Introduction
Angiotensin-converting enzyme inhibitors (ACEIs) are
known as a potent antihypertensive medication since early 1980s,
representing an important and largely prescribed group of
pharmacological compounds (1). They
have been extensively evaluated in clinical trials, demonstrating
significant reduction of morbidity and mortality of patients with
cardiovascular diseases, mainly high blood pressure, myocardial
infarction, heart failure and stroke. It was found that ACEIs
represented the most commonly used antihypertensive medication in
the United States since 2002(2).
Besides their beneficial effects and a general good safety profile,
it was proven that ACEIs might also induce adverse effects in some
patients, most notably chronic cough and angioedema (AE) (3). The occurrence rate of adverse events to
ACEIs is low, with estimated incidence of angioedema between
0.1-2.2%, but the number of suffering patients is relatively high,
since ACEIs is one of the most frequently prescribed classes of
medication worldwide (4,5). Besides angioedema and cough, other less
frequent adverse reactions were mentioned, such as hyperkalemia,
hypotension, liver dysfunction and bone marrow depression.
Adverse reactions induced by ACEIs are secondary to
interference with the renin-angiotensin-aldosterone system (RAAS),
which is responsible for the development and progression of
cardiovascular and renal disorders. ACEIs block
angiotensin-converting enzyme (ACE) activity, decrease conversion
of angiotensin I to angiotensin II and also impact the degradation
metabolism of bradykinin, leading to its increasing accumulation.
The increased level of bradykinin is responsible for cough and
angioedema in some patients, who become intolerant to this class of
medication. Bradykinin is a potent local mediator of vasodilatation
and vascular permeability, with very short half-life, being rapidly
metabolized by peptidases in tissues and serum (6). The mechanism of ACEIs intolerance is
not fully understood and genetic and/or environmental risk factors
are considered to play an important role (7). It was proven that black race and female
sex are genetic risk factors, while other possible mixed genetic
and environmental risk factors are age over 65, history of drug
rash, concomitant seasonal or persistent allergic diseases,
concomitant medication with nonsteroidal anti-inflammatory drugs
(NSAIDs) or immunosuppressive agents and smoking, while diabetes
seems to be a protective factor (8,9).
Angioedema is a potentially severe, life-threatening
adverse event associated with ACEI medication and the reported
number of affected patients is accepted to be continuously
increasing (5). Retrospective
studies from the beginning of ACEI therapy mentioned that
ACEI-induced angioedema (ACEI-AE) represented 30-40% of the
patients with angioedema presenting to emergency departments (ED)
(10). Despite long experience with
this class of medication, ACEIs-AE is still underestimated and
mislabeled as allergic AE or urticaria by many physicians and its
potential for severe outcome is ignored. Recent publications on
angioedema update diagnosis criteria and management recommendations
for both hereditary and acquired angioedema, including mechanisms
and characteristics of ACEI-induced angioedema (11). There are two main forms of acquired
angiodema in clinical practice: Mast cell-mediated and
bradykinin-mediated angioedema (12,13).
The aim of our study was to evaluate clinical
pattern, risk factors, outcome and general management of
ACEI-induced angioedema in a cohort of patients addressed for
allergist evaluation in one university hospital from Romania,
during a period of 32 months.
Materials and methods
A retrospective study of hospital data was performed
of the patients addressed for allergy consultation to the
hospital-based Allergy center during a 32 months period, between
January 2017 to August 2019. The following data were recorded from
the patients files: Age at hospital presentation, sex, indication
for ACEIs therapy, types of ACE inhibitors used, treatment duration
before occurrence of the first AE episode, AE localization,
frequency and recurrence rate, concomitant diseases and medication,
allergist recommendations and management. The study group consisted
of 50 patients who presented for angioedema during the study
period, being treated with ACEIs for variable time periods
previously. The study group represented 56.81% from the total
number of 88 patients addressed to the department for angioedema
without urticaria, during the same period of time. The patients
were aged between 45-83 years, with a mean age of 63 years and the
sex distribution showed a 64% female predominance. From the total
study group of 50 patients, 45 (90%) were treated with ACEIs for
hypertension, 4 patients were treated for chronic heart failure and
one patient had ischemic heart disease associated with chronic
kidney disease. Patients characteristics in terms of age, sex,
hypertension stage and concomitant diseases are showed in Table I. The patients signed an informed
consent for publication of their health data
 | Table IPatients characteristics. |
Table I
Patients characteristics.
