Introduction
Osteoporosis (OP) remains the most common
progressive skeletal disease. This disease limits the activity of
patients (1) and decreases the
quality of life in elders and postmenopausal women (2). The modeling and remodeling of bone is
a dynamic metabolic process primarily mediated by the osteoblasts,
which form new bone by secretion of bone matrix and acceleration of
calcium (Ca2+) deposition, and the osteoclasts, which
resorb old bone by resolving mineralized bone matrix (3,4). In
case of imbalance between osteoblastic bone formation and
osteoclastic bone resorption (5),
which can lead to upregulated bone resorption, downregulated bone
formation or both (6,7), OP can occur.
MicroRNAs (miRNAs) are a subclass of non-coding RNAs
of 19-25 nucleotides in length, which regulate the expression of
target genes at post-transcriptional level (8). Previous studies have reported the
importance of miRNAs in the regulation of skeletal development,
bone formation and homeostasis (9,10). For
example, miR-125b, miR-29a and miR-378 have been demonstrated to be
involved in osteoblastic differentiation (11-13).
Furthermore, miR-21, miR-155 and miR-223 have been found to be
implicated in osteoclastic differentiation (14,15).
In 2015, miR-22-3p expression was found to be decreased in the bone
of patients who suffered a bone fracture (16). In 2016, miR-22-3p was discovered to
be downregulated during the progression of osteoclastic
differentiation (17). In 2018,
miR-22-3p expression was reported to be decreased in the bone of
patients with OP (18). In 2020,
extracellular vesicle-encapsulated miR-22-3p from bone marrow
mesenchymal stem cells were found to induce osteogenic
differentiation via Fat mass- and obesity-associated gene
inhibition (19). However, the
underlying mechanisms of miR-22-3p during the process of osteoclast
differentiation have not yet been reported, which was explored in
the present study.
Materials and methods
Clinical samples
A total of 30 healthy volunteers (mean age,
59.79±6.53 years; age range, 47-68 years) and 30 postmenopausal
women with OP (mean age, 60.23±7.15 years; age range, 49-69 years)
were included in the present study. Blood samples (5 ml) were
collected from each participant for the determination of miR-22-3p
and p38α mitogen-activated protein kinase (MAPK)14 expression. This
study was approved by the Ethics Committee of Chongqing Public
Health Medical Center (approval no. CMC20180106). Each participant
provided a signed informed consent prior to the beginning of the
study.
Bone mineral density (BMD)
measurement
The BMD of each participant was determined at the
lumbar spine (L1-L4) and the left femoral neck through dual-energy
X-ray absorptiometry (DXA) using a Hologic 4500 bone densitometer
(Hologic), according to the World Health Organization criteria
(20). Each scan was handled by a
single technician and repeated thrice with repositioning between
each scan. The precision of the machine, which is presented as
percentage coefficient of variation (CV%), varied between
subregions. CV% for BMD of the lumbar spine (L1-L4) was <1%,
whereas CV% for BMD of the left femoral was <2%.
Isolation and incubation of
CD14+peripheral blood mononuclear cells (PBMCs)
Osteoclasts are primary bone-resorbing cells which
can form from precursor fusion, and CD14+PBMCs are early
progenitors for osteoclasts (21).
Furthermore, osteoclast formation can occur in
CD14+PBMCs with the stimulation of macrophage colony
stimulating factor (M-CSF) and receptor activator of nuclear
factor-κB ligand (RANKL) (22).
Subsequently, the present study used CD14+PBMCs for the
study of OP in vitro. PBMCs were extracted as previously
described (22).
CD14+PBMCs were purified using CD14 antibody-coated
magnetic cell sorting MicroBeads (Miltenyi Biotec GmbH). When
CD14+PBMCs reached 90% purity by flow cytometry, they
were seeded into 48-well plates at the density of
2.5x105 cells/well. CD14+PBMCs were incubated
in α-MEM (Invitrogen; Thermo Fisher Scientific, Inc.) containing
10% FBS (Gibco; Thermo Fisher Scientific, Inc.), penicillin (50
IU/ml) and streptomycin (50 mg/ml) (Gibco; Thermo Fisher
Scientific, Inc.) and placed in an incubator with 5% CO2 at 37˚C
for 2 h.
Osteoclastic differentiation
For the establishment of osteoclastic
differentiation in vitro, CD14+PBMC initial
complete culture medium was changed for α-MEM containing M-CSF (25
ng/ml; R&D Systems, Inc.) and RANKL (25 ng/ml; R&D Systems,
Inc.). The complete culture medium containing differentiation
factors, 10% FBS, penicillin (50 IU/ml) and streptomycin (50 mg/ml)
was refreshed every three days and floating cells were removed.
After incubation for six days, CD14+PBMCs were collected
for subsequent experiments.
Cell transfection
For the overexpression of miR-22-3p,
CD14+PBMCs (2x104 cells/ml; 200 µl) were
seeded into 48-well plates and transfected with miR-NC mimic
(sense, 5'-UUCUCCGAACGUGUCACGU-3' and antisense,
5'-ACGUGACACGUUCGGAGAA-3') or miR-22-3p mimic (sense,
5'-AAAAGCUGCCAGUUGAAGAACUGU-3' and antisense,
5'-ACAGUUCUUCAACUGGCAGCUUUU-3') (50 nmol/l; Guangzhou Ribobio Co.,
Ltd.) using Lipofectamine® 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.). After transfection for 48 h,
CD14+PBMCs were subsequently treated with M-CSF and
RANKL as aforementioned and collected for subsequent
experiments.
To overexpress MAPK14, CD14+PBMCs
(2x104 cells/ml; 200 µl) were seeded into 48-well plates
and transfected with pcDNA3.1 or pcDNA3.1-MAPK14 (2 µg; Sangon
Biotech Co., Ltd.) using Lipofectamine 2000. After transfection for
48 h, CD14+PBMCs were subsequently treated with M-CSF
and RANKL as aforementioned and collected for subsequent
experiments. At 48 h after the transfection, cells were used for
subsequent experiments.
Dual luciferase reporter assay
The binding site between miR-22-3p and MAPK14 was
predicted by TargetScan 7.1 (http://www.targetscan.org/vert_71/). To verify the
interaction between miR-22-3p and MAPK14, CD14+PBMCs
(1x105 cells/ml) were seeded into 24-well plates and
co-transfected with wild type (WT) or mutant (MUT) MAPK14 3'-UTR
(400 ng) which was cloned into psiCHECK2 (Promega Corporation) and
miR-NC mimic or miR-22-3p mimic (20 nmol/l) using Lipofectamine
2000. After incubation for 48 h, CD14+PBMCs were
collected for the determination of luciferase activity with the
dual-luciferase reporter assay (Promega Corporation). The relative
luciferase activity was normalized to Renilla luciferase activity
(Promega corporation).
Cell proliferation assay
CD14+PBMC proliferation was determined
using Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies,
Inc.). CD14+PBMCs were seeded into 96-well
(1x103/well) and incubated for 0, 24, 48 or 72 h. CCK-8
solution (10 µl) was added into each well and incubated for 1 h at
37˚C. Absorbance at 450 nm was determined on a microplate
reader.
