1. Introduction
Respiratory tract infections (RTI) are extremely
common and in developed countries, RTI account for over 20% of all
medical consultations (1). In
children, the figures are even higher. Worldwide, every year, over
1 million children and infants under 5 years die from acute
respiratory infections (2), this
disease represents in developing countries the cause of death of
30% of children under five years (3,4). When
RTI have an increased frequency, over 60% of the cases have a
primary immunodeficiency in humoral immunity. A recent study has
shown that deficiencies in immunoglobulin (Ig) synthesis and
secretion are proven in abnormal levels of IgG, IgA and IgM
(5). Not only Igs are affected, but
also other immune molecules. In children diagnosed with RTI
decreased levels of proinflammatory cytokines were identified,
mainly interferon (IFN)-γ (6).
IFN-stimulated genes (ISGs) are highly involved in the antiviral
immune response (7).
In bacterial infection multidrug-resistant (MDR)
processes are involved in the difficult-to-treat pathogens
(8-10),
but in viral infections this process has just a collateral
importance because a substantial proportion of RTI are actually
acute viral infections. The treatment of these infections relies on
antivirals and on relieving disease symptoms. However, similarly to
bacterial infections, antiviral agents would conserve viral
proteins, that will induce a selective pressure on the viral
particle, leading to development of antiviral resistance (11,12).
As the best ‘treatment’ is prevention, in viral
infections vaccination is the best approach, but the pace of
vaccine development is slow and it has to overcome various hurdles,
e.g., antigenic variations, low efficacy, short-term immune
responses. As we are now faced with one of the major outbreaks of
viral infection of the modern history, vaccine development workflow
can not control the infection spreading (13). Therefore, novel approaches are tested
and designed. One of these novel approaches is passive immunization
(14) where mainly polyclonal
antibodies derived from several sources (e.g., sera of immunized
animals, immunized humans, convalescing patients) can be used
(15,16). In this case also the use of
polyclonal antibodies has some hurdles such as standardization and
patient safety. Monoclonal antibodies (mAbs) is the best choice but
their production costs is still very high, as an example the high
costs of monoclonal antidodies used in melanoma therapies (17). The costs increase further in
respiratory infection because there are viral escape mutants that
would need additional mAbs (18).
Therefore in this complex picture of immunity in viral infection an
interesting approach has risen in recent years - the use of
specific IgY. IgY is produced by birds, reptiles and amphibians
with a function similar to that of mammalian IgGs (19). IgYs are circulating in the sera as
IgGs, but in addition they accumulate 100 times more concentrated
in the egg yolk and are passed to the developing embryo (20).
IgY antibodies extracted from hen eggs have been
used in bacterial (21) and viral
infections therapy (22). It has
some favorable characteristics: it is well tolerated due to human
diet, it can be used in individuals who are allergic to eggs
because the purified IgY is depleted of egg albumin (23), it has a good tolerability. Moreover,
the systemic administration has shown the capability of specific
IgY raised against viruses to protect against actual diseases. For
example, in pig model IgY protected against Rotavirus infection
(24). Systemic and local
administration of IgY in mammals have shown that an anti-IgY
antibody response was generated, mainly consisting of the IgG
subclass. These reports show that IgY is antigenic but this
antibody molecule cannot bind to mammalian Fc receptors, so adverse
effects on this route are minimal (25). More than 20 years ago it was shown
that in mouse model administration of IgY, purified or not, did not
induce an IgE response, hence no allergic response (26) and due to the fact that IgY does not
link to the human complement system or Fc receptors, additional
inflammation upon administration is minimal (27). IgY action is to bind to the bacteria
or virus, and facilitate the elimination of the agent through the
gut preventing bacterial or viral replication and spread (28). Passive immunization with IgY can be
given in humans with active infection as they develop a rapid
response. Moreover, immature infants or immune-suppressed patients
can also benefit from this passive immunization (29,30). IgY
has a high content of sialic acid (31), with increased half-life (32) that suggests IgY to have a longer
circulating half-life, and hence increased anti-pathogen action and
increased efficacy against infections (33).
We aim to highlight in this review that in the quest
to find quick and effective anti-viral therapies, specific IgY can
be another possibility to fight against viral RTI.
2. IgY an established immune-fighter - is
there something new on its (bio) applications?
An antigen attack upon host is followed by Igs or
antibodies production in humans, process sustained by plasma cells
developing thus the humoral immunity (34). Antibodies engage in the fight against
the antigen by an array of mechanisms: neutralization, fixation of
pathogen, complement activation or acting like receptors on B cell
surface (35). Igs have interspecies
differences regarding their structural particularities or classes,
thus the term IgY comes from egg yolk where this Ig type is
produced for assuring immunity in the hen progeny. IgY represents
the functional equivalent of human IgG (29). In the last decade, IgY has gained
scientific attention due to its distinctive biological actions
largely emerged from its structural particularities (36).
Currently there are several commercial applications
where Igs are fully explored in diagnostic and therapy monitoring
assays.
Fighting against bacterial
infections
Immunization of avian with specific bacterial
antigens such as Salmonella sp. would provide a specific IgY
against the inductor antigen. Moreover, due its high stability and
structural unique features IgY has been applied successfully in
diagnostic, prophylactic and therapeutic purposes as well as
immunochemical reagents (34).
In addition to chicken models, there are other
hands-on and inexpensive models such as the quail model where
production of specific IgYs against Salmonella sp. has been
recently reported. These quail anti-Salmonella sp. IgYs
exhibit a high specificity to their matching immunogens, having the
potential to eradicate enterobacterial pathogens. In addition, the
oral ingestion of IgYs represent an efficient alternative for
annihilation of gastrointestinal pathogens as Salmonella
typhimurium and Salmonella enteritidis, bacterial
species that raise major concern in health and food industry
(37).
IgYs has also been proposed as a strategy for
combating infections caused by Pseudomonas aeruginosa known
as a common nosocomial pathogen having antibiotic resistance and a
frequent infection in acute pneumonia and severely burned patients.
