Introduction
As reported, triple-negative breast cancer (TNBC) is
one of the most common malignant tumors and the main cause of
cancer-related mortality in women (1,2). TNBC
is a breast cancer subtype renowned for its capacity to affect
younger women, metastasize early despite optimal adjuvant treatment
and carry a poor prognosis (3,4).
Despite significant progress in the diagnosis and treatment of
TNBC, the long-term survival rate of TNBC patients remains
unsatisfactory (5). Thus, it is of
great significance to make more efforts to reduce the mortality of
TNBC patients. There is an urgent need to further probe potential
regulatory patterns in TNBC development.
Long non-coding RNAs (lncRNAs) are non-coding RNAs
with a length greater than 200 nucleotides (6,7).
Studies have revealed that lncRNAs play important roles in numerous
aspects such as dose compensation effect, epigenetic regulation,
cell cycle regulation and cell differentiation regulation (8-10).
It is often reported that lncRNAs serve as key regulators via
sponging microRNAs (miRNAs) and targeting messenger RNAs (mRNAs)
(6), and ultimately participate in
regulation of cancers. Previous studies have reported that lncRNAs
are identified as potential biomarkers in a plurality of human
cancers (11). For example, lncRNA
NNT-AS1 was revealed to promote proliferative ability in glioma via
sponging miR-494-3p (12).
ST8SIA6-AS1 facilitated proliferative and migrative abilities of
hepatocellular carcinoma via sponging miR-5195-3p (13). Moreover, UCA1 accelerated
endometrial cancer progression via interacting with KLF5(14). Hence, in the present study the aim
was to explore the detailed mechanism by which lncRNAs regulate
TNBC proliferation and migration. Previously, it has been revealed
that small nucleolar RNA host gene 11 (SNHG11) acted as a tumor
promoter in lung (15), prostate
(16) and liver cancer (17), but it has not been studied in TNBC.
Therefore, the present study mainly investigated the role and
regulatory mode of SNHG11 in TNBC.
miRNAs are another subgroup of non-coding RNAs with
a length of 18-25 nucleotides (18-20).
miRNAs can specifically bind with the 3'-untranslated region (UTR)
of mRNAs, thereby modulating gene expression post-transcriptionally
and taking part in various cancer type progressions. For example,
miR-124-3p directly targeted SARM1 to contribute to prostate cancer
progression (21). As a tumor
suppressor in lung cancer, miR-93 downregulated the expression of
TBP-2(22). In recent years,
microRNA 2355-5p (miR-2355-5p) has received a lot of attention in
some cancers. As reported, miR-2355-5p has been revealed to play a
role in the suppression of cancer development in a variety of
cancers, including chondrosarcoma (23) and bladder cancer (24). However, the interaction between
SNHG11 and miR-2355-5p in TNBC has not been elucidated and requires
further investigation.
In summary, the present study was designed to
explore the biological role and regulatory network of SNHG11 in
TNBC. The findings suggested that SNHG11 could be a potential
biomarker for TNBC treatment.
Materials and methods
Tissue samples
A total of 42 pairs of TNBC tissues and adjacent
noncancerous tissues were obtained from TNBC patients who were
clinically and pathologically diagnosed with TNBC at the Jiangsu
Taizhou People's Hospital from July 2018 to July 2019. Written
informed consent was obtained from all participants and study was
approved by the Ethics Committee of Jiangsu Taizhou People's
Hospital (Taizhou, China). The age range of patients with TNBC was
26-67 years, with a mean age of 47.2±5.1 years. The distance
between the tumor tissue edge and the adjacent tissue from which
samples were obtained was >5 cm. The collected tissues were
rapidly frozen in liquid nitrogen and stored at -80˚C for the
following research. All patients had not received anticancer
treatment.
Cell lines
TNBC cell lines BT-20, BT-549, MDA-MB-231, Hs 578T
and an immortalized human epithelial cell line MCF10A were offered
by the American Type Culture Collection (ATCC). In brief, cells
were cultured in Roswell Park Memorial Institute 1640 (RPMI-1640)
supplemented with 10% fetal bovine serum (FBS), 2 mM L-glutamine,
100 U/ml penicillin (Sigma-Aldrich; Merck KGaA) and 100 µg/ml
streptomycin (Sigma-Aldrich; Merck KGaA). The cells were maintained
in a humid atmosphere at 37˚C in the presence of 5%
CO2.