| Mean | 63 years |
|---|
| Age |
|
<65
years | 27 pts (54%) |
|
≥65
years | 23 pts (46%) |
| Sex |
|
Female | 32 pts (64%) |
|
Male | 18 pts (36%) |
| Hypertension
stage |
|
Stage
II | 14 pts (28%) |
|
Stage
III | 36 pts (72%) |
| Concomitant
diseases |
|
Diabetes
mellitus type II | 11 pts (22%) |
|
Chronic
intermittent urticaria | 16 pts (30%) |
|
COPD | 2 pts (4%) |
|
Autoimmune
thyroiditis | 6 pts (12%) |
|
Degenerative
osteoarticular disease | 12 pts (24%) |
Results
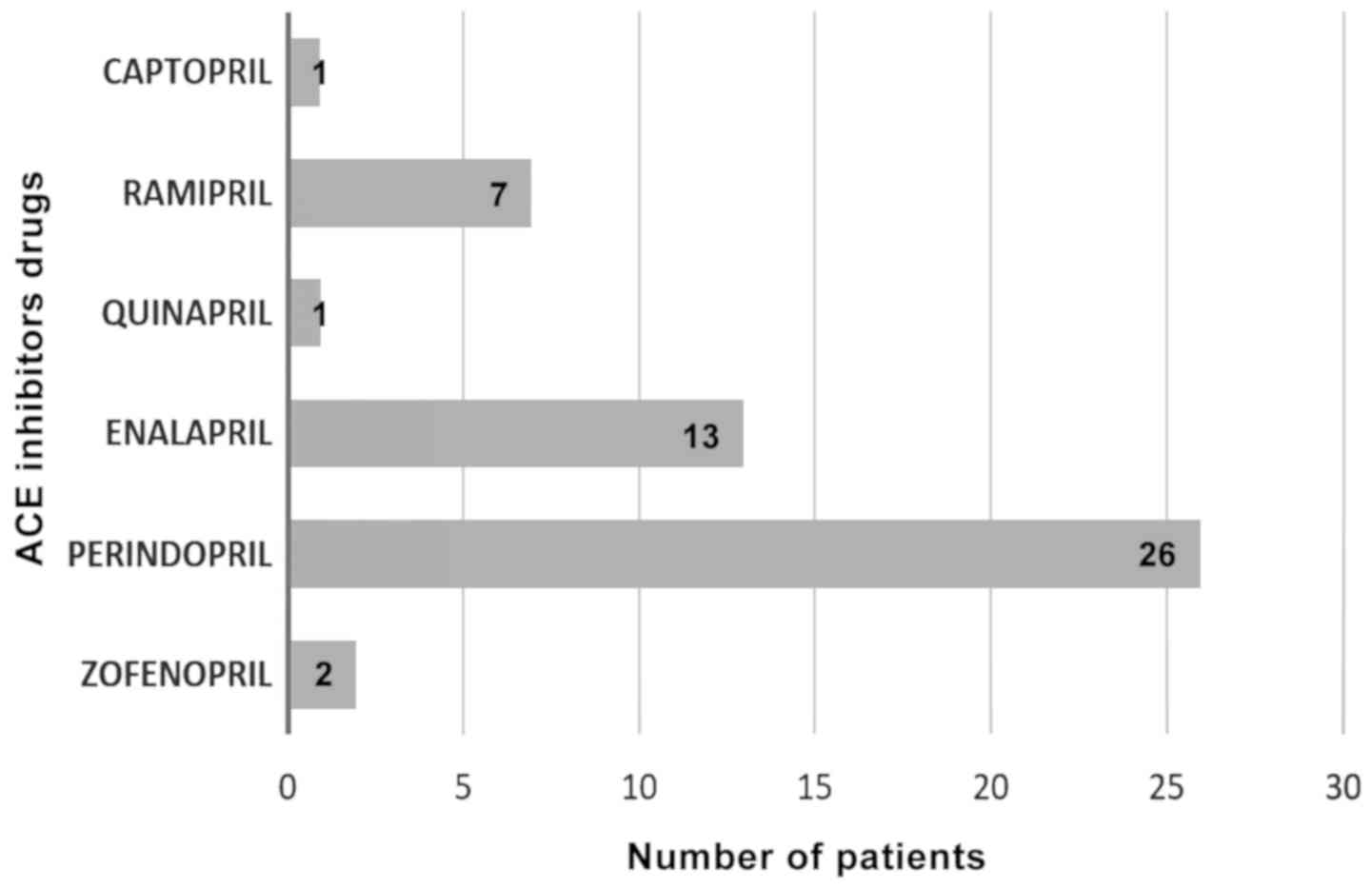
In total, 34 patients out of 50 (68%) received ACEIs
in monotherapy and 16 patients used fixed dose combinations with
ACE inhibitors. The most frequently used pharmacological substances
were perindopril in 52% of patients and enalapril in 26% (Fig. 1).