A protein called PcrV is a vital part of killing machinery
represented by the type III secretion system of P.
aeruginosa and therefore PcrV is viewed as a target for
neutralizing this infectious agent. Thus, recently recombinant PcrV
was used for raising specific IgY. These antibodies displayed a
protective effect in both acute pneumonia and burn wound models and
moreover IgY anti-PcrV has augmented opsonization capacity and
bacterial killing activity of host cells (38). In fact the augmentation of phagocytic
killing via IgY was previously explored in an in vitro study
targeting P. aeruginosa infection in cystic fibrosis (CF)
patients. IgY against P. aeruginosa fulfill their function
by opsonizing the pathogen and thus enhancing the neutrophils
respiratory burst while further enabling bacterial killing. It was
suggested that prophylaxis with anti-P. aeruginosa IgY could
lift the innate immunity of CF patients aiding host neutrophils to
rapidly clear the bacterial agent (39).
In CF the principal contributor of pulmonary failure
is the chronic infection with P. aeruginosa biofilm, which
constantly attracts and activates neutrophils sustaining the
continuous inflammation. It is suggested that IgY favors bacteria
to form aggregates and increase their hydrophobicity enhancing
bacterial killing by neutrophils via phagocytosis (40).
Passive immunization with IgY anti-P.
aeruginosa could reduce the initial airway settlement with
P. aeruginosa in CF patients. Thus in a Balb/c murine P.
aeruginosa pneumonia model administration of specific IgY
significantly reduced the bacterial load at 24 h after infection
along with alleviating the clinical symptoms; in addition an
inflammatory cytokine pattern was noted revealing the lung
inflammation decrease suggesting that immune-prophylaxis with
anti-P. aeruginosa IgY may also function as an adjuvant to
antibiotics in lowering primary colonization of lungs (21). In parasitic diseases the possible
role of IgY in early diagnosis and therapeutics has been tackled by
attempting to obtain polyclonal IgY against parasitic antigens
suitable for immunotherapeutic purposes. Although further studies
in animal models are indispensable and obtaining a monoclonal IgY
anti-parasitic antigens are envisaged, it became obvious that IgY
could stand for immunoassay designing also in parasitology area
(41).
In immunodiagnostic methods, IgY is an excellent
tool in assays involving mammalian sera, due to the discriminative
properties of IgY compared to mammalian IgG. IgY has immunological
properties which makes it very distinct from mammalian IgG but at
the same time very affordable for a plenty of immunological
approaches. Foremost, by lacking the hinge region, IgY is less
structurally flexible than IgG and retains a different protein
content, these structural differences sustaining the differences in
immunological behavior (42). IgY
has poor cross-reactivity to mammalian IgG, does not activate the
complement system similar to IgM/IgG and lacks the reactivity with
mammalian Fc receptor (43).
However, there are still incomplete data regarding
the three-dimensional structure of Fc-IgY raising the question
whether IgY shares a conformational status similar to IgM and IgE
whose Fc regions are significantly flexible. However, the
evolutionary distance between mammals and birds made possible the
feasible generation of IgY against conserved mammal proteins. Thus,
the molecular particularities of IgY raised several functional
advantages recommending IgY as a versatile tool in biotechnological
research, diagnostics and therapeutics (29). One very practical utility is that IgY
can be used to generate a specific antibody when an antigen comes
in small amounts and additionally is low immunogenic in mammals
host (44).
IgY can be generated at low-costs in considerable
amounts through an ecologically friedly approach because it is
produced in egg yolk so no additional procedures on animals are
needed. There are several IgY isolation methods available, mostly
based on precipitation from egg extracts using ammonium sulfate or
polyethylene glycol although protein impurities still remains in
the sample of interest, in addition to time consuming and
complexity required (45,46). Chromatographic methods have become
very popular in recent years because this methodology generates
highly active pure products useful for biomedical applications. A
study published in 2020 reports IgY fragment separations by ion
exchange column using DEAE-Sepharose leading to Fab and Fc
fractions of high purity (88.7 and 90.1%, namely) and intact
activity rendering them easily used for medical purposes (47).
Usually, antibodies and/or their active fragments
produced in mammals are engaged in diagnostic tests. However, due
to animal welfare concerns, technical advantages and the high cost
of production, alternatives to the production of antibodies in
mammals have been investigated (48).
A constant goal for all current approaches involving
IgY applications is to continuously optimize the production and
purification of IgY antibodies from egg yolk to achieve high
quantities and high active products for research and commercial
use.
The most exploited immunoassay format with IgY
remains the ELISA platform. In research as well as in clinic, one
of the most effective and reliable method for rapid detection
remains ELISA which is a versatile, flexible and sensitive method
helpful in monitoring various pathologies including infectious
diseases. Although IgG is the ʻweapon of choiceʼ in
designing such detection formats, in recent years IgY started to
replace IgG because this molecule could be easily obtained, through
a non-invasive way (basically extracted and purified from egg yolk
of chickens immunized with recombinant proteins of interest), in
adequate amounts and at lower costs than IgG.
IgY-based ELISA protocols retain all the features
related to steps, reagents and applications of classic ELISA
formats as IgY molecule can be enzymatically labeled (e.g., with
horseradish peroxidase) and further applied in a wide range of
immunoassay assessments such as bacterial toxins (49) or tumor antigen detection for cancer
diagnosis (50,51).
It is truly noticeable how versatile the use of IgY
could be related to area of application, from viruses and bacteria
to tumor antigens detection and quantification (42). Thus, diagnosis of cholera is somewhat
an intricate attempt. Therefore, a recent study proposed IgY for
targeting two proteins contained by the outer membrane of V.
cholera (protein W and cytotoxin B). The study proposed two
sensitive and specific sandwich ELISA formats, with no
cross-reactivity with other bacteria. It was suggested that these
IgY based ELISAs could constitute a rapid and specific detection
tool for assessing V. cholera in different biological
samples (52).
The use of IgY instead of mouse monoclonal IgG has
the advantage of limiting the risk of false positive results as
well as the reduced costs of the test. For example, an IgY-based
sandwich ELISA was developed to quantify the total PSA level in
human serum as an alternative to commercially available in
vitro diagnosis tests (53).
Taking into account the latest concerns raised by
respiratory infections of viral origin IgY might act as a very
promising detection agent in this area. Thus, recent studies in
swine model suggest that IgY molecule could bind specifically to
viral nucleoprotein of influenza virus, which is known for exerting
a key role in viral replication and therefore emphasizing the IgY
effectiveness for influenza virus diagnosis (48,54).
Is there some spare room for other
methodologies regarding IgY characterization in the last
decade?
