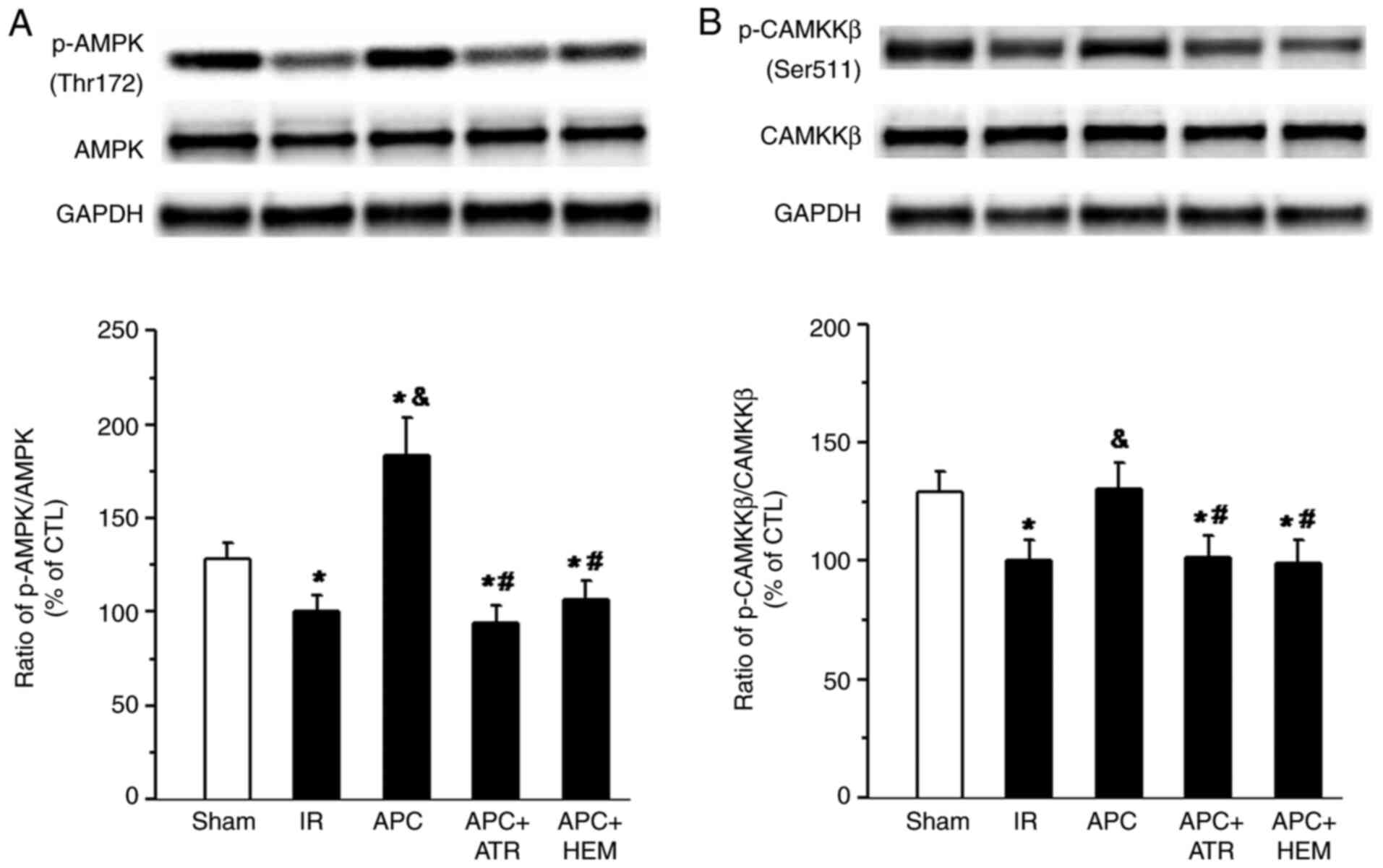
|
1
|
An J, Varadarajan SG, Novalija E and Stowe
DF: Ischemic and anesthetic preconditioning reduces cytosolic
[Ca2+] and improves Ca(2+) responses in intact hearts. Am J Physiol
Heart Circ Physiol. 281:H1508–H1523. 2001.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wang C, Qiao S, Hong L, Sun J, Che T, An J
and Camara AKS: NOS cofactor tetrahydrobiopterin contributes to
anesthetic preconditioning induced myocardial protection in the
isolated ex vivo rat heart. Int J Mol Med. 45:615–622.