Double or triple combinations with ACEIs were:
Perindopril plus indapamide/perindopril plus amlodipine/perindopril
plus indapamide plus amlodipine.
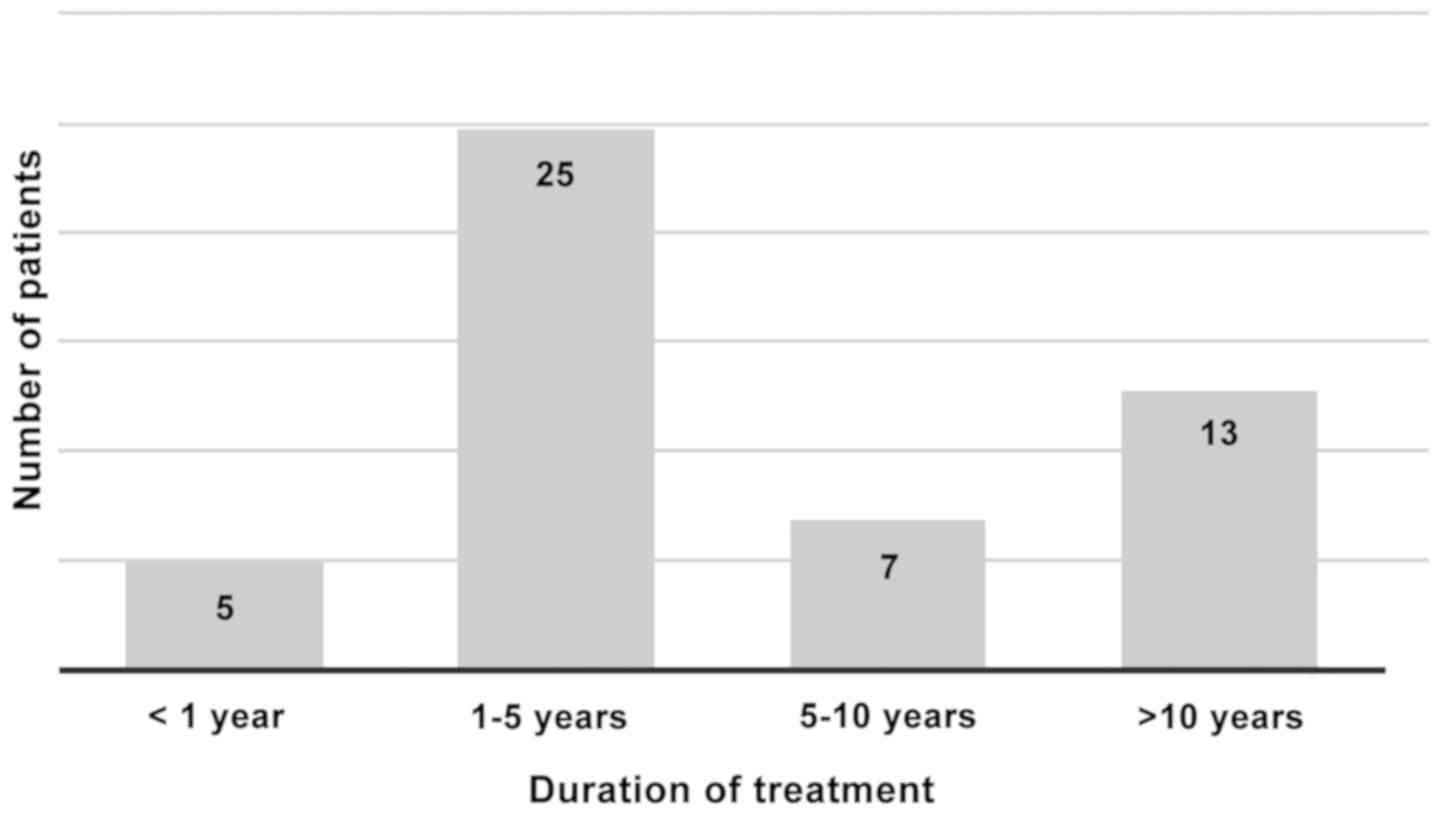
Regarding the duration of ACEI therapy before onset
of angioedema, this was less than one year in only 5 patients
(10%). In one case angioedema occurred during the same day of the
first ACEI intake in one fixed dose combination, in two cases AE
occurred during the first month and in further 2 patients AE
occurred after 7 and 12 months, respectively. In half of the
studied group, the AE episodes occurred within a time period
between 1-5 years after ACEI therapy initiation, in 7 patients
(14%) AE occurred after variable time period between 5-10 years,
while in 13 patients (26%) AE occurred after more than 10 years
(Fig. 2).