Cell transfection
For downregulating SNHG11 in BT-20 and BT-549 cells,
two short hairpin RNAs (sh-RNA#1 and sh-RNA#2; 3 µg) specifically
targeting SNHG11 with sh-NC (cat. no. C02003) as the negative
control were designed and synthesized by Shanghai GenePharma Co.,
Ltd. The sequences of sh-SNHG11#1/2 are as follows: sh-SNHG11#1:
5'-CCTTGGGTCTGGAAACTGTTA-3'; sh-SNHG11#2:
5'-CCGTGTGTGTTATATCATGAT-3'. The full length of CBX5 (mRNA) was
synthesized and subcloned into pcDNA3.1 (Invitrogen; Thermo Fisher
Scientific, Inc.) plasmid to produce pcDNA3.1/CBX5. For miR-2355-5p
overexpression, BT-20 and BT-549 cells were transfected with
miR-2355-5p mimics or (negative control) NC mimics. miR-2355-5p
mimics and NC mimics (cat. no. B01001) were both obtained from
Shanghai GenePharma Co., Ltd. BT-20 and BT-549 cells
(1x106 cells/well) were seeded in 24-well plates and 500
µl DMEM was added to each well. When the cells reached 40-60%
confluence, the aforementioned vectors were transfected into cells
at a final concentration of 50 nM using Lipofectamine®
2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.) at 37˚C
with 5% CO2 according to the manufacturer's
instructions. Cells were harvested 48 h after transfection.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNAs were extracted from tissues and cells
with TRIzol® (Invitrogen; Thermo Fisher Scientific,
Inc.) reagent kits as recommended. Total RNAs were reverse
transcribed into complementary DNA (cDNA) using PrimeScript RT
reagent kits (cat. no. RRO36A; Takara Biotechnology Co., Ltd.)
according to the manufacturer's protocol. A SYBR® Premix
Ex Taq™ II reagent kit (cat. no. RR820A; Takara) was utilized to
perform RT-qPCR with an ABI7500 real-time qPCR system (ABI; Thermo
Fisher Scientific, Inc.). RNU6 (U6) was used as the internal
reference for miR-2355-5p. Glyceraldehyde-3-phosphate dehydrogenase
(GAPDH) was applied as an internal reference for SNHG11 and CBX5.
The relative quantification was calculated with the
2-∆∆Cq method (25). The
following primers were used: SNHG11 forward,
5'-TGGGAGTTGTCATGTTGGGA-3' and reverse, 5'-ACTCGTCACTCTTGGTCTGT-3';
miR-2355-5p forward, 5'-ATTGTCCTTGCTGTTTGGAGAT-3' and reverse,
5'-GCGAGCACAGAATTAATACGAC-3'; CBX5 forward,
5'-GCAGACGTTAGCGTGAGTG-3' and reverse,
5'-GCGGAATTCGGATCCCTCGAGTT-3'; U6 forward,
5'-CTCGCTTCGGCAGCACATA-3' and reverse, 5'-CGCTTCACGAATTTGCGTG-3';
GAPDH forward, 5'-GATCATCAGCAATGCCTCCT-3' and reverse,
5'-TGAGTCCTTCCACGATACCA-3'. The following thermocycling conditions
were used: Initial denaturation at 95˚C for 3 min, followed by 40
cycles at 95˚C for 5 sec, 60˚C for 30 sec and at 72˚C for 45 sec,
before final extension at 72˚C for 3 min.
Cell Counting Kit-8 (CCK-8) assay
Cell proliferation in response to SNHG11 knockdown
was measured with CCK-8 (Dojindo Molecular Technologies, Inc.)
following the manufacturer's instructions. The cells were seeded
into 96-well plates (1x103 cells/well) in triplicate and
at 24, 48 and 72 h. Then, CCK-8 reagent was added, and the cells
were maintained for a further 2 h at 37˚C. The chromogenic reaction
was allowed in a culture incubator for 4 h. The absorption was
recorded at 450 nm by Tecan Infinite F500 microplate reader (Tecan
Group, Ltd.).
Colony formation assay
The colony formation assay was utilized to assess
the cell proliferation. Transfected cells were diluted and seeded
onto 6-well plates with a density of ~1x103 cells/well.
