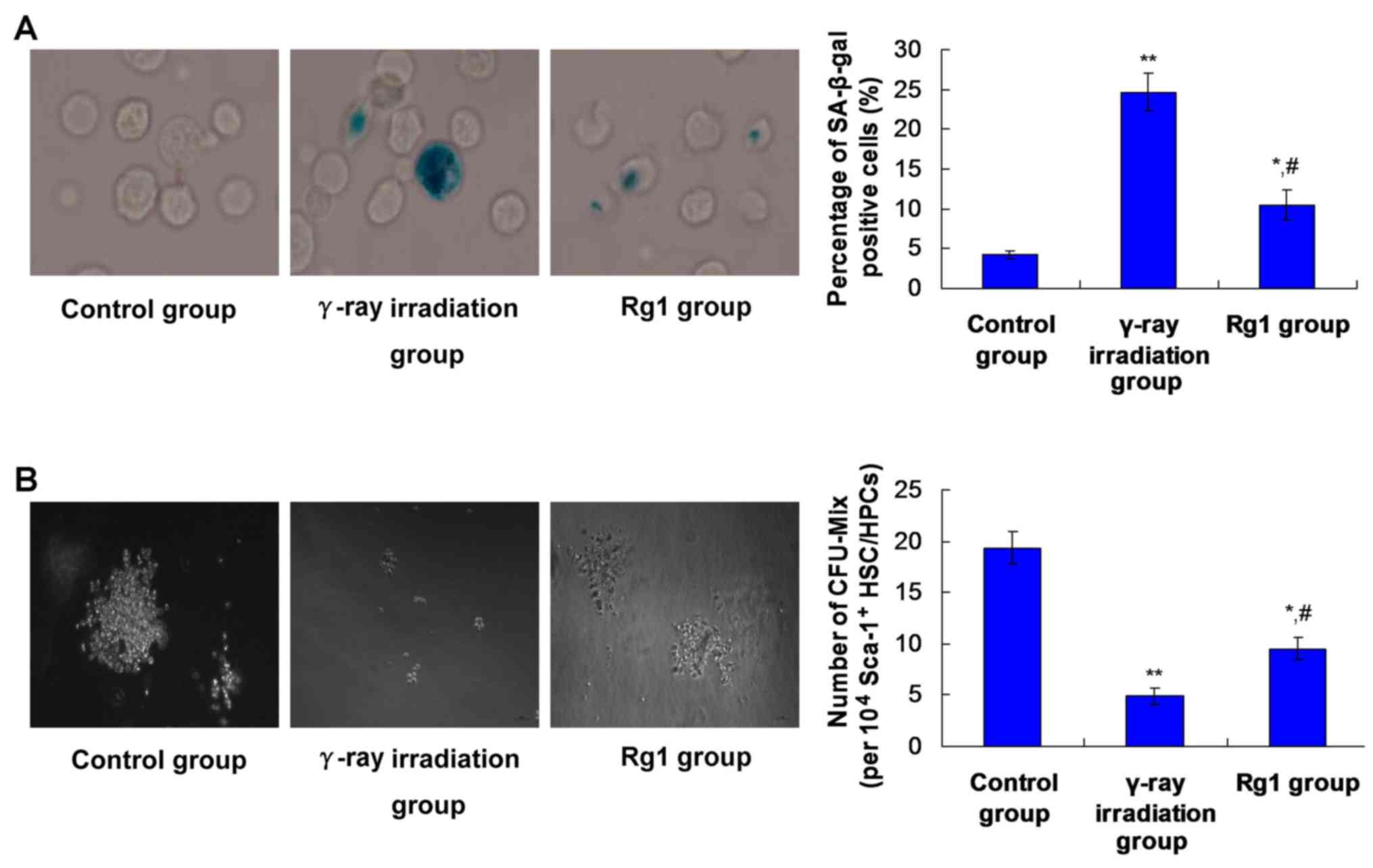
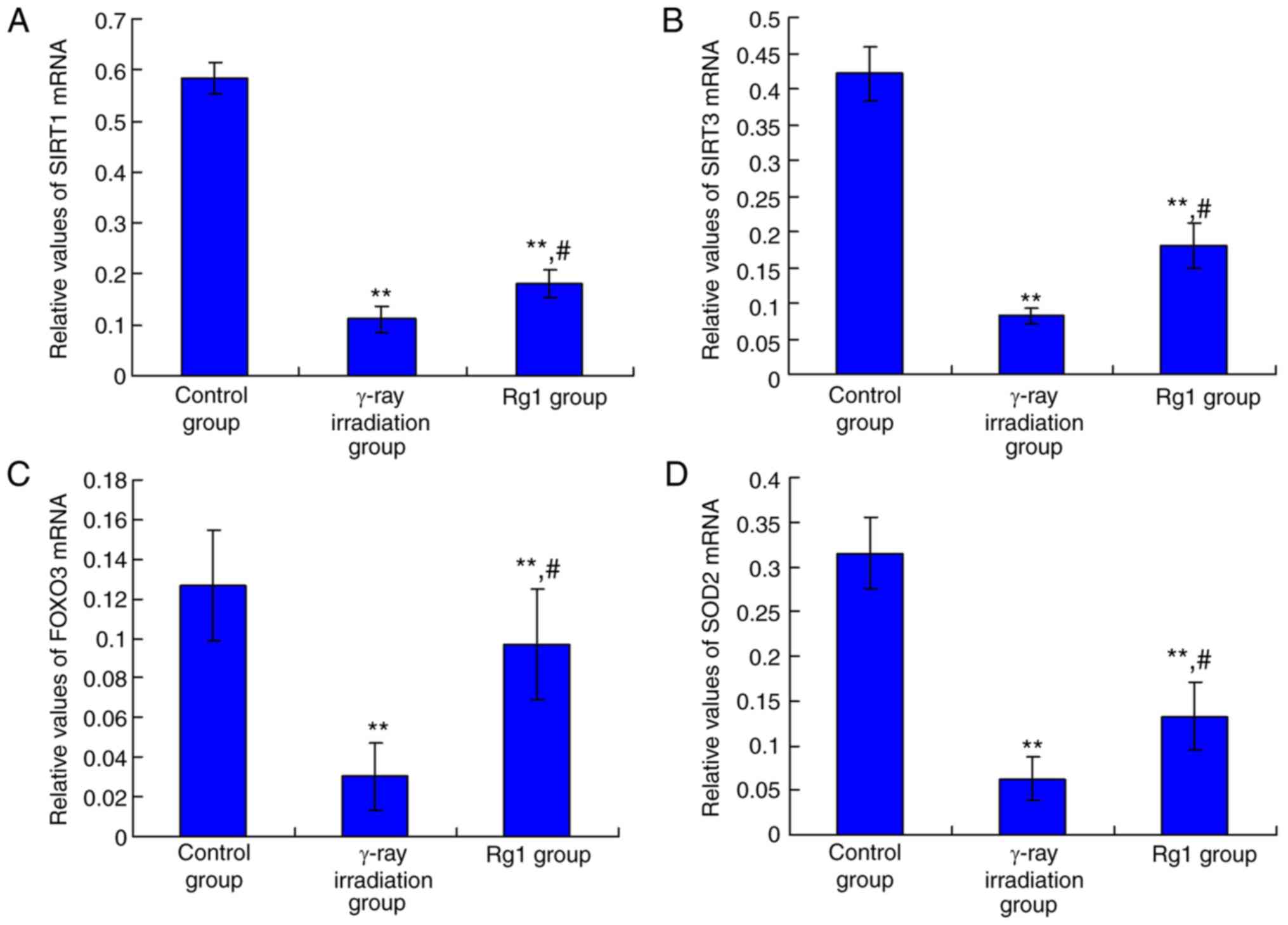
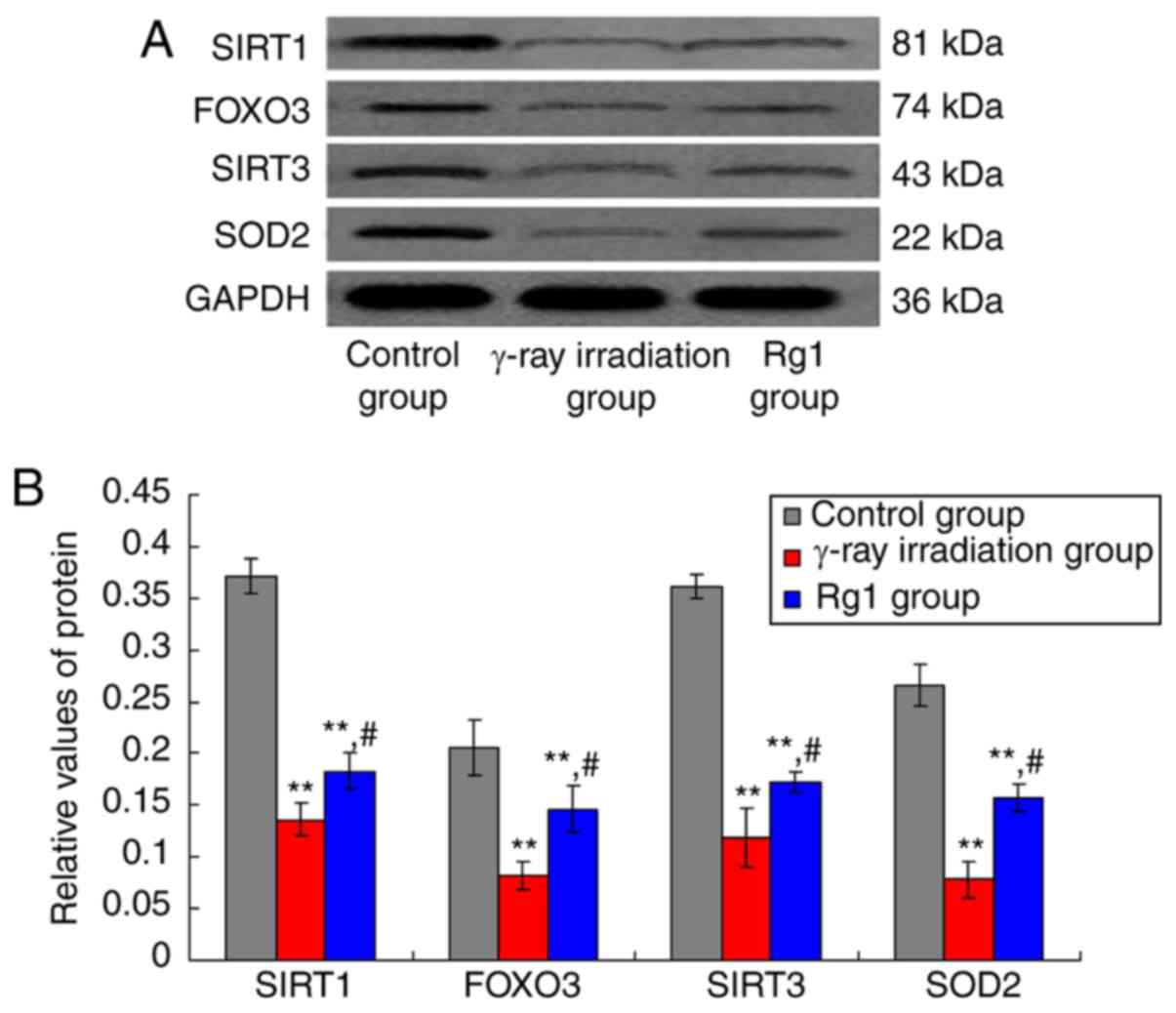
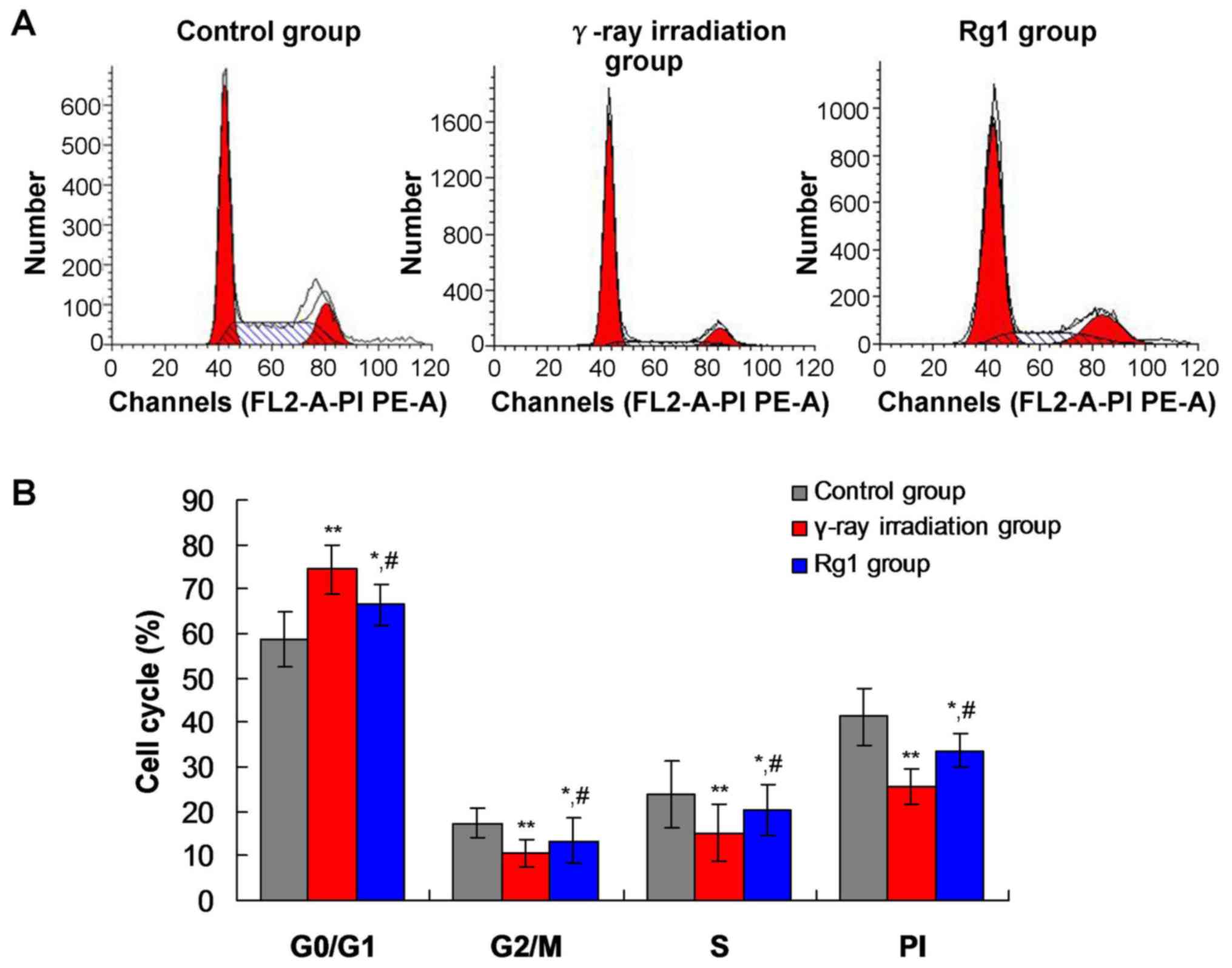
|
1
|
Xu YF, Liang ZJ, Kuang ZJ, Chen JJ, Wu J,
Lu XE, Jiang WW, Fan PL, Tang LY, Li YT, et al: Effect of Suo Quan
Wan on the bladder function of aging rats based on the
β-adrenoceptor. Exp Ther Med. 13:3424–3432. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Li YN, Guo Y, Xi MM, Yang P, Zhou XY, Yin
S, Hai CX, Li JG and Qin XJ: Saponins from Aralia taibaiensis
attenuate D-galactose-induced aging in rats by activating FOXO3a
