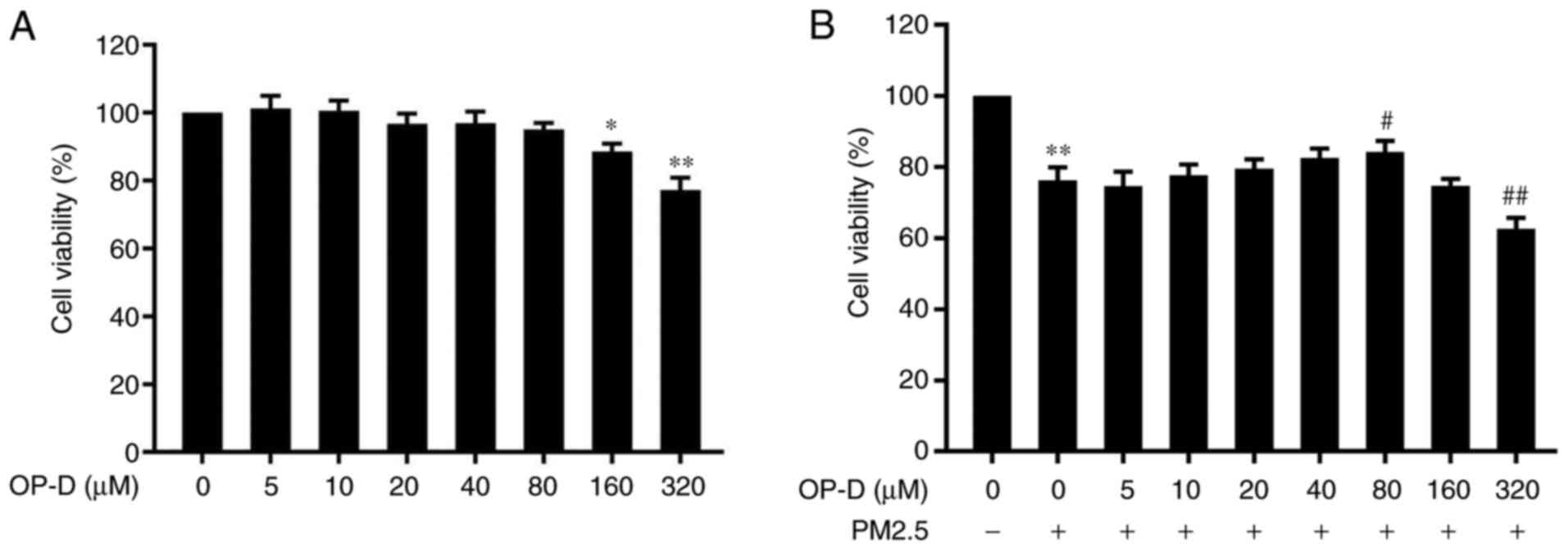
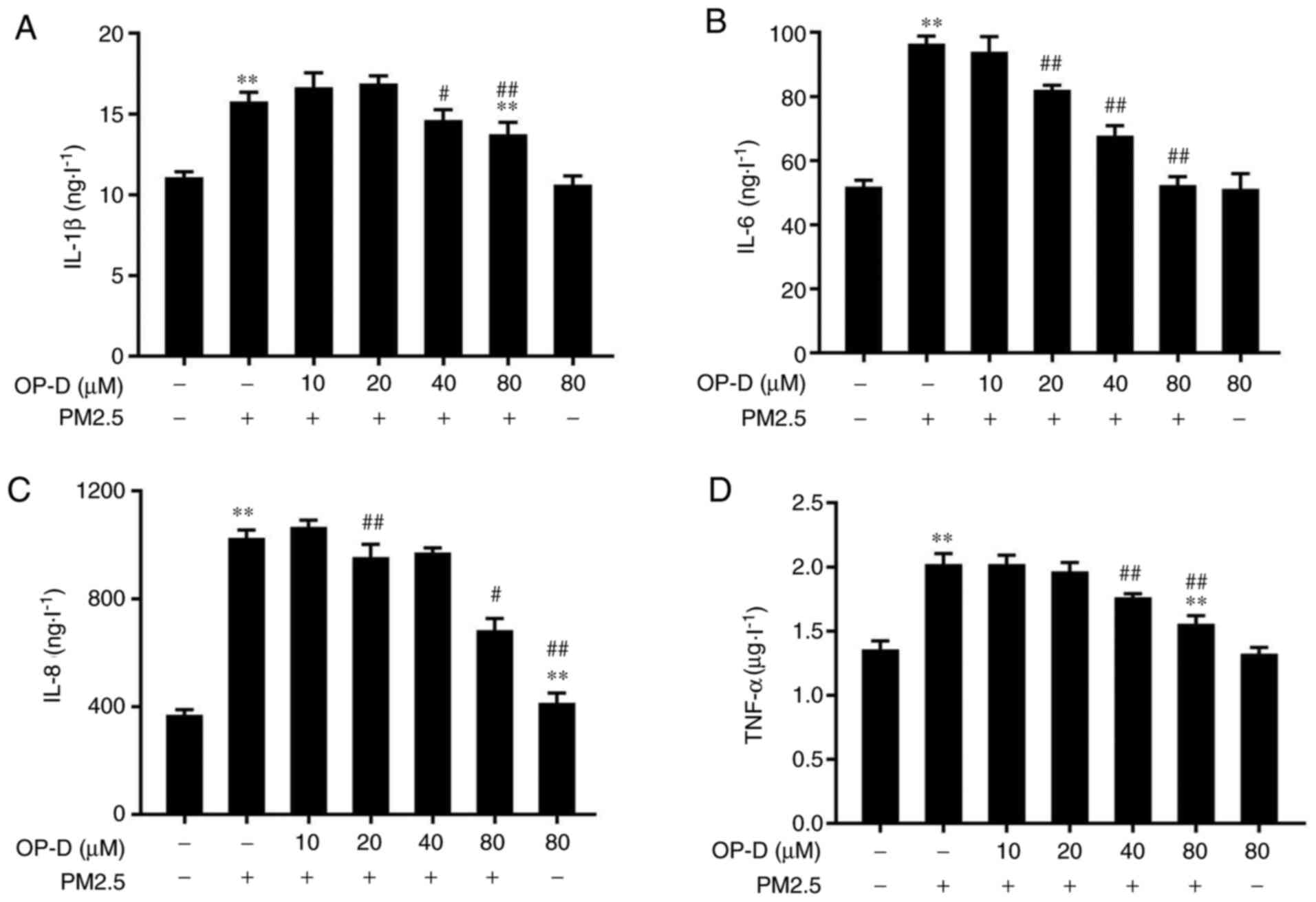
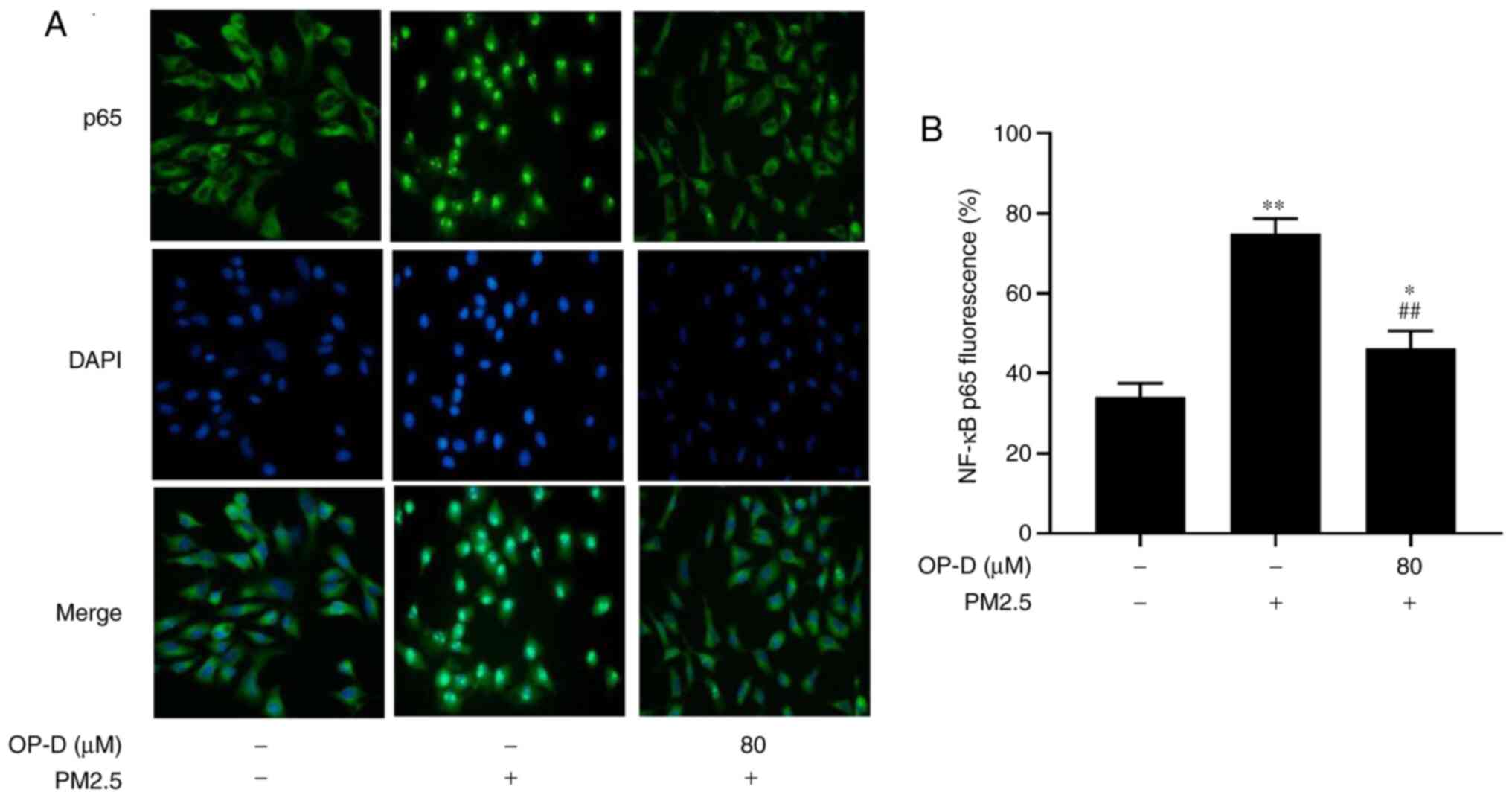
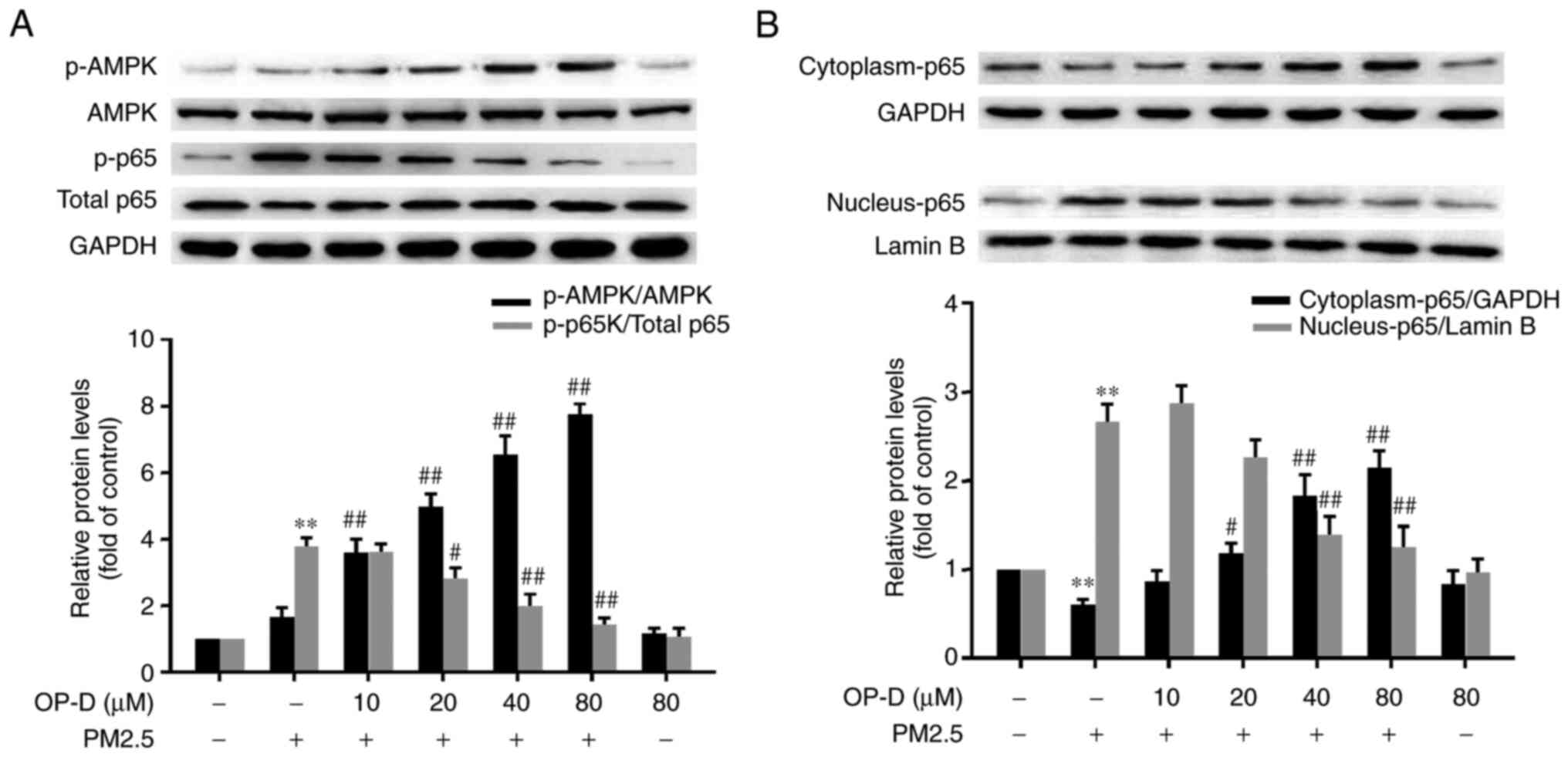
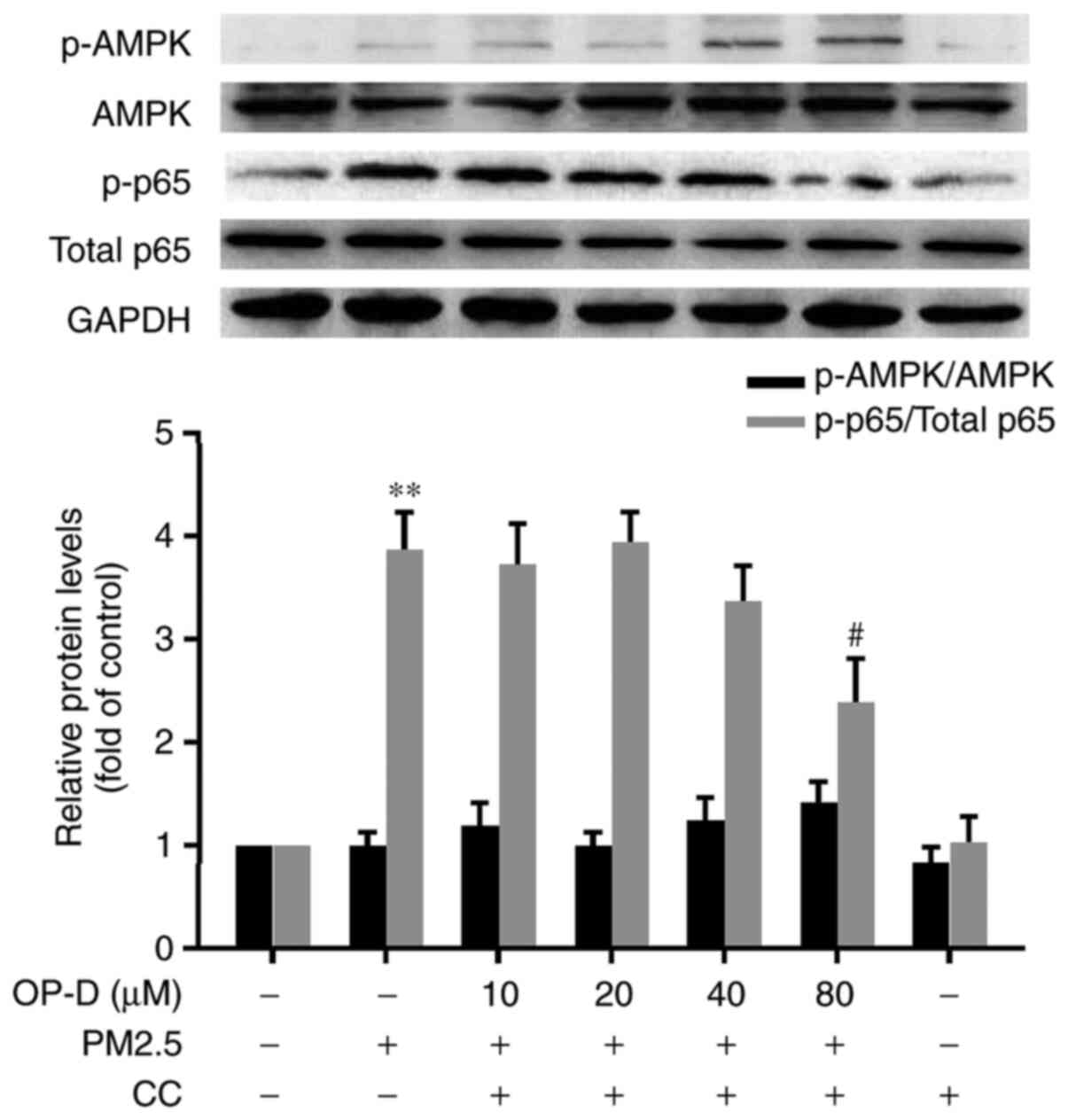
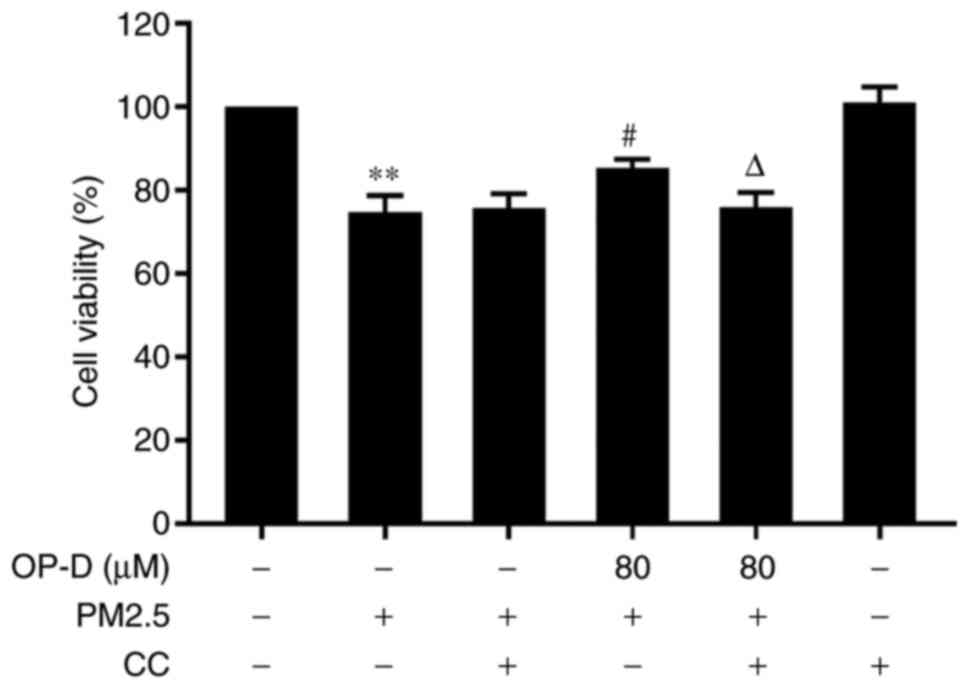
|
1
|
Zheng Q, Liu H, Zhang J and Chen D: The
effect of ambient particle matters on hospital admissions for
cardiac arrhythmia: A multi-city case-crossover study in China.
