Introduction
Lipopolysaccharide (LPS) has been reported to induce
sepsis-associated pulmonary fibrosis by directly activating the
lung fibroblasts and promoting their proliferation, transformation
and subsequent collagen synthesis (1-3).
Pulmonary fibrosis contributes significantly to the high mortality
rates in patients with respiratory distress syndrome (ARDS)
(4). Previous studies have
demonstrated that sepsis-related pulmonary fibrosis and its severe
form, ARDS, are closely associated with metabolic alterations
(5,6). Aerobic glycolysis and the Warburg
effect have been proposed as an important pathogenic mechanism in
the development of pulmonary fibrosis (7,8).
Notably, it has been reported that pulmonary aerobic glycolysis in
the LPS-treated mouse model contributes to the abnormal activation
of lung fibroblasts with excessive collagen deposition (9). Furthermore,
phosphofructokinase-2/fructose-2, 6-bisphosphatase 3 (PFKFB3),
which is a critical regulatory enzyme for glycolysis, could
modulate fructose-2, 6-bisphosphate (Fru-2,6-BP) synthesis, enhance
glucose absorption and promote lactate production in a positive
manner (10). In addition,
PFKFB3-driven aerobic glycolysis has been reported to promote
pulmonary collagen synthesis (5).
Targeting PFKFB3-mediated aerobic glycolysis in lung fibroblasts
may therefore serve as a potentially effective therapy for
LPS-induced pulmonary fibrosis.
Metformin, which is a biguanide anti-hyperglycemic
agent (11), has been demonstrated
to alleviate pulmonary fibrosis in vivo and in vitro.
For example, Gamad et al (12) demonstrated that metformin could
attenuate bleomycin-induced pulmonary fibrosis in rats. Wang et
al (13) demonstrated that
metformin can protect lungs in radiation-induced pulmonary fibrosis
by ameliorating collagen formation. The present study hypothesized
therefore that metformin may serve a crucial role in reducing
LPS-mediated pulmonary fibrosis. However, the underlying mechanism
remains largely unclear.
Previous studies demonstrated that metformin could
modulate glycolysis by activating the AMP-activated protein kinase
(AMPK) (14). Briefly, metformin
has been shown to directly prohibit respiratory-chain complex 1 of
the mitochondrial electron transport chain, resulting in the
decrease in adenosine triphosphate (ATP) synthesis and subsequent
increased adenosine monophosphate (AMP)/ATP or adenosine
diphosphate (ADP)/ATP ratio, leading to AMPK activation by binding
to either AMP or ADP (15,16). Mammalian target of rapamycin (mTOR),
a downstream target of AMPK, can regulate protein synthesis and
cell proliferation via the phosphorylation of eukaryotic
translation initiation factor 4E binding protein 1 (4E-BP1)
(17-19).
However, enhanced AMPK activity can inhibit rat mesangial cell
proliferation and extracellular matrix deposition in high glucose
condition with phosphorylation of mTOR and 4E-BP1(20). A previous study demonstrated that
mTOR activation can promote collagen synthesis in LPS-challenged
lung fibroblasts by upregulating the PFKFB3-meditaed aerobic
glycolysis (9). The present study
therefore hypothesized that metformin, an AMPK agonist, may
alleviate the LPS-induced pulmonary fibrosis by regulating
PFKFB3-mediated aerobic glycolysis.
The present study aimed to verify the hypothesis
that pharmacological interventions on the AMPK/mTOR pathway to
alleviate PFKFB3-related aerobic glycolysis by metformin could
reduce collagen synthesis in an in vitro cellular model of
LPS-treated lung fibroblasts.
Materials and methods
Regents and antibodies
LPS from Escherichia coli O127:B8 was
purchased from Sigma-Aldrich; Merch KGaA. The mTOR activator
3-benzyl-5-((2-nitrophenoxy) methyl)-dihydrofuran-2(3H)-one (3BDO;
cat. no. S8317) and the AMPK inhibitor compound C (cat. no. S7306)
were obtained from Selleck Chemicals. The primary antibodies used
for western blotting were the following: Anti-AMPKα (1:1,000; cat.
no. ab32047; Abcam), anti-phosphorylated-AMPKαThr172
(p-AMPKα; 1:1,000; cat. no. ab133448; Abcam), anti-4E-BP1(1:2,000;
cat. no. ab32024; Abcam), anti-phosphorylated-4E-BP1 (p-4E-BP1;
1:1,000; cat. no. ab278686; Abcam), anti-PFKFB3 (1:2,000; cat. no.
ab218121; Abcam), anti-mTOR (1:1,000, cat. no. 2983; Cell Signaling
Technology, Inc.), anti-phosphorylated-mTOR (p-mTOR, 1:1,000; cat.
no. 2971; Cell Signaling Technology, Inc.), anti-collagen I
(1:1,000; cat. no. ab260043; Abcam), anti-α-smooth muscle actin
(α-SMA, 1:1,000; cat. no. 19245; Cell Signaling Technology, Inc.),
anti-collagen III (1:1,000; cat. no. ab184993; Abcam) and
anti-GAPDH (1:2,000; cat. no. 5174; Cell Signaling Technology,
Inc.). Horseradish peroxidase-conjugated goat anti-rabbit secondary
antibodies (1:2,000; cat. no. A0208; Beyotime Institute of
Biotechnology) were used in this study.
Experimental design and treatment
The human lung fibroblast MRC-5 cell line was
obtained from The Cell Bank of Type Culture Collection of The
Chinese Academy of Sciences. Cells were cultured in minimum
essential medium (MEM; Gibco; Thermo Fisher Scientific, Inc.)
supplemented with 10% FBS (Gibco; Thermo Fisher Scientific, Inc.),
100 U/ml penicillin and 100 µg/ml streptomycin (Thermo Fisher
Scientific, Inc.) and placed at 37˚C in a humidified incubator
containing 5% CO2. MRC-5 cells were seeded into 6-well
culture plates at the density of 3x105 cells/well. The
medium was changed for serum-free MEM for starvation overnight
after reaching 70% confluence, and for complete medium containing
10% FBS. Cells were then treated with a concentration gradient of
metformin (1, 5 and 10 mM; Selleck Chemicals) for 30 min and
incubated with LPS (1 µg/ml) for 48 h. To further investigate the
relationship between AMPK and mTOR, MRC-5 cells were treated with
3BDO (60 µM; Selleck Chemicals) or the AMPK-inhibitor compound C
(50 µM; Selleck Chemicals) for 30 min, followed by metformin (10
mM) for 4 h and LPS (1 µg/ml) for 48 h as reported by Wu et
al (21). The IC50
of lactate levels was 6.0 mM, the IC50 of LDH activation
was 4.7 mM and the IC50 of glucose consumption was 6.3
mM (Fig. S1).