and Nrf2 pathways. Oxid Med Cell Longev.
2014(320513)2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bustos ML, Huleihel L, Kapetanaki MG,
Lino-Cardenas CL, Mroz L, Ellis BM, McVerry BJ, Richards TJ,
Kaminski N, Cerdenes N, et al: Aging mesenchymal stem cells fail to
protect because of impaired migration and antiinflammatory
response. Am J Respir Crit Care Med. 189:787–798. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Montezano AC and Touyz RM: Reactive oxygen
species, vascular Noxs, and hypertension: Focus on translational
and clinical research. Antioxid Redox Signal. 20:164–182.
2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Liochev SI: Reactive oxygen species and
the free radical theory of aging. Free Radic Biol Med. 60:1–4.
2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Milano F, Merriam F, Nicoud I, Li J,
Gooley TA, Heimfeld S, Imren S and Delaney C: Notch-expanded murine
hematopoietic stem and progenitor cells mitigate death from lethal
radiation and convey immune tolerance in mismatched recipients.
Stem Cells Transl Med. 6:566–575. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sadowska-Bartosz I and Bartosz G: Effect
of antioxidants supplementation on aging and longevity. Biomed Res
Int. 2014(404680)2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Guerra-Araiza C, Alvarez-Mejia AL,
Sanchez-Torres S, Farfan-Garcia E, Mondragon-Lozano R,
Pinto-Almazan R and Salgado-Ceballos H: Effect of natural exogenous
antioxidants on aging and on neurodegenerative diseases. Free Radic
Res. 47:451–462. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chu SF and Zhang JT: New achievements in
ginseng research and its future prospects. Chin J Integr Med.