The cells were then incubated at 37˚C for 14 days and fixed with
paraformaldehyde at room temperature for 15 min and stained with
0.1% crystal violet (Beyotime Institute of Biotechnology) at room
temperature for 30 min when colonies (≥50 cells) were visible.
After 48 h, the plates were photographed under a light microscope
(DM1000; Leica Microsystems GmbH), and the number of colonies were
counted (magnification, x100).
Flow cytometric analysis
Briefly, transfected cells were collected and
resuspended with phosphate-buffered saline (PBS). Then, the density
of cells was adjusted to 1x106 cells/ml. Transfected
cells were double stained by propidium iodide and Annexin
V-fluorescein isothiocyanate (20 mg/ml; BD Pharmingen; BD
Biosciences) at room temperature for 10 min in line with the
manufacturer's instructions. Finally, cell apoptosis was detected
via flow cytometry using a FACScan® (BD Biosciences)
equipped with CellQuest Pro software 5.1 (BD Biosciences).
Transwell assay
The capacity for cell migration was assessed using
Transwell chambers (8 µm diameter; Corning, Inc.) without Matrigel.
After being cultured for 48 h, the cells were harvested, washed
with PBS, and resuspended in RPMI-1640 medium without FBS. In
total, 100 µl of suspension containing 5x104 cells was
added into the upper chambers. Transwell chambers were placed into
a 24-well plate that had already been covered with 500 µl of
RPMI-1640 medium containing 10% of FBS. At 24 h after inoculation,
nonmigratory cells were gently removed, and the migratory cells
were fixed with 100% methanol at room temperature for 15 min,
stained with 0.5% crystal violet at room temperature for 20 min,
washed with PBS, and imaged using an inverted microscope
(magnification, x200; Olympus Corporation). The Transwell invasion
assay was similar to the migration assay except that the chamber
was precoated with Matrigel (BD Biosciences) overnight at room
temperature. The migration and invasion abilities were measured by
respectively counting the migrated and invasive cells.
Western blotting
Total proteins in cells were extracted with Radio
Immunoprecipitation Assay (RIPA) lysis buffer containing protease
and phosphatase inhibitors [Roche Diagnostics (Shanghai) Co.,
Ltd.]. Protein concentration was detected by a bicinchoninic acid
kit. Then the proteins (5 µg/sample) were separated using 10%
sodium dodecyl sulfate polyacrylamide gel electrophoresis and
transferred onto a polyvinylidene fluoride (PVDF) membrane. The
membranes were blocked with 5% skim milk powder at room temperature
for 1 h and incubated with the primary antibodies (Abcam) of
antibodies to E-cadherin (cat. no. ab1416; 1:50) and N-cadherin
(cat. no. ab98952; 1:500) and GAPDH (cat. no. ab8245; 1:500) at 4˚C
overnight. Then the membranes were washed with Tris-buffered saline
20% Tween-20 and further incubated with the horseradish
peroxidase-conjugated goat anti-rabbit antibody (cat. no. sc-2357;
1:5,000; Santa Cruz Biotechnology, Inc.) at room temperature for 1
h. Proteins on the membrane were visualized by an enhanced
chemiluminescence detection kit (BB-3501; Amersham Pharmacia
Biotech; Cytiva) and Bio-Rad image analysis system (Bio-Rad
Laboratories, Inc.).
Bioinformatics analysis
The downstream gene of SNHG11 and the downstream
gene of miR-2355-5p were predicted using the starBase database
(http://starbase.sysu.edu.cn/). The
interaction between SNHG11 miR-2355-5p was also predicted using the
starBase database. The binding fragment of miR-2355-5p on the 3'UTR
of CBX5 was predicted using TargetScan (http://www.targetscan.org/vert_72/).
Luciferase reporter assay
To detect the binding ability between SNHG11 (CBX5)
and miR-2355-5p, wild-type sequences of SNHG11 (CBX5 3'UTR) and the
mutant SNHG11 (CBX5 3'UTR) were cloned into the firefly luciferase
gene reporter vector pmiRGLO (Promega Corporation). The plasmid was
synthesized by Invitrogen; Thermo Fisher Scientific, Inc. The
pmirGLO-SNHG11 (CBX5 3'UTR)-Wt or pmirGLO-SNHG11(CBX5 3'UTR)-Mut
was co-transfected with miR-2355-5p mimics or NC mimics (Guangzhou
RiboBio Co., Ltd.) into BT-20 and BT-549 cells. Then, 48 h after
transfection using Lipofectamine® 2000 reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) at 37˚C with 5%
CO2. The luciferase assay was performed using the
dual-luciferase reporter assay system kit (Promega Corporation)
according to the manufacturer's instructions. The luciferase
activity was analyzed using a Modulus single-tube multimode reader
(Promega Corporation) in comparison with Renilla luciferase
activity.
