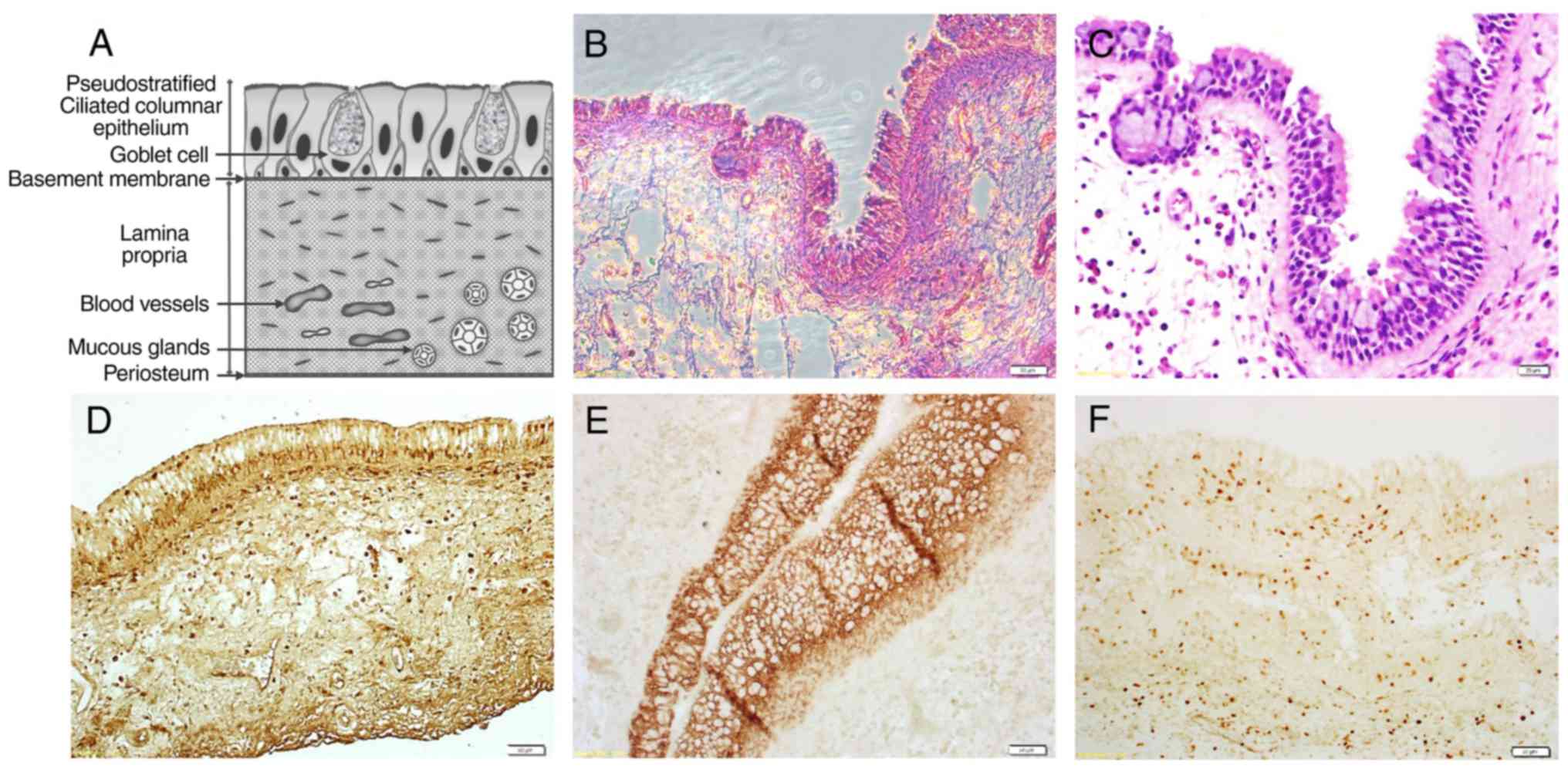
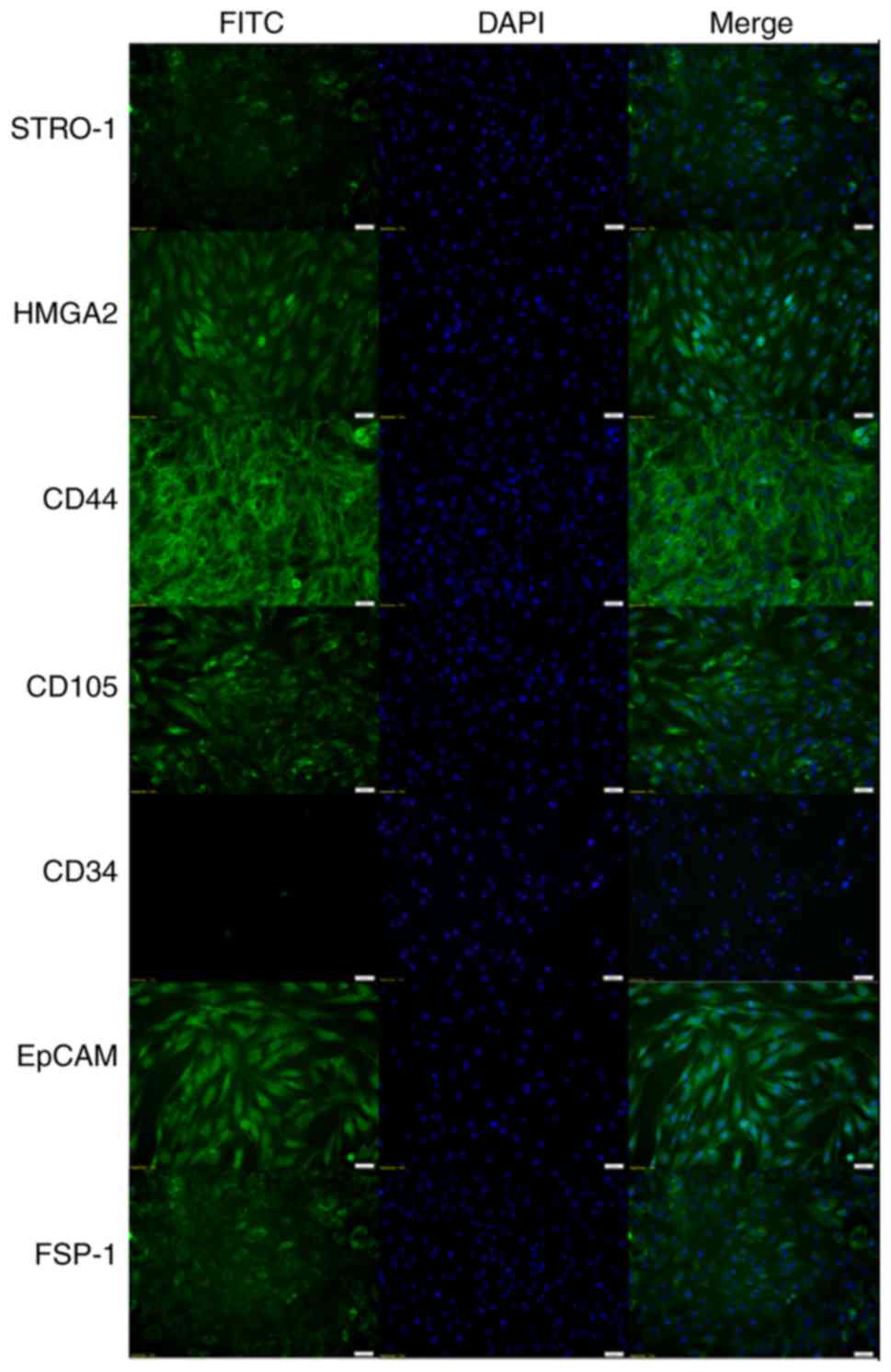
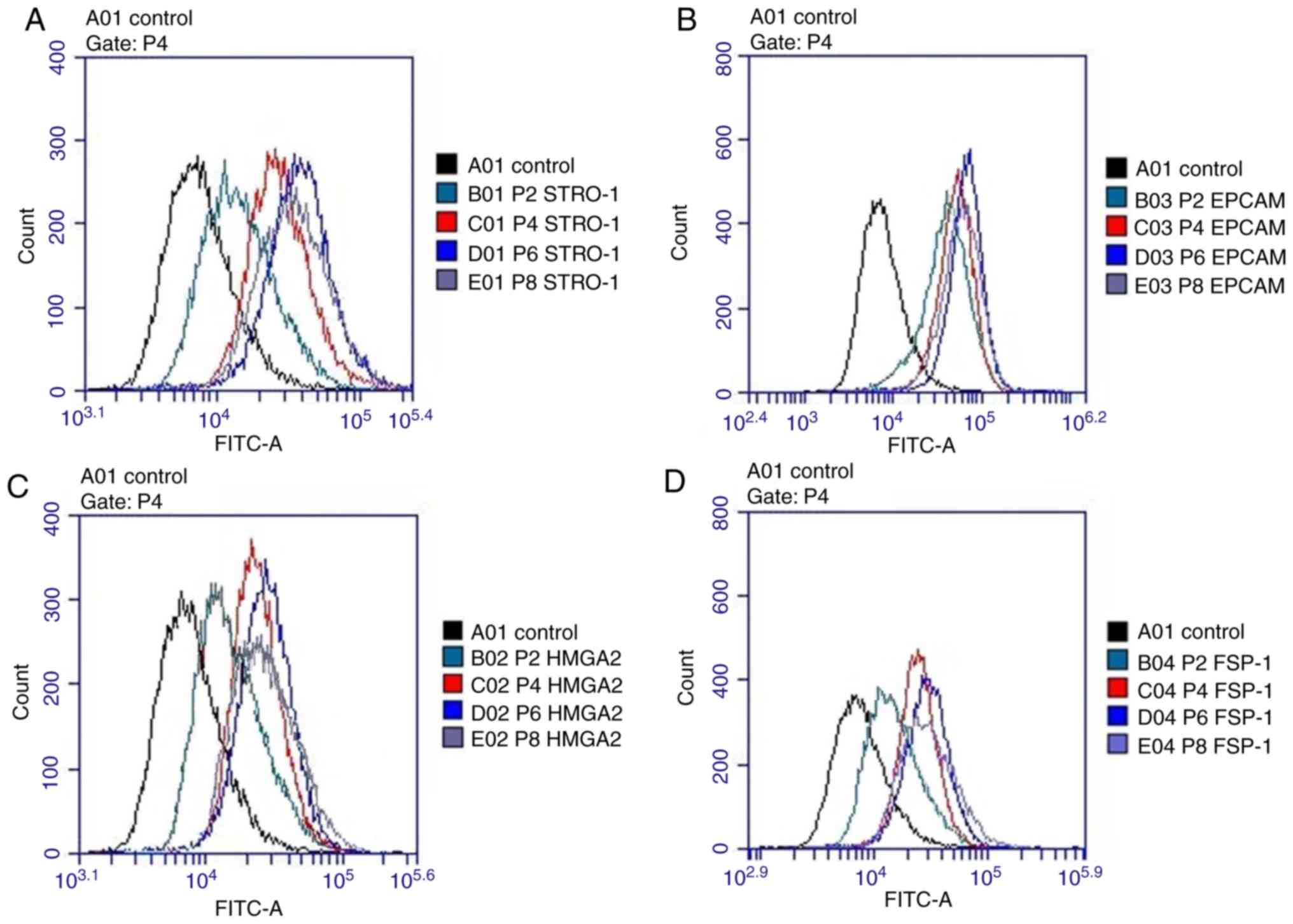
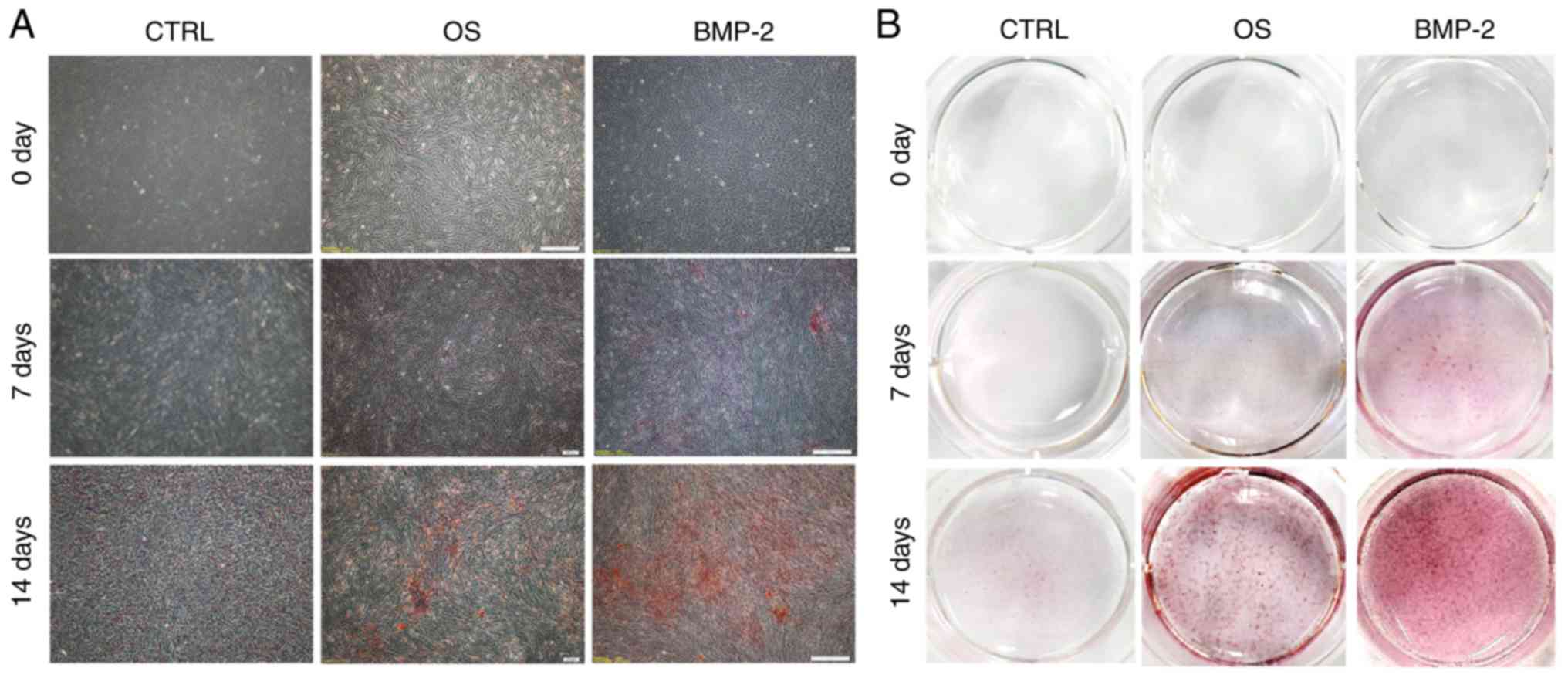
|
1
|
Raghoebar GM, Onclin P, Boven GC, Vissink
A and Meijer HJA: Long-term effectiveness of maxillary sinus floor
augmentation: A systematic review and meta-analysis. J Clin
Periodontol. 46 (Suppl 21):S307–S318. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Stumbras A, Krukis MM, Januzis G and
Juodzbalys G: Regenerative bone potential after sinus floor
elevation using various bone graft materials: A systematic review.
Quintessence Int. 50:548–558. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Helder MN, van Esterik FAS, Kwehandjaja
MD, Ten Bruggenkate CM, Klein-Nulend J and Schulten E: Evaluation
of a new biphasic calcium phosphate for maxillary sinus floor
elevation: Micro-CT and histomorphometrical analyses. Clin Oral
Implants Res. 29:488–498. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Farré-Guasch E, Prins HJ, Overman JR, Ten
Bruggenkate CM, Schulten EA, Helder MN and Klein-Nulend J: Human
maxillary sinus floor elevation as a model for bone regeneration
enabling the application of one-step surgical procedures. Tissue
Eng Part B Rev. 19:69–82. 2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Solar P, Geyerhofer U, Traxler H, Windisch
A, Ulm C and Watzek G: Blood supply to the maxillary sinus relevant
to sinus floor elevation procedures. Clin Oral Implants Res.
10:34–44. 1999.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Riben C and Thor A: Follow-up of the sinus
membrane elevation technique for maxillary sinus implants without
the use of graft material. Clin Implant Dent Relat Res. 18:895–905.
