Introduction
Vaginal infections of bacterial vaginosis (BV) and
vulvovaginal candidiasis (VVC) are common in females, accounting
for almost 80% of all cases of vaginitis also known as
vulvovaginitis, is an inflammation of the vagina and vulva.
Symptoms may include itching, burning, pain, discharge and a bad
odor (1,2). While BV is generally regarded as a mild
disease, it has been indicated to be associated with the occurrence
of endometritis and pelvic inflammatory disease in females without
clinical symptoms of BV and may lead to spontaneous abortion,
premature rupture of the membranes, and premature delivery during
pregnancy (2,3). VVC results from overgrowth of one or
more types of yeast organism (e.g., Candida albicans) that
normally inhabit the vaginal mucosa in small numbers, and symptoms
include external dysuria, pruritus, redness and flocculant vaginal
discharge (2,4). In most cases, standard treatments with
antibiotics or anti-fungals are effective for BV and VVC. However,
the use of antibiotics may cause physiological and
non-physiological changes in patients, and interfere with the
balance of the normal vaginal microbiota. Thus, the common
side-effects of antibiotic treatment are characterized by reduction
or depletion of the Lactobacillus species and the excessive
growth of Candida species. In addition, excessive use of
antibiotics frequently causes the emergence of resistant
strains.
Probiotics are defined as ‘live microorganisms when
administered in adequate amounts confer a health benefit to the
host’ (5). Over the past 2 decades,
accumulating evidence has indicated that the intestinal and
urogenital microflora has a central role in maintaining the health
of human beings (5). In addition,
the use of beneficial bacteria to improve dysbiosis by replacing
pathogenic bacteria or augmenting normal microflora has been
gradually accepted and proven useful in conditions including
necrotizing enterocolitis and antibiotic-resistant infections
(5). The intestinal, vaginal and
urethral microflora have an important role in maintaining health
and preventing gynecologic infections in females, and the use of
probiotics has been extended to the treatment of refractory cases
of female urogenital infections (5).
The use of probiotics has been examined in a number
of studies over the past 2 decades as a method of treating and
reducing the risk and recurrence rate of gynecologic infections in
females, particularly in whom standard treatments are not
effective. Probiotics may protect the vagina from pathogen
colonization through a number of mechanisms, including blocking
potential sites of attachment, production of microbiocidal
substances, e.g. hydrogen peroxide, maintenance of a low pH and
induction of anti-inflammatory cytokine responses in epithelial
cells (3-5).
The most common probiotics used in female patients are of the
Lactobacillus species (3-5).
While numerous clinical trials have been performed
to determine the effectiveness of probiotics for the treatment of
vaginal infections, the results have generally been inconsistent,
with certain studies suggesting an excellent response and other
indicating no effect. Meta-analyses have also provided inconsistent
results. A meta-analysis by Huang et al (3) from 2014 indicated that probiotic
supplementation improves the cure rate for BV. Other previously
published systematic reviews have suggested that the use of
probiotics remains controversial in preventing BV and VVC in adult
females due to evidence limitations (4,6,7). Potential bias on the benefits of
probiotics cannot be ruled out, as the majority of evidence came
from small-scale studies, heterogeneous populations, different
lengths of follow-up and inhomogeneous treatment designs among the
study. Similar views were also expressed by a recently published
systemic review by Hanson et al (4) from 2016 with a focus on urogenital
infections in non-pregnant females, highlighting the requirement of
carefully-planned study stratification upon meta-analysis.
The purpose of the present study was to perform a
meta-analysis of randomized controlled trials (RCTs) and two-armed
prospective studies identified by a thorough systematic review and
meta-analysis of adequately-selected literature to determine the
effect of probiotics for the treatment of common vaginal infections
in non-pregnant adult females.
Materials and methods
Literature search strategy and
inclusion criteria
The present systematic review and meta-analysis was
performed in accordance with the Preferred Reporting Items for
Systematic Reviews and Meta-Analyses guidelines (8). On December 24th, 2018, the Pubmed,
Cochrane and EMBASE databases were searched for all studies
published previously using the following key words: ‘Probiotics’,
‘Lactobacillus’, ‘urogenital infections’, ‘bacterial
vaginosis’, ‘vulvovaginitis’, ‘vaginitis’ and ‘candidiasis’. The
search strategy was (probiotics or Lactobacillus) and
(vaginosis or vulvovaginal candidiasis or vaginitis or
vulvovaginitis or urogenital infections). Articles of interest were
also hand-searched for potentially relevant studies. Searches were
performed by 2 independent reviewers (HSJ and JYC) and any
disagreements were resolved by a third reviewer (TRY). Inclusion
criteria for the analysis were as follows: i) RCTs and two-armed
prospective studies; ii) studies including females with a current
or history of gynecologic infections of BV and/or VCC; iii) studies
that examined probiotic treatment vs. non-probiotics treatment
(control) with or without antibiotics; iv) studies that provided
quantitative data of the outcomes of interest; and v) full-text
articles published in English or Chinese. Exclusion criteria were
as follows: i) Retrospective studies, cohort studies, case series,
letters, comments, editorials, case reports, proceedings, personal
communications and one-arm studies; ii) studies on pediatric
patients, pregnant females or males; iii) studies on healthy
females with/without a history of recurrent urogenital infections.
Studies designed to examine Lactobacillus treatment in
combination with estriol, probiotic agents containing an unknown
number of Lactobacilli or a mixture of multiple types of
non-Lactobacillus bacteria were also excluded.
Data extraction
The following information/data was extracted from
studies that met the inclusion criteria: Name of the first author,
year of publication, study design, number of participants in each
group, participants' age, type of infection, type of interventions,
probiotic agents, probiotic administration, length of follow-up
period and major outcomes (recurrence rate, cure/remission rate
and/or the rate of restoring normal vaginal flora).
Quality assessment
The quality of the RCTs included was assessed using
the Cochrane ‘assessing risk of bias’ table, which consists of 6
domains (random sequence generation, allocation concealment,
blinding of patients and personnel, blinding of outcome assessment,
incomplete outcome data and selective reporting risk) (9). The quality of non-RCTs was assessed
using a Cochrane risk of bias assessment tool for non-randomized
studies of interventions (ACROBAT-NRSI) (10). This tool assesses 7 sources of bias
associated with confounding, selection of participants, measurement
of interventions, departures from intended interventions (10), missing data, measurement of outcomes
and selection of the reported result.
Statistical analysis
Outcome measures for the meta-analysis were
recurrence rate, cure and/or remission rate and restoration rate of
normal flora. The odds ratios (ORs) with 95% CIs were calculated
for each individual study and for all the studies combined. ORs of
<1 for recurrence and ORs of >1 for cure and/or remission
rate and normal flora restoration rate indicated that the probiotic
group was favored. By contrast, ORs of >1 for recurrence and ORs
of <1 for cure and/or remission rate and normal flora
restoration rate indicated the control group was favored. OR=1
indicated that the probiotic and control groups had comparable
outcomes. A χ2-based test of homogeneity was performed
and the inconsistency index (I2) and Q-statistics were
determined. A random effect model (DerSimonian-Laird method) was
considered for the meta-analysis if either the Q statistic of
P<0.10 or I2 value of >50% were derived;
otherwise, a fixed effect model (Mantel-Haenszel method) was
considered for the meta-analysis (11). Heterogeneity determined using the
I2 statistic was defined as follows: 0-24%, no
heterogeneity; 25-49%, moderate heterogeneity; 50-74%, high
heterogeneity; and 75-100%, extreme heterogeneity. When the number
of studies included in a meta-analysis is small, heterogeneity
tests have low statistical power (12) and in this situation, a random-effects
model of analysis is used (13). The
National Research Council recommends the use of random-effects
approaches for meta-analysis and the exploration of sources of
variation in study results (14).
Pooled effects were calculated and a 2-sided P<0.05 was
considered to indicate statistical significance. Sensitivity
analysis was performed using the leave-one-out approach to test the
validity and robustness of the major results (12). All analyses were performed using
Comprehensive Meta-Analysis statistical software, version 2.0
(Biostat).