Environ Health. 17(60)2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Liao J, Yu H, Xia W, Zhang B, Lu B, Cao Z,
Liang S, Hu K, Xu S and Li Y: Exposure to ambient fine particulate
matter during pregnancy and gestational weight gain. Environ Int.
119:407–412. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Sram RJ, Veleminsky M Jr, Veleminsky M Sr
and Stejskalová J: The impact of air pollution to central nervous
system in children and adults. Neuro Endocrinol Lett. 38:389–396.
2017.PubMed/NCBI
|
|
4
|
Xing YF, Xu YH, Shi MH and Lian YX: The
impact of PM2.5 on the human respiratory system. J Thorac Dis.
8:E69–E74. 2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Happo MS, Salonen RO, Hälinen AI, Jalava
PI, Pennanen AS, Kosma VM, Sillanpää M, Hillamo R, Brunekreef B,
Katsouyanni K, et al: Dose and time dependency of inflammatory
responses in the mouse lung to urban air coarse, fine, and
ultrafine particles from six European cities. Inhal Toxicol.
19:227–246. 2007.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wang G, Zhao J, Jiang R and Song W: Rat
lung response to ozone and fine particulate matter (PM2.5)
exposures. Environ Toxicol. 30:343–356. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
de F C Lichtenfels AJ, van der Plaat DA,
de Jong K, van Diemen CC, Postma DS, Nedeljkovic I, van Duijn CM,
Amin N, la Bastide-van Gemert S, de Vries M, et al: Long-term air
pollution exposure, genome-wide DNA methylation and lung function
in the lifelines cohort study. Environ Health Perspect.
126(027004)2018.PubMed/NCBI View
Article : Google Scholar
|
|
8
|
Zhang Y, Wang S, Zhu J, Li C, Zhang T, Liu
H, Xu Q, Ye X, Zhou L and Ye L: Effect of atmospheric PM2.5 on
expression levels of NF-κB genes and inflammatory cytokines
regulated by NF-κB in human macrophage. Inflammation. 41:784–794.
2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
He M, Ichinose T, Yoshida S, Ito T, He C,
Yoshida Y, Arashidani K, Takano H, Sun G and Shibamoto T:
PM2.5-induced lung inflammation in mice: Differences of
inflammatory response in macrophages and type II alveolar cells. J
Appl Toxicol. 37:1203–1218. 2017.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Peixoto CA, Oliveira WH, Araújo SMDR and
Nunes AKS: AMPK activation: Role in the signaling pathways of
neuroinflammation and neurodegeneration. Exp Neurol. 298:31–41.
