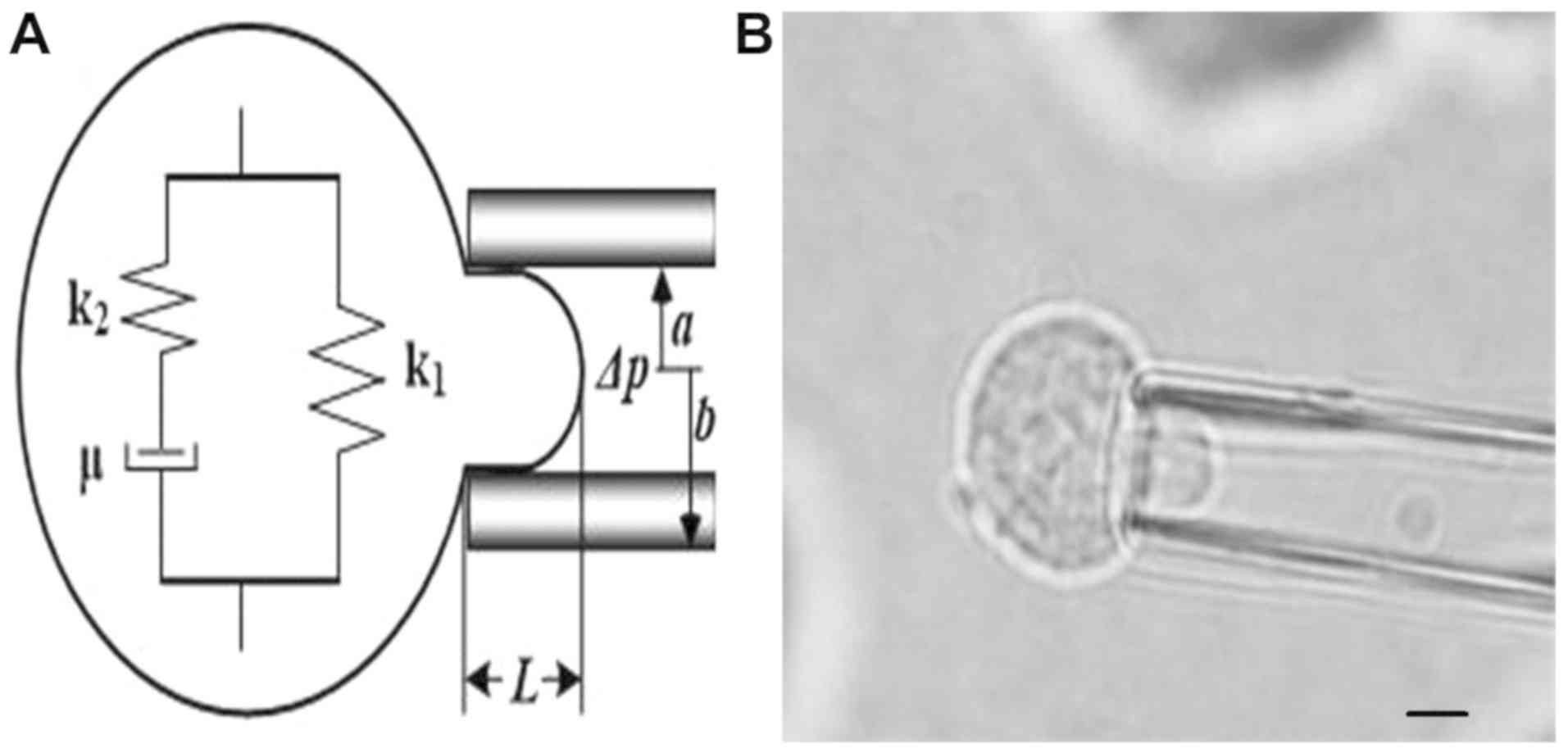
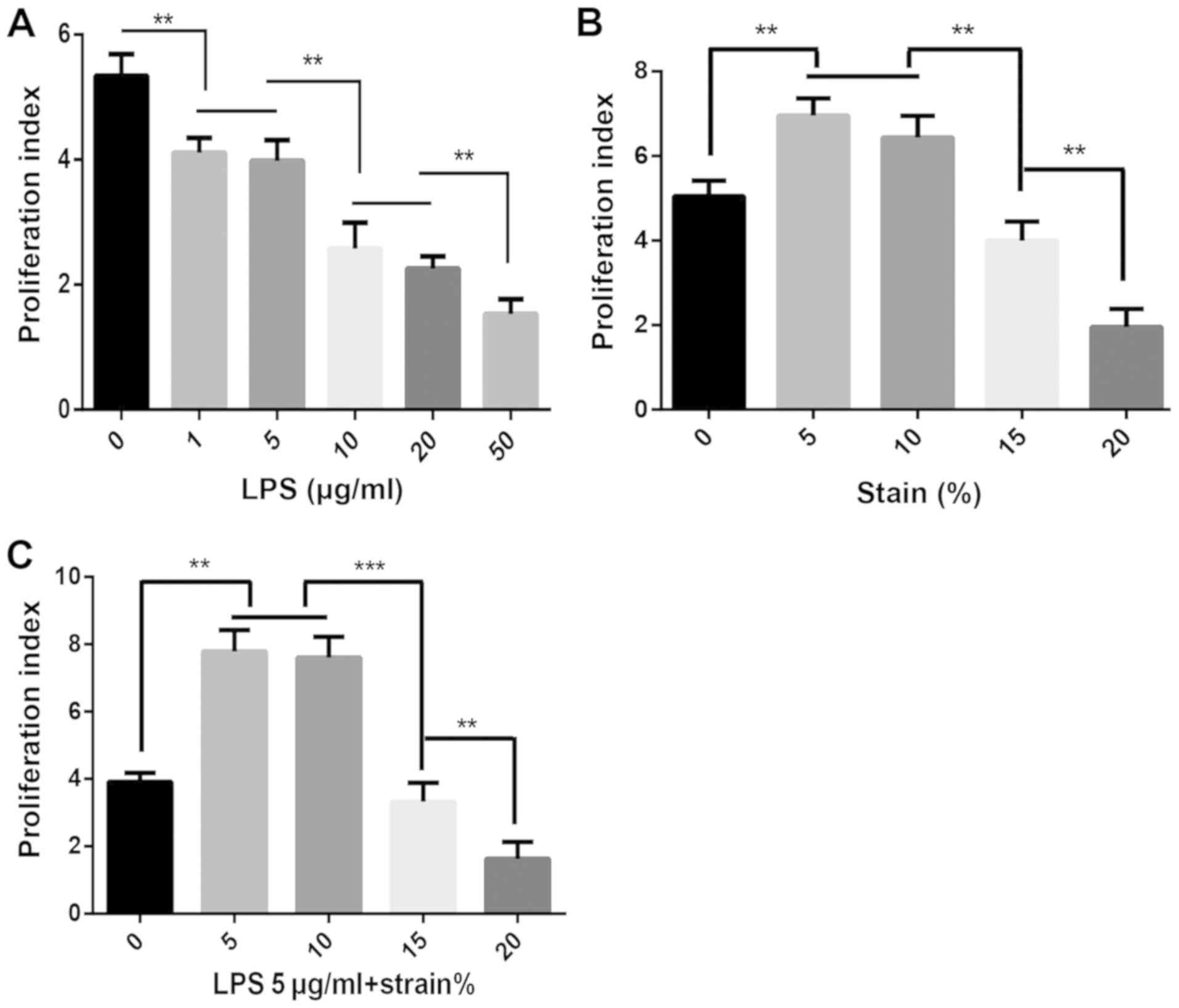
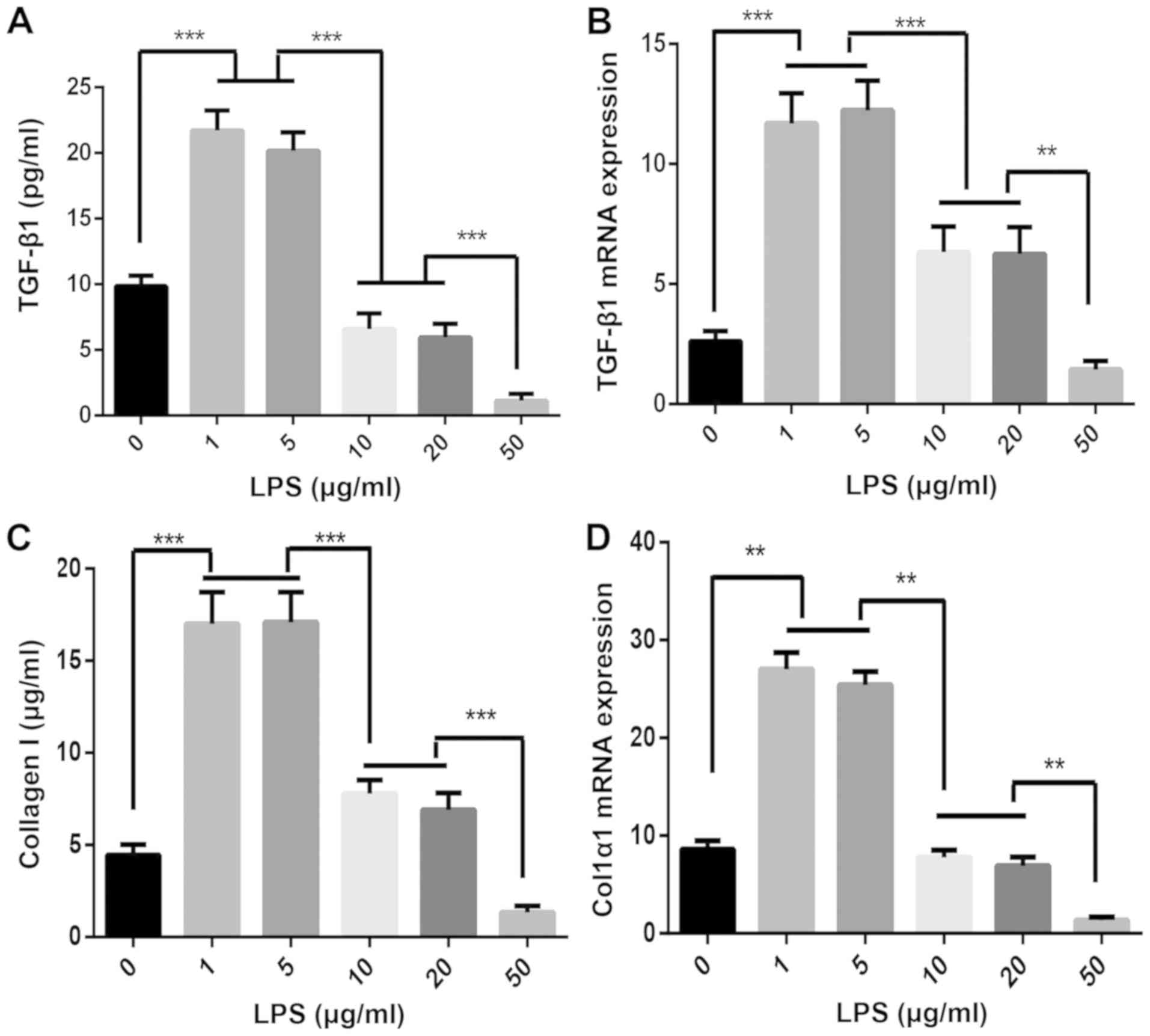
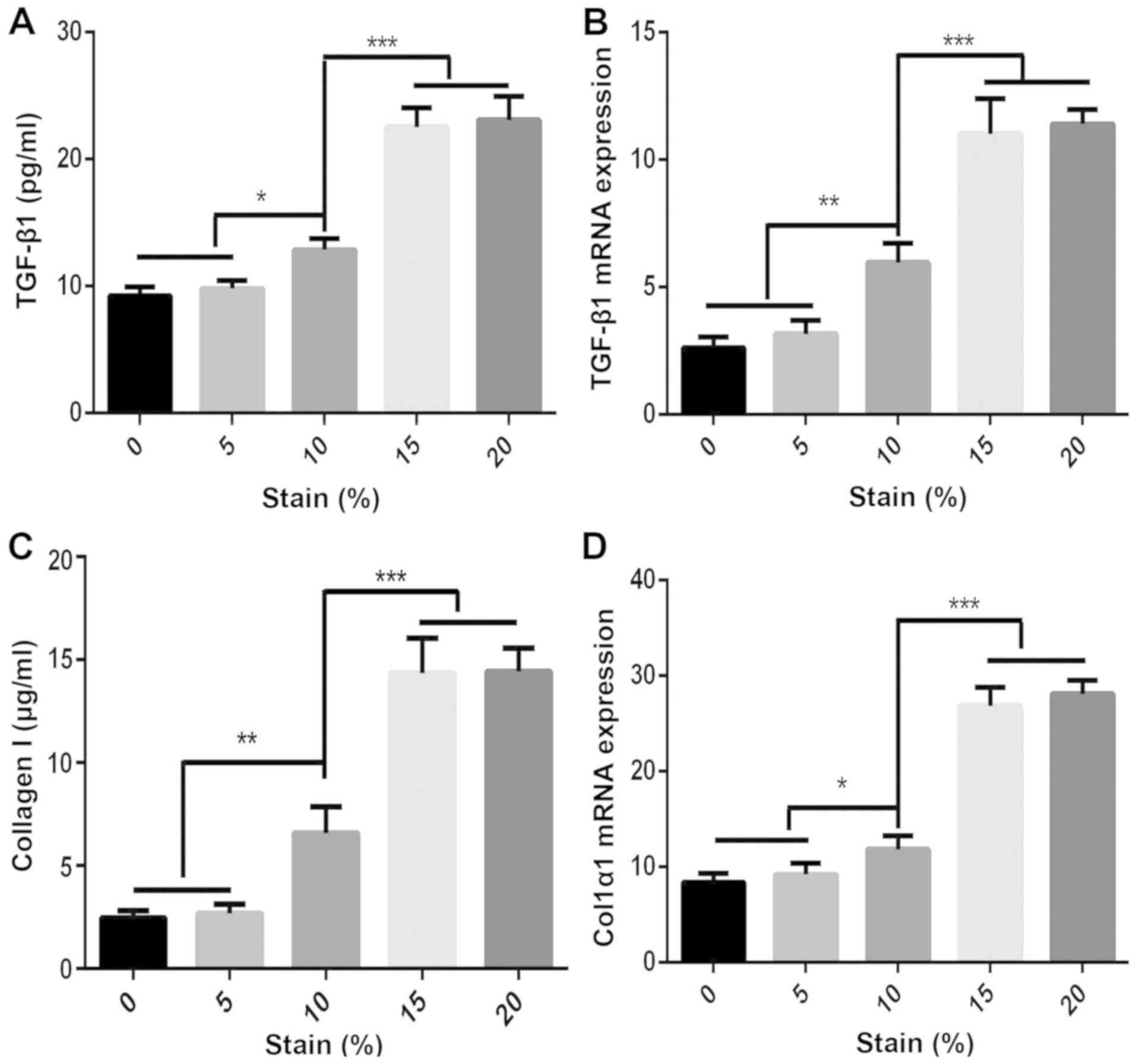
|
1
|
Hu HH, Chen DQ, Wang YN, Feng YL, Cao G,
Vaziri ND and Zhao YY: New insights into TGF-β/Smad signaling in
tissue fibrosis. Chem Biol Interact. 292:76–83. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Nieman GF, Satalin J, Andrews P, Aiash H,
Habashi NM and Gatto LA: Personalizing mechanical ventilation
according to physiologic parameters to stabilize alveoli and
minimize ventilator induced lung injury (VILI). Intensive Care Med
Exp. 5(8)2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bates J and Smith BJ: Ventilator-induced
lung injury and lung mechanics. Ann Transl Med.