2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhang J, Yong Y, Li X, Hu Y, Wang J, Wang
YQ, Song W, Chen WT, Xie J, Chen XM, et al: Vagal modulation of
high mobility group box-1 protein mediates
electroacupuncture-induced cardioprotection in ischemia-reperfusion
injury. Sci Rep. 5(15503)2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Nuntaphum W, Pongkan W, Wongjaikam S,
Thummasorn S, Tanajak P, Khamseekaew J, Intachai K, Chattipakorn
SC, Chattipakorn N and Shinlapawittayatorn K: Vagus nerve
stimulation exerts cardioprotection against myocardial
ischemia/reperfusion injury predominantly through its efferent
vagal fibers. Basic Res Cardiol. 113(22)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhao M, He X, Bi XY, Yu XJ, Gil Wier W and
Zang WJ: Vagal stimulation triggers peripheral vascular protection
through the cholinergic anti-inflammatory pathway in a rat model of
myocardial ischemia/reperfusion. Basic Res Cardiol.
108(345)2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wang Z, Yu L, Wang S, Huang B, Liao K,
Saren G, Tan T and Jiang H: Chronic intermittent low-level
transcutaneous electrical stimulation of auricular branch of vagus
nerve improves left ventricular remodeling in conscious dogs with
healed myocardial infarction. Circ Heart Fail. 7:1014–1021.
2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Uitterdijk A, Yetgin T, te Lintel Hekkert
M, Sneep S, Krabbendam-Peters I, van Beusekom HM, Fischer TM,
Cornelussen RN, Manintveld OC, Merkus D and Duncker DJ: Vagal nerve
stimulation started just prior to reperfusion limits infarct size
and no-reflow. Basic Res Cardiol. 110(508)2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhang R, Wugeti N, Sun J, Yan H, Guo Y,
Zhang L, Ma M, Guo X, Jiao C, Xu W, et al: Effects of vagus nerve
stimulation via cholinergic anti-inflammatory pathway activation on
myocardial ischemia/reperfusion injury in canine. Int J Clin Exp
Med. 7:2615–2623. 2014.PubMed/NCBI
|
|
9
|
Calvillo L, Vanoli E, Andreoli E, Besana
A, Omodeo E, Gnecchi M, Zerbi P, Vago G, Busca G and Schwartz PJ:
Vagal stimulation, through its nicotinic action, limits infarct
size and the inflammatory response to myocardial ischemia and
reperfusion. J Cardiovasc Pharmacol. 58:500–507. 2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kiss A, Tratsiakovich Y, Mahdi A, Yang J,
Gonon AT, Podesser BK and Pernow J: Vagal nerve stimulation reduces
infarct size via a mechanism involving the -7 nicotinic
acetylcholine receptor and downregulation of cardiac and vascular
arginase. Acta Physiol. 221:174–181. 2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Qiao SG, Sun Y, Sun B, Wang A, Qiu J, Hong
L, An JZ, Wang C and Zhang HL: Sevoflurane postconditioning
protects against myocardial ischemia/reperfusion injury by
restoring autophagic flux via an NO-dependent mechanism. Acta
Pharmacol Sin. 40:35–45. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Balligand JL, Kobzik L, Han X, Kaye DM,
Belhassen L, O'Hara DS, Kelly RA, Smith TW and Michel T: Nitric
oxide-dependent parasympathetic signaling is due to activation of
constitutive endothelial (type III) nitric oxide synthase in
cardiac myocytes. J Biol Chem. 270:14582–14586. 1995.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Hao M, Zhu S, Hu L, Zhu H, Wu X and Li Q:
Myocardial ischemic postconditioning promotes autophagy against
ischemia reperfusion injury via the activation of the
nNOS/AMPK/mTOR pathway. Int J Mol Sci. 18(614)2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bi X, He X, Xu M, Zhao M, Yu X, Lu X and
Zang W: Acetylcholine ameliorates endoplasmic reticulum stress in
endothelial cells after hypoxia/reoxygenation via M3 AChR-AMPK
signaling. Cell Cycle. 14:2461–2472. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Xue RQ, Sun L, Yu XJ, Li DL and Zang WJ:
Vagal nerve stimulation improves mitochondrial dynamics via an M3
receptor/CaMKKbeta/AMPK pathway in isoproterenol-induced myocardial