2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Grahame Hardie D: AMP-activated protein
kinase: A key regulator of energy balance with many roles in human
disease. J Intern Med. 276:543–559. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhang J, Hu Y, Wang D, Qin T, Liu C, Liu
X, Sheng X, Chang S, Fan Y, Guo LW and Nguyen TL: The optimization
of sulfation modification conditions for ophiopogonpolysaccharide
based on antiviral activity. Int J Biol Macromol. 51:657–662.
2012.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Liang H, Xing Y, Chen J, Zhang D, Guo S
and Wang C: Antimicrobial activities of endophytic fungi isolated
from Ophiopogon japonicus (Liliaceae). BMC Complement Altern
Med. 12(238)2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kou J, Sun Y, Lin Y, Cheng Z, Zheng W, Yu
B and Xu Q: Anti-inflammatory activities of aqueous extract from
Radix Ophiopogon japonicus and its two constituents. Biol
Pharm Bull. 28:1234–1238. 2005.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chang JM, Shen CC, Huang YL, Chien MY, Ou
JC, Shieh BJ and Chen CC: Five new homoisoflavonoids from the tuber
of Ophiopogon japonicus. J Nat Prod. 65:1731–1733.
2002.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kou J, Tian Y, Tang Y, Yan J and Yu B:
Antithrombotic activities of aqueous extract from Radix
Ophiopogon japonicus and its two constituents. Biol Pharm
Bull. 29:1267–1270. 2006.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Takahama K and Miyata T: Cough-diversity
and the peripheral mechanisms of production. Nihon Yakurigaku
Zasshi. 105:41–52. 1995.(In Japanese). PubMed/NCBI View Article : Google Scholar
|
|
18
|
Qian J, Jiang F, Wang B, Yu Y, Zhang X,
Yin Z and Liu C: Ophiopogonin D prevents H2O2-induced injury in
primary human umbilical vein endothelial cells. J Ethnopharmacol.
128:438–445. 2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhang YY, Meng C, Zhang XM, Yuan CH, Wen
MD, Chen Z, Dong DC, Gao YH, Liu C and Zhang Z: Ophiopogonin D
attenuates doxorubicin-induced autophagic cell death by relieving
mitochondrial damage in vitro and in vivo. J Pharmacol Exp Ther.
352:166–174. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Song L, Li D, Li X, Ma L, Bai X, Wen Z,
Zhang X, Chen D and Peng L: Exposure to PM2.5 induces aberrant
activation of NF-κB in human airway epithelial cells by
downregulating miR-331 expression. Environ Toxicol Pharmacol.
50:192–199. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Monthly report on Urban air quality from
the Ministry of ecology and environment of the People's Republic of
China. http://www.mee.gov.cn/hjzl/dqhj/cskqzlzkyb/index_2.shtml.
Accessed August 11, 2020.
|
|
22
|
Song L, Li D, Gu Y, Li X and Peng L:
Let-7a modulates particulate matter (≤2.5 µm)-induced oxidative
stress and injury in human airway epithelial cells by targeting
arginase 2. J Appl Toxicol. 36:1302–1310. 2016.PubMed/NCBI View
Article : Google Scholar
|
|
23
|
Ogino K, Nagaoka K, Ito T, Takemoto 1,
Okuda T, Nakayama SF, Ogino N, Seki Y, Hamada H, Takashiba S and
Fujikura Y: Involvement of PM2.5-bound protein and metals in
PM2.5-induced allergic airway inflammation in mice. Inhal Toxicol.