Results
Literature search
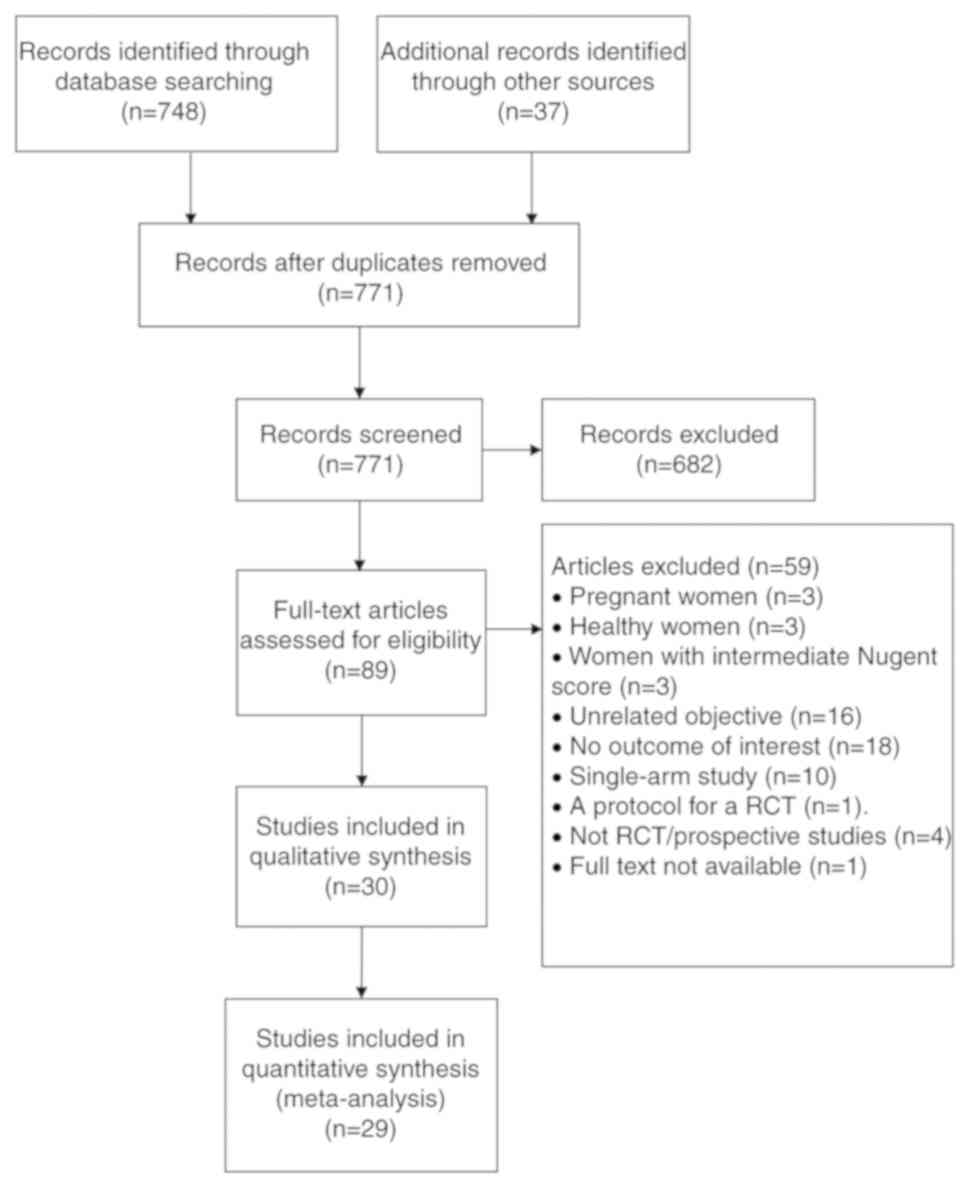
A flow diagram of the study selection process is
provided in Fig. 1. A total of 771
articles were identified by database- and hand-searching with
duplicates removed. After screening by title and abstract, 682
articles were excluded based on inclusion and exclusion criteria.
The full text of the remaining 89 articles was reviewed and 59 were
further excluded for reasons presented in Fig. 1. The remaining 30 articles were
included in the qualitative synthesis, including 20 studies for BV
alone or with other pathogens (15-34),
10 studies for VVC alone (31,35-43)
and 1 study for BV/VVC (44).
Characteristics of the reviewed
studies
Studies were categorized into three types based on
treatment design (Table I): Type I,
antibiotics plus Lactobacillus (probiotic) vs. antibiotic
with or without placebo (control; n=22) (15,17,18,20,22-26,29,31,32,35-41);
type II, Lactobacillus (probiotic) vs. placebo (control; no
antibiotics; n=5) (19,21,27,33,34); and
type III, Lactobacillus (probiotic) vs. antibiotic (control;
n=3) (16,28,30). A
summary of the patients' characteristics and interventions for the
treatment of BV and/or VVC is provided in Table I. The age range of the female
patients included in the analysis was between 18 to 50 years.
Table II presents a summary of the
outcomes of the studies included. Table III provides a summary of the type
of probiotic and the route and dose of administration for the
treatment of vaginitis. Probiotic species included L. rhamnosus
BMX54, L. fermentum, L. plantarum, L.
gasseri, L. plantarum, L. acidophilus, L.
brevis CD2, L. salivarius subsp. Salicinius, L.
delbrueckii subsp. lactis, L. reuteri, P.
acidilactici, L. casei rhamnosus, L. reuteris,
B. bifidum, B. longum, L. crispatus and
Lactobacillus GG either alone or in various combinations
depending on the infection being treated. The route of
administration included oral capsule, vaginal tablet and vaginal
capsule (Table III).
 | Table ISummary of patient characteristics
and interventions for the treatment of bacterial vaginosis. |
Table I
Summary of patient characteristics
and interventions for the treatment of bacterial vaginosis.
| A, Type I,
antibiotics plus Lactobacillus (probiotic) vs. antibiotic
with or without placebo (control) |
|---|
| | | | | | Disease | | | |
|---|
| First author
(publication year) | Study design | Grouping | Number of
patients | Age (years) | Diagnosis | Diagnostic
standard | Intervention | Follow-up time | (Refs.) |
|---|
| Laue (2018) | RCT | Probiotic | 18 | 32.6 | BV | Amsel criteria | Metronidazole plus
Lactobacillus | 4 weeks | (23) |
| | | Control | 18 | 39 | | | Metronidazole plus
placebo | | |
| Davar (2016) | RCT | Probiotic | 28 | 32.3 | VVC | Symptoms and
positive culture | Fluconazole plus
probiotic tablet | 6 months | (36) |
| | | Control | 31 | 31.1 | | | Fluconazole plus
placebo | | |
| Recine (2016) | Prospective | Probiotic | 125 | 29.3 | BV | ≥3 of Amsel
criteriaa | MTZ plus
Lactobacillus | 9 months | (32) |
| | | Control | 125 | 29.5 | | | MTZ plus
placebo | | |
| Heczko (2015) | RCT | Probiotic | 73 | 18-50 | BV/AV | Clinical signs,
NSb | MTZ plus
Lactobacillus | NA | (20) |
| | | Control | 81 | | | | MTZ plus
placebo | | |
| Bradshaw
(2012) | RCT | Probiotic | 140 | 27c | BV | NS 7-10 | MTZ plus
clindamycin cream | 6 months | (17) |
| | | Control | 133 | 27c | | or ≥3 of Amsel
criteria and NS 4-10 | MTZ plus vaginal
pessary containing Lactobacillus | | |
| | | Clindamycin | 135 | 27c | | | MTZ plus placebo
vaginal pessary | | |
| Nouraei (2012) | RCT | Probiotic | 45 | 18-40 | VVC | Symptoms and
culture | Fluconazole plus
probiotic | 5-7 days | (41) |
| | | Control | 45 | | | | Fluconazole plus
placebo | | |
| Ehrström
(2010) | RCT | Probiotic | 60 | 18-45 | BV/VVC | ≥3 Amsel
criteria | Antibiotics plus
vaginal capsule containing Lactobacillus | 1 menstruation | (44) |
| | | Control | 35 | | | | Antibiotics plus
placebo vaginal capsule | | |
| Marcone (2010) | RCT | Probiotic | 24 | N/A | BV | Fulfilled all | MTZ plus
Lactobacillus tablet | 12 months | (25) |
| | | Control | 25 | | | Amsel criteria | MTZ plus placebo
tablet | | |
| Anukam (2009) | RCT | Probiotic | 19 | 18-50 | VVC | Positive
culture | Fluconazole plus
L. rhamnosus GR-1 | 3 months | (35) |
| | | Control | 7 | | | | Fluconazole plus
placebo capsule | | |
| Martinez
(2009a) | RCT | Probiotic | 32 | N/A | BV | ≥3 Amsel
criteria | Tinidazole plus
Lactobacillus | 28 days | (26) |
| | | Control | 32 | | | or NS 7-10 | Tinidazole plus
placebo capsule | | |
| Martinez
(2009b) | RCT | Probiotic | 29 | 29.1±7.5 | VVC | Symptoms and
positive culture | Fluconazole plus
L. rhamnosus GR-1 and L. reuteri RC-14 | 4 wks | (43) |
| | | Control | 26 | 26.9±7.8 | | | Fluconazole plus
placebo | | |
| Yang (2009) | RCT | Probiotic | 44 | 36 (range:
25-48) | VVC | Symptoms and
microscopy | Clotrimazole plus
Lactobacillus | 30 days | (42) |
| | | Control | 42 | | | | Clotrimazole | | |
| Hua (2008) | RCT | Probiotic | 118 | 28.45 | VVC | Symptoms and
microscopy | Miconazole plus
Lactobacillus | 33-37 days | (38) |
| | | Control | 117 | | | | Miconazole | | |
| Larsson (2008) | RCT | Probiotic | 50 | 34.3 | BV | Amsel criteria | Clindamycin plus
Lactobacillus | 6 months | (22) |
| | | Control | 50 | | | | Clindamycin plus
placebo | | |
| Marcone (2008) | RCT | Probiotics | 42 | 18-40 | BV | Fulfilled all | MTZ plus
Lactobacillus tablet | 1 month | (24) |
| | | Control | 42 | | | Amsel criteria | MTZ plus placebo
tablet | | |
| Petricevic
(2008) | RCT | Probiotics | 83 | 32.6 | BV | NS 7-10 | Clindamycin plus
Lactobacillus | 1 month | (29) |
| | | Control | 88 | | | | Clindamycin plus
placebo | | |
| Ma (2007) | RCT | Probiotics | 54 | 26 | VVC | Symptoms and
microscopy | Miconazole plus
Lactobacillus | NA | (39) |
| | | Control | 54 | | | | Miconazole | | |
| Mai (2007) | RCT | Probiotics | 85 | 30.1 (range:
20-47) | VVC | Symptoms and
microscopy | Clotrimazole plus
Lactobacillus | 30 days | (40) |
| | | Control | 84 | | | | Clotrimazole | | |
| Anukam (2006a) | RCT | Probiotic | 65 | 18-44 | BV | NS 7-10 and
positive BV Blue test | MTZ plus
Lactobacillus tablet | 30 days | (15) |
| | | Control | 60 | | | | MTZ plus placebo
tablet | | |
| Han (2006) | RCT | Probiotic | 86 | 37 (range:
19-48) | VVC | Symptoms and
microscopy | Clotrimazole plus
lactobacillus capsule | 30 days | (37) |
| | | Control | 90 | | | | Clotrimazole | | |
| Lin (2006) | RCT | Probiotic | 32 | 30 (range:
20-44) | Trichomonial | Microscopy | MNZ plus
lactobacillus capsule | 30 days | (31) |
| | | Control | 30 | | Vaginitis | | | | |
| | | Probiotic | 53 | | VVC | Microscopy | Cretrozole plus
lactobacillus capsule | | |
| | | Control | 52 | | | | | | |
| | | Probiotic | 59 | | BV | Amsel criteria | MNZ plus
Lactobacillus capsule | | |
| | | Control | 51 | | | | | | |
| Eriksson
(2005) | RCT | Probiotics | 91 | 32c | BV | ≥3 Amsel