15:403–408. 2009.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Xu FT, Li HM, Yin QS, Cui SE, Liu DL, Nan
H, Han ZA and Xu KM: Effect of ginsenoside Rg1 on proliferation and
neural phenotype differentiation of human adipose-derived stem
cells in vitro. Can J Physiol Pharmacol. 92:467–475.
2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chen C, Mu XY, Zhou Y, Shun K, Geng S, Liu
J, Wang JW, Chen J, Li TY and Wang YP: Ginsenoside Rg1 enhances the
resistance of hematopoietic stem/progenitor cells to
radiation-induced aging in mice. Acta Pharmacol Sin. 35:143–150.
2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Dali-Youcef N, Lagouge M, Froelich S,
Koehl C, Schoonjans K and Auwerx J: Sirtuins: The ‘magnificent
seven’, function, metabolism and longevity. Ann Med. 39:335–345.
2007.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Imai S, Johnson FB, Marciniak RA, McVey M,
Park PU and Guarente L: Sir2: An NAD-dependent histone deacetylase
that connects chromatin silencing, metabolism, and aging. Cold
Spring Harb Symp Quant Biol. 65:297–302. 2000.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Buler M, Aatsinki SM, Izzi V and Hakkola
J: Metformin reduces hepatic expression of SIRT3, the mitochondrial
deacetylase controlling energy metabolism. PLoS One.
7(e49863)2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
D'Aquila P, Rose G, Panno ML, Passarino G
and Bellizzi D: SIRT3 gene expression: A link between inherited
mitochondrial DNA variants and oxidative stress. Gene. 497:323–329.
2012.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Finley LW, Haas W, Desquiret-Dumas V,
Wallace DC, Procaccio V, Gygi SP and Haigis MC: Succinate
dehydrogenase is a direct target of sirtuin 3 deacetylase activity.
PLoS One. 6(e23295)2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Libert S and Guarente L: Metabolic and
neuropsychiatric effects of calorie restriction and sirtuins. Annu
Rev Physiol. 75:669–684. 2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yao H, Chung S, Hwang JW, Rajendrasozhan
S, Sundar IK, Dean DA, McBurney MW, Guarente L, Gu W, Ronty M, et
al: SIRT1 protects against emphysema via FOXO3-mediated reduction
of premature senescence in mice. J Clin Invest. 122:2032–2045.
2012.PubMed/NCBI View
Article : Google Scholar
|
|
19
|
Chen Y, Zhang J, Lin Y, Lei Q, Guan KL,
Zhao S and Xiong Y: Tumour suppressor SIRT3 deacetylates and
activates manganese superoxide dismutase to scavenge ROS. EMBO Rep.
12:534–541. 2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Miao L and St Clair DK: Regulation of
superoxide dismutase genes: Implications in disease. Free Radic
Biol Med. 47:344–356. 2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liwski CJ, Padley DJ, Gustafson MP,
Winters JL, Gastineau DA and Jacob EK: Discordant CD34+
cell results in peripheral blood and hematopoietic progenitor
cell-apheresis product: Implications for clinical decisions and
impact on patient treatment. Transfusion. 54:541–544.
2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhou Y, Yang B, Yao X and Wang Y:
Establishment of an aging model of Sca-1+ hematopoietic
stem cell and studies on its relative biological mechanisms. In
Vitro Cell Dev Biol Anim. 47:149–156. 2011.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Schmittgen TD, Zakrajsek BA, Mills AG,
Gorn V, Singer MJ and Reed MW: Quantitative reverse
transcription-polymerase chain reaction to study mRNA decay:
Comparison of endpoint and real-time methods. Anal Biochem.
285:194–204. 2000.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Dimri GP, Lee X, Basile G, Acosta M, Scott
G, Roskelley C, Medrano EE, Linskens M, Rubelj I and Pereira-Smith
O: A biomarker that identifies senescent human cells in culture and
in aging skin in vivo. Proc Natl Acad Sci USA. 92:9363–9367.