ischaemia. J Cell Mol Med. 21:58–71. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Mavropoulos SA, Khan NS, Levy ACJ, Faliks
BT, Sison CP, Pavlov VA, Zhang Y and Ojamaa K: Nicotinic
acetylcholine receptor-mediated protection of the rat heart exposed
to ischemia reperfusion. Mol Med. 23:120–133. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Pickard JMJ, Burke N, Davidson SM and
Yellon DM: Intrinsic cardiac ganglia and acetylcholine are
important in the mechanism of ischaemic preconditioning. Basic Res
Cardiol. 112(11)2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
An J, Rhodes SS, Jiang MT, Bosnjak ZJ,
Tian M and Stowe DF: Anesthetic preconditioning enhances
Ca2+ handling and mechanical and metabolic function
elicited by Na+/Ca2+ exchange inhibition in
isolated hearts. Anesthesiology. 105:541–549. 2006.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Fang L, Xu Z, Lu J, Hong L, Qiao S, Liu L
and An J: Cardioprotective effects of triiodothyronine
supplementation against ischemia reperfusion injury by preserving
calcium cycling proteins in isolated rat hearts. Exp Ther Med.
18:4935–4941. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Sasamori J, Abe Y, Marunouchi T, Manome Y,
Uchibori T and Tanonaka K: Effects of 2-octynyladenosine (YT-146)
on mitochondrial function in ischemic/reperfused rat hearts. Biol
Pharm Bull. 38:1946–1953. 2015.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Intachai K, C Chattipakorn S, Chattipakorn
N and Shinlapawittayatorn K: Revisiting the cardioprotective
effects of acetylcholine receptor activation against myocardial
ischemia/reperfusion injury. Int J Mol Sci. 19(2466)2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhao M, Sun L, Yu XJ, Miao Y, Liu JJ, Wang
H, Ren J and Zang WJ: Acetylcholine mediates AMPK-dependent
autophagic cytoprotection in H9c2 cells during
hypoxia/reoxygenation injury. Cell Physiol Biochem. 32:601–613.
2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Shinlapawittayatorn K, Chinda K, Palee S,
Surinkaew S, Kumfu S, Kumphune S, Chattipakorn S, KenKnight BH and
Chattipakorn N: Vagus nerve stimulation initiated late during
ischemia, but not reperfusion, exerts cardioprotection via
amelioration of cardiac mitochondrial dysfunction. Heart Rhythm.
11:2278–2287. 2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Palee S, Apaijai N, Shinlapawittayatorn K,
Chattipakorn SC and Chattipakorn N: Acetylcholine attenuates
hydrogen peroxide-induced intracellular calcium dyshomeostasis
through both muscarinic and nicotinic receptors in cardiomyocytes.
Cell Physiol Biochem. 39:341–349. 2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Li DL, Liu BH, Sun L, Zhao M, He X, Yu XJ
and Zang WJ: Alterations of muscarinic acetylcholine receptors-2, 4
and α7-nicotinic acetylcholine receptor expression after
ischaemia/reperfusion in the rat isolated heart. Clin Exp Pharmacol
Physiol. 37:1114–1119. 2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Roy A, Fields WC, Rocha-Resende C, Resende
RR, Guatimosim S, Prado VF, Gros R and Prado MA:
Cardiomyocyte-secreted acetylcholine is required for maintenance of
homeostasis in the heart. FASEB J. 27:5072–5082. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kakinuma Y, Akiyama T, Okazaki K, Arikawa
M, Noguchi T and Sato T: A non-neuronal cardiac cholinergic system
plays a protective role in myocardium salvage during ischemic
insults. PLoS One. 7(e50761)2012.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Oikawa S, Kai Y, Tsuda M, Ohata H, Mano A,
Mizoguchi N, Sugama S, Nemoto T, Suzuki K, Kurabayashi A, et al:
Non-neuronal cardiac cholinergic system influences CNS via the
vagus nerve to acquire a stress-refractory propensity. Clin Sci
(Lond). 130:1913–1928. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Pang L, Cai Y, Tang EH, Yan D, Kosuru R,
Li H, Irwin MG, Ma H and Xia Z: Cox-2 inhibition protects against
hypoxia/reoxygenation-induced cardiomyocyte apoptosis via
Akt-dependent enhancement of iNOS expression. Oxid Med Cell Longev.