2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sohn DS, Moon JW, Lee WH, Kim SS, Kim CW,
Kim KT and Moon YS: Comparison of new bone formation in the
maxillary sinus with and without bone grafts: Immunochemical rabbit
study. Int J Oral Maxillofac Implants. 26:1033–1042.
2011.PubMed/NCBI
|
|
8
|
Parra M, Olate S and Cantin M: Clinical
and biological analysis in graftless maxillary sinus lift. J Korean
Assoc Oral Maxillofac Surg. 43:214–220. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Stefanski S, Svensson B and Thor A:
Implant survival following sinus membrane elevation without
grafting and immediate implant installation with a one-stage
technique: An up-to-40-month evaluation. Clin Oral Implants Res.
28:1354–1359. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Cricchio G, Sennerby L and Lundgren S:
Sinus bone formation and implant survival after sinus membrane
elevation and implant placement: A 1- to 6-year follow-up study.
Clin Oral Implants Res. 22:1200–1212. 2011.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Cordaro L, Bosshardt DD, Palattella P, Rao
W, Serino G and Chiapasco M: Maxillary sinus grafting with Bio-Oss
or Straumann Bone Ceramic: Histomorphometric results from a
randomized controlled multicenter clinical trial. Clin Oral
Implants Res. 19:796–803. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Avera SP, Stampley WA and McAllister BS:
Histologic and clinical observations of resorbable and
nonresorbable barrier membranes used in maxillary sinus graft
containment. Int J Oral Maxillofac Implants. 12:88–94.
1997.PubMed/NCBI
|
|
13
|
Graziano A, Benedetti L, Massei G, Cusella
de Angelis MG, Ferrarotti F and Aimetti M: Bone production by human
maxillary sinus mucosa cells. J Cell Physiol. 227:3278–3281.
2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Gruber R, Kandler B, Fuerst G, Fischer MB
and Watzek G: Porcine sinus mucosa holds cells that respond to bone
morphogenetic protein (BMP)-6 and BMP-7 with increased osteogenic
differentiation in vitro. Clin Oral Implants Res. 15:575–580.
2004.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Guo J, Weng J, Rong Q, Zhang X, Zhu S,
Huang D, Li X and Chen S: Investigation of multipotent postnatal
stem cells from human maxillary sinus membrane. Sci Rep.
5(11660)2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Srouji S, Kizhner T, Ben David D,
Riminucci M, Bianco P and Livne E: The Schneiderian membrane
contains osteoprogenitor cells: In vivo and in vitro study. Calcif
Tissue Int. 84:138–145. 2009.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kim SW, Lee IK, Yun KI, Kim CH and Park
JU: Adult stem cells derived from human maxillary sinus membrane
and their osteogenic differentiation. Int J Oral Maxillofac
Implants. 24:991–998. 2009.PubMed/NCBI
|
|
18
|
Berberi A, Al-Nemer F, Hamade E, Noujeim
Z, Badran B and Zibara K: Mesenchymal stem cells with osteogenic
potential in human maxillary sinus membrane: An in vitro study.
Clin Oral Investig. 21:1599–1609. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Cho KS, Park HY, Roh HJ, Bravo DT, Hwang
PH and Nayak JV: Human ethmoid sinus mucosa: A promising novel
tissue source of mesenchymal progenitor cells. Stem Cell Res Ther.
5(15)2014.PubMed/NCBI View
Article : Google Scholar
|
|
20
|
Lin GH, Lim G, Chan HL, Giannobile WV and
Wang HL: Recombinant human bone morphogenetic protein 2 outcomes
for maxillary sinus floor augmentation: A systematic review and
meta-analysis. Clin Oral Implants Res. 27:1349–1359.
2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yamaguchi A, Komori T and Suda T:
Regulation of osteoblast differentiation mediated by bone
morphogenetic proteins, hedgehogs, and Cbfa1. Endocr Rev.
21:393–411. 2000.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Itoh S, Itoh F, Goumans MJ and Ten Dijke
P: Signaling of transforming growth factor-beta family members
through Smad proteins. Eur J Biochem. 267:6954–6967.
2000.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Miyazono K: Positive and negative
regulation of TGF-beta signaling. J Cell Sci. 113:1101–1109.