RNA pull down
Biotinylated SNHG11 (SNHG11 probe-biotin) and
negative control (SNHG11 probe-no biotin) purchased from Sangon
Biotech Co., Ltd., were transfected into BT-20 and BT-549 cells
using Lipofectamine® 2000 reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) for 48 h at room temperature. The cells
were lysed (Roche Diagnostics) and collected after transfection. A
total of 20 µl biotin-labeled RNA and 40 µl streptavidin Dynabeads
(Thermo Fisher Scientific, Inc.) were added to 1 ml of BT-20 and
BT-549 cell supernatant lysate for 6 h at 25°C. Beads
were isolated from the supernatant (centrifugation at 2,500 x g for
5 min at 4°C) and washed with wash buffer (10 mM
Tris-HCl pH 7.5, 1 mM EDTA, 2 M NaCl and 0.1% Tween-20) followed by
centrifugation (2,500 x g for 5 min at 4˚C). Isolated
beads were mixed with 20 µl SDS loading buffer (Beyotime Institute
of Biotechnology) and boiled for 10 min at 100˚C. The
bound RNAs were subjected to RT-qPCR for quantification and
analysis.
RNA immunoprecipitation (RIP)
assay
RIP assay was performed under the instructions of
the EZ-Magna RIP RNA-Binding Protein Immunoprecipitation Kit (EMD
Millipore). Cells in different groups were lysed in lysis buffer (5
mM HEPES; pH 7.4; 85 mM KCl; 0.5% NP40; Thermo Fisher Scientific,
Inc.) which contained protease inhibitor cocktail and RNase
inhibitor. The cells were incubated with RIP buffer containing
magnetic beads coated with Ago2 antibodies (cat. no. MABE56; EMD