Cell apoptosis assay
CD14+PBMC cell apoptosis was determined
using the Annexin V-FITC/propidium iodide (PI) Apoptosis Detection
kit (Sigma-Aldrich; Merck KGaA) on a flow cytometer (BD
Biosciences). Briefly, CD14+PBMCs were collected by
after centrifugation at 1,000 x g for 5 min at 4˚C and resuspended
in 500 µl binding buffer. Subsequently, CD14+PBMCs were
stained with Annexin V-FITC (100 µl) for 15 min and PI (10 µl) for
5 min in the dark. The cell apoptotic rate was quantified on a flow
cytometry and analyzed by CellQuest version 3.3 (BD
Biosciences).
RNA isolation from serum and
cells
Serum samples were obtained from whole blood by
centrifugation at 2,000 x g for 15 min at room temperature after
being incubated at room temperature for 30 min. For detection of
miRNA, total RNA was isolated from serum (200 µl) of participants
or CD14+PBMCs using miRNeasy Serum/Plasma kit (Qiagen
GmbH). For the detection of mRNA, total RNA was isolated from serum
of participants or CD14+PBMCs using TRIzol®
(Invitrogen; Thermo Fisher Scientific, Inc.).
Reverse transcription quantitative
(RT-q)PCR
Complementary DNA (cDNA) was obtained from miRNA and
mRNA using TaqMan MicroRNA Reverse Transcription kit (Thermo Fisher
Scientific, Inc.) and First Strand cDNA Synthesis Kit (Takara Bio,
Inc.), respectively. RT-qPCR reactions were performed as follows:
95˚C for 30 sec, followed by 40 cycles at 95˚C for 5 sec and at
60˚C for 20 sec using SYBR Green qPCR assay kit (Takara Bio, Inc.)
on an ABI 7500 Real-Time PCR System (Applied Biosystems; Thermo
Fisher Scientific, Inc.). The relative expression levels of
miR-22-3p and of MAPK14, tartrate resistant acid phosphatase
(TRAP), nuclear factor of activated T-cells (NFATC1) and cathepsin
K (CTSK) were normalized to endogenous controls U6 or GAPDH,
respectively, and were expressed as 2-∆∆Cq (23). The following primer sequences were
used: GAPDH forward, 5'-GCACCGTCAAGGCTGAG AAC-3' and reverse,
5'-TGGTGAAGACGCCAGTGGA-3'; U6 forward, 5'-GTGCTCGCTTCGGCAGCACAT-3'
and reverse, 5'-AATATGGAACGCTTCACGAAT-3'; CTSK forward,
5'-TCCGCAATCCTTACCGAATA-3' and reverse,
5'-AACTTGAACACCCACATCCTG-3'; NFATC1 forward,
5'-TACCAGCGTTTCACCTACCT-3' and reverse, 5'-CCCTTTTCCTTTCCTTTTCA-3';
TRAP forward, 5'-AGACATCAATGACAAGAGGT-3' and reverse, 5'-AAG
TGCAGGCGGTAGAAAGG-3'; miR-22-3p forward,
5'-AAGCTGCCAGTTGAAGAACTGT-3' and reverse,
5'-ACAGTTCTTCAACTGGCAGCTT-3'; MAPK14 forward,
5'-GAAAAGGGTCTTCTTGGCAGCTT-3' and reverse,
5'-AAGCTGCCAAGAAGACCCTTTTC-3'.
Western blotting
CD14+PBMCs were lysed on ice using RIPA
(Sigma-Aldrich; Merck KGaA) containing PMSF and protease inhibitors
(Roche Diagnostics GmbH). Protein concentration was determined by a
BCA kit (Thermo Fisher Scientific, Inc.). Proteins (20 µg per lane)
were separated by 10% SDS-PAGE and transferred onto PVDF membranes
(EMD Millipore). Membranes were blocked with 5% bovine serum
albumin (Beyotime Institute of Biotechnology) at room temperature
for 1 h and were incubated with primary antibodies against GAPDH
(1:1,000; cat. no. 5174; Cell Signaling Technology, Inc.), MAPK14
(1:1,000; cat. no. 8690; Cell Signaling Technology, Inc.), TRAP
(1:1,000; cat. no. ab65854; Abcam), NFATC1 (1:1,000; cat. no.
ab25916; Abcam), CTSK (1:1,000; cat. no. ab37259; Abcam), p-p65
(1:1,000; cat. no. 3033; Cell Signaling Technology, Inc.) and p65
(1:1,000; cat. no. 8242; Cell Signaling Technology, Inc.) at 4˚C
overnight. Membranes were then incubated with anti-rabbit (1:2,000;
cat. no. 7074) or anti-mouse HRP-conjugated secondary antibody
(1:2,000; cat. no. 7076; both from Cell Signaling Technology, Inc.)
at room temperature for 1 h. Enhanced chemiluminescence reagent
(Bio-Rad Laboratories, Inc.) was used to detect the signal on the
membrane. Densitometry analysis was performed using Image J
software (v1.50; National Institutes of Health).
Statistical analyses
Statistical analyses were performed with the
GraphPad Prism 6.0 (GraphPad Software, Inc.). Data were expressed
as the means ± standard deviation. Unpaired student's t-test was
used to analyze differences between two groups from
CD14+PBMCs and participants. One-way ANOVA followed by
Tukey's post hoc test was used to analyze differences among
multiple groups. Pearson correlation analysis was used to analyze
the relationship between miR-22-3p and MAPK14 expression. P<0.05
was considered to indicate a statistically significant
difference.
Results
Clinicopathological characteristics of
participants
The clinicopathological characteristics of
participants are presented in Table
I. There was no significant difference between the mean age of
postmenopausal women with OP and healthy volunteers (60.23±7.15
years vs. 59.79±6.53 years, respectively). The body mass indexes in
the two groups were also not different (24.23±3.09 vs. 23.59±4.14
in postmenopausal women with OP and healthy volunteers,
respectively). The mean femoral neck BMD and lumbar spine BMD
(0.62±0.13 and 0.61±0.09, respectively) of patients with
postmenopausal OP were significantly lower compared with those of
healthy volunteers (0.89±0.12 and 0.85±0.16, respectively). The
comparison of BMD was not adjusted for age and BMI according to a
previous study (24).
 | Table IClinicopathological characteristics
of participants. |
Table I
Clinicopathological characteristics
of participants.