As already mentioned, IgY purification is currently
achieved by conventional precipitation methods with ammonium
sulphate or PEG and by chromatographic procedures. Sequential
precipitation with 31% ammonium sulfate and 12% polyethylene glycol
(PEG) produces IgY antibodies with above 95% purity without any
loss in immunoreactivity (55).
Additional procedures of purification could be added
to the separation methodologies, such as high-resolution
chromatography accomplished for instance by multi-column systems
such as NGC scout Bio-Rad system. Especially, but not restricted to
mAbs, this system provides certain advantages over conventional
chromatographic columns in terms of automatization, reproducibility
and accessibility. During the process of obtaining biomolecules
intended for biomedical application, high purity and intact
functionality are the first considerations. Moreover a multi-column
automated system could resolve several steps of purification in a
single run, such as separation by affinity purification of a
particular compound from original matrix, size exclusion to resolve
aggregates and finally obtaining the product of interest with
undamaged structure and function (56). By choosing the appropriate column the
compound is loaded, purified and analyzed in a continuous run. High
purification is further explored for designing platforms in which
pathogens could be discriminated or when ELISA type platform are
constructed intended for research or diagnosis.
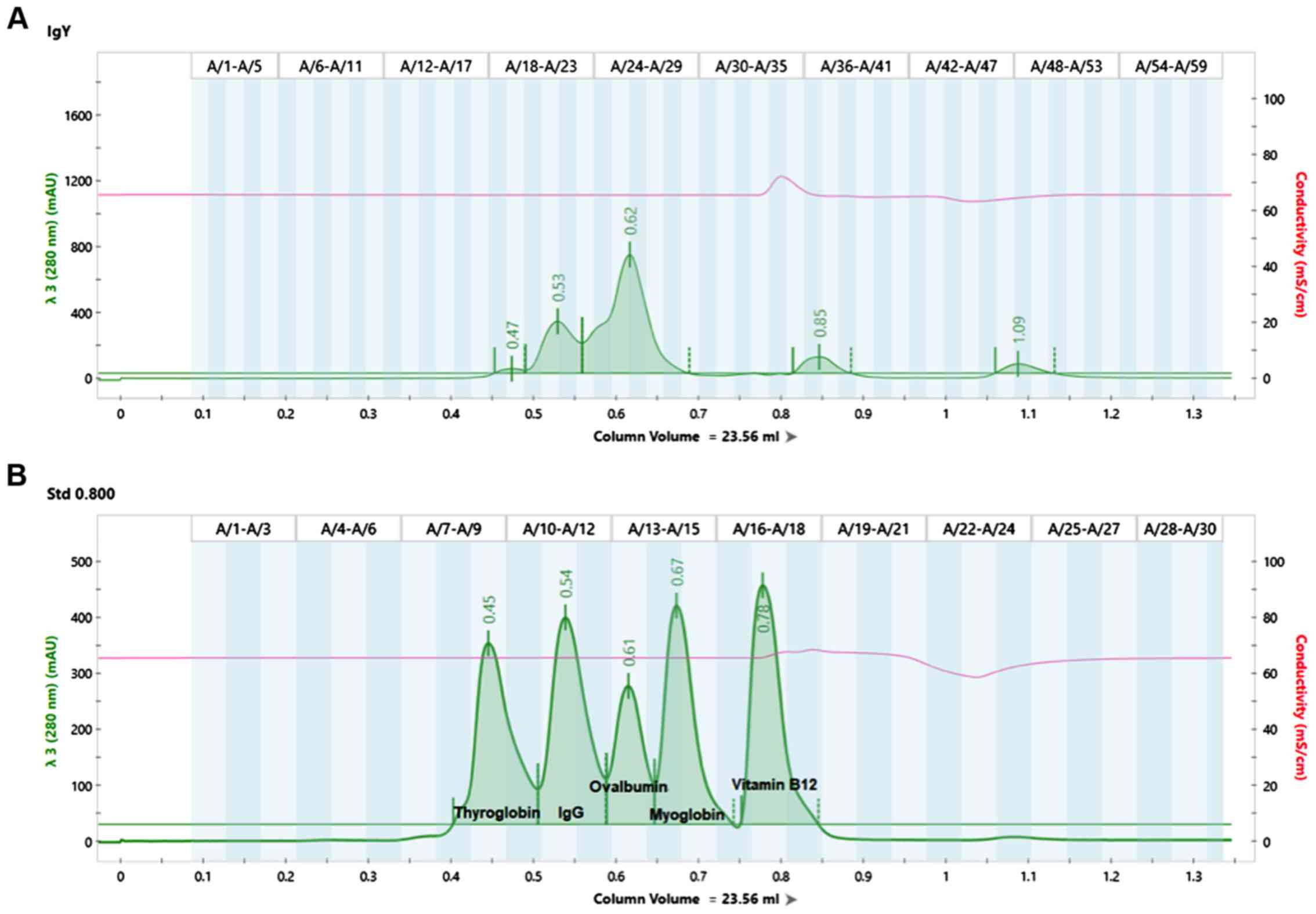
An example of analyzing an IgY extract isolated from
hen egg yolk by the Romanian Company Romvac S.A. using NGC scout
Bio-Rad system is presented in Fig.
1.
3. Passive immunization in infectious
diseases
Passive immunization as general
process
Passive immunization refers to administering already
mature antibodies (Igs) in an attempt to rapidly overcome an
infectious disease (57). Passive
immunity can represent a natural phenomenon and/or can be
artificially induced (58).
The natural passive immunity consists of maternal
antibodies that are transferred to the offspring inducing
protection before the offspring's self-immunity is built up. In
birds, passive imunity is sustained by the IgY from the egg yolk
which enters into the developing embryo (59,60) and
hence sustaining the first 2 weeks immunity in chickens, at the
time when they start to build up their own immunity (61). In mammals passive immunity is
sustained by maternal antibodies that are transferred to the fetus
through the placenta, in humans this process is active especially
during the last 3 months of pregnancy. Then the passive immunity is
sustained by milk that also contains antibodies (62).
Artificially induced passive immunity is sustained
by transferring antibodies by systemic, intravenous, or oral
routes. Passive immunity is limited in time, thus, to sustain this
immunity, preformed antibodies should be re-introduced (63).
