure in adults with congenital heart disease. Rev Recent
Clin Trials. 9:64–67. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chia N, Fulcher J and Keech A:
Beta-blocker, angiotensin-converting enzyme inhibitor/angiotensin
receptor blocker, nitrate-hydralazine, diuretics, aldosterone
antagonist, ivabradine, devices and digoxin [BANDAID(2)]: An
evidence-based mnemonic for the treatment of systolic heart
failure. Intern Med J. 46:653–662. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Spoladore R, Fragasso G, Perseghin G, De
Cobelli F, Esposito A, Maranta F, Calori G, Locatelli M, Lattuada
G, Scifo P, et al: Beneficial effects of beta-blockers on left
ventricular function and cellular energy reserve in patients with
heart failure. Fundam Clin Pharmacol. 27:455–464. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tsujimoto T, Sugiyama T, Shapiro MF, Noda
M and Kajio H: Risk of cardiovascular events in patients with
diabetes mellitus on beta-blockers. Hypertension. 70:103–110. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bakris GL, Fonseca V, Katholi RE, McGill
JB, Messerli FH, Phillips RA, Raskin P, Wright JT Jr, Oakes R,
Lukas MA, et al: Metabolic effects of carvedilol vs. metoprolol in
patients with type 2 diabetes mellitus and hypertension: A
randomized controlled trial. JAMA. 292:2227–2236. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kveiborg B, Hermann TS, Major-Pedersen A,
Christiansen B, Rask-Madsen C, Raunsø J, Køber L, Torp-Pedersen C
and Dominguez H: Metoprolol compared to carvedilol deteriorates
insulin-stimulated endothelial function in patients with type 2
diabetes-a randomized study. Cardiovasc Diabetol. 9:212010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Haas SJ, Vos T, Gilbert RE and Krum H: Are
beta-blockers as efficacious in patients with diabetes mellitus as
in patients without diabetes mellitus who have chronic heart
failure? A meta-analysis of large-scale clinical trials. Am Heart
J. 146:848–853. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rengo G, Cannavo A, Liccardo D, Zincarelli
C, de Lucia C, Pagano G, Komici K, Parisi V, Scala O, Agresta A, et
al: Vascular endothelial growth factor blockade prevents the
beneficial effects of beta-blocker therapy on cardiac function,
angiogenesis, and remodeling in heart failure. Circ Heart Fail.
6:1259–1267. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li X, Zhang X, Wang T, Sun C, Jin T, Yan
H, Zhang J, Li X, Geng T, Chen C, et al: Regulation by bisoprolol
for cardiac microRNA expression in a rat volume-overload heart
failure model. J Nanosci Nanotechnol. 13:5267–5275. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cheng K, Wei MQ, Jia GL, Wang HC, Luan RH,
Guo WY, Li WJ, Zong XJ and Zhou X: Effects of metoprolol and small
intestine RNA on marrow-derived endothelial progenitor cells
applied for autograft transplantation in heart disease. Eur Rev Med
Pharmacol Sci. 18:1666–1673. 2014.PubMed/NCBI
|
|
22
|
Garcia-Egido A, Andrey JL, Puerto JL,
Aranda RM, Pedrosa MJ, López-Sáez JB, Rosety M and Gomez F:
Beta-blocker therapy and prognosis of heart failure patients with
new-onset diabetes mellitus. Int J Clin Pract. 69:550–559. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Couto M and Cates C: Laboratory guidelines
for animal care. Methods Mol Biol. 1920:407–430. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shen YX, Xiao K, Liang P, Ma YW and Huang
X: Improvement on the modified Lowry method against interference of
divalent cations in soluble protein measurement. Appl Microbiol
Biotechnol. 97:4167–4178. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lekawanvijit S, Adrahtas A, Kelly DJ,
Kompa AR, Wang BH and Krum H: Does indoxyl sulfate, a uraemic
toxin, have direct effects on cardiac fibroblasts and myocytes? Eur
Heart J. 31:1771–1779. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Luo JD, Xie F, Zhang WW, Ma XD, Guan JX
and Chen X: Simvastatin inhibits noradrenaline-induced hypertrophy
of cultured neonatal rat cardiomyocytes. Br J Pharmacol.
132:159–164. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nishiga M, Horie T, Kuwabara Y, Nagao K,
Baba O, Nakao T, Nishino T, Hakuno D, Nakashima Y, Nishi H, et al:
MicroRNA-33 controls adaptive fibrotic response in the remodeling
heart by preserving lipid raft cholesterol. Circ Res. 120:835–847.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang M, Zhang WB, Zhu JH, Fu GS and Zhou
BQ: Breviscapine ameliorates hypertrophy of cardiomyocytes induced
by high glucose in diabetic rats via the PKC signaling pathway.