| Variable | Healthy
volunteers | Patients with
OP | P-value |
|---|
| Age, years | 59.79±6.53 | 60.23±7.15 | >0.05 |
| BMI,
kg/m2 | 23.59±4.14 | 24.23±3.09 | >0.05 |
| Femoral neck BMD,
g/cm2 | 0.89±0.12 | 0.62±0.13 | <0.001 |
| Lumbar spine BMD,
g/cm2 | 0.85±0.16 | 0.61±0.09 | <0.001 |
miR-22-3p expression level in
participants and CD14+PBMCs
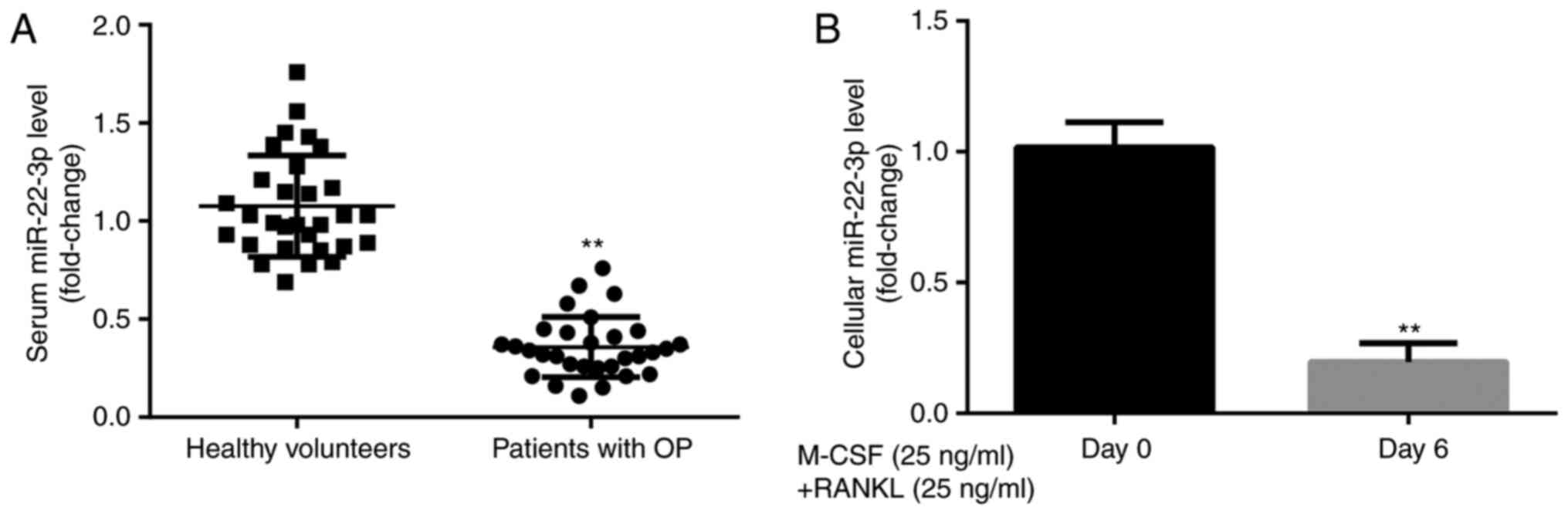
To explore the effects of miR-22-3p during the
development of OP, miR-22-3p expression level was evaluated in
healthy volunteers and patients with postmenopausal OP. The results
from RT-qPCR demonstrated that miR-22-3p expression was
significantly lower in patients with postmenopausal OP compared
with healthy volunteers (Fig. 1A).
Furthermore, miR-22-3p expression level was significantly lower in
CD14+PBMCs treated with M-CSF and RANKL for 6 days
compared with those untreated (Fig.
1B). These results suggested that miR-22-3p may be involved in
the development of OP.
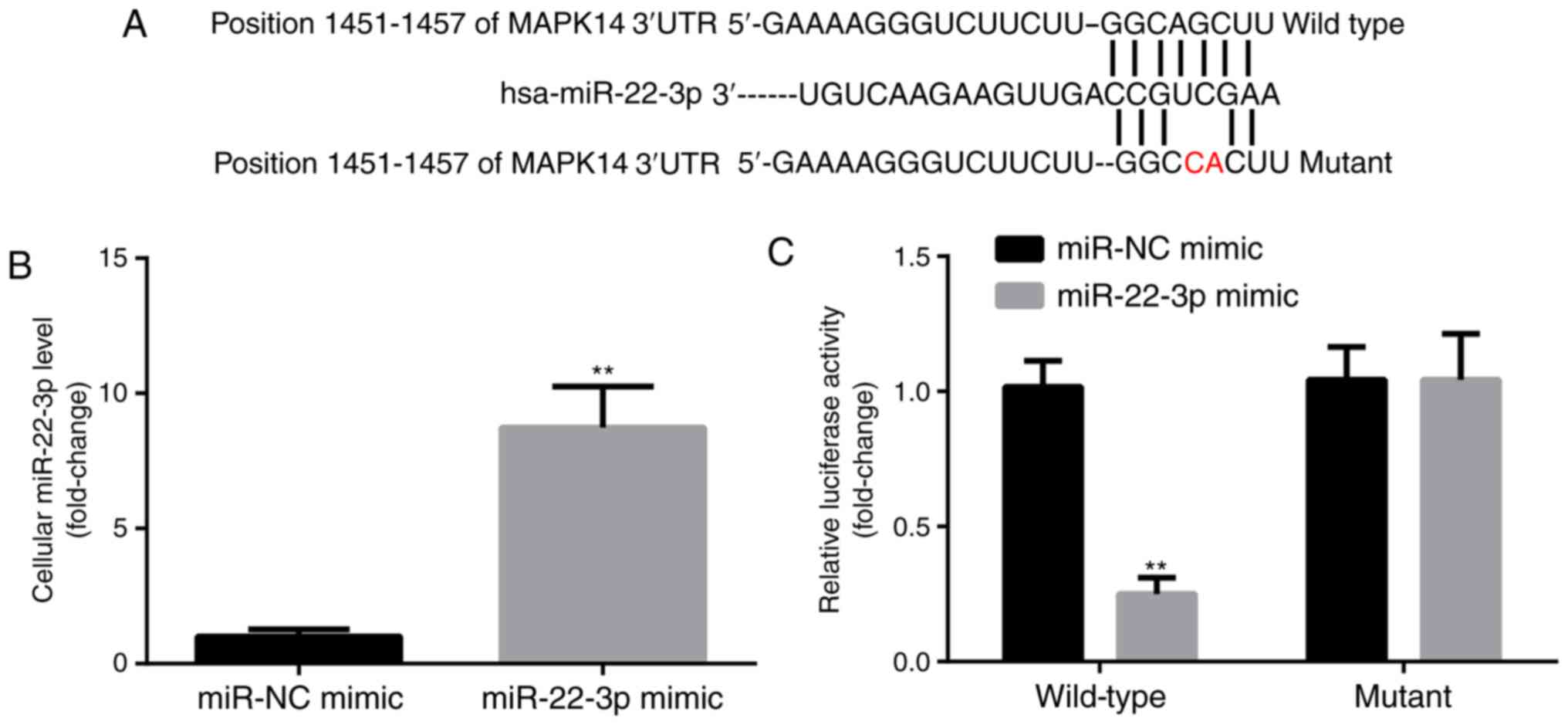
miR-22-3p targets the expression of
MAPK14
MAPK14 was predicted to be a target gene for
miR-22-3p by TargetScan. The predicted pairing between MAPK14 3'UTR
and miR-22-3p is presented in Fig.
2A. In addition, compared with miR-NC mimic group, miR-22-3p
mimic significantly increased miR-22-3p expression level in
CD14+PBMCs (Fig. 2B).