A low-cost and rapid technology to obtain large
scale passive immunity antibodies are IgY from egg yolk via
hyperimmunization of chickens. Briefly this process involves hen
immunization with specific antigens with a specific time schedule
so that specific IgYs accumulate in the egg yolks. Afterwards,
these IgY are extracted (55,64).
Passive immunization has a history of more than 100 years and has
been used in both humans and animals (67,68).
Over 20 years ago, one of the first reports was published showing
that intranasal antibody prophylaxis, was successful against viral
respiratory infections in animal models [e.g., against respiratory
syncytial virus (RSV), influenza virus, Sendai virus], and entered
human clinical trials for influenza A and B viruses, Coxsackie
virus, and rhinoviruses (67). Since
then only non-viral IgY applications have been published such as
oral administration of IgY preventing or treating diseases induced
by Streptococcus mutans (dental carries), E.
coli-diarrhea, gastritis (H. pylori), periodontitis
(P. gingivalis) and oral candidiasis (C. albicans)
infant rotavirus diarrhea (23).
Rotavirus-induced diarrhea containing IgY (Rotamix IgY) was
efficient in pediatric patients diagnosed with non-cholera enteric
pathogen and was proven as a good adjuvant for the management of
acute diarrhea (68). Therefore, IgY
raised for specific viral respiratory infections should enter the
spotlight again.
IgY in passive immunization
IgY utilization in passive immunization has several
advantages. Obtaining IgY from birds is ecologically friendly, the
extract does not induce specific resistance and/or side effects and
due to their specificity would not affect beneficial microbial
population of the host.
Finally, if used as therapy in poultry and livestock
IgY does not stay in the meat that is destined to human consumption
as antibiotics do (69).
Using specific IgY in passive immunization is not
age restricted, it can be applied to a wide range of ages, to a
wide array of individuals characterized by patophysiological
conditions, e.g., women in special conditions like pregnancy and/or
immunodeficient patients.
IgY antibodies are naturally non-toxic and when
lyophilized they can be stored in regular 4˚C or even at room
temperature without losing their efficacy. Production costs are
much lower when compared to standard mAbs and/or vaccines. Then
storing can be done for months and transportation is thus
facilitated in comparison to other biologicals (70). If not extracted the composition of
IgY stored in eggs remains unchanged for at least 1 year at 4˚C
(71). The product, IgY is collected
from eggs and, therefore, does not impede the animal welfare.
From a molecular point of view, IgY has increased
binding avidity for the antigens that generated them in comparison
to mammalian IgG (72). Due to the
evolutionary distance between mammals and birds IgY can be produced
more easily against conserved mammalian molecules than IgG
antibodies (73). For IgY generation
a lower antigen load is necessary to induce the specific immune
response (69).
For mass production, as the production of eggs for
human consumption is already carried out on an industrial scale the
production of eggs containing specific IgY already has its
industrial backbone (74). One
chicken can produce specific IgY of approximately 22 g/year
(45).
4. Learning from severe acute respiratory
syndrome
Almost 20 years ago, coronaviruses (CoVs) were known
to cause disease only in vertebrates but then, the severe acute
respiratory syndrome (SARS)-CoV outburst in China showed the
medical world that nature still has unknown paths (75). The disease spread rapidly worldwide,
causing under 10,000 infections with a 10% mortality rate (76,77).
Through the SARS epidemy in China, passive immunization that used
sera from recovered SARS patients offered positive results
(78). IgY preparations were
obtained with anti-SARS coronavirus action after isolation from egg
yolk obtained from pathogen-free chickens immunized with SARS
coronavirus antigen (78). It was
reported that the obtained IgY had both high purity and biological
activity. In cells model, the obtained IgY neutralized the SARS
coronavirus. The preparation was stable upon lyophilization, that
gave the product good manufacturing qualities (78). Anti-SARS IgY produced upon this
procedure is a good candidate for anti SARS-CoV therapy. The
authors stated that in case of an outbreak, a rapid intervention
can be sustained by production of IgY antibodies. The production of
an IgY antibody takes 1.5 months from hens vaccination to IgY
production, so until a vaccination for an eventual outbreak is put
in use, this rapid response can fulfill an urgent medical need.
Moreover even when a proper vaccination is established, using this
type of passive immunizations can significantly increase the
control of the epidemic (78).
In 2007 a post-SARS outburst paper has shown that
there is SARS-CoV viral reservoir that is actually a biological
bomb waiting to explode (79) and
now, after 15 years we are facing the biggest pandemic since the
Spanish Flu in 1918. The medical world is ʻfast forwardingʼ
research and translating research results to bedside. In this race,
information changes almost in each hour. At present time, in the
race to obtaining quickly a vaccine there are 44 products with 2 in
clinical trial phase I (80), but
these data will be obsolete in the forthcoming month. Nevertheless,
until vaccination enters the application stages there are several
steps and phases to be taken. Therefore, probably by the end of
2020 there will be the most accelerated approved vaccine.
Alternative methods to overcome the outbreak and to therapeutically
sustain infected patients are urgently needed. Passive immunization
with plasma from recovered patients was already approved in USA
(81). In this picture of passive
immunization therapy IgY can have a future role.
Table I summarizes
the main studies that used anti-viral respiratory infection IgY
emphasizing on the experimental models, whether cellular or animal
models.
 | Table IMain studies focusing on in
vitro and in vivo models using anti-viral IgY. |
Table I
Main studies focusing on in
vitro and in vivo models using anti-viral IgY.
| Pathogen | IgY
preparation | Model type | Effect | Refs. |
|---|
| Pandemic influenza
virus A/H1N1 | Ostrich immunized
with swine influenza virus vaccine strain | MDCK cells infected
with pandemic virus | Neutralizing of
viral infectivity in the cells | (82) |
| Influenza B
virus | Hens immunized with
IBV | MDCK cells BALB/c
mouse model | Neutralization of
IBV in MDCK cells Reducing viral replication in the lungs | (83) |
| Influenza A
virus | Hens immunized with
H1N1 virus | MDCK cells Mouse
model | Neutralizing of
viral infectivity in the cells In vivo protection by
reducing the infectious titer of the virus in the lung | (84) |
| Viruses H1N1, H3N2,
and H5N1 strain | Hens immunized with
whole inactivated H1N1, H3N2, and H5N1 | MDCK cell BALB/c
and C.B-17 mice | Neutralization of
viruses in MDCK cells 100% protection against challenge with H5N1
and A/Puerto Rico/8/34 H1N1 | (85) |
| SARS | SPF chickens
immunized with inactivated SARS coronavirus | VERO E6 cells | Neutralizing SARS
coronavirus viral infectivity in the cells | (78) |
| BRSV related to
human syncytial virus | Hens immunized with
BRSV | MDCK cells infected
with A51908 BRSV strain | Neutralization of
viruses in MDCK cells | (86) |
5. Conclusions and future perspectives
Menacing viruses causing pandemics such as COVID-19
claim imperative future measures shaped in therapeutics and novel
vaccines. Large-scale and coordinated effort are needed to minimize
the impact of these threats on human health (87,88).
