Case Report
Open Access
Hydroxyurea‑induced superinfected ulcerations: Two case reports and review of the literature
- Authors:
- Gabriela Mariana Iancu
- Anca Ocneanu
- Maria Rotaru
-
View Affiliations / Copyright
Affiliations:
Dermatology Department, Faculty of Medicine, University Lucian Blaga of Sibiu, 550169 Sibiu, Romania, Dermatology Office, Grivita Medical Center, 810521 Braila, Romania
-
Article Number:
191
|
Published online on:
October 14, 2020
https://doi.org/10.3892/etm.2020.9321
- Expand metrics +
Metrics:
Total
Views: 0
(Spandidos Publications: | PMC Statistics:
)
Metrics:
Total PDF Downloads: 0
(Spandidos Publications: | PMC Statistics:
)
This article is mentioned in:
Abstract
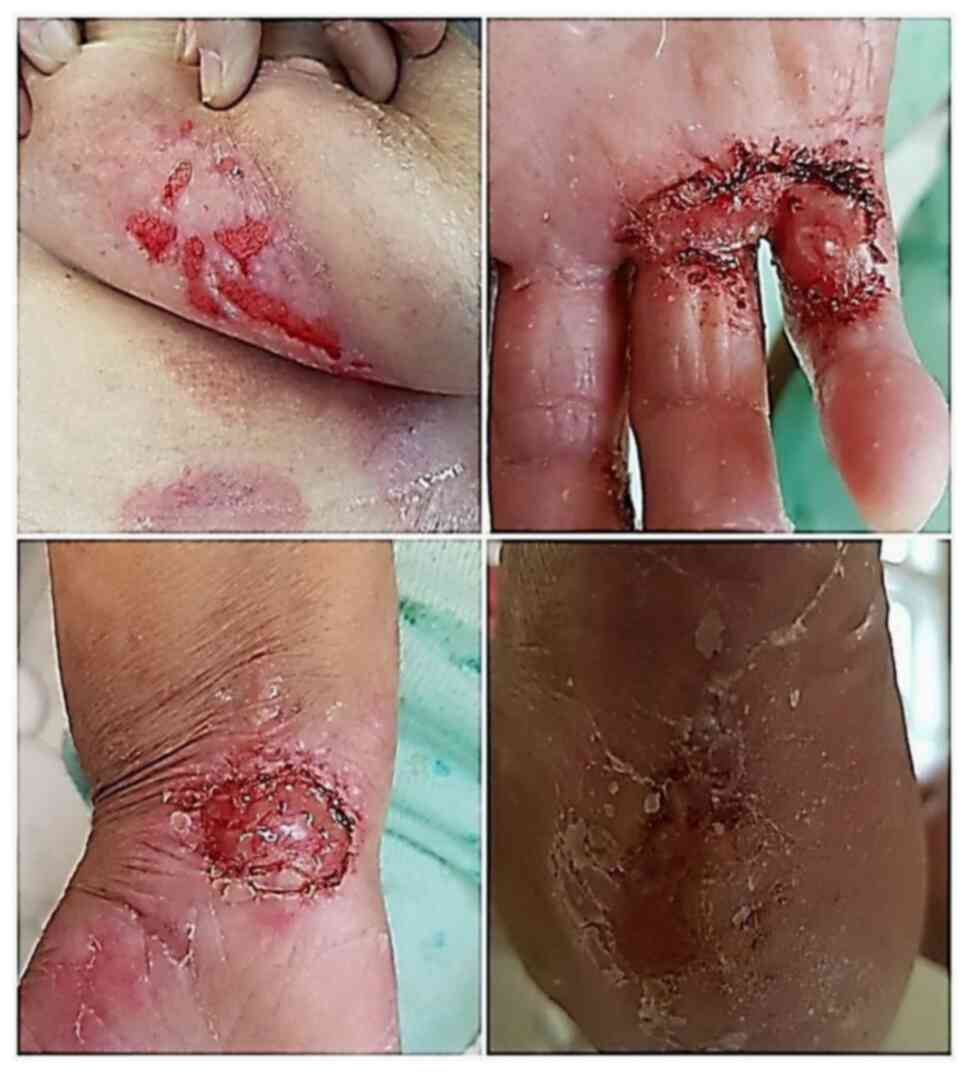
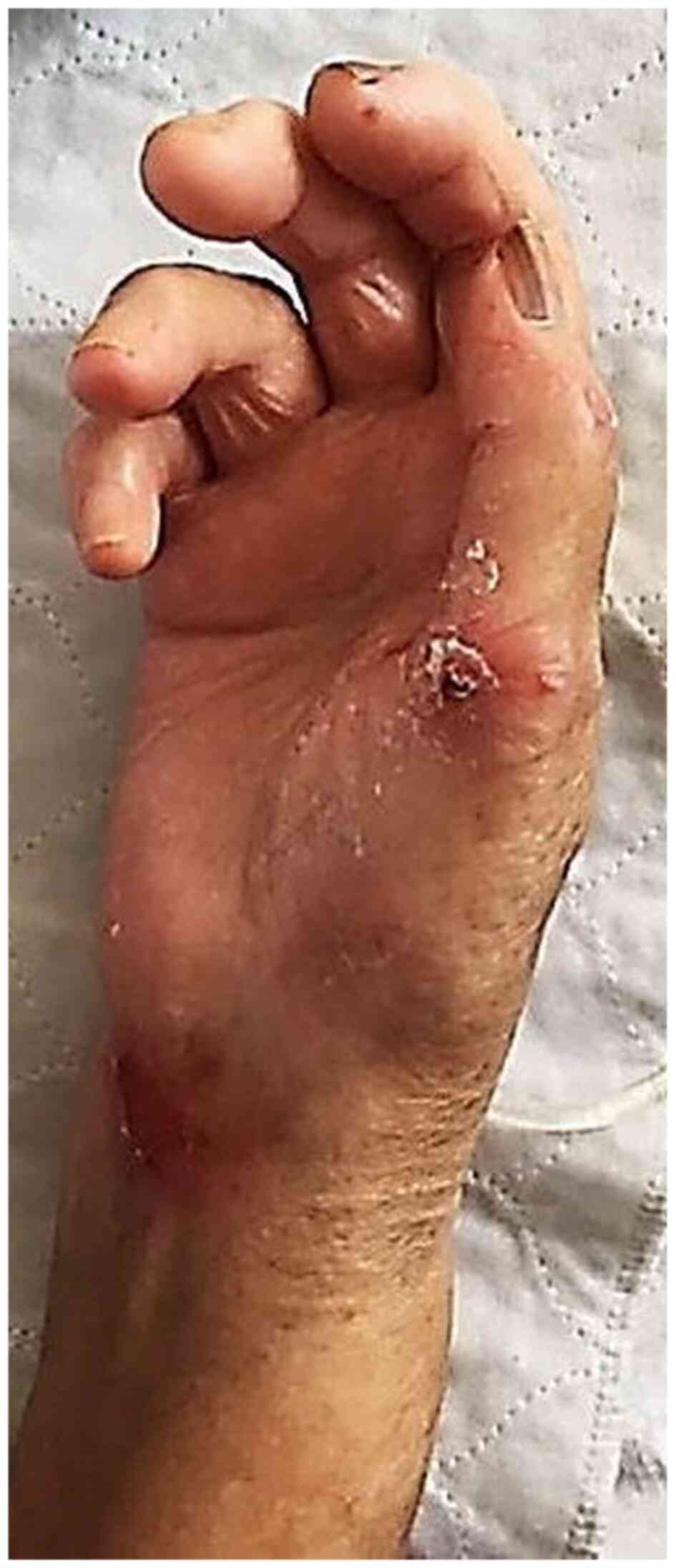
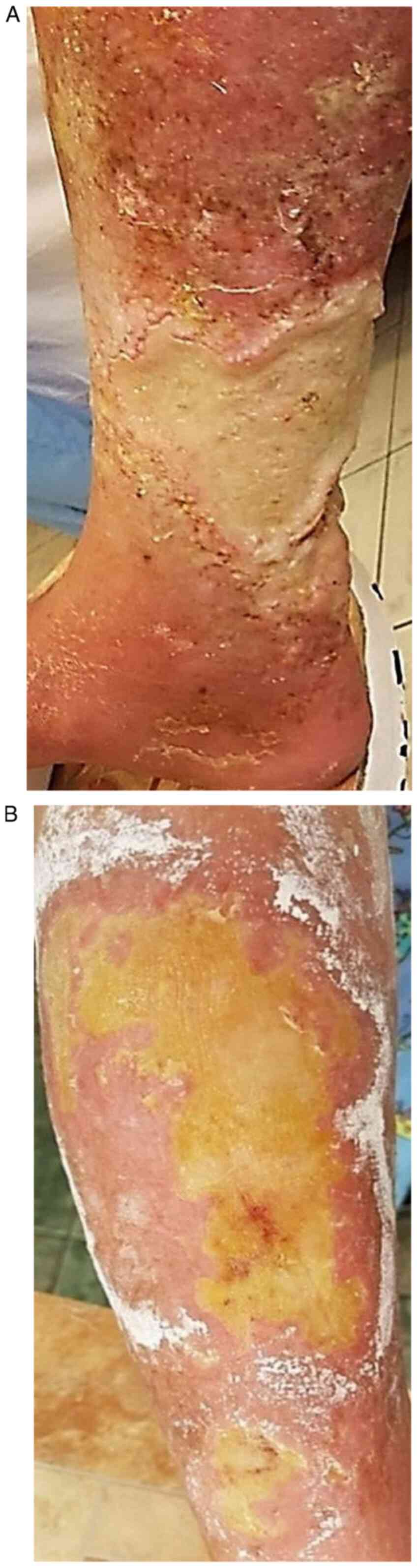
The chronic use of hydroxyurea (HU) in some oncologic and non‑oncologic diseases (psoriasis, sickle cell anemia) can be accompanied by side effects, both systemic and mucocutaneous. The most severe adverse events known in HU therapy are leg ulcers and cutaneous carcinomas. At skin level may also appear: xerosis, persistent pruritus, skin color changes (erythema, hyperpigmentation), cutaneous atrophy. Likewise, oral ulcerations and stomatitis may occur at mucosal level. Hair damage can be expressed through alopecia and nail damage through melanonychia and oncycholysis. First case, a 63‑year‑old woman with severe psoriasis vulgaris and chronic granulocytic leukemia, with 5 years of HU therapy, was admitted to hospital for submammary and palmoplantar ulcers, superinfected with methicillin‑resistant Staphylococcus aureus and Proteus mirabilis. Clinical exam showed that the patient had also cutaneous atrophy, marked palmoplantar xerosis and melanonychia. The second case, a 72‑year‑old woman with