Subsequently, the interaction between miR-22-3p and MAPK14 in
CD14+PBMCs was detected by dual luciferase reporter
assay. Compared with miR-NC mimic group, miR-22-3p mimic
significantly decreased the luciferase activity of the WT-MAPK14
but not MUT-MAPK14 (Fig. 2C). These
findings confirmed the potential interaction between miR-22-3p and
MAPK14.
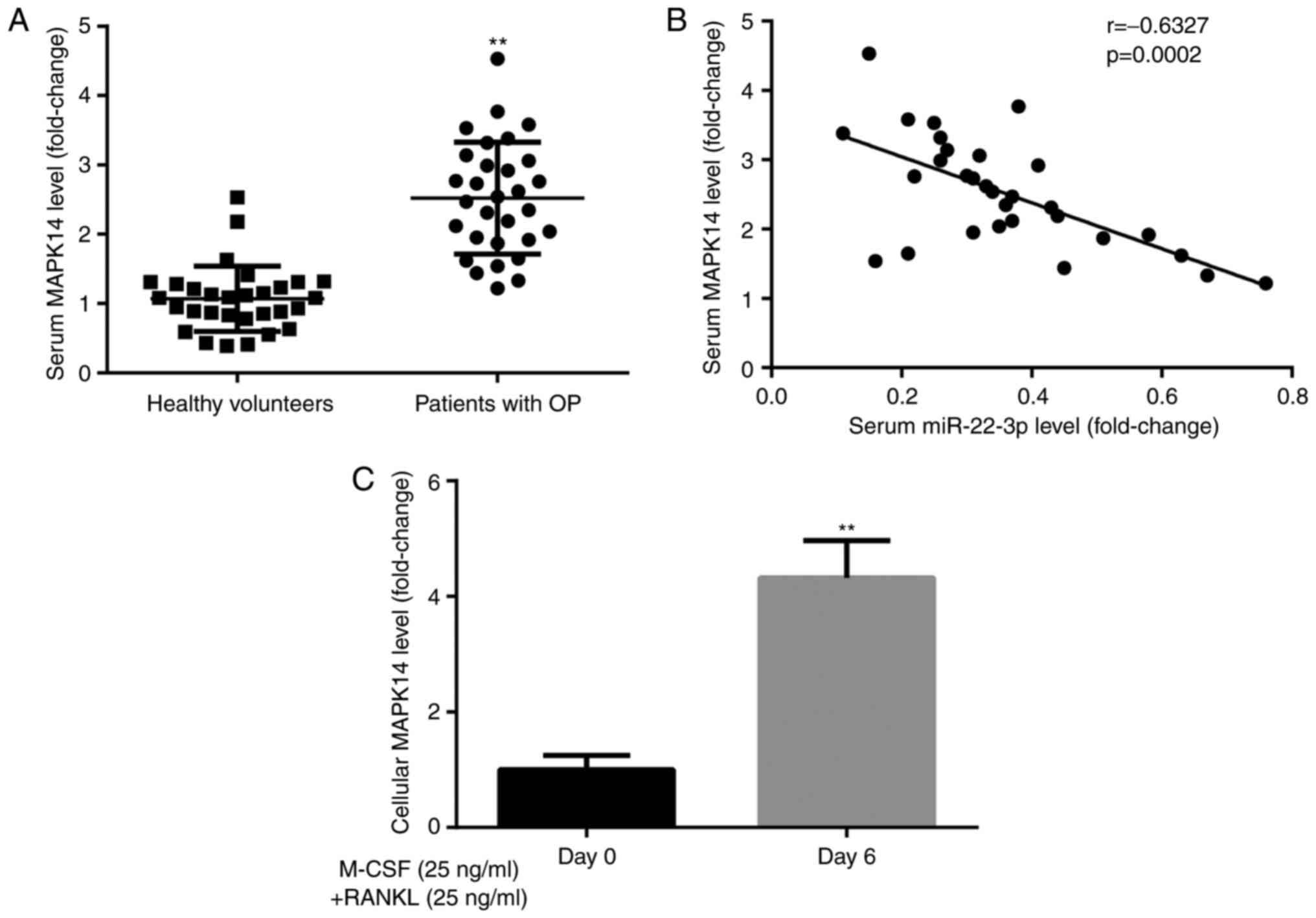
MAPK14 expression in participants and
CD14+PBMCs
To explore the effects of MAPK14 during the
development of OP, MAPK14 expression was evaluated in healthy
volunteers and patients with postmenopausal OP. The results from
RT-qPCR demonstrated that MAPK14 expression was significantly
higher in patients with postmenopausal OP compared with healthy
volunteers (Fig. 3A). Furthermore,
Pearson correlation analysis demonstrated that miR-22-3p and MAPK14
expression levels were negatively correlated in patients with
postmenopausal OP (Fig. 3B).
In addition, results from RT-qPCR showed that MAPK14
expression level was significantly higher in CD14+PBMCs
treated with M-CSF and RANKL for 6 days compared with those
untreated (Fig. 3C). These findings
suggested that MAPK14 may be implicated in the development of
OP.
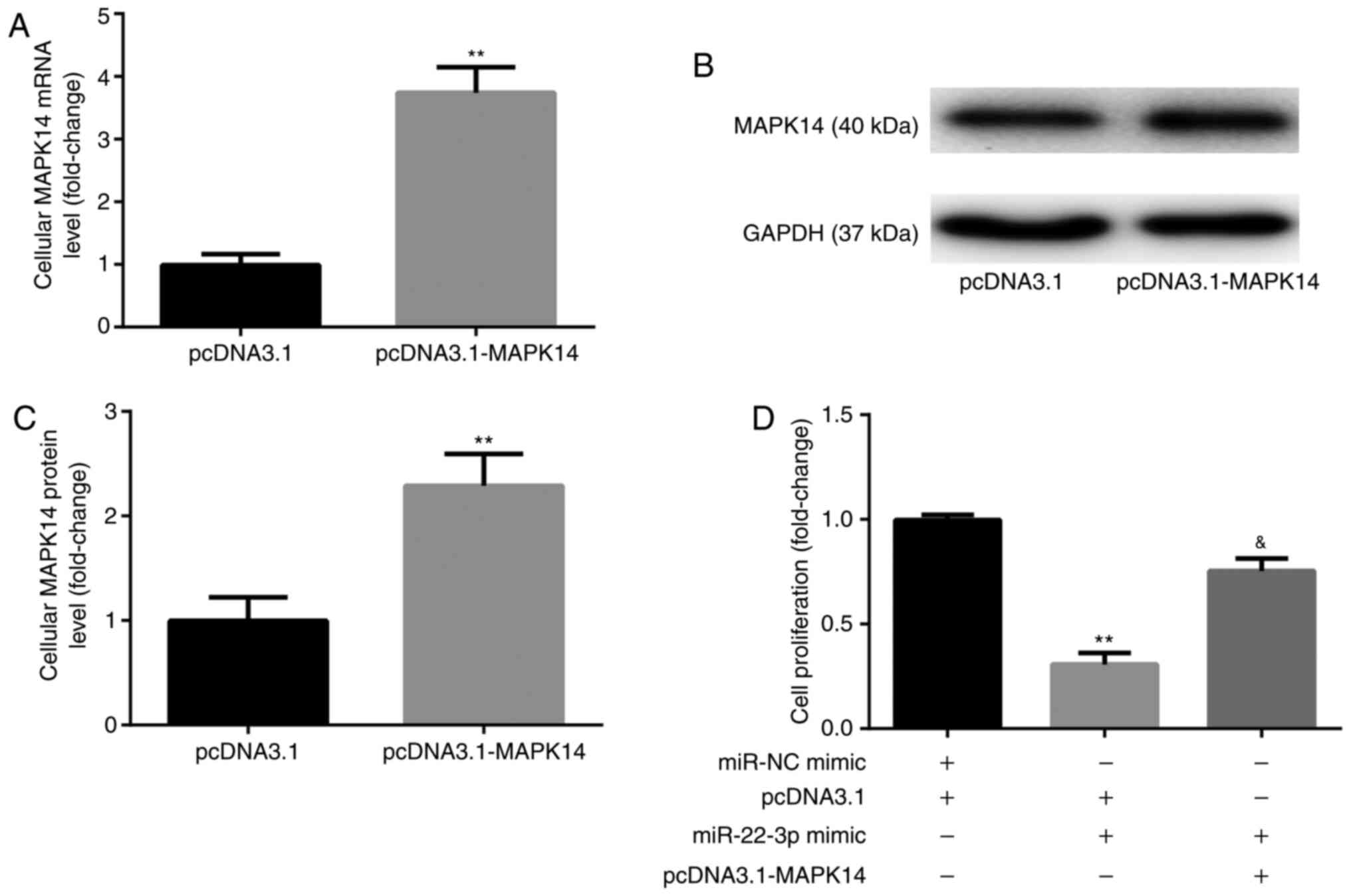
miR-22-3p mimic inhibits
CD14+PBMC proliferation by targeting MAPK14
Prior to the assessment of cell proliferation,
CD14+PBMCs were transfected with pcDNA3.1-MAPK14 to
overexpress MAPK14. The results from RT-qPCR and western blotting
demonstrated that the mRNA (Fig.