Lactate dehydrogenase (LDH) activity
assay
The activity of LDH, which is a key enzyme in the
last step of glycolytic pathway (9). LDH activity in the MRC-5 cell lysate
was measured using the LDH Enzymatic Assay Kit (cat. no. A020-2-2;
Nanjing Jiancheng Bioengineering Institute) according to the
manufacturers' instructions.
Measurement of glucose consumption and
lactate in culture medium
Glucose and lactate concentration in culture medium
were measured with the Glucose and Lactate
Colorimetric/Fluorometric Assay Kits (cat. nos. K606 and K607,
respectively; both from BioVision, Inc.), respectively, according
to the manufacturers' instructions.
Mitochondrial complex I activity
assay
Mitochondrial complex I activity was measured with
the Complex I Activity Assay kit (cat. no. ab109721; Abcam)
according to the manufacturers' instructions. The extracted cell
proteins were loaded onto 96-well plates and incubated with
mitochondrial complex I antibody for 3 h at room temperature. Each
experiment was performed in five parallel wells and the mean
optical density (OD 450 nm) values were measured using the Nano
Drop spectrophotometer.
Western blotting
Cells were lysed using RIPA lysis buffer at 4˚C
(cat. no. P0013C; Beyotime Institute of Biotechnology) supplemented
with protease and phosphatase inhibitors (Roche Diagnostics) and
then centrifuged at 12,000 x g for 15 min at 4˚C. Protein
concentration was determined with the bicinchoninic acid assay
(Thermo Fisher Scientific, Inc.). Proteins (50 µg) were separated
by 6-12% SDS-PAGE, transferred onto PVDF membranes, membranes were
blocked with 5% bovine serum albumin (cat. no. ST025; Beyotime
Institute of Biotechnology) in TBS with 0.1% Tween-20 for 1 h at
room temperature and then incubated with primary antibodies at 4˚C
for 15 h. Membranes were then incubated with secondary antibodies
at room temperature for 1 h. Bands were detected using enhanced
chemiluminescence substrate (Beyotime Institute of Biotechnology).
Relative expression levels were normalized to endogenous control
using ImageLab™ software version 2.0.0.27 (Bio-Rad Laboratories,
Inc.).
Statistical analysis
Data were presented as the means ± standard
deviation and analyzed using GraphPad Prism 7 software (GraphPad
Software, Inc.). One-way ANOVA followed by Tukey's post hoc test