Acta Pharmacol Sin. 30:1081–1091. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang M, Zhang WB, Zhu JH, Fu GS and Zhou
BQ: Breviscapine ameliorates cardiac dysfunction and regulates the
myocardial Ca(2+)-cycling proteins in streptozotocin-induced
diabetic rats. Acta Diabetol. 47 (Suppl 1):S209–S218. 2010.
View Article : Google Scholar
|
|
30
|
Song X, Qian X, Shen M, Jiang R, Wagner
MB, Ding G, Chen G and Shen B: Protein kinase C promotes cardiac
fibrosis and heart failure by modulating galectin-3 expression.
Biochim Biophys Acta. 1853:513–521. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kawamura N, Kubota T, Kawano S, Monden Y,
Feldman AM, Tsutsui H, Takeshita A and Sunagawa K: Blockade of
NF-kappaB improves cardiac function and survival without affecting
inflammation in TNF-alpha-induced cardiomyopathy. Cardiovasc Res.
66:520–529. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lu J, Liu F, Chen F, Jin Y, Chen H, Liu D
and Cui W: Amlodipine and atorvastatin improve ventricular
hypertrophy and diastolic function via inhibiting TNF-α, IL-1β and
NF-κB inflammatory cytokine networks in elderly spontaneously
hypertensive rats. Biomed Pharmacother. 83:330–339. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hers I, Tavaré JM and Denton RM: The
protein kinase C inhibitors bisindolylmaleimide I (GF 109203×) and
IX (Ro 31-8220) are potent inhibitors of glycogen synthase kinase-3
activity. FEBS Lett. 460:433–436. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Catley MC, Cambridge LM, Nasuhara Y, Ito
K, Chivers JE, Beaton A, Holden NS, Bergmann MW, Barnes PJ and
Newton R: Inhibitors of protein kinase C (PKC) prevent activated
transcription: Role of events downstream of NF-kappaB DNA binding.
J Biol Chem. 279:18457–18466. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Abe J: Role of PKCs and NF-kappaB
activation in myocardial inflammation: Enemy or ally? J Mol Cell
Cardiol. 43:404–408. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Guo Y, Zhuang X, Huang Z, Zou J, Yang D,
Hu X, Du Z, Wang L and Liao X: Klotho protects the heart from
hyperglycemia-induced injury by inactivating ROS and NF-κB-mediated
inflammation both in vitro and in vivo. Biochim Biophys Acta Mol
Basis Dis. 1864:238–251. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ren XM, Zuo GF, Wu W, Luo J, Ye P, Chen SL
and Hu ZY: Atorvastatin alleviates experimental diabetic
cardiomyopathy by regulating the GSK-3β-PP2Ac-NF-κB signaling axis.
PLoS One. 11:e01667402016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Freund C, Schmidt-Ullrich R, Baurand A,
Dunger S, Schneider W, Loser P, El-Jamali A, Dietz R, Scheidereit C
and Bergmann MW: Requirement of nuclear factor-kappaB in
angiotensin II- and isoproterenol-induced cardiac hypertrophy in
vivo. Circulation. 111:2319–2325. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Higuchi Y, Otsu K, Nishida K, Hirotani S,
Nakayama H, Yamaguchi O, Matsumura Y, Ueno H, Tada M and Hori M:
Involvement of reactive oxygen species-mediated NF-kappa B
activation in TNF-alpha-induced cardiomyocyte hypertrophy. J Mol
Cell Cardiol. 34:233–240. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Higuchi Y, Chan TO, Brown MA, Zhang J,
DeGeorge BR Jr, Funakoshi H, Gibson G, McTiernan CF, Kubota T,
Jones WK and Feldman AM: Cardioprotection afforded by NF-kappaB
ablation is associated with activation of Akt in mice
overexpressing TNF-alpha. Am J Physiol Heart Circ Physiol.
290:H590–H598. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hattori Y, Hattori S, Akimoto K, Nishikimi
T, Suzuki K, Matsuoka H and Kasai K: Globular adiponectin activates
nuclear factor-kappaB and activating protein-1 and enhances
angiotensin II-induced proliferation in cardiac fibroblasts.