Millipore). IgG (cat. no. 12-370; EMD Millipore) group was applied
as a negative control group. After incubation at 4˚C for 2 h, the
coprecipitated RNA was eluted from the beads and measured by PCR
analysis.
Statistical analysis
The data were obtained and presented as the mean ±
standard deviation. SPSS 20.0 software (IBM Corp.) and Graphpad
Prism 7.0 software (GraphPad Software, Inc.) were used for data
evaluation and charting. One-way analysis of variance (ANOVA)
followed by a Tukey's post hoc test or Student's t-test was
performed for comparison between groups. A log rank test was
performed for Kaplan-Meier survival curves. In addition, the
relationship among genes was determined by Spearman correlation
analysis. All experiments were repeated three times. P<0.05 was
considered to indicate a statistically significant difference.
Results
SNHG11 upregulation is significantly
reflected in TNBC and represents an unsatisfactory prognosis
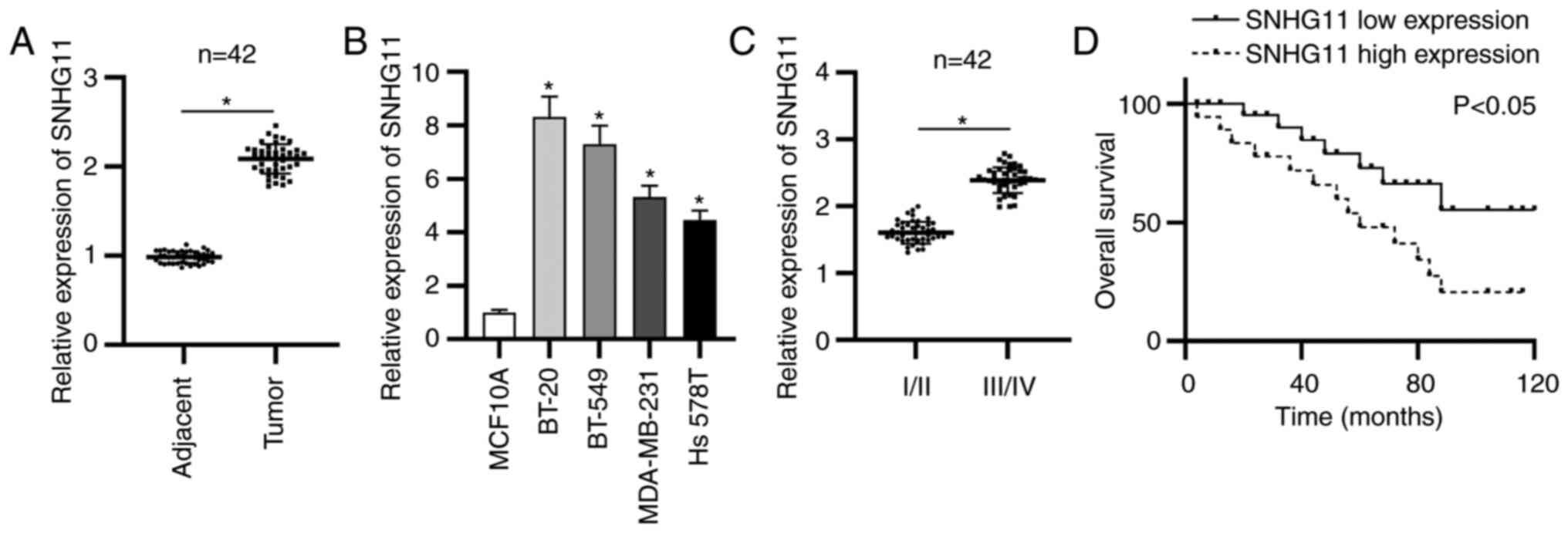
To probe role of lncRNA SNHG11 in TNBC, the SNHG11
expression in TNBC was first detected via RT-qPCR analysis. The
data revealed that SNHG11 expression was significantly upregulated
in TNBC tissues (Fig. 1A). SNHG11
overexpression was also revealed in 4 human TNBC cells compared
with that of normal mammary epithelial cells (MCF10A) (Fig. 1B). In addition, a marked increase of
SNHG11 expression was presented at advanced stages of patients with
TNBC (Fig. 1C). Moreover,