was used for comparison of three groups or more. P<0.05 was
considered to indicate a statistically significant difference.
Results
Metformin inhibits collagen synthesis
in LPS-treated lung fibroblasts by regulating PFKFB3-mediated
aerobic glycolysis
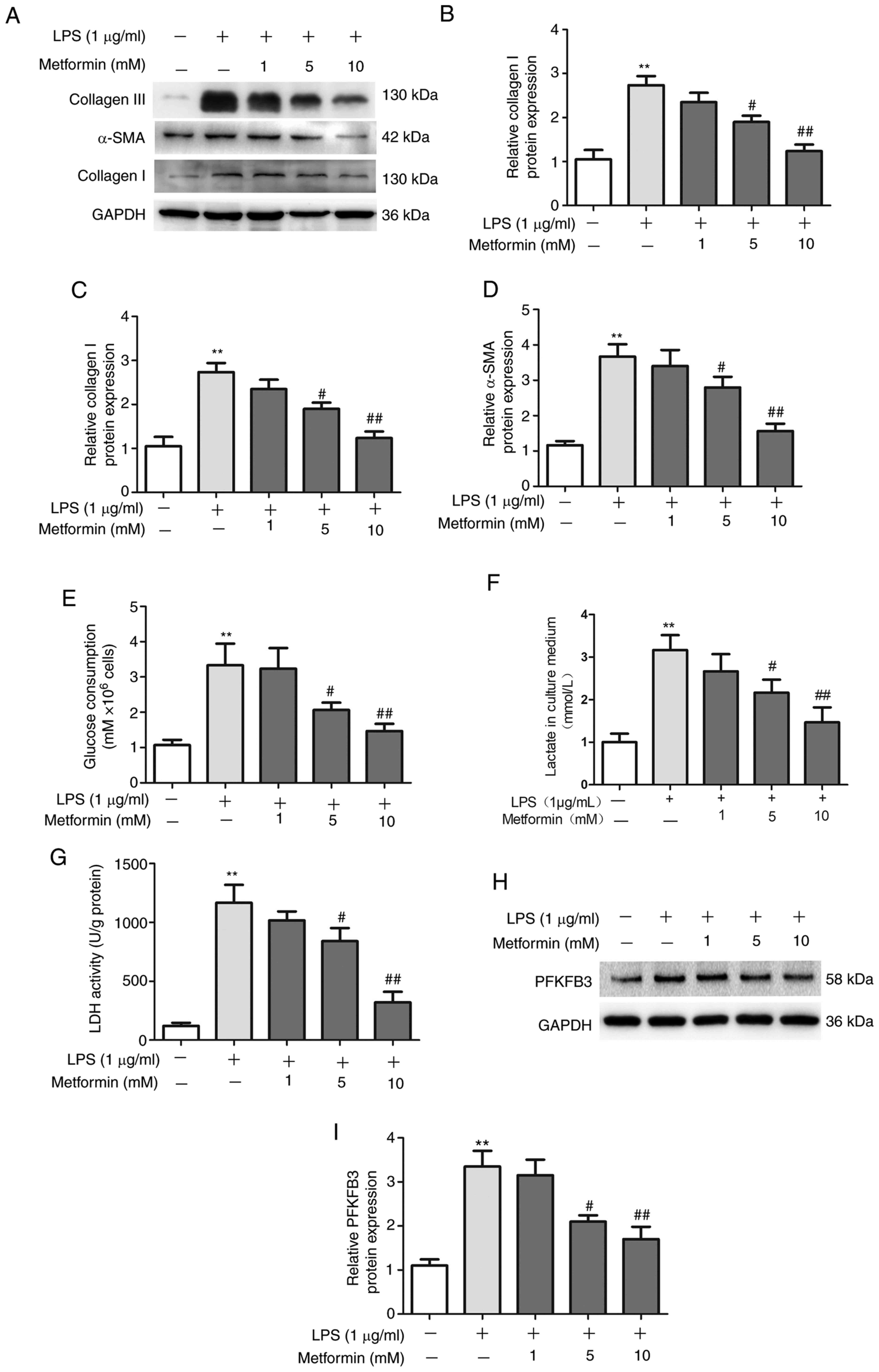
In the present study, metformin at the concentration
of 10 mM could successfully decrease the expression of α-SMA,
collagen III and collagen I in lung fibroblasts following LPS
treatment, which indicated the inhibition of collagen synthesis
(Fig. 1A-D). Furthermore, LPS
treatment significantly increased cell glucose consumption
(Fig. 1E), enhanced lactate
production (Fig. 1F) and promoted
LDH activation (Fig. 1G), all of
which could be reversed with metformin treatment at the
concentration of 10 mM. This finding suggested that metformin may
inhibit collagen synthesis by regulating aerobic glycolysis in
LPS-treated lung fibroblasts. Furthermore, LPS significantly
upregulated PFKFB3 expression in MRC-5 cells compared with negative
controls. However, PFKFB3 expression in lung fibroblasts was
decreased and returned to the baseline level following treatment
with 10 mM metformin (Fig. 1H and
I). These results indicated that
metformin may successfully inhibit collagen synthesis in
LPS-treated lung fibroblasts by regulating PFKFB3-mediated aerobic
glycolysis.
Metformin suppresses collagen
synthesis in LPS-treated lung fibroblasts by blocking the
activation of the mTOR/PFKFB3 signaling pathway
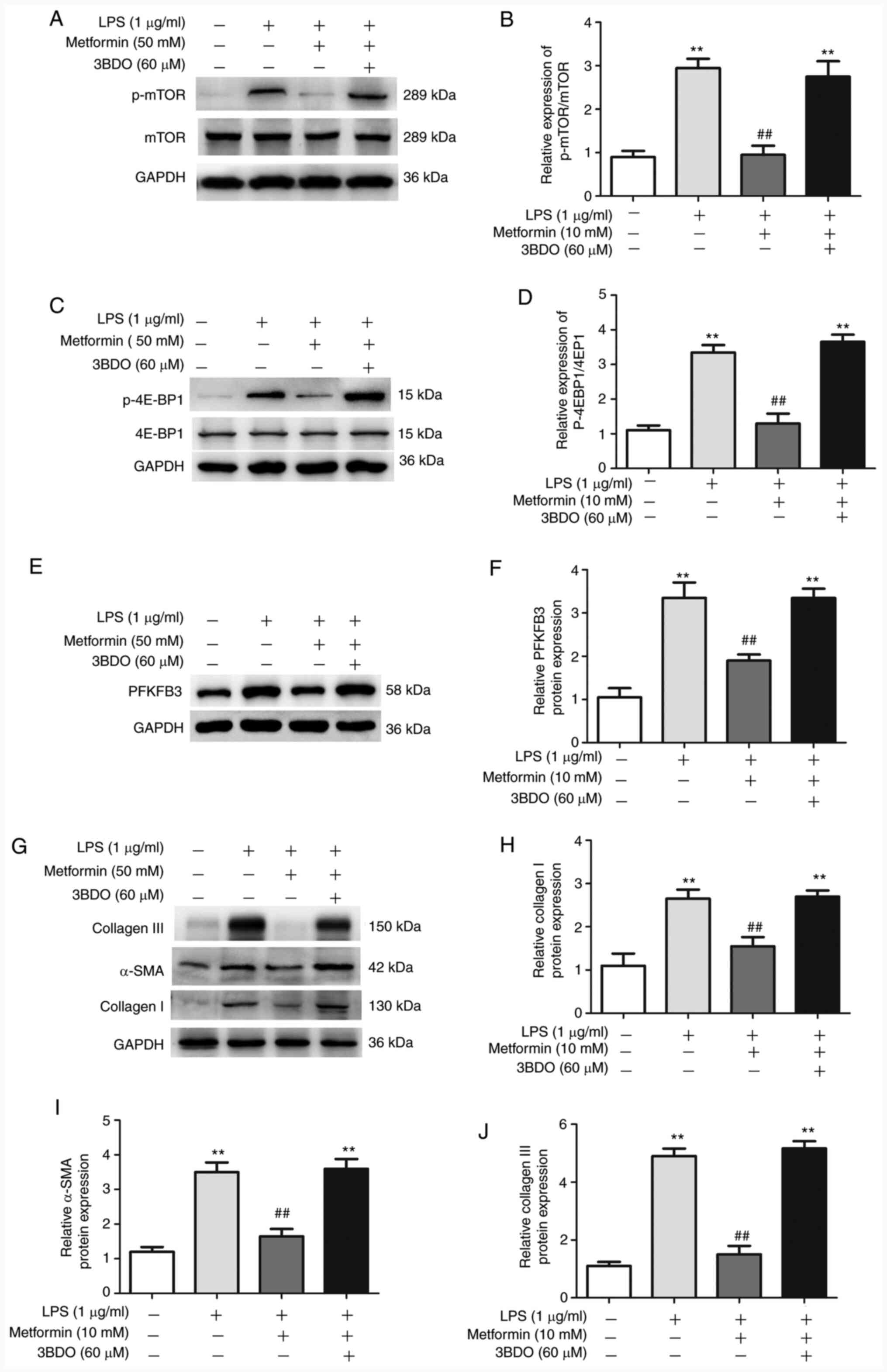
Activation of the mTOR signaling pathway promotes
aerobic glycolysis in the lung fibroblasts by upregulating PFKFB3
expression (9). In the present
study, expression of mTOR downstream pathway proteins, including
p-mTOR (mTOR activation) and p-4E-BP1 (4E-BP1 activation), was
significantly increased after LPS treatment; however, treatment
with metformin reversed these effects (Fig. 2A-D). The mTOR activator 3BDO was
subsequently used to better characterize the essential role of mTOR
in LPS-induced PFKFB3 overexpression. The results demonstrated that
3BDO could inhibit the function of metformin to decrease the
expression of p-mTOR, p-4E-BP1 and PFKFB3 (Fig. 2A-D), indicating that mTOR activation
may upregulate PFKFB3 expression in LPS-treated lung fibroblasts.