The patients continued to take ACEIs after AE onset,
except three cases who discontinued ACEIs and replaced it with
angiotensin-receptor blocker (ARBs) before hospital
presentation.
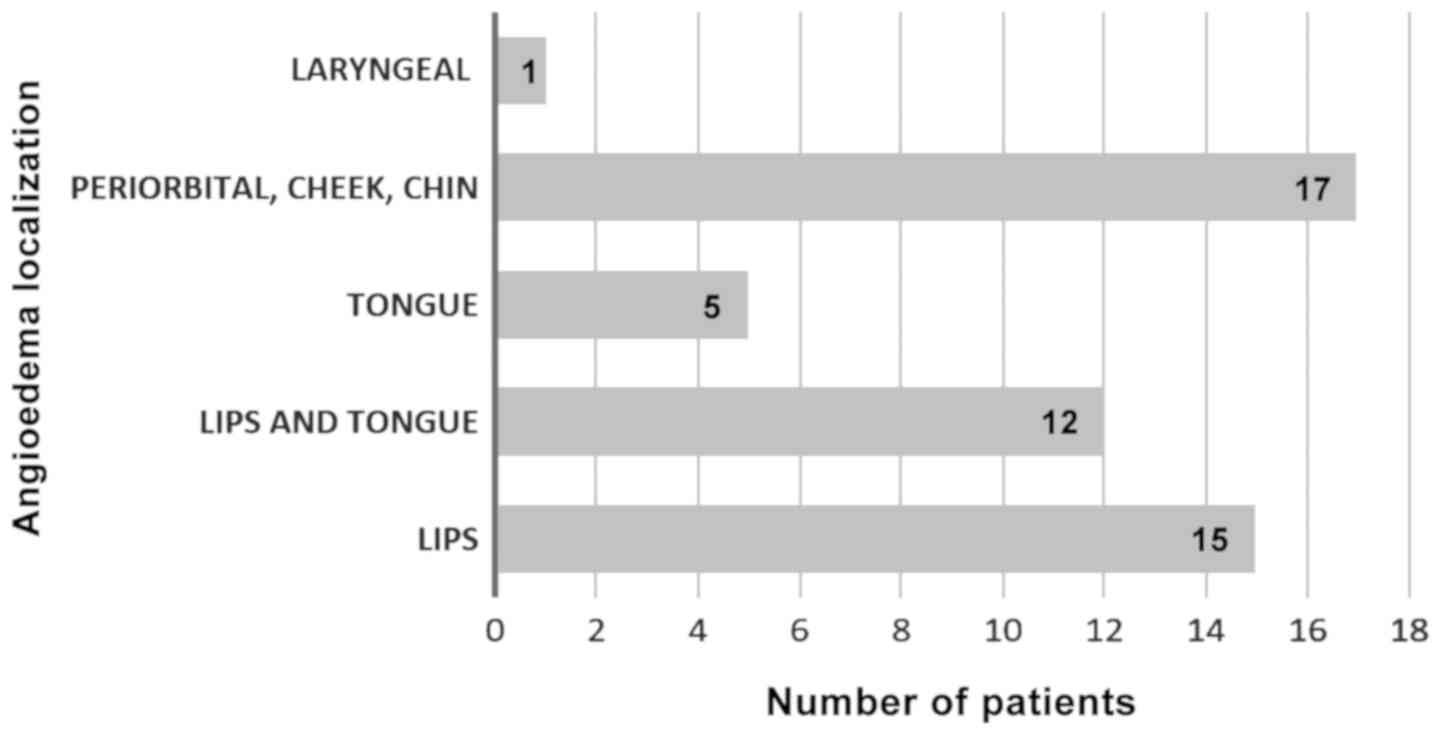
Regarding the site of angioedema, all patients
presented with facial angioedema, 15 patients had angioedema of the
lips, 12 patients had angioedema of both lips and tongue and 5
patients developed tongue angioedema alone. Laryngeal edema was
found in one case only (Fig. 3).