Kaplan-Meier analysis revealed that TNBC patients with a high level
of SNHG11 exhibited poorer overall survival compared to patients
with a low expression level of SNHG11 (Fig. 1D). Collectively, SNHG11 was highly
expressed in TNBC and high SNHG11 expression was related to a poor
prognosis.
SNHG11 aggravates cell proliferative
and migrative capabilities of TNBC cells
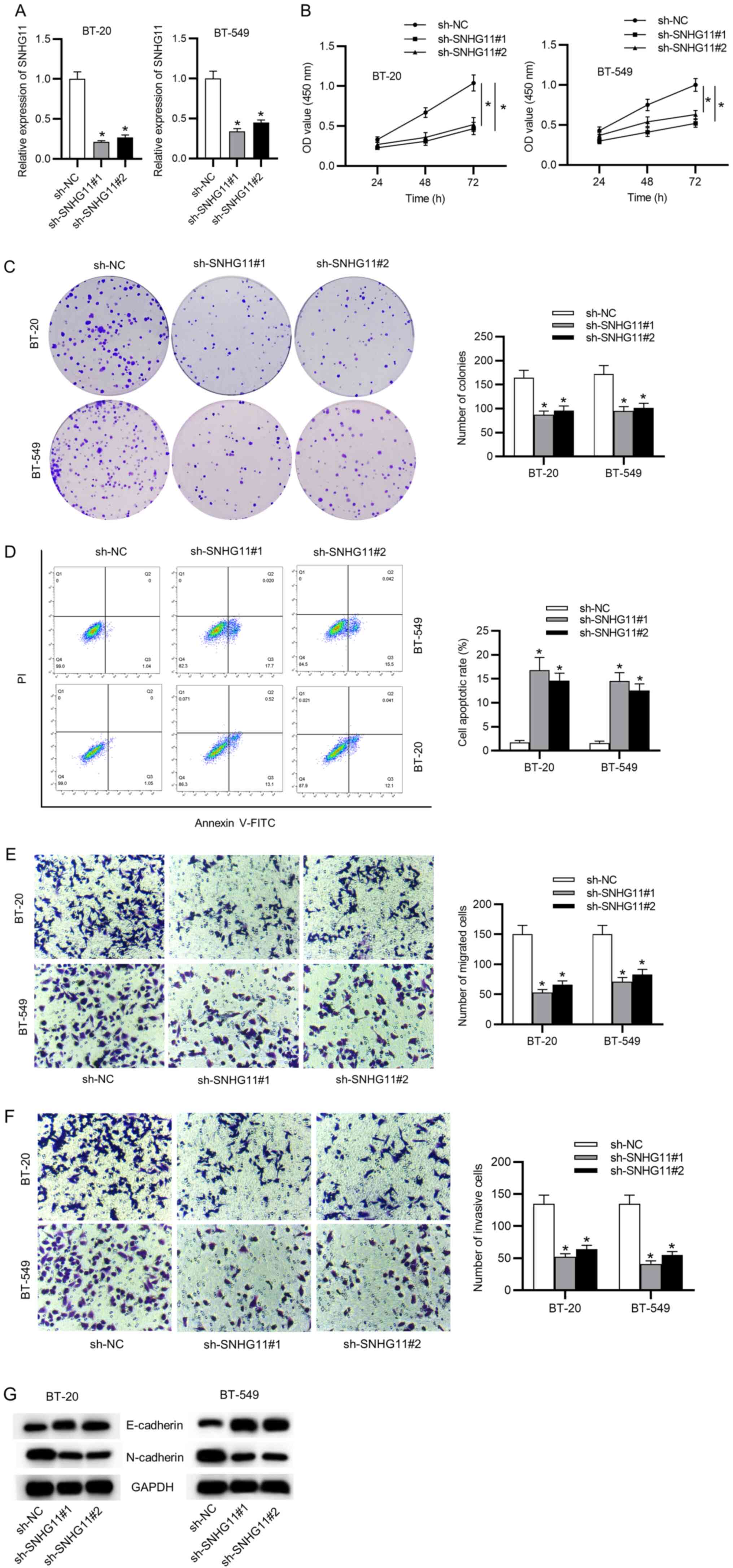
To explore possible biological roles of SNHG11 in
tumorigenesis and development of TNBC, SNHG11 expression was first
knocked down using sh-SNHG11#1 or sh-SNHG11#2. In succession,
silencing efficacy of sh-SNHG11 in BT-20 and BT-549 cells was
confirmed with RT-qPCR analysis (Fig.
2A). Then, it was determined that SNHG11 knockdown inhibited
cell viability through CCK-8 results (Fig. 2B). It had an inhibitory effect on
colony formation (Fig. 2C), while
it induced cell apoptosis in TNBC cells (Fig. 2D). In addition, Transwell
experiments revealed that SNHG11 knockdown significantly hampered
cell migration and invasion capacities in TNBC (Fig. 2E and F). Western blot analysis examined the
levels of epithelial-mesenchymal transition (EMT) markers
(E-cadherin and N-cadherin). As presented in Fig. 2G, the expression of the epithelial
marker E-cadherin was markedly upregulated, and the mesenchymal
marker N-cadherin expression was obviously reduced by silencing of
SNHG11 in TNBC. Collectively, SNHG11 accelerated the cell growth
and migration in TNBC.