30:498–508. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ogino K, Nagaoka K, Okuda T, Oka A, Kubo
M, Eguchi E and Fujikura Y: PM2.5-induced airway inflammation and
hyperresponsiveness in NC/Nga mice. Environ Toxicol. 32:1047–1054.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yue W, Tong L, Liu X, Weng X, Chen X, Wang
D, Dudley SC, Weir EK, Ding W, Lu Z, et al: Short term Pm2.5
exposure caused a robust lung inflammation, vascular remodeling,
and exacerbated transition from left ventricular failure to right
ventricular hypertrophy. Redox Biol. 22(101161)2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Li R, Zhou R and Zhang J: Function of
PM2.5 in the pathogenesis of lung cancer and chronic airway
inflammatory diseases. Oncol Lett. 15:7506–7514. 2018.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Akther T, Ahmed M, Shohel M, Ferdousi FK
and Salam A: Particulate matters and gaseous pollutants in indoor
environment and association of ultra-fine particulate matters
(PM1) with lung function. Environ Sci Pollut Res Int.
26:5475–5484. 2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Jeong SC, Cho Y, Song MK, Lee E and Ryu
JC: Epidermal growth factor receptor (EGFR)-MAPK-nuclear
factor(NF)-κB-IL8: A possible mechanism of particulate matter(PM)
2.5-induced lung toxicity. Environ Toxicol. 32:1628–1636.
2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Li R, Zhao L, Tong J, Yan Y and Xu C: Fine
particulate matter and sulfur dioxide coexposures induce rat lung
pathological injury and inflammatory responses Via TLR4/p38/NF-κB
pathway. Int J Toxicol. 36:165–173. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Tilborghs S, Corthouts J, Verhoeven Y,
Arias D, Rolfo C, Trinh XB and Dam PA: The role of nuclear
factor-kappa B signaling in human cervical cancer. Crit Rev Oncol
Hematol. 120:141–150. 2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Leung TH, Hoffmann A and Baltimore D: One
nucleotide in a kappaB site can determine cofactor specificity for
NF-kappaB dimers. Cell. 118:453–464. 2004.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Mukherjee SP, Behar M, Birnbaum HA,
Hoffmann A, Wright PE and Ghosh G: Analysis of the RelA: CBP/p300
interaction reveals its involvement in NF-κB-driven transcription.
PLoS Biol. 11(e1001647)2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Dagher Z, Garcon G, Billet S, Verdin A,
Ledoux F, Courcot D, Aboukais A and Shirali P: Role of nuclear
factor-kappa B activation in the adverse effects induced by air
pollution particulate matter (PM2.5) in human epithelial lung cells
(L132) in culture. J Appl Toxicol. 27:284–290. 2007.PubMed/NCBI View
Article : Google Scholar
|
|
34
|
Jacquemin B, Lanki T, Yli-Tuomi T, Vallius
M, Hoek G, Heinrich J, Timonen K and Pekkanen J: Source
category-specific PM2.5 and urinary levels of Clara cell protein
CC16. The ULTRA study. Inhal Toxicol. 21:1068–1076. 2009.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Longhin E, Holme JA, Gutzkow KB, Arlt VM,
Kucab JE, Camatini M and Gualtieri M: Cell cycle alterations
induced by urban PM2.5 in bronchial epithelial cells:
Characterization of the process and possible mechanisms involved.
Part Fibre Toxicol. 10(63)2013.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Ke R, Xu Q, Li C, Luo L and Huang D:
Mechanisms of AMPK in the maintenance of ATP balance during energy
metabolism. Cell Biol Int. 42:384–392. 2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Cheng XY, Li YY, Huang C, Li J and Yao HW:
AMP-activated protein kinase reduces inflammatory responses and
cellular senescence in pulmonary emphysema. Oncotarget.
8:22513–22523. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Zhang J, Zhang Y, Xiao F, Liu Y, Wang J,
Gao H, Rong S, Yao Y, Li J and Xu G: The peroxisome
proliferator-activated receptor γ agonist pioglitazone prevents
NF-κB activation in cisplatin nephrotoxicity through the reduction
of p65 acetylation via the AMPK-SIRT1/p300 pathway. Biochem
Pharmacol. 101:100–111. 2016.PubMed/NCBI View Article : Google Scholar
|