4A) and protein (Fig. 4B and
C) expression of MAPK14 was
significantly increased following transfection with pcDNA3.1-MAPK14
in CD14+PBMCs compared with pcDNA3.1 group, which
confirmed the successful transfection of pcDNA3.1-MAPK14 into
CD14+PBMCs.
The results from CCK-8 showed that at day 6 of
incubation with M-CSF and RANKL, miR-22-3p mimic significantly
decreased CD14+PBMC proliferation compared with miR-NC
mimic + pcDNA3.1 group, which was significantly rescued following
co-transfection with pcDNA3.1-MAPK14 (Fig. 4D).
miR-22-3p mimic promotes
CD14+PBMC apoptosis by targeting MAPK14
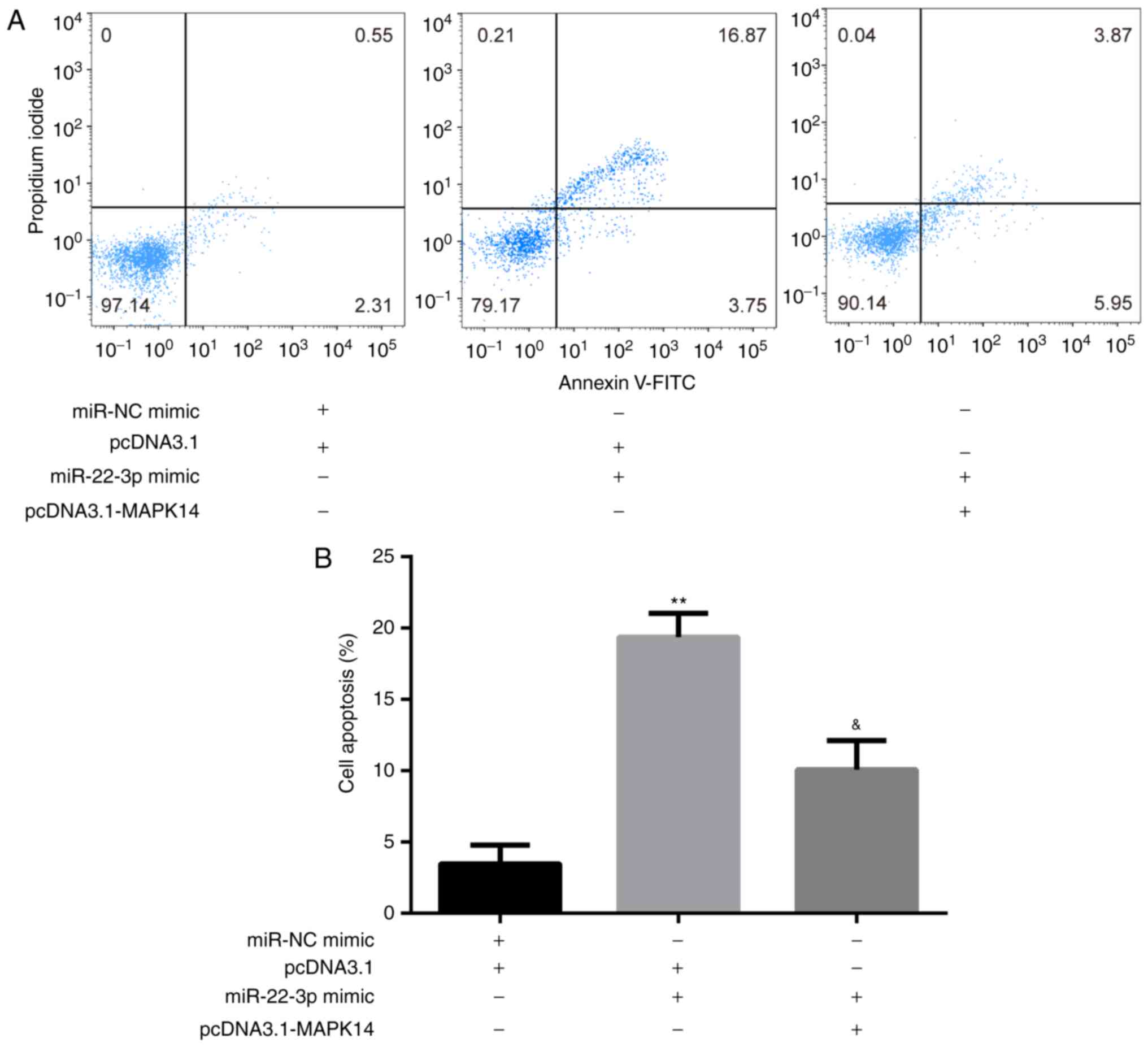
The results from flow cytometry showed that at day 6
of incubation of M-CSF and RANKL, miR-22-3p mimic significantly
increased CD14+PBMC apoptosis compared with miR-NC mimic
+ pcDNA3.1 group, which was significantly rescued following
co-transfection with pcDNA3.1-MAPK14 (Fig. 5A and B).