Diabetes. 56:804–808. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Dawn B, Guo Y, Rezazadeh A, Wang OL, Stein
AB, Hunt G, Varma J, Xuan YT, Wu WJ, Tan W, et al: Tumor necrosis
factor-alpha does not modulate ischemia/reperfusion injury in naive
myocardium but is essential for the development of late
preconditioning. J Mol Cell Cardiol. 37:51–61. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhu XX, Niu XL, Chen DZ, Zhou XD, Pei JM,
Zhu MZ, Guo J, Zhu XL and Wang WQ: Inhibitory effects of
rosiglitazone against endothelin-1-induced proliferation of rat
cardiac myocytes: The role of PKC-c-fos pathway. Nan Fang Yi Ke Da
Xue Xue Bao. 28:1056–1060. 2008.(In Chinese). PubMed/NCBI
|
|
44
|
Maniar R, Pecherskaya A, Ila R and Solem
M: PKC alpha-dependent regulation of the IGF1 receptor in adult and
embryonic rat cardiomyocytes. Mol Cell Biochem. 275:15–24. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rizzi E, Guimaraes DA, Ceron CS, Prado CM,
Pinheiro LC, Martins-Oliveira A, Gerlach RF and Tanus-Santos JE:
β1-Adrenergic blockers exert antioxidant effects, reduce matrix
metalloproteinase activity, and improve renovascular
hypertension-induced cardiac hypertrophy. Free Radic Biol Med.
73:308–317. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Amanfu RK and Saucerman JJ: Modeling the
effects of β1-adrenergic receptor blockers and polymorphisms on
cardiac myocyte Ca2+ handling. Mol Pharmacol. 86:222–230. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sharma V, Dhillon P, Wambolt R, Parsons H,
Brownsey R, Allard MF and McNeill JH: Metoprolol improves cardiac
function and modulates cardiac metabolism in the
streptozotocin-diabetic rat. Am J Physiol Heart Circ Physiol.
294:H1609–H1620. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chen RJ, Chu H and Tsai LW: Impact of
beta-blocker initiation timing on mortality risk in patients with
diabetes mellitus undergoing noncardiac surgery: A nationwide
population-based cohort study. J Am Heart Assoc. 6:e0043922017.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Fröhlich H, Torres L, Täger T, Schellberg
D, Corletto A, Kazmi S, Goode K, Grundtvig M, Hole T, Katus HA, et
al: Bisoprolol compared with carvedilol and metoprolol succinate in
the treatment of patients with chronic heart failure. Clin Res
Cardiol. 106:711–721. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Arnold SV, Spertus JA, Lipska KJ, Lanfear
DE, Tang F, Grodzinsky A, McGuire DK, Gore MO, Goyal A, Maddox TM
and Kosiborod M: Type of β-blocker use among patients with versus
without diabetes after myocardial infarction. Am Heart J.
168:273–279.e1. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zhao L, Xu C and Xu J: Autoantibodies
against β1 receptor and AT1 receptor in type 2 diabetes patients
with left ventricular dilatation. Cardiology. 129:191–196. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Saran V, Sharma V, Wambolt R, Yuen VG,
Allard M and McNeill JH: Combined metoprolol and ascorbic acid
treatment prevents intrinsic damage to the heart during diabetic
cardiomyopathy. Can J Physiol Pharmacol. 92:827–837. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wai B, Kearney LG, Hare DL, Ord M, Burrell
LM and Srivastava PM: Beta blocker use in subjects with type 2
diabetes mellitus and systolic heart failure does not worsen
glycaemic control. Cardiovasc Diabetol. 11:142012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Hirst JA, Farmer AJ, Feakins BG, Aronson
JK and Stevens RJ: Quantifying the effects of diuretics and
β-adrenoceptor blockers on glycaemic control in diabetes mellitus-a
systematic review and meta-analysis. Br J Clin Pharmacol.
79:733–743. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Phan D, Stratton MS, Huynh QK and McKinsey
TA: A novel protein kinase C target site in protein kinase D is
phosphorylated in response to signals for cardiac hypertrophy.
Biochem Biophys Res Commun. 411:335–341. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wang N, Guan P, Zhang JP, Li YQ, Chang YZ,
Shi ZH, Wang FY and Chu L: Fasudil hydrochloride hydrate, a
Rho-kinase inhibitor, suppresses isoproterenol-induced heart
failure in rats via JNK and ERK1/2 pathways. J Cell Biochem.
112:1920–1929. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kovacs K, Hanto K, Bognar Z, Tapodi A,
Bognar E, Kiss GN, Szabo A, Rappai G, Kiss T, Sumegi B and Gallyas
F Jr: Prevalent role of Akt and ERK activation in cardioprotective
effect of Ca(2+) channel- and beta-adrenergic receptor blockers.
Mol Cell Biochem. 321:155–164. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Fujioka H, Yoshihara S, Tanaka T, Fukumoto
T, Kuroiwa A, Tanonaka K, Hayashi M and Takeo S: Enhancement of
post-hypoxic contractile and metabolic recovery of perfused rat
hearts by dl-propranolol: Possible involvement of non-beta-receptor
mediated activity. J Mol Cell Cardiol. 23:949–962. 1991. View Article : Google Scholar : PubMed/NCBI
|