Joining these efforts, additional therapeutics open the perspective
of using passive immunization with IgY on large scale as adjuvant
therapy in viral respiratory infection. This endeavor should focus
on aspects of increased production, improved hen immunization
protocols, improved IgY extraction and increased antibody yield.
Another point to be taken into consideration in the future is that
chicken IgY have good monoclonality, therefore are more specific
and have higher affinity in comparison to the ones obtained from
immunizing mammals (89). As
compared to IgGs, obtaining genetically engineered single chain
fragment variable for IgY (IgY-scFv) is easier as these fragments
were already implemented in diagnostics and therapy (29,90).
Therefore, using specific IgY in viral respiratory
infections have good premise and can be expanded to other
infectious diseases.
In conclusion, far from being an obsolete chapter in
immunodiagnostics, the IgY-technology is revised as a new frontier
for research and clinic as already a wide range of IgY applications
has been effectively confirmed in both human and animal health. The
molecular particularities of IgY give them functional advantages
recommending IgY as a versatile tool in biotechnological research,
diagnostics and therapeutics. The main advantage is that IgY
methodology is a reliable and nature friendly alternative for
obtaining mammalian antibodies, as it is a non-invasive procedure.
The aria of application is continuously enlarging from bacterial
and viral infections to tumor biology. Specific anti-viral IgY has
already been tested in several designs, thus it is worth pointing
out that in the actual COVID-19 pandemic context, respiratory
infections need an enlarged arsenal of therapeutic approaches and
definitely IgY roles should be exploited in depth.
Acknowledgements
Authors would like to thank Dr Alexandra Livescu and
Dr Ovidiu Geicu (Dialab Solutions) for their invaluable technical
assistance in NGC scout Bio-Rad system separations.
Funding
This study was supported by IntelBiomed project,
SMIS code 105631, ID: P_40_197, Grant No. 52/2016.
Availability of data and materials
The information analyzed during the current study is
available from the corresponding author on reasonable request.
Authors' contributions
All authors substantially contributed to the writing
and revision of the work and read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable
Competing interests
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
The other authors declare that they have no competing
interests.
References
|
1
|
Bryce J, Boschi-Pinto C, Shibuya K and
Black RE: WHO Child Health Epidemiology Reference Group: WHO
estimates of the causes of death in children. Lancet.
365:1147–1152. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Qazi S, Aboubaker S, MacLean R, Fontaine
O, Mantel C, Goodman T, Young M, Henderson P and Cherian T: Ending
preventable child deaths from pneumonia and diarrhoea by 2025.
Development of the integrated Global Action Plan for the Prevention
and Control of Pneumonia and Diarrhoea. Arch Dis Child. 100 (Suppl
1):S23–S28. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ujunwa F and Ezeonu C: Risk factors for
acute respiratory tract infections in under-five children in enugu
Southeast Nigeria. Ann Med Health Sci Res. 4:95–99. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Munteanu AN, Surcel M, Constantin C and
Neagu M: Syncytial virus respiratory infections in children -
immunological aspects. Rev Biol Biomed Sci. 2:29–39. 2019.
|
|
5
|
Pasternak G, Lewandowicz-Uszyńska A and
Pentoś K: Disorders of humoral immunity in children with IgG
subclass deficiency and recurrent respiratory infections. Adv Exp
Med Biol. 1108:99–106. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ivanova ON, Argunova EF, Alekseev SN,
Ystugina TV, Varfolomeev AR, Troev IP, Kononova IV and Egorova VE:
Adaptive mechanisms of the immune system in children in far north.
Wiad Lek. 68:534–536. 2015.PubMed/NCBI
|
|
7
|
Liu Z, Qin Q, Wu C, Li H, Shou J, Yang Y,
Gu M, Ma C, Lin W, Zou Y, et al: Downregulated NDR1 protein kinase
inhibits innate immune response by initiating an miR146a-STAT1
feedback loop. Nat Commun. 9(2789)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mogayzel PJ Jr, Naureckas ET, Robinson KA,
Brady C, Guill M, Lahiri T, Lubsch L, Matsui J, Oermann CM, Ratjen
F, et al: Cystic Fibrosis Foundation Pulmonary Clinical Practice
Guidelines Committee: Cystic Fibrosis Foundation pulmonary
guideline. pharmacologic approaches to prevention and eradication
of initial Pseudomonas aeruginosa infection. Ann Am Thorac Soc.
11:1640–1650. 2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Mogayzel PJ Jr, Naureckas ET, Robinson KA,
Mueller G, Hadjiliadis D, Hoag JB, Lubsch L, Hazle L, Sabadosa K
and Marshall B: Pulmonary Clinical Practice Guidelines Committee:
Cystic fibrosis pulmonary guidelines. Chronic medications for
maintenance of lung health. Am J Respir Crit Care Med. 187:680–689.
2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Pasteur MC, Bilton D and Hill AT: British
Thoracic Society Bronchiectasis non-CF Guideline Group: British
Thoracic Society guideline for non-CF bronchiectasis. Thorax. 65
(Suppl 1):i1–i58. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
McCaskill JL, Ressel S, Alber A, Redford
J, Power UF, Schwarze J, Dutia BM and Buck AH: Broad-spectrum
inhibition of respiratory virus infection by MicroRNA mimics
targeting p38 MAPK signaling. Mol Ther Nucleic Acids. 7:256–266.
2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Ge Y and Sun S: Estimation of coronavirus
disease case-fatality risk in real time. Emerg Infect Dis: Apr 21,
2020 (Epub ahead of print).
|
|
13
|
Corti D, Passini N, Lanzavecchia A and
Zambon M: Rapid generation of a human monoclonal antibody to combat
Middle East respiratory syndrome. J Infect Public Health.