miR-22-3p mimic inhibits
CD14+PBMC differentiation by targeting MAPK14
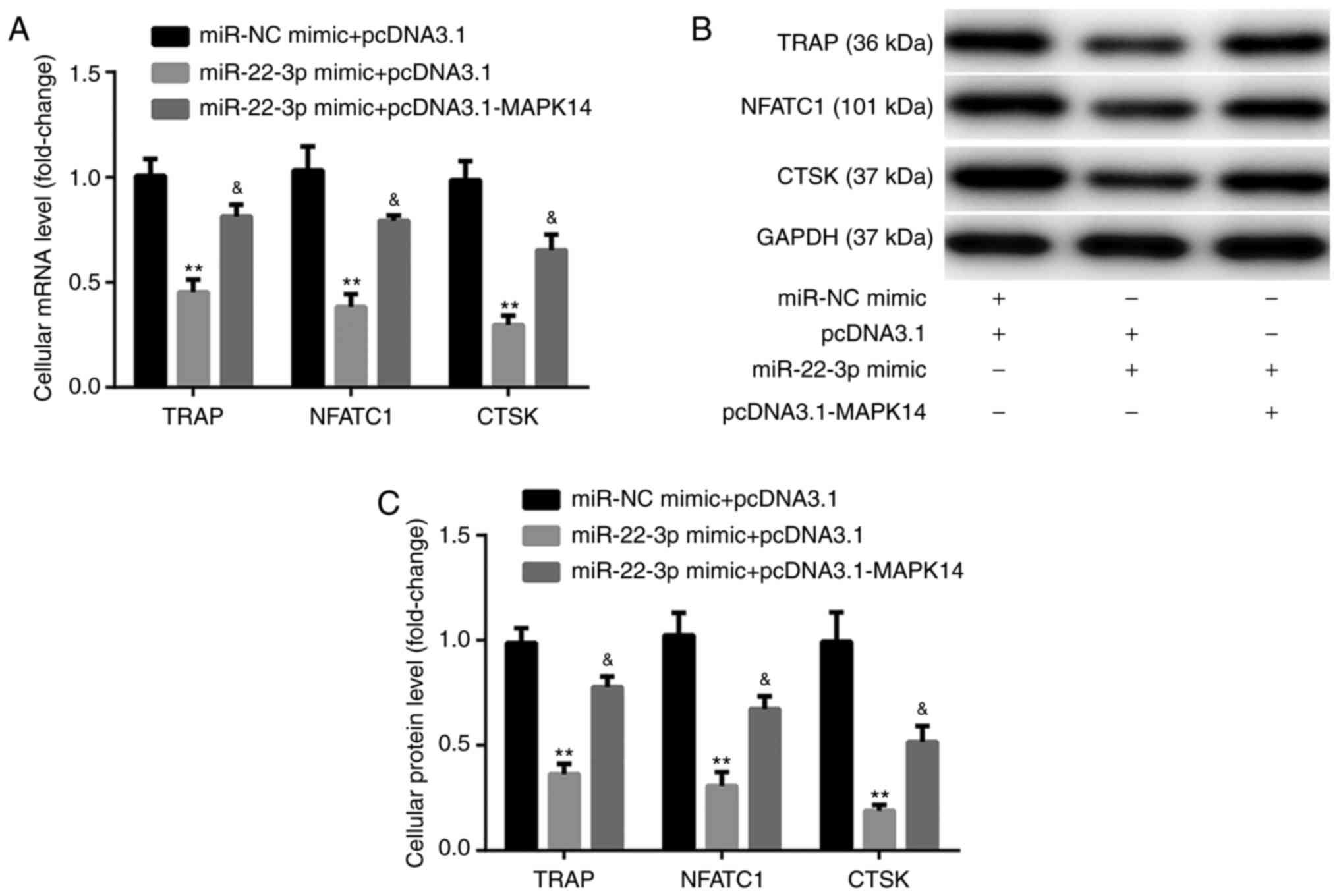
The results from RT-qPCR and western blotting
exhibited that at day 6 after incubation with M-CSF and RANKL,
miR-22-3p mimic significantly decreased the mRNA and protein
expression of the osteoclast markers, including TRAP, NFATC1, and
CTSK, compared with miR-NC mimic + pcDNA3.1 group. These
observations were reversed following co-transfection with
pcDNA3.1-MAPK14 (Fig. 6A-C). These
findings indicated that miR-22-3p mimic may attenuate OP via
reducing the osteoclastic proliferation and differentiation, while
inducing osteoclastic apoptosis by targeting MAPK14.
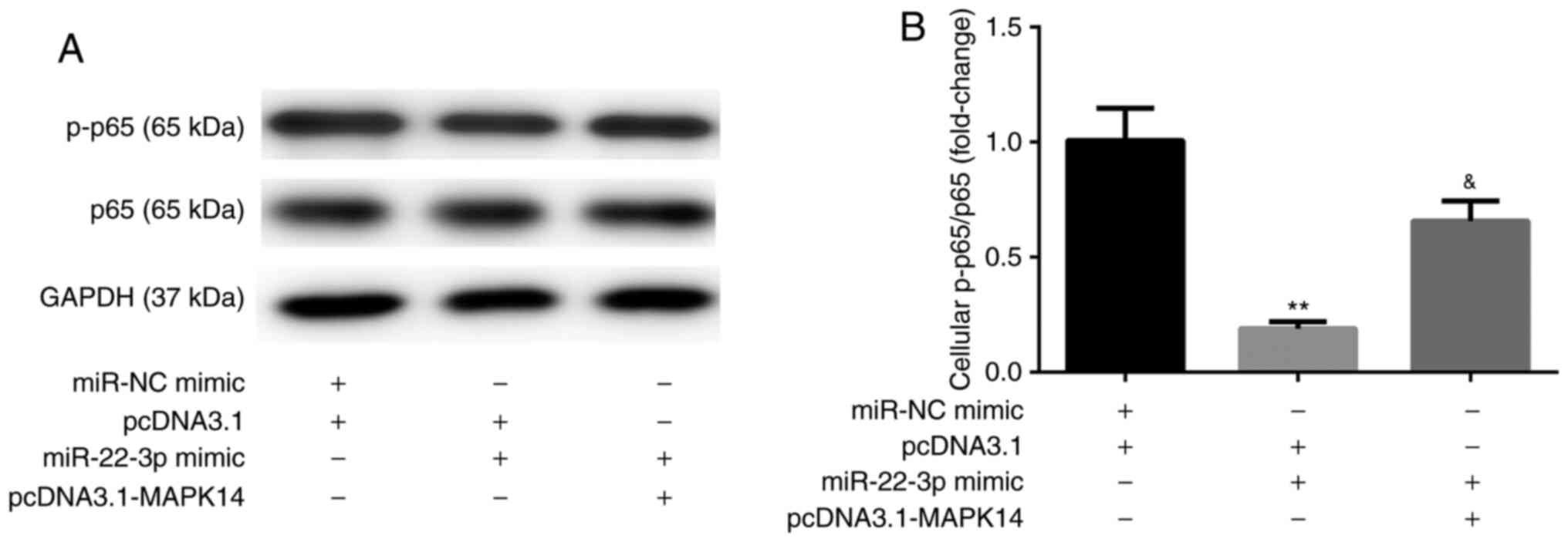
miR-22-3p mimic inhibits NF-κB
activity by targeting MAPK14
The results from western blotting showed that, at
day 6 after incubation with M-CSF and RANKL, miR-22-3p mimic
decreased the protein expression of NF-κB p-p65 compared with
miR-NC mimic + pcDNA3.1 group. These observations were reversed
following co-transfection with pcDNA3.1-MAPK14 (Fig. 7A and B). These results suggested that miR-22-3p
mimic may attenuate OP by targeting MAPK14 and inactivating NF-κB
p65.