SNHG11 sponges miR-2355-5p in
TNBC
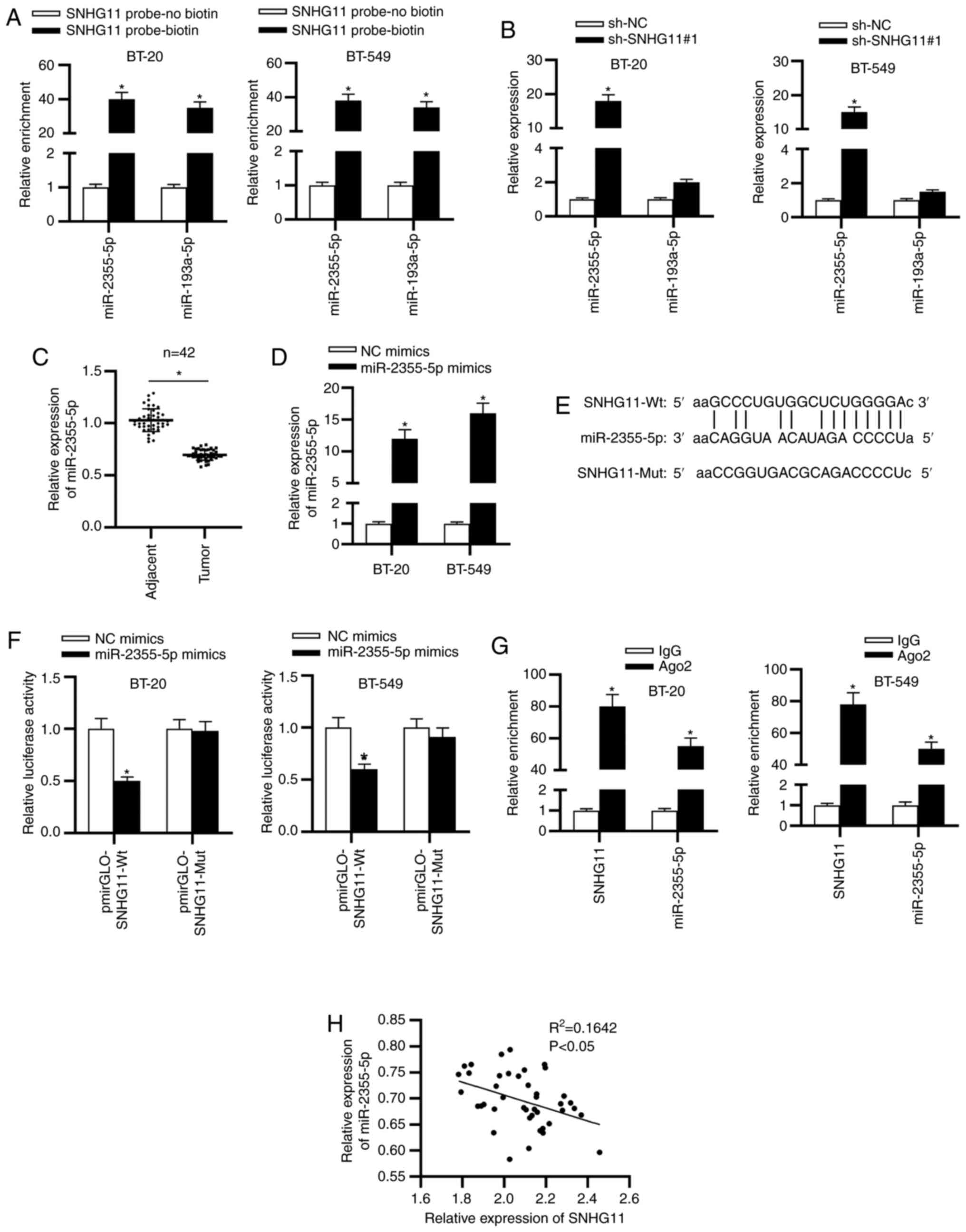
To probe the underlying mechanism of SNHG11 in TNBC,
starBase v2.0 was used and 2 miRNAs (miR-2355-5p and miR-193a-5p)
(condition: Pan-cancer: 8 cancer types) which may bind with SNHG11
were identified. To select the qualified miRNA, a pull down assay
was performed. As revealed in Fig.
3A, both miR-2355-5p and miR-193a-5p were enriched in the
SNHG11-probe biotin group compared with the SNHG11-probe no biotin
group. Next, it was revealed that only miR-2355-5p could be
upregulated with sh-SNHG11#1 (Fig.
3B). Thus, miR-2355-5p was identified to be the target of
SNHG11 and selected for the following experiments. It was revealed
from RT-qPCR analysis that miR-2355-5p expression exhibited a
significant decrease in TNBC tissues (Fig. 3C). The overexpression efficacy of
miR-2355-5p was examined as revealed in Fig. 3D. The binding sequence of
miR-2355-5p with SNHG11 was predicted by starBase (Fig. 3E). To verify interaction between
SNHG11 and miR-2355-5p, luciferase reporter and RIP assays were
conducted. The results indicated that the luciferase activity of
SNHG11-Wt reporters was significantly decreased by miR-2355-5p
mimics, whereas SNHG11-Mut reporters presented no marked change
(Fig. 3F). Based on the results
from the RIP assay, it was revealed that SNHG11 and miR-2355-5p
were significantly enriched in Ago2 group (Fig. 3G). Additionally, a negative
correlation was revealed between SNHG11 and miR-2355-5p in TNBC
tissues (Fig. 3H). Hence, it was
concluded that miR-2355-5p was sponged by SNHG11 in TNBC.