9:231–235. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sparrow E, Friede M, Sheikh M and
Torvaldsen S: Therapeutic antibodies for infectious diseases. Bull
World Health Organ. 95:235–237. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Graham BS and Ambrosino DM: History of
passive antibody administration for prevention and treatment of
infectious diseases. Curr Opin HIV AIDS. 10:129–134.
2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Shen C, Wang Z, Zhao F, Yang Y, Li J, Yuan
J, Wang F, Li D, Yang M, Xing L, et al: Treatment of 5
critically ill patients with COVID-19 with convalescent plasma.
JAMA: Mar 27, 2020 (Epub ahead of print).
|
|
17
|
Ancuceanu R and Neagu M: Immune based
therapy for melanoma. Indian J Med Res. 143:135–144.
2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Casadevall A, Dadachova E and Pirofski LA:
Passive antibody therapy for infectious diseases. Nat Rev
Microbiol. 2:695–703. 2004.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Warr GW, Magor KE and Higgins DA: IgY:
Clues to the origins of modern antibodies. Immunol Today.
16:392–398. 1995.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Carlander D, Stålberg J and Larsson A:
Chicken antibodies: A clinical chemistry perspective. Ups J Med
Sci. 104:179–189. 1999.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Thomsen K, Christophersen L, Bjarnsholt T,
Jensen PO, Moser C and Høiby N: Anti-Pseudomonas aeruginosa IgY
antibodies augment bacterial clearance in a murine pneumonia model.
J Cyst Fibros. 15:171–178. 2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Nguyen HH, Tumpey TM, Park HJ, Byun Y-H,
Tran LD, Nguyen VD, Kilgore PE, Czerkinsky C, Katz JM, Seong BL, et
al: Prophylactic and therapeutic efficacy of avian antibodies
against influenza virus H5N1 and H1N1 in mice. PLoS One.
5(e10152)2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Rahman S, Van Nguyen S, Icatlo FC Jr,
Umeda K and Kodama Y: Oral passive IgY-based immunotherapeutics: A
novel solution for prevention and treatment of alimentary tract
diseases. Hum Vaccin Immunother. 9:1039–1048. 2013.PubMed/NCBI View
Article : Google Scholar
|
|
24
|
Vega CG, Bok M, Vlasova AN, Chattha KS,
Fernández FM, Wigdorovitz A, Parreño VG, Saif LJ and Salmon H: IgY
antibodies protect against human Rotavirus induced diarrhea in the
neonatal gnotobiotic piglet disease model. PLoS One.
7(e42788)2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Torché AM, Le Dimna M, Le Corre P,
Mesplède A, Le Gal S, Cariolet R and Le Potier MF: Immune responses
after local administration of IgY loaded-PLGA microspheres in
gut-associated lymphoid tissue in pigs. Vet Immunol Immunopathol.
109:209–217. 2006.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Akita E, Jang C, Kitts D and Nakai S:
Evaluation of allergenicity of egg yolk immunoglobulin Y and other
egg proteins by passive cutaneous anaphylaxis. Food Agric Immunol.
11:191–201. 1999.
|
|
27
|
Kovacs-Nolan J and Mine Y: Egg yolk
antibodies for passive immunity. Annu Rev Food Sci Technol.
3:163–182. 2012.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Xu Y, Li X, Jin L, Zhen Y, Lu Y, Li S, You
J and Wang L: Application of chicken egg yolk immunoglobulins in
the control of terrestrial and aquatic animal diseases: A review.
Biotechnol Adv. 29:860–868. 2011.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zhang X, Calvert RA, Sutton BJ and Doré
KA: IgY: A key isotype in antibody evolution. Biol Rev Camb Philos
Soc. 92:2144–2156. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Muller S, Schubert A, Zajac J, Dyck T and
Oelkrug C: IgY antibodies in human nutrition for disease
prevention. Nutr J. 14(109)2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Gilgunn S, Millán Martín S, Wormald MR,
Zapatero-Rodríguez J, Conroy PJ, O'Kennedy RJ, Rudd PM, Saldova R
and Mondelli MU: Comprehensive N-Glycan profiling of avian
immunoglobulin Y. PLoS One. 11(e0159859)2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Liu L: Antibody glycosylation and its
impact on the pharmacokinetics and pharmacodynamics of monoclonal
antibodies and Fc-fusion proteins. J Pharm Sci. 104:1866–1884.
2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Abbas AT, El-Kafrawy SA, Sohrab SS and
Azhar EIA: IgY antibodies for the immunoprophylaxis and therapy of
respiratory infections. Hum Vaccin Immunother. 15:264–275.
2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Amro WA, Al-Qaisi W and Al-Razem F:
Production and purification of IgY antibodies from chicken egg
yolk. J Genet Eng Biotechnol. 16:99–103. 2018.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Hoffman W, Lakkis FG and Chalasani G: B
cells, antibodies, and more. Clin J Am Soc Nephrol. 11:137–154.
2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Michael A, Meenatchisundaram S,
Parameswari G, Subbraj T, Selvakumaran R and Ramalingam S: Chicken
egg yolk antibodies (IgY) as an alternative to mammalian
antibodies. Indian J Sci Technol. 3:468–474. 2010.
|
|
37
|
Esmailnejad A, Abdi-Hachesoo B, Hosseini
Nasab E and Shakoori M: Production, purification, and evaluation of
quail immunoglobulin Y against Salmonella typhimurium and
Salmonella enteritidis. Mol Immunol. 107:79–83. 2019.
|
|
38
|
Ranjbar M, Behrouz B, Norouzi F and
Mousavi Gargari SL: Anti-PcrV IgY antibodies protect against
Pseudomonas aeruginosa infection in both acute pneumonia and burn
wound models. Mol Immunol. 116:98–105. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Thomsen K, Christophersen L, Jensen PØ,
Bjarnsholt T, Moser C and Høiby N: Anti-Pseudomonas aeruginosa IgY
antibodies promote bacterial opsonization and augment the
phagocytic activity of polymorphonuclear neutrophils. Hum Vaccin
Immunother. 12:1690–1699. 2016.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Thomsen K, Christophersen L, Bjarnsholt T,
Jensen PØ, Moser C and Høiby N: Anti-Pseudomonas aeruginosa IgY
antibodies induce specific bacterial aggregation and
internalization in human polymorphonuclear neutrophils. Infect
Immun. 83:2686–2693. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Thirumalai D, Visaga Ambi S, Vieira-Pires
RS, Xiaoying Z, Sekaran S and Krishnan U: Chicken egg yolk antibody
(IgY) as diagnostics and therapeutics in parasitic infections - A
review. Int J Biol Macromol. 136:755–763. 2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Pereira EPV, van Tilburg MF, Florean EOPT
and Guedes MIF: Egg yolk antibodies (IgY) and their applications in
human and veterinary health: A review. Int Immunopharmacol.