criteria | Clindamycin plus
tampons containing Lactobacillus | NA | (18) |
| | | Control | 96 | 32c | | | Clindamycin plus
placebo tampons | | |
| B, Type II studies:
Lactobacillus (probiotic) vs. placebo (control) |
| | | | | | Disease | | | |
| First author
(publication year) | Study design | Grouping | Number of
patients | Age (years) | Diagnosis | Diagnostic
standard | Intervention | Follow-up time | (Refs.) |
| Vicariotto
(2014) | RCT | Probiotic | 24 | 34.7 | BV | ≥3 of Amsel
criteria |
Lactobacillus | 56 days | (33) |
| | | Control | 10 | | | or NS 7-10 | Placebo tablet | | |
| Vujic (2013) | RCT | Probiotic | 395 | N/A | BV and | ≥3 Amsel
criteria |
Lactobacillus capsule | 44.16 days | (34) |
| | | Control | 149 | | Other vaginal
infections | or NS 7-10 | Placebo
capsule | | |
| Hemalatha
(2012) | RCT | Probiotic | 34 | N/A | BV | NS 7-10 |
Lactobacillus | 9 days | (21) |
| | | Control | 27 | | | | Placebo tablet | | |
| Mastromarino
(2009) | RCT | Probiotics | 18 | 33 | BV | NS 7-10 | Vaginal tablets
containing Lactobacillus | 2 weeks | (27) |
| | | Control | 16 | 35 | | | Placebo vaginal
tablets | | |
| Hallen (1992) | RCT | Probiotics | 28 | 24 | BV | ≥3 Amsel
criteria | Vaginal tablets
containing Lactobacillus | 7-10 days; 20-40
days | (19) |
| | | Control | 29 | | | | Placebo vaginal
tablets | | |
| C, Type III
studies: Lactobacillus (probiotic) vs. antibiotics
(control) |
| | | | | | Disease | | | |
| First author
(publication year) | Study design | Grouping | Number of
patients | Age (years) | Diagnosis | Diagnostic
standard | Intervention | Follow-up time | (Refs.) |
| Ling (2013) | RCT | Probiotic | 25 | N/A | BV | Amsel criteria and
NS | Intravaginal
Lactobacillus | 30 days | (30) |
| | | Control | 30 | | | | MTZ | | |
| Anukam (2006b) | RCT | Probiotic | 20 | N/A | BV | NS7-10 and positive
BV Blue test |
Lactobacillus capsule | 30 days | (16) |
| | | Control | 20 | | (Symptomatic) | | MTZ | | |
| Parent (1996) | RCT | Probiotics | 16 | 31.1 | BV | ≥3 Amsel
criteria |
Lactobacillus tablet | 28 days | (28) |
| | | Control | 16 | 34.4 | | | MTZ plus placebo
tablet | | |
 | Table IISummary of the outcomes in the
meta-analysis. |
Table II
Summary of the outcomes in the
meta-analysis.
| A, Type I |
|---|
| First author
(year) | Disease type | Patients (n) | Intervention | Recurrence | Cure/remission | Restored normal
flora | (Refs.) |
|---|
| Laue (2018) | BV | 18 | Probiotic | | 16(100) | | (23) |
| | | 18 | Control | | 13 (76.5) | | |
| Davar (2016) | VVC | 28 | Probiotic | 2 (7.2) | | | (36) |
| | | 31 | Control | 11 (35.5) | | | |
| Recine (2016) | BV | 125 | Probiotic | | | 2 mo: 113 (90.4) 6
mo: 106 (74.6) 9 mo: 118 (79.7) | (32) |
| | | 125 | Control | | | 2 mo: 99 (79.2) 6
mo: 36 (25.4) 9 mo: 30 (20.3) | |
| Heczko (2015) | BV/AV | 73 | Probiotic | 33 (45.2) | | | (20) |
| | | 81 | Control | 38 (47.0) | | | |
| Bradshaw
(2012) | BV | 140 | Clindamycin | 42(30) | | 92 (65.7) | (17) |
| | | 133 | Probiotic | 37 (27.8) | | 63 (47.4) | |
| | | 135 | Control | 36 (26.7) | | 63 (46.7) | |
| Nouraei (2012) | VVC | 45 | Probiotic | | 42 (93.3) | | (41) |
| | | 45 | Control | | 37 (82.2) | | |
| Ehrström
(2010) | BV/VVC | 60 | Probiotic | 1 mo: 13
(22.4) | 1 mo: 47(78) 2 mo:
23 (38.1) 6 mo: 35 (58.4) | | (44) |
| | | 35 | Control | 1 mo: 10 (29.4) 2
mo: 13 (38.1) 6 mo: 20 (56.6) | 1 mo: 25(71) | | |
| Marcone (2010) | BV | 24 | Probiotic | | | 6 mo: 18(74) | (25) |
| | | | | | | 12 mo: 16(69) | |
| | | 25 | Control | | | 6 mo: 24(96) 12 mo:
23(91) |
| Anukam (2009) | VVC | 19 | Probiotic | | 15(79) | | (35) |
| | | 7 | Control | | 3(43) | | |
| Martinez
(2009a) | BV | 32 | Probiotic | 4 (12.5) | 28 (87.5) | 24(75) | (26) |
| | | 32 | Control | 15 (46.9) | 16(50) | 11 (34.4) | |
| Martinez
(2009b) | VVC | 29 | Probiotic | 3 (10.3) | | | (43) |
| | | 26 | Control | 10 (38.5) | | | |
| Yang (2009) | VVC | 44 | Probiotic | 3 (7.1) | 42 (92.86) | | (42) |
| | | 42 | Control | 7 (16.7) | 38 (83.33) | | |
| Hua (2008) | VVC | 118 | Probiotic | 4 (4.8) | 83 (70.34) | | (38) |
| | | 117 | Control | 11 (13.9) | 79 (67.52) | | |
| Larsson (2008) | BV | 50 | Probiotics | | 24 (64.9) | | (22) |
| | | 50 | Control | | 18 (46.2) | | |
| Marcone (2008) | BV | 42 | Probiotics | | 1 mon: 22(96) 6
mon: 23(98) | 30 d: 37(88) 90 d:
37(88) 180 d: 35(83) | (24) |
| | | 42 | Control | | 1 mon: 21(91) 6 mo:
17(74) | 30 d: 34(81) 90 d:
30(71) 180 d: 28(67) | |
| Petricevic
(2008) | BV | 83 | Probiotics | | 1 mon: 83(100) | 69 (83.1) | (29) |
| | | 88 | Control | | 1 mon: 35
(39.8) | 31(35.2) | |
| Ma (2007) | VVC | 54 | Probiotics | | 46 (85.2) | | (39) |
| | | 54 | Control | | 38 (70.4) | | |
| Mai (2007) | VVC | 85 | Probiotics | 5 (5.9) | 80 (94.1) | | (40) |
| | | 84 | Control | 13 (15.5) | 70 (83.3) | | |
| Anukam (2006a) | BV | 65 | Probiotic | 0 (0) | 8(12) | 57(88) | (15) |
| | | 60 | Control | 17(28) | 19(32) | 24(40) | |
| Han (2006) | VVC | 86 | Probiotic | 3 (3.9) | 74 (96.10) | | (37) |
| | | 90 | Control | 9 (13.0) | 60 (86.96) | | |
| Lin (2006) | VVC | 53 | Probiotic | 2 (3.8) | 52 (98.1) | | (31) |
| | | 52 | Control | 13 (25.0) | 49 (94.2) | | |
| Lin (2006) | BV | 59 | Probiotic | 1 (1.7) | 58 (98.3) | | (31) |
| | | 51 | Control | 12 (23.5) | 47 (92.2) | | |
| Eriksson
(2005) | BV | 91 | Probiotics | | 52 (56.8) | | (18) |
| | | 96 | Control | | 58 (60.2) | | |
| B, Type II |
| First author
(year) | Disease type | Patients (n) | Intervention | Recurrence | Cure/remission | Restored normal
flora | (Refs.) |
| Vicariotto
(2014) | BV | 24 | Probiotic | Day 28: 2 (8.3) Day
56: 4 (16.7) | Day 28: 22 (91.7)
Day 56: 20 (83.3) | | (33) |
| | | 10 | Control | Day 28: 8(80) Day
56: 9(90) | Day 28: 2(20) Day
56: 1(10) | | |
| Vujic (2013) | BV and other
infection | 395 | Probiotic | | | 1.5 mo: 243 (61.5)
3 mo: 202 (51.1) | (34) |
| | | 149 | Control | | | 1.5 mo: 40 (26.8) 3
mo: 31 (20.8) |
| Hemalatha
(2012) | BV | 34 | Probiotic | 7(21) | | 11(32) | (21) |
| | | 27 | Control | 7(26) | | 7(26) | |
| Mastromarino
(2009) | BV | 18 | Probiotics | | 11(61) | 9(50) | (27) |
| | | 16 | Control | | 3 (18.75) | 1 (6.25) | |
| Hallen (1992) | BV | 28 | Probiotics | | 7-10 d: 16 (57.1)
20-40 d: 0 (0) | | (19) |
| | | 29 | Control | | 7-10 d: 3 (10.3)
20-40 d: 0 (0) | | |
| C, Type III |
| First author
(year) | Disease type | Patients (n) | Intervention | Recurrence | Cure/remission | Restored normal
flora | (Refs.) |
| Ling (2013) | BV | 25 | Probiotic | | | | (30) |
| | | 30 | Control | | | | |
| Anukam (2006b) | BV | 20 | Probiotic | 2(10) | 15(75) | 11(55) | (16) |
| | | 20 | Control | 9(45) | 9(45) | 6(30) | |
| Parent (1996) | BV | 16 | Probiotics | | 14 (87.5) | | (28) |
| | | 16 | Control | | 4 (22.2) | | |
 | Table IIITypes of probiotics and route and
dose of administration of probiotics for treatment. |
Table III
Types of probiotics and route and
dose of administration of probiotics for treatment.