In addition, 3BDO could also reverse the effects of metformin to
decrease collagen synthesis (Fig.
2G-J). These results suggested that metformin may inhibit
collagen synthesis in LPS-treated lung fibroblasts by regulating
the mTOR/PFKFB3 pathway.
Metformin activates mitochondrial
complex I/p-AMPK expression in LPS-treated lung fibroblasts
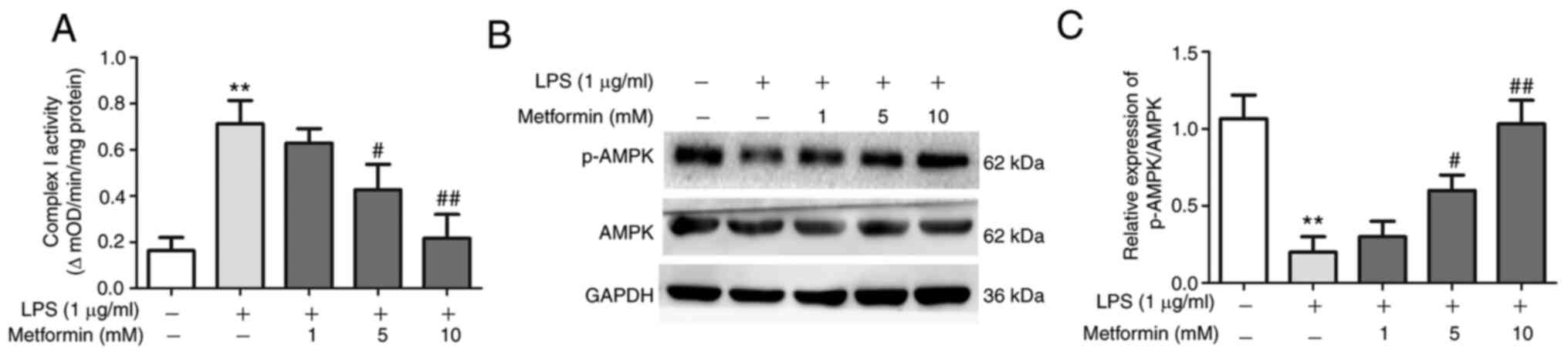
Metformin promotes AMPK phosphorylation by
decreasing the activity of mitochondrial complex I (22). In the present study, the activity of
mitochondrial complex I was increased in LPS-treated lung
fibroblasts, whereas it was decreased after treatment with
metformin for 48 h in a concentration-dependent manner (Fig. 3A). In addition, p-AMPK protein
expression was significantly decreased in LPS-treated lung
fibroblasts; however, its expression gradually increased after
treatment with increasing concentrations of metformin. Treatment
with 10 mM metformin could stimulate p-AMPK expression back to
normal baseline level (Fig. 3B and
C). Taken together, these findings
indicated that 10 mM metformin could successfully activate p-AMPK
expression in LPS-treated lung fibroblasts.
Metformin inhibits collagen synthesis
in lung fibroblasts by regulating AMPK/mTOR pathway
To better investigate the regulatory role of
metformin on aerobic glycolysis in LPS-treated lung fibroblasts,
the AMPK inhibitor compound C was administered with metformin to
LPS-treated lung fibroblasts. The results demonstrated that
pre-treatment with compound C could reverse the inhibitory effect
of metformin on the PFKFB3-mediated aerobic glycolysis in lung
fibroblasts, as indicated by the higher levels of glucose
consumption (Fig. 4A), increased
lactate production (Fig. 4B) and
enhanced LDH activation (Fig. 4C)
as well as the increase in PFKFB3 expression (Fig. 4D and E). In addition, compound C successfully
prevented metformin from upregulating p-AMPK (Fig. 4F and G) and downregulating p-mTOR (Fig. 4H and I) and p-4E-BP1 (Fig. 4J and K) in LPS-treated lung fibroblasts.
Furthermore, the decrease in collagen synthesis, as indicated by
reduced α-SMA, collagen I and collagen III expression due to
metformin treatment of LPS-induced lung fibroblasts, was abolished
after treatment with compound C (Fig.
4L-O). These results suggested that metformin may prevent
PFKFB3-related aerobic glycolysis from enhancing collagen synthesis
in lung fibroblasts by regulating AMPK/mTOR pathway.