73:293–303. 2019.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Dunkelberger JR and Song WC: Complement
and its role in innate and adaptive immune responses. Cell Res.
20:34–50. 2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Munhoz LS, Vargas GDÁ, Fischer G, Lima M,
Esteves PA and Hübner SO: Avian IgY antibodies: Characteristics and
applications in immunodiagnostic. Cienc Rural. 44:153–160.
2014.
|
|
45
|
Pauly D, Chacana PA, Calzado EG, Brembs B
and Schade R: IgY technology: Extraction of chicken antibodies from
egg yolk by polyethylene glycol (PEG) precipitation. J Vis Exp.
2011(3084)2011.PubMed/NCBI View
Article : Google Scholar
|
|
46
|
Polanowski A, Zabłocka A, Sosnowska A,
Janusz M and Trziszka T: Immunomodulatory activity accompanying
chicken egg yolk immunoglobulin Y. Poult Sci. 91:3091–3096.
2012.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Zhou X, Wang Y, Ahn DU and Cai Z: An easy
and simple separation method for Fc and Fab fragments from chicken
immunoglobulin Y (IgY). J Chromatogr B Analyt Technol Biomed Life
Sci. 1141(122011)2020.PubMed/NCBI View Article : Google Scholar
|
|
48
|
da Silva MC, Schaefer R, Gava D, Souza CK,
da Silva Vaz I Jr, Bastos AP and Venancio EJ: Production and
application of anti-nucleoprotein IgY antibodies for influenza A
virus detection in swine. J Immunol Methods. 461:100–105.
2018.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Nagaraj S, Ramlal S, Kingston J and Batra
HV: Development of IgY based sandwich ELISA for the detection of
staphylococcal enterotoxin G (SEG), an egc toxin. Int J Food
Microbiol. 237:136–141. 2016.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Grzywa R, Łupicka-Słowik A, Walczak M,
Idzi M, Bobrek K, Boivin S, Gaweł A, Stefaniak T, Oleksyszyn J and
Sieńczyk M: Highly sensitive detection of cancer antigen 15-3 using
novel avian IgY antibodies. ALTEX. 31:43–52. 2014.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Ferreira A Jr, Santos JP, Bassi PB, Bittar
JFF and Bittar ER: IgY-technology applied to studies of toxoplasma
gondii infection. In: Toxoplasmosis. IntechOpen, 2017. https://www.intechopen.com/books/toxoplasmosis/igy-technology-applied-to-studies-of-toxoplasma-gondii-infection.
Accessed June 14, 2017.
|
|
52
|
Bayat M, Khabiri A and Hemati B:
Development of IgY-based sandwich ELISA as a robust tool for rapid
detection and discrimination of toxigenic Vibrio cholerae.
Can J Infect Dis Med Microbiol. 2018(e4032531)2018.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Łupicka-Słowik A, Grzywa R, Leporowska E,
Procyk D, Oleksyszyn J and Sieńczyk M: Development and evaluation
of an immunoglobulin Y-based ELISA for measuring prostate specific
antigen in human serum. Ann Lab Med. 39:373–380. 2019.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Eisfeld AJ, Neumann G and Kawaoka Y: At
the centre: Influenza A virus ribonucleoproteins. Nat Rev
Microbiol. 13:28–41. 2015.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Rajic A, Stehmann C, Autelitano DJ, Vrkic
AK, Hosking CG, Rice GE and Ilag LL: Protein depletion using IgY
from chickens immunised with human protein cocktails. Prep Biochem
Biotechnol. 39:221–247. 2009.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Elms P: Advantages of multidimensional
(Multi-D) chromatography using the NGC™ chromatography system over
traditional sequential chromatography. Bulletin 6694. Bio-Rad
Laboratories, Inc., Hercules, CA, 2016.
|
|
57
|
Casadevall A and Scharff MD: Return to the
past: The case for antibody-based therapies in infectious diseases.
Clin Infect Dis. 21:150–161. 1995.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Baxter D: Active and passive immunity,
vaccine types, excipients and licensing. Occup Med (Lond).
57:552–556. 2007.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Patterson R, Youngner JS, Weigle WO and
Dixon FJ: The metabolism of serum proteins in the hen and chick and
secretion of serum proteins by the ovary of the hen. J Gen Physiol.
45:501–513. 1962.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Rose ME, Orlans E and Buttress N:
Immunoglobulin classes in the hen's egg: Their segregation in yolk
and white. Eur J Immunol. 4:521–523. 1974.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Hamal KR, Burgess SC, Pevzner IY and Erf
GF: Maternal antibody transfer from dams to their egg yolks, egg
whites, and chicks in meat lines of chickens. Poult Sci.
85:1364–1372. 2006.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Helmenstine AM: An introduction to active
immunity and passive immunity. ThoughtCo, 2019. https://www.thoughtco.com/active-immunity-and-passive-immunity-4134137.
Accessed April 29, 2020.
|
|
63
|
Chalghoumi R, Marcq C, Théwis A,
Portetelle D and Beckers Y: Effects of feed supplementation with
specific hen egg yolk antibody (immunoglobin Y) on Salmonella
species cecal colonization and growth performances of challenged
broiler chickens. Poult Sci. 88:2081–2092. 2009.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Gadde U, Rathinam T and Lillehoj HS:
Passive immunization with hyperimmune egg-yolk IgY as prophylaxis
and therapy for poultry diseases - A review. Anim Health Res Rev.
16:163–176. 2015.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Eibl MM: History of immunoglobulin
replacement. Immunol Allergy Clin North Am. 28:737–764, viii.
2008.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Hsu JL and Safdar N: Polyclonal
immunoglobulins and hyperimmune globulins in prevention and
management of infectious diseases. Infect Dis Clin North Am.