| A, Disease type,
BV |
|---|
| First author
(year) | Probiotic
regimen | Brand | Dosage and
duration | Route of
administration | Length of follow-up
period | (Refs.) |
|---|
| Laue (2018) |
Lactobacillus | Verum | 125 g yoghurt
containing (besides L. delbrueckii ssp. bulgaricus and S.
thermophilus) living strains L. crispatus LbV 88, L.
gasseri LbV 150N, L. jensenii LbV 116 and L.
rhamnosus LbV96, each 1x107 cfu/ml; placebo was 125
g chemically acidified milk. Twice daily for 4wks | Oral | 4 wks | (23) |
| Recine (2016) | L. rhamnosus
BMX54 | NORMOGIN | Once a day for 10
d, twice a week for 15 d and once every 5 d for 7 mo as maintenance
therapy | Vaginal tablet | 9 mo | (32) |
| Heczko (2015) | L.
fermentum, L. plantarum, and L. gasseri | prOVag | One capsule daily
for 10 d perimenstrually | Oral | Approximately 4
menstrual periods | (20) |
| Vicariotto
(2014) | L. fermentum
plus L. plantarum | N/A | Once a day for 7
nights, followed by 1 tablet every 3 nights for 3 wks and 1 tablet
per wk | Vaginal tablet | 56 d | (33) |
| Bradshaw
(2012) | L.
acidophilus | N/A | 12 nights | Vaginal tablet | 6 mo | (17) |
| Hemalatha
(2012) | L. brevis
CD2, L. salivarius subsp. Salicinius, L.
plantarum | Florisia | 8 nights | Vaginal tablet | 9 d | (21) |
| Ling (2013) | L.
delbrueckii subsp. lactis | N/A | 10 d | Vaginal
capsule | 30 d | (30) |
| Vujic (2013) | L. rhamnosus
and L. reuteri | Lactogyn | Twice daily | Oral | 6 mo | (34) |
| Ehrström
(2010) | L gasseri,
L. fermentum, L. casei subsp. rhamnosus and
P. acidilactici | N/A | 2 capsules daily
for 5 d | Vaginal
capsule | 6 mo | (44) |
| Marcone (2010) | L.
rhamnosus | Normogin | Once a week for 6
mo of a capsule containing 40 mg of L. rhamnosus (N40000
CFU; Normogin), beginning 8 d after MTZ discontinued | Oral | 12 mo | (25) |
| Martinez
(2009a) | GR-1, RC-14 | | 2 Capsule daily for
the following 4 wks | Vaginal
capsule | 28 d | (26) |
| Mastromarino
(2009) | L. brevis,
L. salivarius subsp. salicinius, and L.
plantarum | N/A | Daily for 7 d | Vaginal tablet | 2 wks | (27) |
| Larsson (2008) | L. gasseri
and L. rhamnosus |
EcoVag® | 10 d during 3
menstrual cycles | Vaginal
capsule | 6 mo | (22) |
| Marcone (2008) | L.
rhamnosus | N/A | Once a week at
bedtime for 2 mo starting 1 wk after the last antibiotic
administration | Vaginal tablet | | (24) |
| Petricevic
(2008) | L.
caseirhamnosus | N/A | 7 d | Vaginal
capsule | 1 mo | (29) |
| Anukam (2006a) | L. rhamnosus
and L. reuteri | N/A | 1-30 d | Vaginal tablet | 30 d | (15) |
| Anukam (2006b) | L. rhamnosus
and L. reuteri | N/A | 2 capsules for 5
d | Vaginal tablet | 30 d | (16) |
| Eriksson
(2005) | L. gasseri,
L. caseivarrhamnosus and L. fermentum | Medipharm AB | During the
following menstruation | Tampon containing
lactobacilli | After the second
menstrual period (~1 mo) | (18) |
| Parent (1996) | L.
acidophilus | Gynoflor | 1-2 tablets daily
for 6 d | Vaginal tablet | 28 d | (28) |
| Hallen (1992) | L.
acidophilus (Vivag) | Vigag | Twice daily for 6
d | Vaginal
capsule | 7-10 d, 20-40
d | (19) |
| Lin (2006) |
Lactobacillus capsule | | Once daily for 7
d | Vaginal
capsule | 30 d | (31) |
| B, Disease type,
VVC |
| First author
(year) | Probiotic
regimen | Brand | Dosage and
duration | Route of
administration | Length of follow-up
period | (Refs.) |
| Davar (2016) | L.
acidophilus, B. bifidum and B. longum | Pro-Digest | Twice daily for 10
d | Oral | 6 mo | (36) |
| Anukam (2009) | L. rhamnosus
and L. reuteri | N/A | Once daily for 3
mo | Oral | 3 mo | (35) |
| Martinez
(2009b) | L. rhamnosus
and L. reuteri | N/A | Once daily for 28
d | Oral | 4 wks | (43) |
| Nouraei (2012) | | protexin | 20 capsules within
an interval of 72 h (3 d) | Oral | 5-7 d | (41) |
| Yang (2009) |
Lactobacillus capsule | Ding-jun-sheng | Once daily for 10
d | Vaginal
capsule | 30 d | (42) |
| Hua (2008) |
Lactobacillus capsule | Ding-jun-sheng | Once daily for 10
d | Vaginal
capsule | 33-37 d | (38) |
| Ma (2007) |
Lactobacillus capsule | N/A | 0.5 g once daily
for 7 d | Vaginal
capsule | N/A | (39) |
| Mai (2007) |
Lactobacillus capsule | Ding-jun-sheng | 0.25 g capsule,
once daily for 10 d | Vaginal
capsule | 30 d | (40) |
| Han (2006) |
Lactobacillus capsule | Ding-jun-sheng | Once daily for 10
d | Vaginal
capsule | 30 d | (37) |
| Lin (2006) |
Lactobacillus capsule | N/A | Once daily for 7
d | Vaginal
capsule | 30 d | (31) |
Meta-analysis
The detailed treatment outcomes of all studies
reviewed are summarized in Table
II. The majority of studies adopted a type I treatment design
for BV and/or VVC infections and those with 1- and/or 6-months
follow-up data were included in the meta-analysis. These comprised
of a total of 21 articles (10 articles on BV, 9 studies on VVC and
2 on BV/VVC) (15,17,18,20,22-26,29,31,32,35-41).
The total number of patients evaluated in the 21 type I studies was
1,788 (probiotic test group, n=910; control group, n=878). These
type I studies were the major focus of the present meta-analysis,
while type II and III studies were analyzed separately for
supplementation.