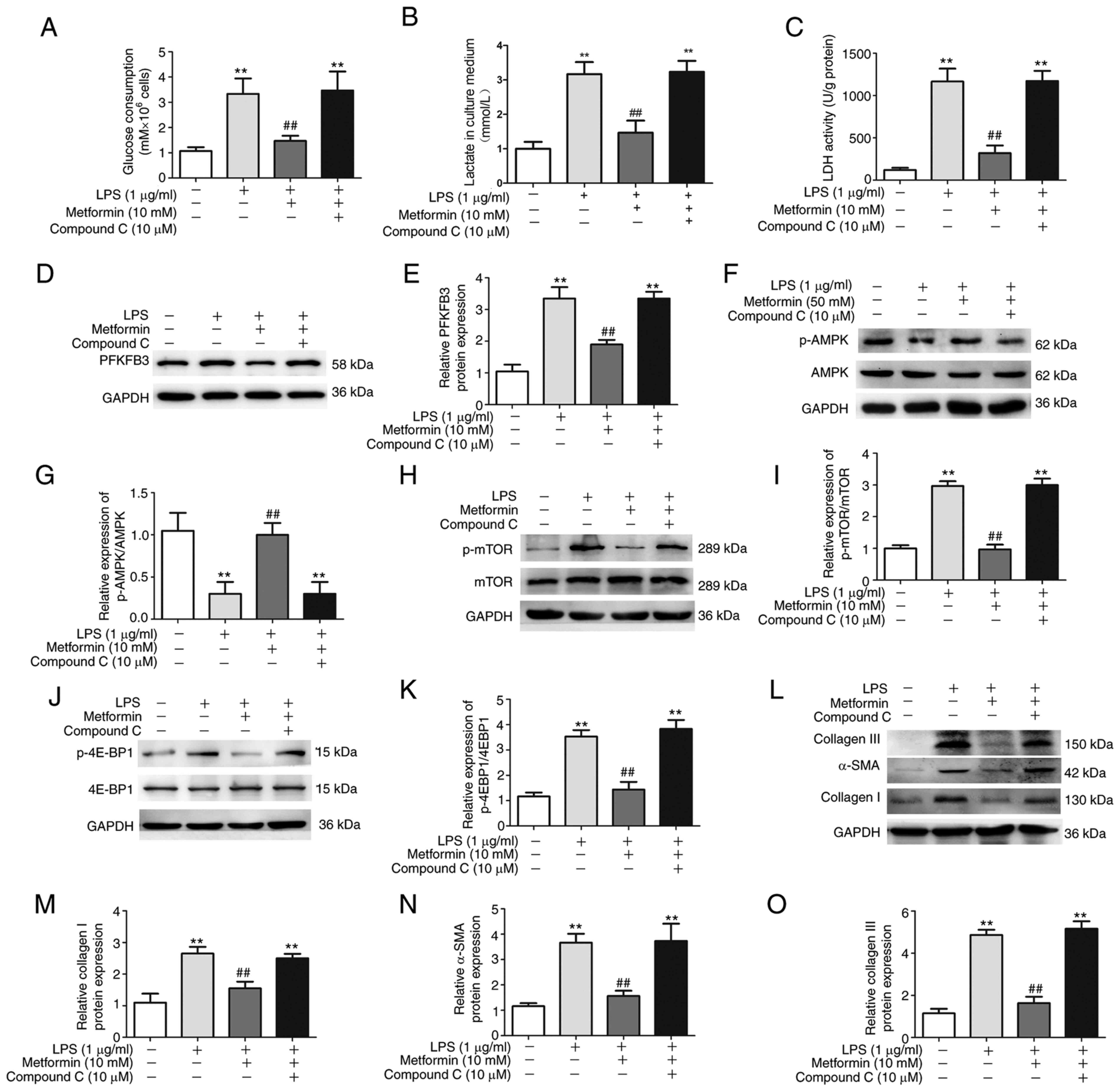 | Figure 4Metformin prevents collagen synthesis
in lung fibroblasts by regulating AMPK/mTOR pathway. Lung
fibroblasts were pre-treated with the AMPK inhibitor compound C at
a concentration of 50 µM for 30 min, followed by metformin
treatment (10 mM) for 4 h and LPS (1 µg/ml) for 48 h. (A) Changes
in glucose consumption, (B) lactate in cell culture medium and (C)
intracellular LDH activity. (D) Representative western blotting
images and (E) quantitative analysis showing the expression of
PFKFB3 protein. (F) Representative western blotting images and (G)
quantitative analysis showing the expression of p-AMPK/AMPK. (H)
Representative western blotting images and (I) quantitative
analysis showing the expression of p-mTOR/mTOR. (J) Representative
western blotting images and (K) quantitative analysis showing the
expression of p-4E-BP1/4E-BP1 in lung fibroblasts. (L)
Representative western blotting images and quantitative analysis
showing the expression of (M) collagen I, (N) α-SMA and (O)
collagen III protein in lung fibroblasts. Data were expressed as
the means ± standard error of the mean (n=3).
**P<0.01 vs. Control group. ##P<0.01
vs. LPS group. mTOR, mammalian target of rapamycin; LPS,
lipopolysaccharide; p, phosphorylated; LDH, lactate dehydrogenase;
PFKFB3, 6-phosphofructo-2-kinase/fructose-2, 6-biphosphatase 3;
α-SMA, α-smooth muscle actin. |
Discussion
Previous studies have reported that metformin can
alleviate bleomycin-induced idiopathic pulmonary fibrosis both
in vivo and in vitro (12,23).
However, whether metformin could have the same protective effect in
inhibiting LPS-associated pulmonary fibrosis are still unknown and
the underlying mechanism remains unclear. The results from the
present study demonstrated that 10 mM metformin could successfully
inhibit collagen synthesis in LPS-treated lung fibroblasts, which
was similar to a previous studies reporting that 10 mM metformin
can inhibit TGF-β-induced myofibroblast collagen synthesis
(24). Abnormality in glucose
metabolism, especially aerobic glycolysis, has been considered to
serve a crucial role in the aberrant activation of lung fibroblasts
and to contribute to the development and deterioration of pulmonary
fibrosis (25,26). PFKFB3 protein initiates the aerobic
glycolysis by improving glucose uptake, enhancing LDH activity and
increasing lactate production (27). In addition, the PFKFB3-mediated
glycolytic reprogramming has been reported to promote the
extracellular matrix production by lung fibroblasts (9,28). In
the present study, 10 mM metformin inhibited the PFKFB3-mediated
aerobic glycolysis, as evidenced by the decreased glucose uptake,
inhibited LDH activity and decreased lactate production in lung
fibroblasts following LPS treatment. Taken together, these results
provide direct evidence that metformin may prevent collagen
synthesis by lung fibroblasts via regulating PFKFB3-mediated
glycolysis, suggesting that a pharmacological intervention of
metformin may have some potential therapeutic significance for
LPS-induced pulmonary fibrosis.
Emerging evidence has demonstrated that the mTOR
signaling pathway is vital in both the aerobic glycolysis (29,30)
and collagen synthesis of lung fibroblasts (31,32).