Regarding the frequency of AE episodes, about half
of the study group (24 patients) presented for allergist evaluation
after 1 or 2 episodes of angioedema and 26 patients presented after
3 or more AE episodes. The AE recurrence rate was lower, ~1-3 AE
episodes per year in cases of patients with longer AE history
before hospital presentation. Only 18 patients had AE at the time
of hospital presentation, including the patient with laryngeal AE
confirmed by ENT examination.
It was found that 16 patients (32%) declared history
of chronic mild urticaria, but not concomitant with AE episodes and
12 patients (24%) declared intermittent intake of non-steroidal
anti-inflammatory drugs (NSAIDs) for chronic osteo-articular
symptoms, which was coincident with AE episodes in only two
cases.
Written documentation regarding medical attitude at
ED could be found in less than half of the patients, with
confirmation of adrenaline administration in only six cases.
Allergist recommendations on hospital presentation
consisted of monitoring until complete resolution of AE, immediate
discontinuation of ACEIs and replacement with other
antihypertensive therapy, mostly diuretics and/or calcium blockers
and ARBs in five cases, evaluation of severity and possible risk
factors for AE, treatment with systemic corticosteroids and
antihistamines. The clinical outcome was good in all cases, with
almost complete remission of AE in less than 24 h. In 6 patients
(12%) recurrence of AE episodes was recorded during the first month
after discontinuation of ACEIs, but with reduced intensity and
duration.
Discussion
The study showed that ACEI-induced angioedema
represented more than half of the patients with acquired angioedema
addressed to one hospital-based Allergy center during a period of
32 months. Almost all of the patients from the study group were
referred by their general practitioners (GPs) with diagnosis of
urticaria or other skin allergies, being mislabeled as allergy in
most of the cases. It was found that a significant number of
patients had a long history of angioedema, until diagnosis
confirmation, more than 10 years after initiation of ACEI
treatment. This is a longer period comparing with published data
from the literature, reporting that ACEI-AE can appear from few
hours up to 8 years after ACEI therapy initiation (14). Previous publications showed that AE
most often occurred within the first month of ACEI treatment and
~27% of cases occurred after more than 6 months after treatment
initiation (10). Regarding the risk
factors for ACEI induced angioedema mentioned in the literature,
the results of this study showed a 64% female predominance, with
not relevant contribution of age >65 years, co-morbidities or
concomitant medication, possible due to rather small study
group.
Data from the literature showed that ACEI-related
angioedema has an increasing prevalence and severity during the
last decades, being underestimated in clinical practice (15). It was proven that failure to
recognize the relation between angioedema and ACEI treatment may
lead to unnecessary morbidity and mortality, caused by the lack of
medical provider knowledge and low frequency of this type of
reaction. A published survey of physician's knowledge, including
cardiologists, GPs and allergists, demonstrated a rather poor
awareness of the ACEI adverse reactions, with correct responses in
about half of the group (16).
Majority of the patients with angioedema are considered having food
or drug allergy and treated with antihistamines, corticosteroids
and epinephrine, despite unclear response. Difficulties in
recognizing allergies and discriminating between allergic and
intolerant patients are due to limited university or postgraduate
training of medical staff in the field of allergic diseases,
despite increasing prevalence and severity of this pathology
(17). A recent local study
investigating knowledge and attitude of GPs in the field of drug
allergies revealed a significant need for training and updated
information in allergic diseases, as well as for interdisciplinary
collaboration (18).