25:773–788. 2011.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Weltzin R and Monath TP: Intranasal
antibody prophylaxis for protection against viral disease. Clin
Microbiol Rev. 12:383–393. 1999.PubMed/NCBI
|
|
68
|
Rahman S, Higo-Moriguchi K, Htun KW,
Taniguchi K, Icatlo FC Jr, Tsuji T, Kodama Y, Van Nguyen S, Umeda
K, Oo HN, et al: Randomized placebo-controlled clinical trial of
immunoglobulin Y as adjunct to standard supportive therapy for
rotavirus-associated diarrhea among pediatric patients. Vaccine.
30:4661–4669. 2012.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Li X, Wang L, Zhen Y, Li S and Xu Y:
Chicken egg yolk antibodies (IgY) as non-antibiotic production
enhancers for use in swine production: A review. J Anim Sci
Biotechnol. 6(40)2015.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Thu HM, Myat TW, Win MM, Thant KZ, Rahman
S, Umeda K, Nguyen SV, Icatlo FC Jr, Higo-Moriguchi K, Taniguchi K,
et al: Chicken egg yolk antibodies (IgY) for prophylaxis and
treatment of rotavirus diarrhea in human and animal neonates: A
concise review. Han-gug Chugsan Sigpum Hag-hoeji. 37:1–9.
2017.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Jensenius JC, Andersen I, Hau J, Crone M
and Koch C: Eggs: Conveniently packaged antibodies. Methods for
purification of yolk IgG. J Immunol Methods. 46:63–68.
1981.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Ikemori Y, Peralta RC, Kuroki M, Yokoyama
H and Kodama Y: Research note: Avidity of chicken yolk antibodies
to enterotoxigenic Escherichia coli fimbriae. Poult Sci.
72:2361–2365. 1993.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Gassmann M, Thömmes P, Weiser T and
Hübscher U: Efficient production of chicken egg yolk antibodies
against a conserved mammalian protein. FASEB J. 4:2528–2532.
1990.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Sharma JM: Introduction to poultry
vaccines and immunity. Adv Vet Med. 41:481–494. 1999.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Perlman S and Netland J: Coronaviruses
post-SARS: Update on replication and pathogenesis. Nat Rev
Microbiol. 7:439–450. 2009.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Rota PA, Oberste MS, Monroe SS, Nix WA,
Campagnoli R, Icenogle JP, Peñaranda S, Bankamp B, Maher K, Chen
MH, et al: Characterization of a novel coronavirus associated with
severe acute respiratory syndrome. Science. 300:1394–1399.
2003.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Drosten C, Günther S, Preiser W, van der
Werf S, Brodt HR, Becker S, Rabenau H, Panning M, Kolesnikova L,
Fouchier RAM, et al: Identification of a novel coronavirus in
patients with severe acute respiratory syndrome. N Engl J Med.
348:1967–1976. 2003.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Fu CY, Huang H, Wang XM, Liu YG, Wang ZG,
Cui SJ, Gao HL, Li Z, Li JP and Kong XG: Preparation and evaluation
of anti-SARS coronavirus IgY from yolks of immunized SPF chickens.
J Virol Methods. 133:112–115. 2006.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Cheng VC, Lau SK, Woo PC and Yuen KY:
Severe acute respiratory syndrome coronavirus as an agent of
emerging and reemerging infection. Clin Microbiol Rev. 20:660–694.
2007.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Gavi, The Vaccine Alliance: The latest in
the COVID-19 vaccine race. https://www.gavi.org/vaccineswork/covid-19-vaccine-race.
Accessed March 24, 2020.
|
|
81
|
Mail Online: FDA approves first US
coronavirus treatment: Doctors across the US can now treat the
sickest Americans with plasma from recovered patients. https://www.dailymail.co.uk/health/article-8147631/Can-blood-coronavirus-survivors-treat-newly-ill.html.
Accessed March 24, 2020.
|
|
82
|
Tsukamoto M, Hiroi S, Adachi K, Kato H,
Inai M, Konishi I, Tanaka M, Yamamoto R, Sawa M, Handharyani E, et
al: Antibodies against swine influenza virus neutralize the
pandemic influenza virus A/H1N1. Mol Med Rep. 4:209–214.
2011.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Wen J, Zhao S, He D, Yang Y, Li Y and Zhu
S: Preparation and characterization of egg yolk immunoglobulin Y
specific to influenza B virus. Antiviral Res. 93:154–159.
2012.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Yang YE, Wen J, Zhao S, Zhang K and Zhou
Y: Prophylaxis and therapy of pandemic H1N1 virus infection using
egg yolk antibody. J Virol Methods. 206:19–26. 2014.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Wallach MG, Webby RJ, Islam F,
Walkden-Brown S, Emmoth E, Feinstein R and Gronvik KO:
Cross-protection of chicken immunoglobulin Y antibodies against
H5N1 and H1N1 viruses passively administered in mice. Clin Vaccine
Immunol. 18:1083–1090. 2011.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Ferella A, Bellido D, Chacana P and
Wigdorovitz A: DusSantos MJ and Mozgovoj MV: Chicken egg yolk
antibodies against bovine respiratory syncytial virus neutralize
the virus in vitro. Procedia Vaccinol. 6:33–38. 2012.
|
|
87
|
Tsitoura E, Bibaki E, Bolaki M, Vasarmidi
E, Trachalaki A, Symvoulakis EK, Spandidos DA and Antoniou KM:
[Comment] Treatment strategies to fight the new coronavirus
SARS-CoV-2: A challenge for a Rubik's Cube solver. Exp Ther Med (In
Press).
|
|
88
|
Docea AO, Tsatsakis A, Albulescu D,
Cristea O, Zlatian O, Vinceti M, Moschos SA, Tsoukalas D, Goumenou
M, Drakoulis N, et al: A new threat from an old enemy: Re-emergence
of coronavirus (Review). Int J Mol Med. 45:1631–1643.
2020.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Lee W, Syed Atif A, Tan SC and Leow CH:
Insights into the chicken IgY with emphasis on the generation and
applications of chicken recombinant monoclonal antibodies. J
Immunol Methods. 447:71–85. 2017.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Spillner E, Braren I, Greunke K, Seismann
H, Blank S and du Plessis D: Avian IgY antibodies and their
recombinant equivalents in research, diagnostics and therapy.
Biologicals. 40:313–322. 2012.PubMed/NCBI View Article : Google Scholar
|