primary thrombocytemia, treated with HU for 3 years, presented with necrotic leg ulcers that were superinfected with Pseudomonas aeruginosa, Enterobacter and E. Coli. The patient associates cellulitis, microbial eczema and xeroderma. In both cases, after HU discontinuation, systemic antibiotics, topical epithelizing agents and emollients, the ulcers had a slow favorable evolution. In our cases, the ulcers appeared after 5, respectively 3 years of HU therapy. It is stressed that in the first case, which had associated psoriasis, after 1 year of 1 g of HU/day, the psoriatic lesions completely disappeared. The severe progression of the ulcers was also favored by the superinfection of the ulcers with 2, respectively, 3 identified germs for which appropriate systemic antibiotics was required.
View References
|
1
|
Gwilt PR and Tracewell WG:
Pharmacokinetics and pharmacodynamics of hydroxyurea. Clin
Pharmacokinet. 34:347–358. 1998.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Yarbro JW: Mechanism of action of
hydroxyurea. Semin Oncol. 19 (3 Suppl 9):S1–S10. 1992.PubMed/NCBI
|
|
3
|
National Library of Medicine (US),
National Center for Biotechnology Information: PubChem Compound
Summary for CID 3657, Hydroxyurea. urihttps://pubchem.ncbi.nlm.nih.gov/compound/Hydroxyureasimplehttps://pubchem.ncbi.nlm.nih.gov/compound/Hydroxyurea.
Accessed August 4, 2020.
|
|
4
|
Chaine B, Neonato MG, Girot R and
Aractingi S: Cutaneous adverse reactions to hydroxyurea in patients
with sickle cell disease. Arch Dermatol. 137:467–470.
2001.PubMed/NCBI
|
|
5
|
Dissemond J and Körber A:
Hydroxyurea-induced ulcers on the leg. CMAJ.
180(1132)2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
França ER, Teixeira MA, Matias Kde F,
Antunes DE, Braz Rde A and Silva CE: Cutaneous effects after