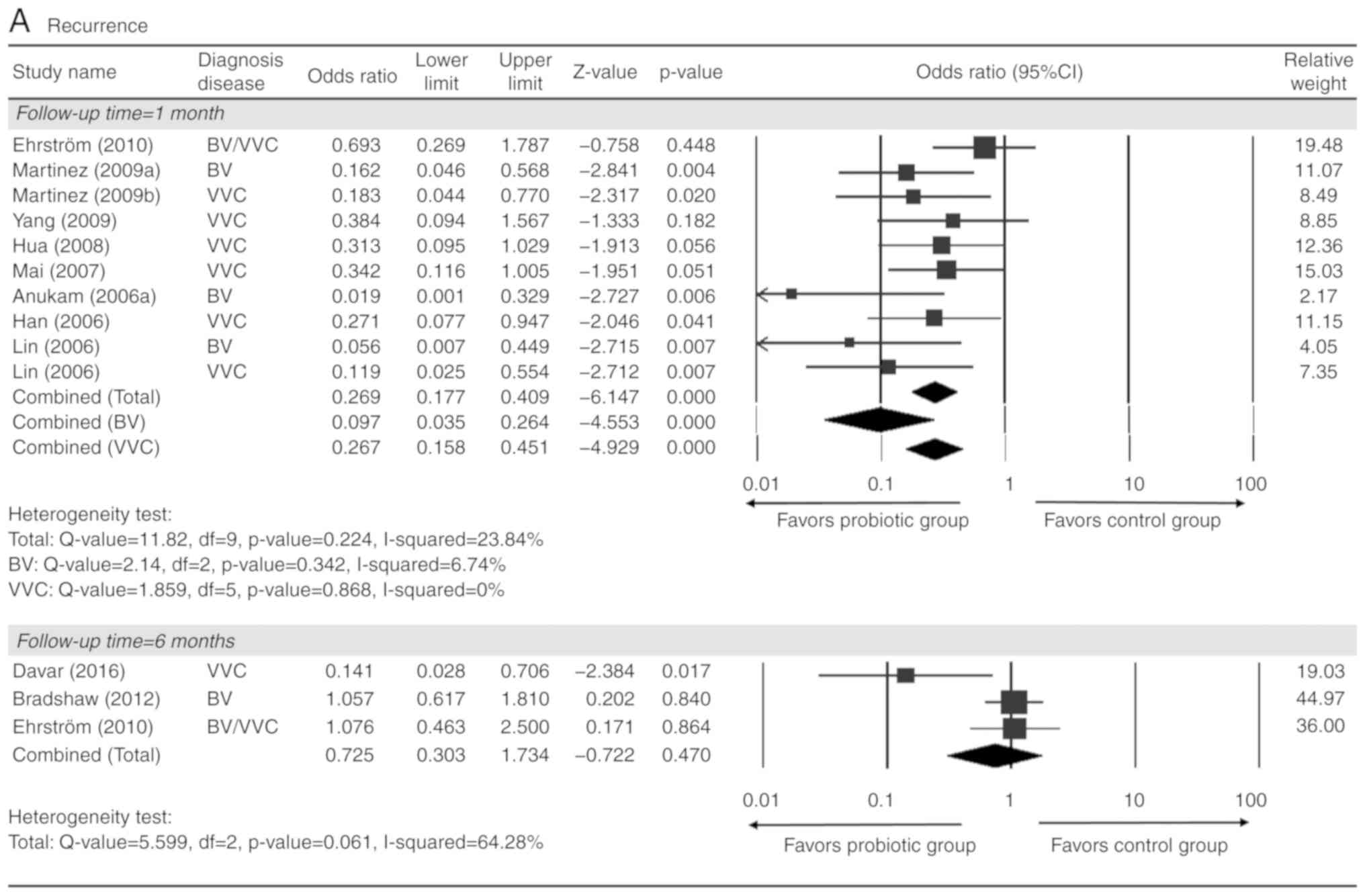
With respect to recurrence at 1 month after
treatment, 9 studies [2 on BV alone (15,26), 5
on VVC alone (37,38,40,42,43) and
2 on BV/VVC (31,44)] with complete quantitative data were
included in the present meta-analysis. A total of 1,220 patients
were evaluated (probiotic test group, n=631; control group, n=589).
There was no heterogeneity present among all 9 studies or those on
either BV or VVC (total: Q=11.82, I2=24%; BV: Q=2.14,
I2=7%; VVC: Q=1.86, I2=0%; Fig. 2A). The analysis indicated that
patients in the probiotic group had a significantly lower
recurrence rate than those in the control group (pooled OR=0.27,
95% CI: 0.18-0.41; Fig. 2A). A
favorable outcome associated with the probiotics group was also
observed when analyzing BV and VVC individually (BV: Pooled
OR=0.10, 95% CI: 0.04-0.26; VVC: Pooled OR=0.27, 95% CI: 0.16-0.45;
all P<0.001; Fig. 2A). However,
there was no significant difference in the recurrence rate between
the probiotic and control groups at 6 months after treatment
(Fig. 2A).
With respect to cure or remission after treatments,
a total of 12 studies were included. These comprised 12 studies
with 1-month follow-up results [6 for BV alone (15,18,23,24,26,29), 4
for VVC alone (37,38,40,42) and
2 for BV/VVC (31,44)] and 2 studies (22,24) with
6-month follow-up for BV alone. In the 12 studies with 1-month
follow-up outcomes, 1,643 patients in total were evaluated
(probiotic test group, n=836; control group, n=807). There was
moderate to high heterogeneity among the 12 studies with 1-month
follow-up (total: Q=52.69, I2=77%; BV: Q=47.02,
I2=87. %; VVC: Q=5.45, I2=27%), as well as
between studies with 6-month follow-up (Q=1.70, I2=40%).
The analysis indicated that probiotic treatment was favorable among
all studies and those focusing on VVC alone 1 month after treatment
(total: Pooled OR=2.28, 95% CI: 1.21-4.32, P=0.011; VVC: Pooled
OR=1.72, 95% CI: 1.13-2.64, P=0.012), as well as 6 months after
treatment of BV (pooled OR=2.58, 95% CI: 1.07-6.23, P=0.036;
Fig. 2B). However, there was no
significant difference in the cure rate at 1 month for BV (pooled
OR=2.59; 95% CI: 0.76-8.85; P=0.129; Fig. 2B).
With respect to restoration of the normal flora, 4
studies (15,24,26,29) had
complete quantitative data at 1 month and 4 studies (17,24,25,32) at 6
months after treatments for BV, and were included in the analysis.
High heterogeneity existed among the studies on the restoration of
normal flora at 1 month and 6 months after treatment (1 month:
Q=17.28, I2=83%; 6 months: Q=47.86, I2=94%).
The analysis indicated that patients in the probiotic group had a
significantly higher rate of normal flora restoration at 1 month
after treatment (pooled OR=4.55, 95% CI: 1.44-14.36, P=0.010).
However, there were no differences in the normal flora restoration
rate between the two groups at 6 months after treatment (Fig. 2C).
Additional analyses were performed for type II
(19,21,27,33,34) or
type III (16,28) studies that had at least one follow-up
outcome. These studies all focused on BV and had varied
heterogeneity (Recurrence: Q=7.98; I2=87%; Cure or
remission: Q=1.94; I2=0%; Restored normal flora: Q=4.37;
I2=54% for type II and Cure or remission: Q=2.58;
I2=61%; for type III). Patients with BV given type II
treatments in the probiotic group were indicated to have a higher
cure or remission rate and normal flora restoration rate than those
in the control group (cure/remission rate: Pooled OR=12.44, 95% CI:
4.86-31.89, P<0.001; normal flora restoration rate: Pooled
OR=3.32, 95% CI: 1.11-9.97, P=0.033). In BV patients given type III
treatments, the probiotic group had a higher cure/remission rate
than the control group (cure/remission rate: Pooled OR=8.39, 95%
CI: 1.32-53.23, P=0.024; Table
IV).
 | Table IVExtra meta-analysis for recurrence,
cure or remission, and restored normal flora for patients with BV
given type II and type III treatment. |
Table IV
Extra meta-analysis for recurrence,
cure or remission, and restored normal flora for patients with BV
given type II and type III treatment.