2016(3453059)2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Li XD, Yang YJ, Geng YJ, Zhao JL, Zhang
HT, Cheng YT and Wu YL: Phosphorylation of endothelial NOS
contributes to simvastatin protection against myocardial no-reflow
and infarction in reperfused swine hearts: Partially via the PKA
signaling pathway. Acta Pharmacol Sin. 33:879–887. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Cao J, Xie H, Sun Y, Zhu J, Ying M, Qiao
S, Shao Q, Wu H and Wang C: Sevoflurane post-conditioning reduces
rat myocardial ischemia reperfusion injury through an increase in
NOS and a decrease in phopshorylated NHE1 levels. Int J Mol Med.
36:1529–1537. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Ge ZD, Pravdic D, Bienengraeber M, Pratt
PF Jr, Auchampach JA, Gross GJ, Kersten JR and Warltier DC:
Isoflurane postconditioning protects against reperfusion injury by
preventing mitochondrial permeability transition by an endothelial
nitric oxide synthase-dependent mechanism. Anesthesiology.
112:73–85. 2010.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Sun L, Lu J, Yu XJ, Li DL, Xu XL, Wang B,
Ren KY, Liu JK and Zang WJ: Adenine sulfate improves cardiac
function and the cardiac cholinergic system after myocardial
infarction in rats. J Pharmacol Sci. 115:205–213. 2011.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Waid DK, Chell M and El-Fakahany EE: M(2)
and M(4) muscarinic receptor subtypes couple to activation of
endothelial nitric oxide synthase. Pharmacology. 61:37–42.
2000.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Dong Y, Zhang M, Liang B, Xie Z, Zhao Z,
Asfa S, Choi HC and Zou MH: Reduction of AMP-activated protein
kinase alpha2 increases endoplasmic reticulum stress and
atherosclerosis in vivo. Circulation. 121:792–803. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kataoka Y, Shibata R, Ohashi K, Kambara T,
Enomoto T, Uemura Y, Ogura Y, Yuasa D, Matsuo K, Nagata T, et al:
Omentin prevents myocardial ischemic injury through AMP-activated
protein kinase- and Akt-dependent mechanisms. J Am Coll Cardiol.
63:2722–2733. 2014.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Viollet B, Horman S, Leclerc J, Lantier L,
Foretz M, Billaud M, Giri S and Andreelli F: AMPK inhibition in
health and disease. Crit Rev Biochem Mol Biol. 45:276–295.
2010.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Mukherjee D, Ghosh AK, Bandyopadhyay A,
Basu A, Datta S, Pattari SK, Reiter RJ and Bandyopadhyay D:
Melatonin protects against isoproterenol-induced alterations in
cardiac mitochondrial energy-metabolizing enzymes, apoptotic
proteins, and assists in complete recovery from myocardial injury
in rats. J Pineal Res. 53:166–179. 2012.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Groenendyk J, Sreenivasaiah PK, Kim DH,
Agellon LB and Michalak M: Biology of endoplasmic reticulum stress
in the heart. Circ Res. 107:1185–1197. 2010.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lin E and Symons JA: Volatile anaesthetic
myocardial protection: A review of the current literature. HSR Proc
Intensive Care Cardiovasc Anesth. 2:105–109. 2010.PubMed/NCBI
|
|
41
|
Turek Z, Sykora R, Matejovic M and Cerny
V: Anesthesia and the microcirculation. Semin Cardiothorac Vasc
Anesth. 13:249–258. 2009.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Harvey KL, Hussain A and Maddock HL:
Ipratropium bromide-mediated myocardial injury in in vitro models
of myocardial ischaemia/reperfusion. Toxicol Sci. 138:457–467.
2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Buchholz B, Donato M, Perez V, Deutsch
ACR, Höcht C, Del Mauro JS, Rodríguez M and Gelpi RJ: Changes in
the loading conditions induced by vagal stimulation modify the
myocardial infarct size through sympathetic-parasympathetic
interactions. Pflugers Arch. 467:1509–1522. 2015.PubMed/NCBI View Article : Google Scholar
|