Discussion
Abnormal expression of miRNAs has been shown to be
associated with the process of OP (16), and certain miRNAs are implicated in
multiple biological processes, including bone remolding (25). For example, estrogen can
downregulate miR-21 biogenesis and upregulate its target gene Fas
ligand, inhibiting therefore osteoclastogenesis and inducing
osteoclastic apoptosis (26,27).
Furthermore, miR-223, of which expression is decreased during
RAW264.7 osteoclast differentiation, represses osteoclastogenesis
by targeting nuclear factor I A (28,29).
In addition, Linc02349 can induce the osteogenesis of human
umbilical cord-derived stem cells by sponging miR-25-3p and
miR-33b-5p (30). The present study
aimed therefore to determine the effect of miR-22-3p on OP in
osteoclast cells. It was demonstrated that miR-22-3p inactivated
p38/NF-κB pathway by MAPK14 inhibition, thus inhibiting
osteoclastic proliferation and differentiation, while promoting
osteoclastic apoptosis.
Originally, the present study demonstrated that
miR-22-3p expression was lower in patients with postmenopausal OP
and CD14+PBMCs treated with M-CSF and RANKL for 6 days
compared with healthy volunteers and untreated
CD14+PBMCs, respectively. These findings suggested the
involvement of miR-22-3p in the development of OP, which was
consistent with a previous study reporting miR-22-3p downregulation
during osteoclast differentiation (17). Subsequently, the target genes for
miR-22-3p had to be investigated.
In the present study, MAPK14 was verified as a
target of miR-22-3p in CD14+PBMCs. The p38 MAPKs
represent a class of four paralogous mammalian genes, including
p38α/MAPK14, p38β/MAPK11, p38γ/MAPK12 and p38δ/MAPK13, which
together with ERK1/2 and ERK5 and the c-Jun N-terminal kinases,
belong to a wider family of serine-threonine and tyrosine kinases
that regulate numerous cellular processes (31). Activated p38 MAPKs can phosphorylate
and activate 200-300 downstream targets (32). In addition, p38 MAPK14/11 controls
the entry into primitive endoderm differentiation during the
development of preimplantation mouse embryo (33). Furthermore, p38α MAPK can promote
bone loss. For example, specific p38α inhibitors can prevent bone
loss in postmenopausal OP (34),
p38α MAPK promotes bone loss induced by ovariectomy via increasing
RANKL in osteoblast lineage cells (35), and p38α MAPK induces the
proliferation and differentiation of osteoclast progenitors as well
as bone remodeling (36).
Similarly, the present study demonstrated that MAPK14 expression
was higher in patients with postmenopausal OP and
CD14+PBMCs treated with M-CSF and RANKL for 6 days
compared with healthy volunteers and untreated
CD14+PBMCs, respectively.
Subsequently, the present study showed that
miR-22-3p mimic significantly decreased osteoclastic proliferation
and induced osteoclastic apoptosis by targeting MAPK14.
Furthermore, the expression of the osteoclast markers TRAP, NFATC1,
and CTSK (37) was significantly
decreased by miR-22-3p mimic, which was rescued following MAPK14
overexpression. These results demonstrated the involvement of
miR-22-3p and MAPK14 in osteoclastic proliferation, apoptosis and
differentiation. Although a previous study reported that miR-22-3p
targets MAPK14 in Huntington's disease (38), the present study verified for the
first time their relationship in OP, which is of great importance.
However, the effects of miR-22-3p and MAPK14 in the regulation of
the potential signaling pathways are unknown.
As functional cytokines during osteoclastic
differentiation in mammals, M-CSF and RANKL can activate NF-κB
pathway, which in turn induces the osteoclastic survival and
differentiation (39,40). The present study reported that
miR-22-3p mimic could inactivate NF-κB p65 by targeting MAPK14.
The current study presented the following
limitations: i) Data on resorptive activity and images of
osteoclast maturation were lacking; ii) in vivo experiments
were lacking; and iii) miRNA-22-3p downregulation experiments to
verify the present findings were missing. Future investigation will
address these limitations.
In summary, the present study demonstrated that
miR-22-3p mimic could attenuate OP via reducing osteoclastic
proliferation and differentiation, while inducing osteoclastic
apoptosis by targeting MAPK14 and inactivating NF-κB. These
findings may provide a potential therapeutic target for patients
with OP.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Chongqing
Medical Research Program (grant no. zy201602149).
Availability of materials and methods
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XJ, MY and WH performed the experiments and data
analysis. SC conceived the project, supervised the experiments and
data analysis, and prepared the manuscript. XJ, MY, WH and SC
confirmed the authenticity of all the raw data. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
Chongqing Public Health Medical Center (approval no. CMC20180106).
Each participant provided signed informed consent prior to the
beginning of the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that there have no competing
interests.
References
|
1
|
Cosman F, de Beur SJ, LeBoff MS, Lewiecki
EM, Tanner B, Randall S and Lindsay R: Erratum to: Clinician's
guide to prevention and treatment of osteoporosis. Osteoporos Int.
26:2045–2047. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Nguyen BN, Hoshino H, Togawa D and
Matsuyama Y: Cortical thickness index of the proximal femur: A
radiographic parameter for preliminary assessment of bone mineral
density and osteoporosis status in the age 50 years and over
population. Clin Orthop Surg. 10:149–156. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Xiao W, Wang Y, Pacios S, Li S and Graves
DT: Cellular and molecular aspects of bone remodeling. Front Oral
Biol. 18:9–16. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Karsenty G: Transcriptional control of
skeletogenesis. Annu Rev Genomics Hum Genet. 9:183–196.
2008.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Teitelbaum SL: Bone resorption by
osteoclasts. Science. 289:1504–1508. 2000.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Manolagas SC: From estrogen-centric to
aging and oxidative stress: A revised perspective of the
pathogenesis of osteoporosis. Endocr Rev. 31:266–300.
2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rachner TD, Khosla S and Hofbauer LC:
Osteoporosis: Now and the future. Lancet. 377:1276–1287.
2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Landgraf P, Rusu M, Sheridan R, Sewer A,
Iovino N, Aravin A, Pfeffer S, Rice A, Kamphorst AO, Landthaler M,
et al: A mammalian microRNA expression atlas based on small RNA
library sequencing. Cell. 129:1401–1414. 2007.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Gámez B, Rodriguez-Carballo E and Ventura
F: MicroRNAs and post-transcriptional regulation of skeletal
development. J Mol Endocrinol. 52:R179–R197. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lian JB, Stein GS, van Wijnen AJ, Stein
JL, Hassan MQ, Gaur T and Zhang Y: MicroRNA control of bone
formation and homeostasis. Nat Rev Endocrinol. 8:212–227.