| Outcome/study
[Author name (year) (Refs.)] | Treatment type | Time-point | Odds ratio | Lower limit | Upper limit | Z-value | P-value | Heterogeneity
test |
|---|
| Recurrence | | | | | | | | Q=7.977, df=1,
P=0.005, I2=87.46% |
|
Vicariotto
(2014) (33) | BV-Type II | 1 mon | 0.023 | 0.003 | 0.189 | -3.498 | <0.001 | |
|
Hemalatha
(2012) (21) | BV-Type II | 9 d | 0.757 | 0.230 | 2.492 | -0.459 | 0.646 | |
|
Combined | | | 0.147 | 0.005 | 4.529 | -1.096 | 0.273 | |
| Cure or
remission | | | | | | | | Q=1.939, df=2,
P=0.379, I2=0% |
|
Vicariotto
(2014) (33) | BV-Type II | 1 mon | 44.019 | 5.280 | 366.956 | 3.498 | <0.001 | |
|
Mastromarino
(2009) (27) | BV-Type II | 14 d | 6.806 | 1.412 | 32.809 | 2.390 | 0.017 | |
|
Hallen
(1992) (19) | BV-Type II | 40 d | 11.560 | 2.821 | 47.366 | 3.401 | 0.001 | |
|
Combined | | | 12.444 | 4.856 | 31.888 | 5.251 | <0.001 | |
| Restored normal
flora | | | | | | | | Q=4.366, df=2,
P=0.113, I2=54.19% |
|
Vujic (2013)
(34) | BV-Type II | 3 mon | 3.983 | 1.603 | 9.895 | 2.977 | 0.003 | |
|
Hemalatha
(2012) (21) | BV-Type II | 9 d | 1.339 | 0.436 | 4.113 | 0.510 | 0.610 | |
|
Mastromarino
(2009) (27) | BV-Type II | 14 d | 15.000 | 1.621 | 138.821 | 2.385 | 0.017 | |
|
Combined | | | 3.319 | 1.105 | 9.974 | 2.137 | 0.033 | |
| Restored normal
flora | | | | | | | | |
|
Anukam
(2006b) (16) | BV-Type III | 1 mon | 2.852 | 0.777 | 10.467 | 1.580 | 0.114 | Not assessed |
| Recurrence | | | | | | | | |
|
Anukam
(2006b) (16) | BV-Type III | 1 mon | 0.136 | 0.025 | 0.748 | -2.294 | 0.022 | Not assessed |
| Cure or
remission | | | | | | | | Q=2.577, df=1,
P=0.108, I2=61.19% |
|
Anukam
(2006b) (16) | BV-Type III | 1 mon | 3.667 | 0.958 | 14.028 | 1.898 | 0.058 | |
|
Parent
(1996) (28) | BV-Type III | 28d | 24.532 | 3.693 | 162.947 | 3.312 | 0.001 | |
|
Combined | | | 8.393 | 1.323 | 53.227 | 2.257 | 0.024 | |
Quality assessment
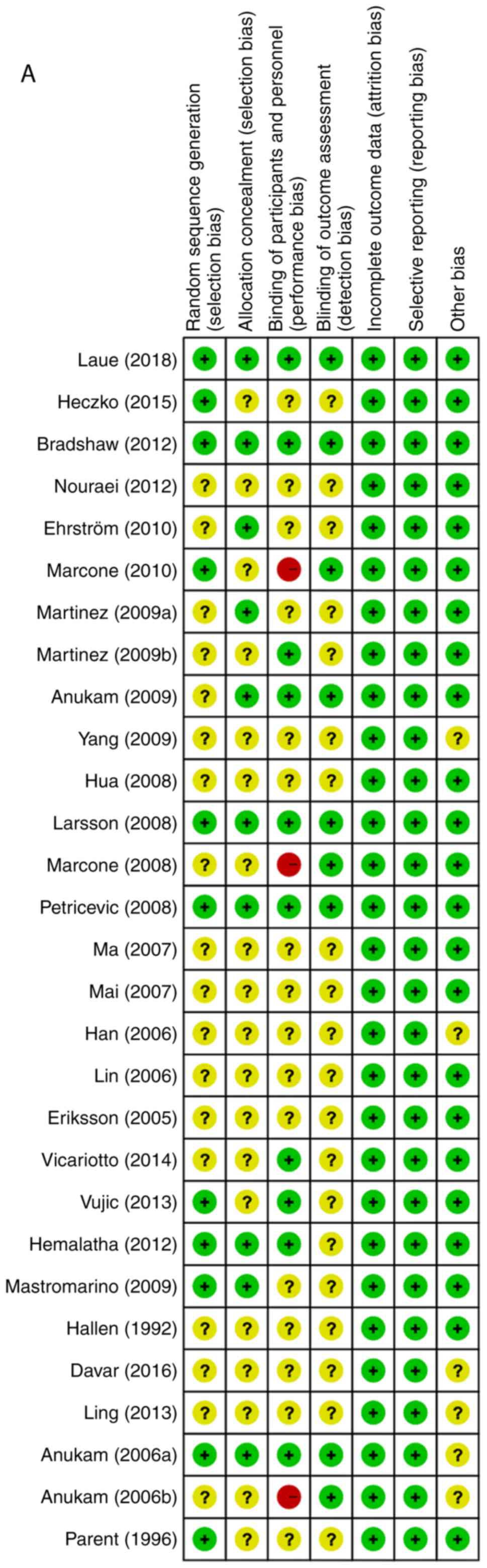
The risk of bias assessment for individual studies
is provided in Fig. 3, including the
potential risk of individual studies (Fig. 3A and B) and the overall risk (Fig. 3C and D). Overall, the studies had a low risk of
attrition bias and reporting bias, and low or unclear risk of
selection bias and detection bias. Furthermore, 3 studies had a
high risk of performance bias due to improper blinding of
participants and researchers.
Sensitivity analysis
Sensitivity analyses were performed on the major
results using the leave-one-out approach, in which the
meta-analysis was performed with each study removed in turn
(Table V). The direction of combined
estimates on recurrence rates and cure/remission rates at 1 month
and normal flora restoration rates at 6 months did not vary
markedly with the removal of the studies, indicating that the
meta-analysis had good reliability and supported that there was no
or little inter-study heterogeneity. However, for normal flora
restoration rates at 1 month, the study of Marcone et al
(24) from 2008 may have had a
disproportionate effect on the pooled OR, as the difference became
more significant and greater when this study was not included in
the meta-analysis, while the three other studies had no such
effect.
 | Table VSensitivity analysis. |
Table V
Sensitivity analysis.
| A, Recurrence at 1
month |
|---|
| | Statistics with
study removed | |
|---|
| Author name
(year) | Odds ratio | Lower limit | Upper limit | Z-value | P-value | (Refs.) |
|---|
| Ehrström
(2010) | 0.214 | 0.135 | 0.342 | -6.478 | <0.001 | (44) |
| Martinez
(2009a) | 0.287 | 0.184 | 0.447 | -5.516 | <0.001 | (26) |
| Martinez
(2009b) | 0.279 | 0.180 | 0.432 | -5.720 | <0.001 | (43) |
| Yang (2009) | 0.260 | 0.168 | 0.403 | -6.023 | <0.001 | (42) |
| Hua (2008) | 0.264 | 0.169 | 0.412 | -5.848 | <0.001 | (38) |
| Mai (2007) | 0.258 | 0.164 | 0.407 | -5.848 | <0.001 | (40) |
| Anukam (2006a) | 0.286 | 0.187 | 0.436 | -5.809 | <0.001 | (15) |
| Han (2006) | 0.269 | 0.173 | 0.420 | -5.797 | <0.001 | (37) |
| Lin (2006) | 0.288 | 0.188 | 0.441 | -5.718 | <0.001 | (31) |
| Lin (2006) | 0.288 | 0.186 | 0.444 | -5.622 | <0.001 | (31) |
| B, Cure or
remission at 1 month |
| | Statistics with
study removed | |
| Author name
(year) | Odds ratio | Lower limit | Upper limit | Z-value | P-value | (Refs.) |
| Laue (2018) | 2.165 | 1.131 | 4.146 | 2.330 | 0.020 | (23) |
| Ehrström
(2010) | 2.416 | 1.197 | 4.879 | 2.461 | 0.014 | (44) |
| Martinez
(2009a) | 2.062 | 1.069 | 3.979 | 2.158 | 0.031 | (26) |
| Yang (2009) | 2.271 | 1.150 | 4.486 | 2.361 | 0.018 | (42) |
| Hua (2008) | 2.521 | 1.209 | 5.256 | 2.467 | 0.014 | (38) |
| Marcone (2008) | 2.286 | 1.179 | 4.431 | 2.447 | 0.014 | (24) |
| Petricevic
(2008) | 1.818 | 1.047 | 3.155 | 2.123 | 0.034 | (29) |
| Mai (2007) | 2.224 | 1.117 | 4.427 | 2.275 | 0.023 | (40) |
| Han (2006) | 2.764 | 1.523 | 5.015 | 3.345 | 0.001 | (37) |
| Lin (2006) | 2.197 | 1.114 | 4.333 | 2.271 | 0.023 | (31) |
| Lin (2006) | 2.199 | 1.136 | 4.259 | 2.337 | 0.019 | (31) |
| Eriksson
(2005) | 2.252 | 1.160 | 4.372 | 2.398 | 0.016 | (18) |
| Anukam (2006a) | 2.578 | 1.280 | 5.193 | 2.651 | 0.008 | (15) |
| C, Restoration of
normal flora at 1 month |
| | Statistics with
study removed | |
| Author name
(year) | Odds ratio | Lower limit | Upper limit | Z-value | P-value | (Refs.) |
| Martinez
(2009a) | 4.121 | 0.853 | 19.905 | 1.762 | 0.078 | (26) |
| Marcone (2008) | 8.705 | 5.274 | 14.368 | 8.464 | <0.001 | (24) |
| Petricevic
(2008) | 3.442 | 0.646 | 18.335 | 1.448 | 0.148 | (29) |
| Anukam (2006a) | 3.284 | 0.692 | 15.591 | 1.496 | 0.135 | (15) |
| D, Restoration of
normal flora at 6 months |
| | Statistics with
study removed | |
| Author name
(year) | Odds ratio | Lower limit | Upper limit | Z-value | P-value | (Refs.) |
| Recine (2016) | 0.536 | 0.195 | 1.477 | -1.205 | 0.228 | (32) |
| Bradshaw
(2012) | 0.861 | 0.062 | 11.885 | -0.112 | 0.911 | (17) |
| Marcone (2010) | 1.599 | 0.293 | 8.737 | 0.542 | 0.588 | (25) |
| Marcone (2008) | 1.312 | 0.197 | 8.715 | 0.281 | 0.779 | (24) |
Discussion
The overall summary of the qualitative analysis of
the 30 studies suggests that probiotic treatments are useful for
managing common vaginal infections, particularly BV and VVC.