2000.PubMed/NCBI
|
|
24
|
Lee MH, Kim YJ, Kim HJ, Park HD, Kang AR,
Kyung HM, Sung JH, Wozney JM, Kim HJ and Ryoo HM: BMP-2-induced
Runx2 expression is mediated by Dlx5, and TGF-beta 1 opposes the
BMP-2-induced osteoblast differentiation by suppression of Dlx5
expression. J Biol Chem. 278:34387–34394. 2003.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Lee MH, Kwon TG, Park HS, Wozney JM and
Ryoo HM: BMP-2-induced Osterix expression is mediated by Dlx5 but
is independent of Runx2. Biochem Biophys Res Commun. 309:689–694.
2003.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Nakashima M and Reddi AH: The application
of bone morphogenetic proteins to dental tissue engineering. Nat
Biotechnol. 21:1025–1032. 2003.PubMed/NCBI View
Article : Google Scholar
|
|
27
|
Bowler D and Dym H: Bone morphogenic
protein: Application in implant dentistry. Dent Clin North Am.
59:493–503. 2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Srouji S, Ben-David D, Lotan R, Riminucci
M, Livne E and Bianco P: The innate osteogenic potential of the
maxillary sinus (Schneiderian) membrane: An ectopic tissue
transplant model simulating sinus lifting. Int J Oral Maxillofac
Surg. 39:793–801. 2010.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Sohn DS and Moon YS: Histomorphometric
study of rabbit's maxillary sinus augmentation with various graft
materials. Anat Cell Biol. 51 (Suppl 1):S1–S12. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Valentin-Opran A, Wozney J, Csimma C,
Lilly L and Riedel GE: Clinical evaluation of recombinant human
bone morphogenetic protein-2. Clin Orthop Relat Res 110-120,
2002.
|
|
31
|
Haugen HJ, Lyngstadaas SP, Rossi F and
Perale G: Bone grafts: Which is the ideal biomaterial? J Clin
Periodontol. 46 (Suppl 21):S92–S102. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Jung RE, Glauser R, Scharer P, Hammerle
CH, Sailer HF and Weber FE: Effect of rhBMP-2 on guided bone
regeneration in humans. Clin Oral Implants Res. 14:556–568.
2003.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Freitas RM, Spin-Neto R, Marcantonio
Junior E, Pereira LA, Wikesjö UM and Susin C: Alveolar ridge and
maxillary sinus augmentation using rhBMP-2: A systematic review.
Clin Implant Dent Relat Res. 17 (Suppl 1):e192–e201.
2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Boyne PJ, Lilly LC, Marx RE, Moy PK,
Nevins M, Spagnoli DB and Triplett RG: De novo bone induction by
recombinant human bone morphogenetic protein-2 (rhBMP-2) in
maxillary sinus floor augmentation. J Oral Maxillofac Surg.
63:1693–1707. 2005.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Torrecillas-Martinez L, Monje A, Pikos MA,
Ortega-Oller I, Suarez F, Galindo-Moreno P and Wang HL: Effect of
rhBMP-2 upon maxillary sinus augmentation: A comprehensive review.
Implant Dent. 22:232–237. 2013.PubMed/NCBI View Article : Google Scholar
|
|
36
|
James AW, LaChaud G, Shen J, Asatrian G,
Nguyen V, Zhang X, Ting K and Soo C: A review of the clinical side
effects of bone morphogenetic protein-2. Tissue Eng Part B Rev.
22:284–297. 2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Lee KB, Taghavi CE, Murray SS, Song KJ,
Keorochana G and Wang JC: BMP induced inflammation: A comparison of
rhBMP-7 and rhBMP-2. J Orthop Res. 30:1985–1994. 2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Woo EJ: Adverse events reported after the
use of recombinant human bone morphogenetic protein 2. J Oral
Maxillofac Surg. 70:765–767. 2012.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Pjetursson BE, Tan WC, Zwahlen M and Lang
NP: A systematic review of the success of sinus floor elevation and
survival of implants inserted in combination with sinus floor
elevation. J Clin Periodontol. 35 (Suppl 8):S216–S240.
2008.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Mohan N, Wolf J and Dym H: Maxillary sinus
augmentation. Dent Clin North Am. 59:375–388. 2015.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Tawil G, Barbeck M, Unger R, Tawil P and
Witte F: Sinus floor elevation using the lateral approach and
window repositioning and a xenogeneic bone substitute as a grafting
material: A histologic, histomorphometric, and radiographic
analysis. Int J Oral Maxillofac Implants. 33:1089–1096.