6(378)2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gattinoni L, Marini JJ, Collino F, Maiolo
G, Rapetti F, Tonetti T, Vasques F and Quintel M: The future of
mechanical ventilation: Lessons from the present and the past. Crit
Care. 21(183)2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Souma K, Shichino S, Hashimoto S, Ueha S,
Tsukui T, Nakajima T, Suzuki HI, Shand FHW, Inagaki Y, Nagase T and
Matsushima K: Lung fibroblasts express a miR-19a-19b-20a
sub-cluster to suppress TGF-β-associated fibroblast activation in
murine pulmonary fibrosis. Sci Rep. 8(16642)2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Lv Z, Wang Y, Liu YJ, Mao YF, Dong WW,
Ding ZN, Meng GX, Jiang L and Zhu XY: NLRP3 inflammasome activation
contributes to mechanical stretch-induced endothelial-mesenchymal
transition and pulmonary fibrosis. Crit Care Med. 46:e49–e58.
2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Xie Y, Wang M, Cheng M, Gao Z and Wang G:
The viscoelastic behaviors of several kinds of cancer cells and
normal cells. J Mech Behav Biomed Mater. 91:54–58. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Wang G and Chen W: Effects of mechanical
stimulation on viscoelasticity of rabbit scleral fibroblasts after
posterior scleral reinforcement. Exp Biol Med (Maywood).
237:1150–1154. 2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Xie Y, Liu X, Wang S, Wang M and Wang G:
Proper mechanical stimulation improve the chondrogenic
differentiation of mesenchymal stem cells: Improve the
viscoelasticity and chondrogenic phenotype. Biomed Pharmacother.
115(108935)2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Maca J, Jor O, Holub M, Sklienka P, Burša
F, Burda M, Janout V and Ševčík P: Past and present ARDS mortality
rates: A systematic review. Respir Care. 62:113–122.
2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Pais FM, Sinha P, Liu KD and Matthay MA:
Influence of clinical factors and exclusion criteria on mortality
in ARDS observational studies and randomized controlled trials.
Respir Care. 63:1060–1069. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang X, Lai R, Su X, Chen G and Liang Z:
Edaravone attenuates lipopolysaccharide-induced acute respiratory
distress syndrome associated early pulmonary fibrosis via
amelioration of oxidative stress and transforming growth
factor-β1/Smad3 signaling. Biochem Biophys Res Commun. 495:706–712.
2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yen S, Preissner M, Bennett E, Dubsky S,
Carnibella R, O'Toole R, Roddam L, Jones H, Dargaville PA, Fouras A
and Zosky GR: The link between regional tidal stretch and lung
injury during mechanical ventilation. Am J Respir Cell Mol Biol,
2018.
|
|
15
|
Shimbori C, Upagupta C, Bellaye PS, Ayaub
EA, Sato S, Yanagihara T, Zhou Q, Ognjanovic A, Ask K, Gauldie J,
et al: Mechanical stress-induced mast cell degranulation activates
TGF-β1 signalling pathway in pulmonary fibrosis. Thorax.
74:455–465. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
White ES: Lung extracellular matrix and
fibroblast function. Ann Am Thorac Soc. 12 (Suppl 1):S30–S33.
2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Erdogan B and Webb DJ: Cancer-associated
fibroblasts modulate growth factor signaling and extracellular
matrix remodeling to regulate tumor metastasis. Biochem Soc Trans.
45:229–236. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wu Y, Zhuang J, Zhao D, Zhang F, Ma J and
Xu C: Cyclic stretch-induced the cytoskeleton rearrangement and
gene expression of cytoskeletal regulators in human periodontal
ligament cells. Acta Odontol Scand. 75:507–516. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Bartolák-Suki E, Imsirovic J, Nishibori Y,
Krishnan R and Suki B: Regulation of mitochondrial structure and
dynamics by the cytoskeleton and mechanical factors. Int J Mol Sci.
18(E1812)2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Manou D, Caon I, Bouris P,
Triantaphyllidou IE, Giaroni C, Passi A, Karamanos NK, Vigetti D
and Theocharis AD: The complex interplay between extracellular
matrix and cells in tissues. Methods Mol Biol. 1952:1–20.
2019.PubMed/NCBI View Article : Google Scholar
|