However, patient populations, treatment protocols, endpoints and
follow-up time-points exhibited a marked variation. The results of
the meta-analysis indicated that probiotics as a supplement of
antibiotic/anti-fungal treatments (as observed in type I studies)
reduced the recurrence rate and increased the cure/remission rate
in non-pregnant adult females at 1 month after treatment. With less
evident data, the normal bacterial flora restoration rate was also
increased by probiotic-supplemented treatments in BV. The
short-term benefits of probiotics were further supported by
individual analysis of BV and VVC, although probiotics
supplementary to standard treatments did not increase the
cure/remission rate in BV and the post-treatment normal bacterial
restoration rate in VVC was lacking. However, observations at 6
months post-treatment were less frequently reported. In line with
the results demonstrated by probiotic-supplemented treatments,
probiotics alone without antibiotics may have clinical benefits in
promoting the cure/remission rate and normal flora restoration
rates in BV.
To the best of our knowledge, the present
meta-analysis was the first to review and analyze the effect of
probiotics in common vaginal infections reported by RCTs or
appropriately-controlled studies. Furthermore, only few studies
have evaluated the benefits of probiotics in vaginal infection
stratified by treatment regimen. The quantitative data of the
present study are supported by conclusions from two published
systemic reviews, which examined the overall effect of probiotics
in females with urogenital infections qualitatively. In 2009, Abad
and Safdar (6) identified 25
studies that used Lactobacillus-containing probiotics to
either prevent or treat a urogenital infection [BV, VVC and urinary
tract infections (UTI)]. Of the 25 studies, 18 used
Lactobacillus preparations for the treatment or prevention
of urogenital infections and 7 focused solely on vaginal
colonization (6). Of the 18 studies,
only 8 studies included patients with BV, 4 included patients with
VVC, 5 included patients with UTI and 1 was on multiple infections
(6). Overall, Lactobacilli
were beneficial for the treatment of BV, while no clear benefit was
observed for VVC or UTI (6). A more
recent systematic review published in 2016 investigated probiotics
for the treatment and prevention of urogenital infections in
females (4). A total of 20 studies
(published from 2008 to 2015) were identified, with 14 examining
BV, 2 examining VCC, 3 examining UTI and 1 examining human
papillomavirus (HPV) (4). While the
studies reviewed by Hanson et al (4) in 2016 were heterogeneous with respect
to study type, design, intervention and outcomes and varied in
quality (4 of good quality, 9 of fair and 7 of poor quality), the
authors still made to the conclusion that the use of probiotics may
be effective for the treatment and prevention of BV, recurrent
candidiasis or UTI, as well as HPV lesions. In the current review,
an analysis of quantitative outcomes from a total of 1,788 patients
with common vaginal infections was presented, with focus on BV and
VVC that are most directly impacted by an imbalanced
microflora/dysbiosis.
One prior meta-analysis examined the use of
probiotics for treating BV. In a meta-analysis published in 2014,
Huang et al (3) indicated
that the use of probiotic supplementation significantly improved
the cure rate in adult females with BV [risk ratio (RR)=1.53; 95%
CI: 1.19-1.97]. When only 9 high-quality studies were included in
the analysis, the RR increased slightly to 1.60 (95% CI: 1.16-2.22)
(3). Of note, when a subgroup
analysis was performed, a single treatment with probiotics may only
be effective for short-term follow-up (≤1 month) but not for
long-term follow-up (>1 month) (3), which was consistent with the present
result that no difference between two groups in recurrence rate and
cure/remission rates was determined at 6 months after the
treatment. In a meta-analysis by Huang et al (3) from 2014, the eligible articles were
searched up to May 2013 and the studies included in the
meta-analysis were also heterogeneous. In the present
meta-analysis, the literature search was further updated to
December 24th, 2018, and studies all except one RCT analyzed in the
previous study by Huang et al (3) from 2014 were included. This particular
RCT was excluded from the present study due to its study design for
healthy females with a history of BV (45); furthermore, it had different
follow-up time-points from other studies analyzed in the present
study and was deemed unsuitable for analysis of post-treatment
outcomes.
A recent meta-analysis study suggested that,
although probiotics appeared effective in treating VVC, relevant
studies were not sufficient in number (5-7 studies included for
each analysis) or of comparable quality (7). In the present study, which focused on
common vaginal infections as a whole, only studies with comparable
treatment designs and study follow-up schedules were included in
the meta-analysis. Furthermore, the major results of the present
study were based on >10 RCTs or prospective studies with control
arms. In 2006, Falagas et al (46) reported on several clinical trials on
VVC that support the effectiveness of Lactobacilli
administered either orally or intravaginally in decreasing
colonization of C. albicans or preventing vaginal
candidiasis. However, most of the relevant clinical trials had
methodological problems, including small sample size, no control
group (single-arm) and included females without confirmed recurrent
VVC. All of the studies on VVC reviewed in the present
meta-analysis were designed to compare Lactobacillus
capsule-supplemented anti-fungal treatments (probiotic group) with
anti-fungal agents alone (control group). Despite the follow-up
period ranging from <1 week to 6 months among the studies
included, only those with comparable follow-up schedules were
included in the present meta-analysis. The outcome supports the
effectiveness of Lactobacilli in decreasing the recurrence
rate and improving the cure rate.
The primary limitation of the present study has
already been mentioned-the large heterogeneity between studies with
respect to the patient population, type of treatment, probiotic
strains and outcome follow-ups. However, it was sought to overcome
this by carefully-planned stratification based on treatment design
and follow-up schedules. The major results on short-term benefits
of combined therapy of antibiotics/anti-fungals with probiotics was
further confirmed by the sensitivity test. By contrast, the limited
sample size and heterogeneous study design prevented us from a
reliable subgroup analysis of long-term benefits and of probiotics
treatment alone without antibiotic/anti-fungal agents.
In conclusion, the results of the present study
confirm the results of other reports in a quantitative manner,
namely that probiotics as a supplement to conventional
pharmacological treatments are effective in the short term for the
treatment of common vaginal infections in non-pregnant adult
females. However, high-quality evidence for the effectiveness of
probiotics alone in recurrent or curative vaginal infections is
limited. Further high-quality clinical trials are necessary to
identify the most effective probiotic strains, the most effective
treatment regimens (with or without antibiotics) and the
subpopulations of females (e.g. pre-menopausal vs. post-menopausal)
that may benefit the most from probiotics.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
HSJ conceived and designed the current study,
defined the content of the research, conducted literature research,
performed statistical analysis and prepared and edited the
manuscript. TRY is the guarantor of study integrity, designed the
current study, defined the content of the research and reviewed the
manuscript. JYC conducted literature research, acquired data and
performed statistical analysis. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing
interests.
References
|
1
|
Kent HL: Epidemiology of vaginitis. Am J
Obstet Gynecol. 165:1168–1176. 1991.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Mills BB: Vaginitis: Beyond the Basics.
Obstet Gynecol Clin North Am. 44:159–177. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Huang H, Song L and Zhao W: Effects of
probiotics for the treatment of bacterial vaginosis in adult women:
A meta-analysis of randomized clinical trials. Arch Gynecol Obstet.
289:1225–1234. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hanson L, VandeVusse L, Jermè M, Abad CL
and Safdar N: Probiotics for treatment and prevention of urogenital
infections in women: A systematic review. J Midwifery Womens
Health. 61:339–355. 2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Reid G: Probiotic use in an infectious
disease setting. Expert Rev Anti Infect Ther. 15:449–455.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Abad CL and Safdar N: The role of
Lactobacillus probiotics in the treatment or prevention of
urogenital infections-a systematic review. J Chemother. 21:243–252.
2009.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Xie HY, Feng D, Wei DM, Mei L, Chen H,
Wang X and Fang F: Probiotics for vulvovaginal candidiasis in
non-pregnant women. Cochrane Database Syst Rev.
11(CD010496)2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Liberati A, Altman DG, Tetzlaff J, Mulrow
C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J
and Moher D: The PRISMA statement for reporting systematic reviews
and meta-analyses of studies that evaluate health care
interventions: Explanation and elaboration. J Clin Epidemiol.
62:e1–e34. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Higgins JP: Commentary: Heterogeneity in
meta-analysis should be expected and appropriately quantified. Int
J Epidemiol. 37:1158–1160. 2008.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Sterne JAC, Higgins JPT, Reeves BC on
behalf of the development group for ACROBAT- NRSI. A Cochrane Risk
Of Bias Assessment Tool: for Non-Randomized Studies of
Interventions (ACROBAT- NRSI), Version 1.0.0, 24 September 2014.
http://www.riskofbias.info.
|
|
11
|
Deeks JJ, Higgins JPT and DG A: Chapter
10: Analysing data and undertaking meta-analyses. In: Cochrane
Handbook for Systematic Reviews of Interventions version 6.0
(updated July 2019). Higgins JPT, Thomas J, Chandler J, et
al. (eds.) Cochrane, 2019.
|
|
12
|
Hardy RJ and Thompson SG: Detecting and
describing heterogeneity in meta-analysis. Stat Med. 17:841–856.
1998.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Takkouche B, Cadarso-Suárez C and
Spiegelman D: Evaluation of old and new tests of heterogeneity in
epidemiologic meta-analysis. Am J Epidemiol. 150:206–215.
1999.PubMed/NCBI View Article : Google Scholar
|
|
14
|
National Research Council: Combining
Information: Statistical issues and opportunities for research. The
National Academies Press, Washington, DC, 1992.
https://doi.org/10.17226/20865.
|
|
15
|
Anukam K, Osazuwa E, Ahonkhai I, Ngwu M,
Osemene G, Bruce AW and Reid G: Augmentation of antimicrobial
metronidazole therapy of bacterial vaginosis with oral probiotic
Lactobacillus rhamnosus GR-1 and Lactobacillus
reuteri RC-14: Randomized, double-blind, placebo controlled trial.