2018.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Baksh D, Song L and Tuan RS: Adult
mesenchymal stem cells: Characterization, differentiation, and
application in cell and gene therapy. J Cell Mol Med. 8:301–316.
2004.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kelly MP, Vaughn OL and Anderson PA:
Systematic review and meta-analysis of recombinant human bone
morphogenetic protein-2 in localized alveolar ridge and maxillary
sinus augmentation. J Oral Maxillofac Surg. 74:928–939.
2016.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Väänänen HK: Mesenchymal stem cells. Ann
Med. 37:469–479. 2005.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Dennis JE, Carbillet JP, Caplan AI and
Charbord P: The STRO-1+ marrow cell population is multipotential.
Cells Tissues Organs. 170:73–82. 2002.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Shi S and Gronthos S: Perivascular niche
of postnatal mesenchymal stem cells in human bone marrow and dental
pulp. J Bone Miner Res. 18:696–704. 2003.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Chaudhury H, Raborn E, Goldie LC and
Hirschi KK: Stem cell-derived vascular endothelial cells and their
potential application in regenerative medicine. Cells Tissues
Organs. 195:41–47. 2012.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Litvinov SV, Velders MP, Bakker HA,
Fleuren GJ and Warnaar SO: Ep-CAM: A human epithelial antigen is a
homophilic cell-cell adhesion molecule. J Cell Biol. 125:437–446.
1994.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Strutz F, Okada H, Lo CW, Danoff T, Carone
RL, Tomaszewski JE and Neilson EG: Identification and
characterization of a fibroblast marker: FSP1. J Cell Biol.
130:393–405. 1995.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Draenert FG, Nonnenmacher AL, Kammerer PW,
Goldschmitt J and Wagner W: BMP-2 and bFGF release and in vitro
effect on human osteoblasts after adsorption to bone grafts and
biomaterials. Clin Oral Implants Res. 24:750–757. 2013.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Kao DW, Kubota A, Nevins M and Fiorellini
JP: The negative effect of combining rhBMP-2 and Bio-Oss on bone
formation for maxillary sinus augmentation. Int J Periodontics
Restorative Dent. 32:61–67. 2012.PubMed/NCBI
|
|
53
|
Nguyen V, Meyers CA, Yan N, Agarwal S,
Levi B and James AW: BMP-2-induced bone formation and neural
inflammation. J Orthop. 14:252–256. 2017.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Huang RL, Yuan Y, Tu J, Zou GM and Li Q:
Opposing TNF-α/IL-1β- and BMP-2-activated MAPK signaling pathways
converge on Runx2 to regulate BMP-2-induced osteoblastic
differentiation. Cell Death Dis. 5(e1187)2014.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Aggarwal S and Pittenger MF: Human
mesenchymal stem cells modulate allogeneic immune cell responses.
Blood. 105:1815–1822. 2005.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Sotiropoulou PA and Papamichail M: Immune
properties of mesenchymal stem cells. Methods in molecular biology.
407:225–243. 2007.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Gonzalez-Rey E, Gonzalez MA, Varela N,
O'Valle F, Hernandez-Cortes P, Rico L, Büscher D and Delgado M:
Human adipose-derived mesenchymal stem cells reduce inflammatory
and T cell responses and induce regulatory T cells in vitro in
rheumatoid arthritis. Ann Rheum Dis. 69:241–248. 2010.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Prockop DJ and Oh JY: Mesenchymal
stem/stromal cells (MSCs): Role as guardians of inflammation. Mol
Ther. 20:14–20. 2012.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Castro-Manrreza ME: Participation of
mesenchymal stem cells in the regulation of immune response and
cancer development. Bol Med Hosp Infant Mex. 73:380–387.
2016.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Zara JN, Siu RK, Zhang X, Shen J, Ngo R,
Lee M, Li W, Chiang M, Chung J, Kwak J, et al: High doses of bone
morphogenetic protein 2 induce structurally abnormal bone and
inflammation in vivo. Tissue Eng Part A. 17:1389–1399.
2011.PubMed/NCBI View Article : Google Scholar
|