Angioedema without urticaria may raise significant
problems in clinical practice, mainly due to unclear etiopathology,
variable clinical presentation and potential for severe outcome
(19). The classic clinical
presentation of ACEI-AE is recurrent episodes of lips and tongue
edema, without urticaria and pruritus (20). History of atopy or concomitant
allergies are usually missing and clinical outcome is not
significantly influenced by antihistamines and corticosteroids, due
to non-allergic underlining mechanism (21). The late onset as well as variable
clinical pattern of ACEI-AE can be confusing for both patients and
medical professionals, who should take into consideration the
potential role of risk factors and triggers (22). Depending on associated risk factors
or concomitant diseases and medications, patients may have an
increased risk for severe forms, even fatal angioedema due to
laryngeal edema (23). This
life-threatening reaction is possible even after the first dose of
an ACEI, with higher risk in female patients and in
African-American population (24).
It has been proven that African-American patients have an increased
sensitivity to bradykinin compared to Caucasian patients and that
patients with heart failure have higher rates of angioedema due to
ACEI medication (25,26). In rare cases of acquired angioedema
in adult patients, this may be first attributed to ACEI therapy,
but later revealing another mechanism and pathology, such is the
acquired C1-inhibitor deficiency due to hematological neoplasia or
autoimmune or unknown diseases (27). Regarding therapeutic approach of
ACEI-induced angioedema, several new drugs used for the treatment
of hereditary angioedema, such as icatibant (a bradykinin receptor
antagonist), have been evaluated, due to similar pathophysiological
mechanism (9).
The main limitation of our study is the small study
group and experience of one allergy center only, during a rather
short period of time.
It was concluded that acquired, non-allergic
angioedema induced by ACEI therapy is a frequent and possible
severe condition in medical practice and the awareness of both
patients and medical staff is still low. It is important that all
clinicians have adequate information and training with regard to
safety profile of ACEIs and the most frequent possible adverse
events, such as recurrent angioedema and chronic cough. A
standardized medical attitude, including detailed written
documentation of medication at emergency departments and improved
interdisciplinary collaboration in this field are needed.
Acknowledgements
Not applicable.
Funding
Not funding was received.
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
PML designed the current study, analyzed and
interpreted datasets and wrote the manuscript. VFA composed and
analyzed the datasets. CB, AM and DB contributed to the study
design, drafting and revision of the manuscript. All authors read
and approved the final version of the manuscript.
Ethics approval and consent to
participate
The patients signed an informed consent for
publication of their health data.
Patient consent for publication
Not applicable.
Competing interests
The authors declare no competing interests in
relation with this study and manuscript.
References
|
1
|
Smith RE and Ashiya M: Antihypertensive
therapies. Nat Rev Drug Discov. 6:597–598. 2007.PubMed/NCBI View
Article : Google Scholar
|
|
2
|
Gu Q, Paulose-Ram R, Dillon C and Burt V:
Antihypertensive medication use among US adults with hypertension.
Circulation. 113:213–221. 2006.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Dunlap ME and Peterson RC: ACE inhibitors
vs ARBs: Is one class better for heart failure. Cleve Clin J Med.
69:433–438. 2002.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Cicardi M, Zingale LC, Bergamaschini L and
Agostoni A: Angioedema associated with angiotensin-converting
enzyme inhibitor use: Outcome after switching to a different
treatment. Arch Intern Med. 164:910–913. 2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Banerji A, Clark S, Blanda M, LoVecchio F,
Snyder B and Camargo CA Jr: Multicenter study of patients with
angiotensin-converting enzyme inhibitor-induced angioedema who
present to the emergency department. Ann Allergy Asthma Immunol.
100:327–332. 2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Nussberger J, Cugno M and Cicardi M:
Bradykinin-mediated angioedema. N Engl J Med. 347:621–622.
2002.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Morimoto T, Gandhi TK, Fiskio JM, Seger
AC, So JW, Cook EF, Fukui T and Bates DW: An evaluation of risk
factors for adverse drug events associated with
angiotensin-converting enzyme inhibitors. J Eval Clin Pract.
10:499–509. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hoover T, Lippmann M, Grouzmann E, Marceau
F and Herscu P: Angiotensin converting enzyme inhibitor induced
angio-oedema: A review of the pathophysiology and risk factors.