Previous studies confirmed that mTOR activation promotes aerobic
glycolysis, mostly by upregulating PFKFB3 expression (33,34).
In addition, active 4E-BP1, as the mTOR downstream target, is
negatively associated with glycolysis (35). In the present study, 10 mM metformin
successfully inhibited mTOR and 4-EBP1 activation. However, the
mTOR activator 3BDO prevented metformin from decreasing PFKFB3
expression and inhibiting collagen synthesis in LPS-treated lung
fibroblasts. These findings indicated that metformin may reverse
PFKFB3-mediated collagen synthesis in LPS-treated lung fibroblasts
through regulating the mTOR pathway.
Metformin affects cell aerobic glycolysis metabolism
by mainly regulating the AMPK activation (36). Mitochondrial complex I as the direct
target of metformin can negatively regulate the phosphorylation of
AMPK (23), and AMPK activation is
frequently downregulated in pulmonary fibrosis (37). These results were also observed in
the present study where AMPK expression was decreased in
LPS-associated lung fibroblasts. In the present study, 10 mM
metformin successfully activated the AMPK signaling pathway in
LPS-treated lung fibroblasts by inhibiting the complex I activity
and promoting p-AMPK expression. Subsequently, AMPK, which is cell
energy regulator, may inhibit aerobic glycolysis by regulating the
mTOR pathway (38). Furthermore,
AMPK activation by metformin leads to the phosphorylation and
activation of the tuberous sclerosis complex 1/2, which in turn
inhibits mTOR phosphorylation and activation (39). In the present study, after blocking
AMPK expression with compound C with resultant activation of mTOR
pathway, metformin unexpectedly failed to inhibit collagen
synthesis and PFKFB3 expression in LPS-treated lung fibroblasts. In
addition, the PFKFB3-mediated aerobic glycolysis (indicated by
glucose uptake and lactate production) in LPS-treated lung
fibroblasts was also not inhibited with the co-administration of
compound C and metformin. Taken together, these findings suggested
that metformin efficacy to attenuate collagen synthesis in
LPS-treated lung fibroblasts may be mediated by aerobic glycolysis
inhibition and AMPK activation. This result was consistent with a
previous study reporting that metformin can alleviate
sepsis-induced acute lung injury via restoring AMPK-dependent
suppression of mTOR (21). The
results from the present study demonstrated that metformin could
prevent PFKFB3-related aerobic glycolysis from enhancing collagen
synthesis in lung fibroblasts by regulating AMPK/mTOR pathway.
In the present study, MRC-5 cells were treated with
serum-free MEM for overnight starvation prior to LPS treatment in
order to synchronize cultured cells in the G1 phase and improve
cell sensitivity to LPS treatment. A previous study reported that
autophagy is enhanced and becomes a driving force for oxidative
phosphorylation and ATP production when glycolysis is inhibited
(40). In addition, LPS promotes
lung fibroblast proliferation through autophagy inhibition via
activation of the PI3K-Akt-mTOR pathway (41), which in turn mediates lung
fibroblast aerobic glycolysis and collagen synthesis in LPS-induced
pulmonary fibrosis (9).
Subsequently, LPS promotes the aerobic glycolysis by suppressing
lung fibroblast autophagy that contributes to pulmonary
fibrosis.
The present study had some limitations. Firstly,
pulmonary fibrosis is not limited to fibroblast activation with
collagen fiber secretion and deposition (42). Previous studies have reported that
numerous types of cell, including alveolar epithelium and
macrophages, are also actively implicated in the etiopathogenesis
of pulmonary fibrosis (43). The
present study mainly focused on the regulation of lung fibroblasts
by metformin following LPS exposure and failed to investigate the
potential effect of metformin in other types of cell. Secondly, it
takes at least 30 days for LPS to induce typical pulmonary fibrosis
in mice (32). However, we observed
the effect of metformin on aerobic glycolysis in lung fibroblasts
after LPS exposure for only 48 h, which may not mirror the effect
of a single dose of metformin on pulmonary fibrosis for a longer
time in vivo. It remains unclear whether the effects of
metformin on pulmonary fibrosis is transient or long-term. Although
Wang et al (44) reported
that metformin inhibits collagen synthesis in TGF-β1-stimulated
HFL-1 cells for 72 h, how long the effect of metformin on
LPS-treated lung fibroblasts lasts for remains unknown. In
addition, only cell experiments were performed in the present
study, which may not reflect the mechanism in animal models. The
effect and duration of metformin on animal models of pulmonary
fibrosis should therefore be further investigated.
In conclusion, the present study demonstrated that
metformin may decrease collagen synthesis in LPS-treated lung
fibroblasts via inhibiting PFKFB3-related aerobic glycolysis and
regulating AMPK/mTOR pathway.
Supplementary Material
The dose-response curve of metformin
on the LPS-exposed pulmonary fibroblasts. Gradient dosage of
metformin (0.5, 1.0, 2.5, 5.0, 10.0, 15.0 and 20.0mM) were added
into the LPS-exposed pulmonary fibroblasts. The median inhibition
concentration (IC50) was calculated to estimate metformin on
regulating aerobic glycolysis in the LPS-exposed pulmonary
fibroblasts. The IC50 of lactate levels, LDH activation and glucose
consumption was 6.0mM (A), 4.7mM (B), and 6.3mM (C),
respectively.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Top-level
Clinical Discipline Project of Shanghai Pudong (grant no.
PWYgf2018-05).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CJT and JX wrote the first draft of manuscript. HYY
and XBW contributed to the conception and design of the research.