Microbes Infect. 8:1450–1454. 2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Anukam KC, Osazuwa E, Osemene GI,
Ehigiagbe F, Bruce AW and Reid G: Clinical study comparing
probiotic Lactobacillus GR-1 and RC-14 with metronidazole
vaginal gel to treat symptomatic bacterial vaginosis. Microbes
Infect. 8:2772–2776. 2006.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Bradshaw CS, Pirotta M, De Guingand D,
Hocking JS, Morton AN, Garland SM, Fehler G, Morrow A, Walker S,
Vodstrcil LA and Fairley CK: Efficacy of oral metronidazole with
vaginal clindamycin or vaginal probiotic for bacterial vaginosis:
Randomised placebo-controlled double-blind trial. PLoS One.
7(e34540)2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Eriksson K, Carlsson B, Forsum U and
Larsson PG: A double-blind treatment study of bacterial vaginosis
with normal vaginal lactobacilli after an open treatment with
vaginal clindamycin ovules. Acta Derm Venereol. 85:42–46.
2005.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Hallen A, Jarstrand C and Påhlson C:
Treatment of bacterial vaginosis with lactobacilli. Sex Transm Dis.
19:146–148. 1992.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Heczko PB, Tomusiak A, Adamski P, Jakimiuk
AJ, Stefanski G, Mikołajczyk-Cichońska A, Suda-Szczurek M and Strus
M: Supplementation of standard antibiotic therapy with oral
probiotics for bacterial vaginosis and aerobic vaginitis: A
randomised, double-blind, placebo-controlled trial. BMC Womens
Health. 15(115)2015.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hemalatha R, Mastromarino P, Ramalaxmi BA,
Balakrishna NV and Sesikeran B: Effectiveness of vaginal tablets
containing lactobacilli versus pH tablets on vaginal health and
inflammatory cytokines: A randomized, double-blind study. Eur J
Clin Microbiol Infect Dis. 31:3097–3105. 2012.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Larsson PG, Stray-Pedersen B, Ryttig KR
and Larsen S: Human lactobacilli as supplementation of clindamycin
to patients with bacterial vaginosis reduce the recurrence rate; a
6-month, double-blind, randomized, placebo-controlled study. BMC
Womens Health. 8(3)2008.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Laue C, Papazova E, Liesegang A,
Pannenbeckers A, Arendarski P, Linnerth B, Domig KJ, Kneifel W,
Petricevic L and Schrezenmeir J: Effect of a yoghurt drink
containing Lactobacillus strains on bacterial vaginosis in
women-a double-blind, randomised, controlled clinical pilot trial.
Benef Microbes. 9:35–50. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Marcone V, Calzolari E and Bertini M:
Effectiveness of vaginal administration of Lactobacillus
rhamnosus following conventional metronidazole therapy: How to
lower the rate of bacterial vaginosis recurrences. New Microbiol.
31:429–433. 2008.PubMed/NCBI
|
|
25
|
Marcone V, Rocca G, Lichtner M and
Calzolari E: Long-term vaginal administration of Lactobacillus
rhamnosus as a complementary approach to management of
bacterial vaginosis. Int J Gynaecol Obstet. 110:223–226.
2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Martinez RC, Franceschini SA, Patta MC,
Quintana SM, Gomes BC, De Martinis EC and Reid G: Improved cure of
bacterial vaginosis with single dose of tinidazole (2 g),
Lactobacillus rhamnosus GR-1, and Lactobacillus reuteri RC-14: a
randomized, double-blind, placebo-controlled trial. Can J
Microbiol. 55:133–138. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Mastromarino P, Macchia S, Meggiorini L,
Trinchieri V, Mosca L, Perluigi M and Midulla C: Effectiveness of
Lactobacillus-containing vaginal tablets in the treatment of
symptomatic bacterial vaginosis. Clin Microbiol Infect. 15:67–74.
2009.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Parent D, Bossens M, Bayot D, Kirkpatrick
C, Graf F, Wilkinson FE and Kaiser RR: Therapy of bacterial
vaginosis using exogenously-applied Lactobacilli acidophili
and a low dose of estriol: A placebo-controlled multicentric
clinical trial. Arzneimittelforschung. 46:68–73. 1996.PubMed/NCBI
|
|
29
|
Petricevic L and Witt A: The role of
Lactobacillus casei rhamnosus Lcr35 in restoring the normal
vaginal flora after antibiotic treatment of bacterial vaginosis.
BJOG. 115:1369–1374. 2008.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ling Z, Liu X, Chen W, Luo Y, Yuan L, Xia
Y, Nelson KE, Huang S, Zhang S, Wang Y, et al: The restoration of
the vaginal microbiota after treatment for bacterial vaginosis with
metronidazole or probiotics. Microb Ecol. 65:773–780.
2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lin H and Meng XB: Clinical application of
Lactasin capsules in vaginitis. J Practical Medicine. 22:1927–1928.
2006.
|
|
32
|
Recine N, Palma E, Domenici L, Giorgini M,
Imperiale L, Sassu C, Musella A, Marchetti C, Muzii L and Benedetti
Panici P: Restoring vaginal microbiota: Biological control of
bacterial vaginosis. A prospective case-control study using
Lactobacillus rhamnosus BMX 54 as adjuvant treatment against
bacterial vaginosis. Arch Gynecol Obstet. 293:101–107.
2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Vicariotto F, Mogna L and Del Piano M:
Effectiveness of the two microorganisms Lactobacillus
fermentum LF15 and Lactobacillus plantarum LP01,
formulated in slow-release vaginal tablets, in women affected by
bacterial vaginosis: A pilot study. J Clin Gastroenterol. 48 (Suppl
1):S106–S112. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Vujic G, Jajac Knez A, Despot Stefanovic V
and Kuzmic Vrbanovic V: Efficacy of orally applied probiotic
capsules for bacterial vaginosis and other vaginal infections: A
double-blind, randomized, placebo-controlled study. Eur J Obstet
Gynecol Reprod Biol. 168:75–79. 2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Anukam KC, Duru MU, Eze CC, Egharevba J,
Aiyebelehin A, Bruce A and Reid G: Oral use of probiotics as an
adjunctive therapy to fluconazole in the treatment of yeast
vaginitis: A study of Nigerian women in an outdoor clinic.
Microbial Ecol Health Dis. 21:72–77. 2009.
|
|
36
|
Davar R, Nokhostin F, Eftekhar M, Sekhavat
L, Bashiri Zadeh M and Shamsi F: Comparing the recurrence of
vulvovaginal candidiasis in patients undergoing prophylactic
treatment with probiotic and placebo during the 6 months.
Probiotics Antimicrob Proteins. 8:130–133. 2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Han YX and Zhao SW: Clinical observation
of Ding JunSheng and clotrimazole vaginal tablet in the treatment
ofvulvovaginal candidiasis. Anh Med J. 27:528–529. 2006.
|
|
38
|
Hua Y, Lin M, Wang L and Xia L:
Observation of curative effect of miconazole and
Lactobacillus on vulvovaginal candidiasis treated. Chin J
Microecol. 20:386–387. 2008.
|
|
39
|
Ma L and Li L: Miconazole plus Lactasin
capsules to treat vulvovaginal candidiasis (54 cases). Herald Med.
26:1041–1042. 2007.
|
|
40
|
Mai XY: Analysis of probiotic
Lactobacillus's effect on vulvovaginal candidiasis. Chin J
Microecol. 19:362–363. 2007.
|
|
41
|
Nouraei S, Amir Ali Akbari S, Jorjani M,
Alavi Majd H, Afrakhteh M, Ghafoorian A and Tafazzoli Harandi H:
Comparison between fluconazole with oral protexin combination and
fluconazole in the treatment of vulvovaginal candidiasis. ISRN
Obstet Gynecol. 2012(375806)2012.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Yang Q, Zhao WF and Zheng JP: Clinical
observat ion of mycobacterium lacticola preparation in treatment of
vaginal vini candidiasis using clotrimazole effervescent tablets.
Chin Hosp Pharm J. 29:1377–1379. 2009.
|
|
43
|
Martinez RC, Franceschini SA, Patta MC,
Quintana SM, Candido RC, Ferreira JC, De Martinis EC and Reid G:
Improved treatment of vulvovaginal candidiasis with fluconazole
plus probiotic Lactobacillus rhamnosus GR‐1 and Lactobacillus
reuteri RC‐14. Lett Appl Microbiol. 48:269–274. 2009.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Ehrstrom S, Daroczy K, Rylander E,
Samuelsson C, Johannesson U, Anzèn B and Påhlson C: Lactic acid
bacteria colonization and clinical outcome after probiotic
supplementation in conventionally treated bacterial vaginosis and
vulvovaginal candidiasis. Microbes Infect. 12:691–699.
2010.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Ya W, Reifer C and Miller LE: Efficacy of
vaginal probiotic capsules for recurrent bacterial vaginosis: A
double-blind, randomized, placebo-controlled study. Am J Obstet
Gynecol. 203:120.e1–e6. 2010.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Falagas ME, Betsi GI and Athanasiou S:
Probiotics for prevention of recurrent vulvovaginal candidiasis: A
review. J Antimicrob Chemother. 58:266–272. 2006.PubMed/NCBI View Article : Google Scholar
|