2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mizuno Y, Yagi K, Tokuzawa Y,
Kanesaki-Yatsuka Y, Suda T, Katagiri T, Fukuda T, Maruyama M, Okuda
A, Amemiya T, et al: miR-125b inhibits osteoblastic differentiation
by down-regulation of cell proliferation. Biochem Biophys Res
Commun. 368:267–272. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wang FS, Chuang PC, Lin CL, Chen MW, Ke
HJ, Chang YH, Chen YS, Wu SL and Ko JY: MicroRNA-29a protects
against glucocorticoid-induced bone loss and fragility in rats by
orchestrating bone acquisition and resorption. Arthritis Rheum.
65:1530–1540. 2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kahai S, Lee SC, Lee DY, Yang J, Li M,
Wang CH, Jiang Z, Zhang Y, Peng C and Yang BB: MicroRNA miR-378
regulates nephronectin expression modulating osteoblast
differentiation by targeting GalNT-7. PLoS One.
4(e7535)2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Xia Z, Chen C, Chen P, Xie H and Luo X:
MicroRNAs and their roles in osteoclast differentiation. Front Med.
5:414–419. 2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
van Wijnen AJ, van de Peppel J, van
Leeuwen JP, Lian JB, Stein GS, Westendorf JJ, Oursler MJ, Im HJ,
Taipaleenmäki H, Hesse E, et al: MicroRNA functions in osteogenesis
and dysfunctions in osteoporosis. Curr Osteoporos Rep. 11:72–82.
2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Weilner S, Skalicky S, Salzer B, Keider V,
Wagner M, Hildner F, Gabriel C, Dovjak P, Pietschmann P,
Grillari-Voglauer R, et al: Differentially circulating miRNAs after
recent osteoporotic fractures can influence osteogenic
differentiation. Bone. 79:43–51. 2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ma Y, Shan Z, Ma J, Wang Q, Chu J, Xu P,
Qin A and Fan S: Validation of downregulated microRNAs during
osteoclast formation and osteoporosis progression. Mol Med Rep.
13:2273–2280. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Mäkitie RE, Hackl M, Niinimäki R, Kakko S,
Grillari J and Mäkitie O: Altered microRNA profile in osteoporosis
caused by impaired WNT signaling. J Clin Endocrinol Metab.
103:1985–1996. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhang X, Wang Y, Zhao H, Han X, Zhao T, Qu
P, Li G and Wang W: Extracellular vesicle-encapsulated miR-22-3p
from bone marrow mesenchymal stem cell promotes osteogenic
differentiation via FTO inhibition. Stem Cell Res Ther.
11(227)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
World Health Organization. Assessment of
fracture risk and its application to screening for postmenopausal
osteoporosis. Report of a WHO Study Group. World Health Organ Tech
Rep Ser. 843:1–129. 1994.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hemingway F, Cheng X, Knowles HJ, Estrada
FM, Gordon S and Athanasou NA: In vitro generation of mature human
osteoclasts. Calcif Tissue Int. 89:389–395. 2011.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Sørensen MG, Henriksen K, Schaller S,
Henriksen DB, Nielsen FC, Dziegiel MH and Karsdal MA:
Characterization of osteoclasts derived from CD14+
monocytes isolated from peripheral blood. J Bone Miner Metab.
25:36–45. 2007.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Cheng P, Chen C, He HB, Hu R, Zhou HD, Xie
H, Zhu W, Dai RC, Wu XP, Liao EY, et al: miR-148a regulates
osteoclastogenesis by targeting V-maf musculoaponeurotic
fibrosarcoma oncogene homolog B. J Bone Miner Res. 28:1180–1190.
2013.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Jing D, Hao J, Shen Y, Tang G, Li ML,
Huang SH and Zhao ZH: The role of microRNAs in bone remodeling. Int
J Oral Sci. 7:131–143. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
García Palacios V, Robinson LJ, Borysenko
CW, Lehmann T, Kalla SE and Blair HC: Negative regulation of
RANKL-induced osteoclastic differentiation in RAW264.7 cells by
estrogen and phytoestrogens. J Biol Chem. 280:13720–13727.
2005.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Sugatani T and Hruska KA: Down-regulation
of miR-21 biogenesis by estrogen action contributes to osteoclastic
apoptosis. J Cell Biochem. 114:1217–1222. 2013.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kagiya T and Nakamura S: Expression
profiling of microRNAs in RAW264.7 cells treated with a combination
of tumor necrosis factor alpha and RANKL during osteoclast
differentiation. J Periodontal Res. 48:373–385. 2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Shibuya H, Nakasa T, Adachi N, Nagata Y,
Ishikawa M, Deie M, Suzuki O and Ochi M: Overexpression of
microRNA-223 in rheumatoid arthritis synovium controls osteoclast
differentiation. Mod Rheumatol. 23:674–685. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Cao L, Liu W, Zhong Y, Zhang Y, Gao D, He
T, Liu Y, Zou Z, Mo Y, Peng S, et al: Linc02349 promotes
osteogenesis of human umbilical cord-derived stem cells by acting
as a competing endogenous RNA for miR-25-3p and miR-33b-5p. Cell
Prolif. 53(e12814)2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Cargnello M and Roux PP: Activation and
function of the MAPKs and their substrates, the MAPK-activated
protein kinases. Microbiol Mol Biol Rev. 75:50–83. 2011.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Cuadrado A and Nebreda AR: Mechanisms and
functions of p38 MAPK signalling. Biochem J. 429:403–417.
2010.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Thamodaran V and Bruce AW: p38 (Mapk14/11)
occupies a regulatory node governing entry into primitive endoderm
differentiation during preimplantation mouse embryo development.
Open Biol. 6(160190)2016.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Caverzasio J, Higgins L and Ammann P:
Prevention of trabecular bone loss induced by estrogen deficiency
by a selective p38alpha inhibitor. J Bone Miner Res. 23:1389–1397.
2008.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Thouverey C and Caverzasio J: Ablation of
p38α MAPK signaling in osteoblast lineage cells protects mice from
bone loss induced by estrogen deficiency. Endocrinology.
156:4377–4387. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Cong Q, Jia H, Li P, Qiu S, Yeh J, Wang Y,
Zhang ZL, Ao J, Li B and Liu H: p38α MAPK regulates proliferation
and differentiation of osteoclast progenitors and bone remodeling
in an aging-dependent manner. Sci Rep. 7(45964)2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Boyle WJ, Simonet WS and Lacey DL:
Osteoclast differentiation and activation. Nature. 423:337–342.
2003.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Jovicic A, Zaldivar Jolissaint JF, Moser
R, Silva Santos MF and Luthi-Carter R: MicroRNA-22 (miR-22)
overexpression is neuroprotective via general anti-apoptotic
effects and may also target specific Huntington's disease-related
mechanisms. PLoS One. 8(e54222)2013.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kwon M, Kim JM, Lee K, Park SY, Lim HS,
Kim T and Jeong D: Synchronized cell cycle arrest promotes
osteoclast differentiation. Int J Mol Sci. 17(1292)2016.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Boyce BF: Advances in osteoclast biology
reveal potential new drug targets and new roles for osteoclasts. J
Bone Miner Res. 28:711–722. 2013.PubMed/NCBI View Article : Google Scholar
|