CJT, JX, HYY and XBW contributed to the experiments and analysis of
the data. CJT, JX, HYY and XBW confirm the authenticity of all the
raw data. All authors critically revised the manuscript and agreed
to be fully accountable for ensuring the integrity and accuracy of
the work, and read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Xie T, Xu Q, Wan H, Xing S, Shang C, Gao Y
and He Z: Lipopolysaccharide promotes lung fibroblast proliferation
through autophagy inhibition via activation of the PI3K-Akt-mTOR
pathway. Lab Invest. 99:625–633. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gu N, Xing S, Chen S, Zhou Y, Jiang T,
Jiao Y, Gao Y, Yu W, He Z and Wen D: Lipopolysaccharide induced the
proliferation of mouse lung fibroblasts by suppressing FoxO3a/p27
pathway. Cell Biol Int. 42:1311–1320. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhou WQ, Wang P, Shao QP and Wang J:
Lipopolysaccharide promotes pulmonary fibrosis in acute respiratory
distress syndrome (ARDS) via lincRNA-p21 induced inhibition of
Thy-1 expression. Mol Cell Biochem. 419:19–28. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Martin C, Papazian L, Payan MJ, Saux P and
Gouin F: Pulmonary fibrosis correlates with outcome in adult
respiratory distress syndrome. A study in mechanically ventilated
patients. Chest. 107:196–200. 1995.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Xie N, Tan Z, Banerjee S, Cui H, Ge J, Liu
RM, Bernard K, Thannickal VJ and Liu G: Glycolytic reprogramming in
myofibroblast differentiation and lung fibrosis. Am J Respir Crit
Care Med. 192:1462–1474. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhong WJ, Yang HH, Guan XX, Xiong JB, Sun
CC, Zhang CY, Luo XQ, Zhang YF, Zhang J, Duan JX, et al: Inhibition
of glycolysis alleviates lipopolysaccharide-induced acute lung
injury in a mouse model. J Cell Physiol. 234:4641–4654.
2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Schruf E, Schroeder V, Kuttruff CA, Weigle
S, Krell M, Benz M, Bretschneider T, Holweg A, Schuler M, Frick M,
et al: Human lung fibroblast-to-myofibroblast transformation is not
driven by an LDH5-dependent metabolic shift towards aerobic
glycolysis. Respir Res. 20(87)2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kang YP, Lee SB, Lee JM, Kim HM, Hong JY,
Lee WJ, Choi CW, Shin HK, Kim DJ, Koh ES, et al: Metabolic
profiling regarding pathogenesis of idiopathic pulmonary fibrosis.
J Proteome Res. 15:1717–1724. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hu X, Xu Q, Wan H, Hu Y, Xing S, Yang H,
Gao Y and He Z: PI3K-Akt-mTOR/PFKFB3 pathway mediated lung
fibroblast aerobic glycolysis and collagen synthesis in
lipopolysaccharide-induced pulmonary fibrosis. Lab Invest.
100:801–811. 2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Cao Y, Zhang X, Wang L, Yang Q, Ma Q, Xu
J, Wang J, Kovacs L, Ayon RJ, Liu Z, et al: PFKFB3-mediated
endothelial glycolysis promotes pulmonary hypertension. Proc Natl
Acad Sci USA. 116:13394–13403. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kheirollahi V, Wasnick RM, Biasin V,
Vazquez-Armendariz AI, Chu X, Moiseenko A, Weiss A, Wilhelm J,
Zhang JS, Kwapiszewska G, et al: Metformin induces lipogenic
differentiation in myofibroblasts to reverse lung fibrosis. Nat
Commun. 10(2987)2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gamad N, Malik S, Suchal K, Vasisht S,
Tomar A, Arava S, Arya DS and Bhatia J: Metformin alleviates
bleomycin-induced pulmonary fibrosis in rats: Pharmacological
effects and molecular mechanisms. Biomed Pharmacother.
97:1544–1553. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang J, Wang Y, Han J, Mei H, Yu D, Ding
Q, Zhang T, Wu G, Peng G and Lin Z: Metformin attenuates
radiation-induced pulmonary fibrosis in a murine model. Radiat Res.
188:105–113. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Liu Y, Bai F, Liu N, Zhang B, Qin F, Tu T,
Li B, Li J, Ma Y, Ouyang F and Liu Q: Metformin improves lipid
metabolism and reverses the Warburg effect in a canine model of
chronic atrial fibrillation. BMC Cardiovasc Disord.
20(50)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wang Y, An H, Liu T, Qin C, Sesaki H, Guo
S, Radovick S, Hussain M, Maheshwari A, Wondisford FE, et al:
Metformin improves mitochondrial respiratory activity through
activation of AMPK. Cell Rep. 29:1511–1523.e1515. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Hawley SA, Ross FA, Chevtzoff C, Green KA,
Evans A, Fogarty S, Towler MC, Brown LJ, Ogunbayo OA, Evans AM and
Hardie DG: Use of cells expressing gamma subunit variants to
identify diverse mechanisms of AMPK activation. Cell Metab.
11:554–565. 2010.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Mossmann D, Park S and Hall MN: mTOR
signalling and cellular metabolism are mutual determinants in
cancer. Nat Rev Cancer. 18:744–757. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Mousavizadeh R, Hojabrpour P, Eltit F,
McDonald PC, Dedhar S, McCormack RG, Duronio V, Jafarnejad SM and
Scott A: β1 integrin, ILK and mTOR regulate collagen synthesis in
mechanically loaded tendon cells. Sci Rep. 10(12644)2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ma XM and Blenis J: Molecular mechanisms
of mTOR-mediated translational control. Nat Rev Mol Cell Biol.
10:307–318. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
20
|
Lv C, Wu C, Zhou YH, Shao Y, Wang G and
Wang QY: Alpha lipoic acid modulated high glucose-induced rat
mesangial cell dysfunction via mTOR/p70S6K/4E-BP1 pathway. Int J
Endocrinol. 2014(658589)2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Wu K, Tian R, Huang J, Yang Y, Dai J,
Jiang R and Zhang L: Metformin alleviated endotoxemia-induced acute
lung injury via restoring AMPK-dependent suppression of mTOR. Chem
Biol Interact. 291:1–6. 2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Owen MR, Doran E and Halestrap AP:
Evidence that metformin exerts its anti-diabetic effects through
inhibition of complex 1 of the mitochondrial respiratory chain.
Biochem J. 348:607–614. 2000.PubMed/NCBI
|
|
23
|
Xiao H, Huang X, Wang S, Liu Z, Dong R,
Song D and Dai H: Metformin ameliorates bleomycin-induced pulmonary
fibrosis in mice by suppressing IGF-1. Am J Transl Res. 12:940–949.