1995.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tang YL, Zhou Y, Wang YP, Wang JW and Ding
JC: SIRT6/NF-κB signaling axis in ginsenoside Rg1-delayed
hematopoietic stem/progenitor cell senescence. Int J Clin Exp
Pathol. 8:5591–5596. 2015.PubMed/NCBI
|
|
26
|
Giralt A and Villarroya F: SIRT3, a
pivotal actor in mitochondrial functions: Metabolism, cell death
and aging. Biochem J. 444:1–10. 2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Atkins KM, Thomas LL, Barroso-Gonzalez J,
Thomas L, Auclair S, Yin J, Kang H, Chung JH, Dikeakos JD and
Thomas G: The multifunctional sorting protein PACS-2 regulates
SIRT1-mediated deacetylation of p53 to modulate p21-dependent
cell-cycle arrest. Cell Rep. 8:1545–1557. 2014.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Stein GH, Beeson M and Gordon L: Failure
to phosphorylate the retinoblastoma gene product in senescent human
fibroblasts. Science. 249:666–669. 1990.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Chen X, Zhang J, Fang Y, Zhao C and Zhu Y:
Ginsenoside Rg1 delays tert-butyl hydroperoxide-induced premature
senescence in human WI-38 diploid fibroblast cells. J Gerontol A
Biol Sci Med Sci. 63:253–264. 2008.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Asadullina NR, Usacheva AM and Gudkov SV:
Protection of mice against X-ray injuries by the post-irradiation
administration of inosine-5'-monophosphate. J Radiat Res.
53:211–216. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zhu J, Mu X, Zeng J, Xu C, Liu J, Zhang M,
Li C, Chen J, Li T and Wang Y: Ginsenoside Rg1 prevents cognitive
impairment and hippocampus senescence in a rat model of
D-galactose-induced aging. PLoS One. 9(e101291)2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Hu W, Jing P, Wang L, Zhang Y, Yong J and
Wang Y: The positive effects of Ginsenoside Rg1 upon the
hematopoietic microenvironment in a D-Galactose-induced aged rat
model. BMC Complement Altern Med. 15(119)2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhou Y, Yang B, Jiang R, Yao X and Wang
YP: Mechanism of ginsenoside Rg1 in the delayed senescence of
hematopoietic stem cell. Zhonghua Yi Xue Za Zhi. 90:3421–3425.
2010.(In Chinese). PubMed/NCBI
|
|
34
|
Wang Y, Liu L, Pazhanisamy SK, Li H, Meng
A and Zhou D: Total body irradiation causes residual bone marrow
injury by induction of persistent oxidative stress in murine
hematopoietic stem cells. Free Radic Biol Med. 48:348–356.
2010.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kamminga LM, van Os R, Ausema A, Noach EJ,
Weersing E, Dontje B, Vellenga E and de Haan G: Impaired
hematopoietic stem cell functioning after serial transplantation
and during normal aging. Stem Cells. 23:82–92. 2005.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kwon Y, Kim J, Lee CY and Kim H:
Expression of SIRT1 and SIRT3 varies according to age in mice. Anat
Cell Biol. 48:54–61. 2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Brown K, Xie S, Qiu X, Mohrin M, Shin J,
Liu Y, Zhang D, Scadden DT and Chen D: SIRT3 reverses
aging-associated degeneration. Cell Rep. 3:319–327. 2013.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Lu J, Cheng K, Zhang B, Xu H, Cao Y, Guo
F, Feng X and Xia Q: Novel mechanisms for superoxide-scavenging
activity of human manganese superoxide dismutase determined by the
K68 key acetylation site. Free Radic Biol Med. 85:114–126.
2015.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Chang HC and Guarente L: SIRT1 and other
sirtuins in metabolism. Trends Endocrinol Metab. 25:138–145.
2014.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Morris BJ, Willcox DC, Donlon TA and
Willcox BJ: FOXO3: A major gene for human longevity-a mini-review.
Gerontology. 61:515–525. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Das S, Mitrovsky G, Vasanthi HR and Das
DK: Antiaging properties of a grape-derived antioxidant are
regulated by mitochondrial balance of fusion and fission leading to
mitophagy triggered by a signaling network of
Sirt1-Sirt3-Foxo3-PINK1-PARKIN. Oxid Med Cell Longev.
2014(345105)2014.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Gao J, Wang Y, Zhao X, Chen P and Xie L:
MicroRNA-204-5p-mediated regulation of SIRT1 contributes to the
delay of epithelial cell cycle traversal in diabetic corneas.
Invest Ophthalmol Vis Sci. 56:1493–1504. 2015.PubMed/NCBI View Article : Google Scholar
|