Clin Exp Allergy. 40:50–61. 2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Terreehorst I, Reitsma S and Cohn DM:
Current treatment of Angioedema induced by ACE inhibitors. Curr
Treat Options Allergy. 6:18–26. 2019.
|
|
10
|
Gabb GM, Ryan P, Wing LM and Hutchinson
KA: Epidemiological study of angioedema and ACE inhibitors. Aust N
Z J Med. 26:777–782. 1996.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Cicardi M, Aberer W, Banerji A, Bas M,
Bernstein JA, Bork K, Caballero T, Farkas H, Grumach A, Kaplan AP,
et al: HAWK under the patronage of EAACI (European Academy of
Allergy and Clinical Immunology): Classification, diagnosis, and
approach to treatment for angioedema: Consensus report from the
Hereditary Angioedema International Working Group. Allergy.
69:602–616. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Craig TJ, Bernstein JA, Farkas H, Bouillet
L and Boccon-Gibod I: Diagnosis and treatment of
bradykinin-mediated angioedema: Outcomes from an angioedema expert
consensus meeting. Int Arch Allergy Immunol. 165:119–127.
2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Nedelea I and Deleanu D: Isolated
angioedema: An overview of clinical features and etiology (Review).
Exp Ther Med. 17:1068–1072. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Agostoni A, Cicardi M, Cugno M, Zingale
LC, Gioffré D and Nussberger J: Angioedema due to
angiotensin-converting enzyme inhibitors. Immunopharmacology.
44:21–25. 1999.
|
|
15
|
Zingale LC, Beltrami L, Zanichelli A,
Maggioni L, Pappalardo E, Cicardi B and Cicardi M: Angioedema
without urticaria: A large clinical survey. CMAJ. 175:1065–1070.
2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Lombardi C, Crivellaro M, Dama A, Senna G,
Gargioni S and Passalacqua G: Are physicians aware of the side
effects of angiotensin-converting enzyme inhibitors?: A
questionnaire survey in different medical categories. Chest.
128:976–979. 2005.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Shakib S, Caughey GE, Fok JS and Smith WB:
Adverse drug reaction classification by health professionals:
Appropriate discrimination between allergy and intolerance? Clin
Transl Allergy. 9(18)2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Leru PM: Drug allergies in primary care
practice in Romania: A questionnaire-based survey. Allergy Asthma
Clin Immunol. 10(16)2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Aberer W: Angioedema is not just ‘deep
urticaria’ but an entity of its own. Allergy. 69:549–552.
2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Agostoni A and Cicardi M: Drug-induced
angioedema without urticaria. Drug Saf. 24:599–606. 2001.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cicardi M and Zanichelli A: Acquired
angioedema. Allergy Asthma Clin Immunol. 6(14)2010.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Schiller PI, Messmer SL, Haefeli WE,
Schlienger RG and Bircher AJ: Angiotensin-converting enzyme
inhibitor-induced angioedema: Late onset, irregular course, and
potential role of triggers. Allergy. 52:432–435. 1997.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Krogh Nielsen T, Bygum A and Rye Rasmussen
E: Life-threatening angio-oedema after the first dose of an ACE
inhibitor-not an anaphylactic reaction. BMJ Case Rep. 2016(pii:
bcr2016214364)2016.doi: 10.1136/bcr-2016-214364. PubMed/NCBI View Article : Google Scholar
|
|
24
|
Dean DE, Schultz DL and Powers RH:
Asphyxia due to angiotensin converting enzyme (ACE) inhibitor
mediated angioedema of the tongue during the treatment of
hypertensive heart disease. J Forensic Sci. 46:1239–1243.
2001.PubMed/NCBI
|
|
25
|
Makani H, Messerli FH, Romero J,
Wever-Pinzon O, Korniyenko A, Berrios RS and Bangalore S:
Meta-analysis of randomized trials of angioedema as an adverse
event of renin-angiotensin system inhibitors. Am J Cardiol.
110:383–391. 2012.PubMed/NCBI View Article : Google Scholar
|
|
26
|
McDowell SE, Coleman JJ and Ferner RE:
Systematic review and meta-analysis of ethnic differences in risks
of adverse reactions to drugs used in cardiovascular medicine. BMJ.
332:1177–1181. 2006.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Leru PM, Anton VF and Bumbea H: Nine year
follow-up of a rare case of angioedema due to acquired C1-inhibitor
deficiency with late onset and good response to attenuated
androgen. Allergy Asthma Clin Immunol. 14(69)2018.PubMed/NCBI View Article : Google Scholar
|