2020.PubMed/NCBI
|
|
24
|
Sato N, Takasaka N, Yoshida M, Tsubouchi
K, Minagawa S, Araya J, Saito N, Fujita Y, Kurita Y, Kobayashi K,
et al: Metformin attenuates lung fibrosis development via NOX4
suppression. Respir Res. 17(107)2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Xu JN, Li JY, Yu ZH, Rao HW, Wang S and
Lan HB: HMGB1 promotes HLF-1 proliferation and ECM production
through activating HIF1-α-regulated aerobic glycolysis. Pulm
Pharmacol Ther. 45:136–141. 2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Cho SJ, Moon JS, Lee CM, Choi AM and
Stout-Delgado HW: Glucose transporter 1-dependent glycolysis is
increased during aging-related lung fibrosis, and phloretin
inhibits lung fibrosis. Am J Respir Cell Mol Biol. 56:521–531.
2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Gong Y, Lan H, Yu Z, Wang M, Wang S, Chen
Y, Rao H, Li J, Sheng Z and Shao J: Blockage of glycolysis by
targeting PFKFB3 alleviates sepsis-related acute lung injury via
suppressing inflammation and apoptosis of alveolar epithelial
cells. Biochem Biophys Res Commun. 491:522–529. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kottmann RM, Trawick E, Judge JL, Wahl LA,
Epa AP, Owens KM, Thatcher TH, Phipps RP and Sime PJ: Pharmacologic
inhibition of lactate production prevents myofibroblast
differentiation. Am J Physiol Lung Cell Mol Physiol.
309:L1305–L1312. 2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Cheng SC, Quintin J, Cramer RA, Shepardson
KM, Saeed S, Kumar V, Giamarellos-Bourboulis EJ, Martens JH, Rao
NA, Aghajanirefah A, et al: mTOR- and HIF-1α-mediated aerobic
glycolysis as metabolic basis for trained immunity. Science.
345(1250684)2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wu F, Gao P, Wu W, Wang Z, Yang J, Di J,
Jiang B and Su X: STK25-induced inhibition of aerobic glycolysis
via GOLPH3-mTOR pathway suppresses cell proliferation in colorectal
cancer. J Exp Clin Cancer Res. 37(144)2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Liu MW, Su MX, Tang DY, Hao L, Xun XH and
Huang YQ: Ligustrazin increases lung cell autophagy and ameliorates
paraquat-induced pulmonary fibrosis by inhibiting PI3K/Akt/mTOR and
hedgehog signalling via increasing miR-193a expression. BMC Pulm
Med. 19(35)2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Wan H, Xie T, Xu Q, Hu X, Xing S, Yang H,
Gao Y and He Z: Thy-1 depletion and integrin β3
upregulation-mediated PI3K-Akt-mTOR pathway activation inhibits
lung fibroblast autophagy in lipopolysaccharide-induced pulmonary
fibrosis. Lab Invest. 99:1636–1649. 2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Feng Y and Wu L: mTOR up-regulation of
PFKFB3 is essential for acute myeloid leukemia cell survival.
Biochem Biophys Res Commun. 483:897–903. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Li Z, Liu J, Que L and Tang X: The
immunoregulatory protein B7-H3 promotes aerobic glycolysis in oral
squamous carcinoma via PI3K/Akt/mTOR pathway. J Cancer.
10:5770–5784. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Suginohara T, Wakabayashi K, Ato S and
Ogasawara R: Effect of 2-deoxyglucose-mediated inhibition of
glycolysis on the regulation of mTOR signaling and protein
synthesis before and after high-intensity muscle contraction.
Metabolism. 114(154419)2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Duca FA, Cote CD, Rasmussen BA,
Zadeh-Tahmasebi M, Rutter GA, Filippi BM and Lam TK: Metformin
activates a duodenal Ampk-dependent pathway to lower hepatic
glucose production in rats. Nat Med. 21:506–511. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
37
|
Cho SJ, Moon JS, Lee CM, Choi AM and
Stout-Delgado HW: Glucose transporter 1-dependent glycolysis is
increased during aging-related lung fibrosis, and phloretin
inhibits lung fibrosis. Am J Respir Cell Mol Biol. 56:521–531.
2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Zhang L, Yang H, Zhang W, Liang Z, Huang
Q, Xu G, Zhen X and Zheng LT: Clk1-regulated aerobic glycolysis is
involved in glioma chemoresistance. J Neurochem. 142:574–588.
2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kim J, Kundu M, Viollet B and Guan KL:
AMPK and mTOR regulate autophagy through direct phosphorylation of
Ulk1. Nat Cell Biol. 13:132–141. 2011.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Kawaguchi M, Aoki S, Hirao T, Morita M and
Ito K: Autophagy is an important metabolic pathway to determine
leukemia cell survival following suppression of the glycolytic
pathway. Biochem Biophys Res Commun. 474:188–192. 2016.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Xie T, Xu Q, Wan H, Xing S, Shang C, Gao Y
and He Z: Lipopolysaccharide promotes lung fibroblast proliferation
through autophagy inhibition via activation of the PI3K-Akt-mTOR
pathway. Lab Invest. 99:625–633. 2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Thannickal VJ, Toews GB, White ES, Lynch
JP III and Martinez FJ: Mechanisms of pulmonary fibrosis. Annu Rev
Med. 55:395–417. 2004.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Hou J, Ji J, Chen X, Cao H, Tan Y, Cui Y,
Xiang Z and Han X: Alveolar epithelial cell-derived Sonic hedgehog
promotes pulmonary fibrosis through OPN-dependent alternative
macrophage activation. FEBS J: Dec 13, 2020 (Epub ahead of print).
doi: https://doi.org/10.1111/febs.15669.
|
|
44
|
Wang Y, Lin C, Han R, Lu C, Li L, Hu C,
Feng M, Chen H and He Y: Metformin attenuates TGF-β1-induced
pulmonary fibrosis through inhibition of transglutaminase 2 and
subsequent TGF-β pathways. 3 Biotech. 10(287)2020.PubMed/NCBI View Article : Google Scholar
|