prolonged use of hydroxyurea in polycythemia Vera. An Bras
Dermatol. 86:751–754. 2011.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Branisteanu DE, Nichifor M, Dorobat CM,
Branisteanu DC, Petrariu FD, Molodoi AD, Radu DC and Boda D: Use of
textile biomaterials for the topic treatment of chronic venous
disease. Rom Biotechnol Lett. 20:10618–10625. 2015.
|
|
8
|
Grigore O, Mihailescu AI, Solomon I, Boda
D and Caruntu C: Role of stress in modulation of skin neurogenic
inflammation. Exp Ther Med. 17:997–1003. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Oakley A: Hydroxyurea. DermNet NZ, New
Zealand, 2001. urihttps://www.dermnetnz.org/topics/hydroxyureasimplehttps://www.dermnetnz.org/topics/hydroxyurea.
Accessed June 23, 2019.
|
|
10
|
Hoff NP, Akanay-Diesel S, Pippirs U,
Schulte KW and Hanneken S: Cutaneous side effects of hydroxyurea
treatment for polycythemia Vera. Hautarzt. 60:783–787.
2009.PubMed/NCBI View Article : Google Scholar : (In German).
|
|
11
|
Soutou B and Aractingi S:
Myeloproliferative disorder therapy: Assessment and management of
adverse events - a dermatologist's perspective. Hematol Oncol. 27
(Suppl 1):S11–S13. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
12
|
Gropper CA, Don PC and Sadjadi MM: Nail
and skin hyperpigmentation associated with hydroxyurea therapy for
polycythemia Vera. Int J Dermatol. 32:731–733. 1993.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Neynaber S, Wolff H, Plewig G and Wienecke
R: Longitudinal melanonychia induced by hydroxyurea therapy. J
Dtsch Dermatol Ges. 2:588–5891. 2004.PubMed/NCBI View Article : Google Scholar : (In German).
|
|
14
|
Wiechert A, Reinhard G, Tüting T, Uerlich
M, Bieber T and Wenzel J: Multiple skin cancers in a patient
treated with hydroxyurea. Hautarzt. 60:651–652, 654.
2009.PubMed/NCBI View Article : Google Scholar : (In German).
|