CBX5 was a downstream target of
miR-2355-5p
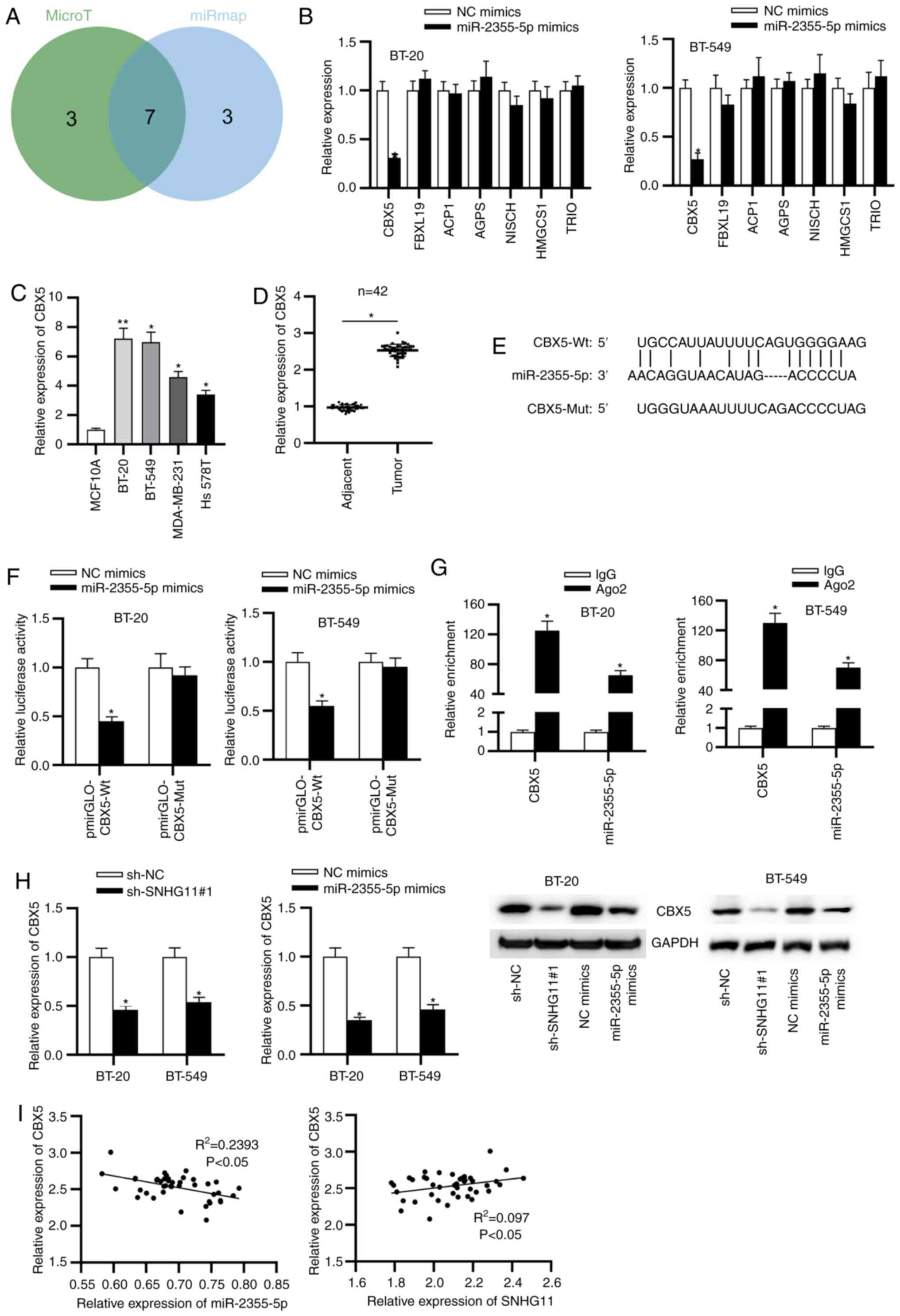
To determine the target of miR-2355-5p, starBase
v2.0 was used to obtain potential downstream targets of
miR-2355-5p. There were 7 potential mRNAs that were identified as
follows: CBX5, FBXL19, ACP1, AGPS, NISCH, HMGCS1 and TRIO (Fig. 4A). After miR-2355-5p upregulation,
the expression levels of the aforementioned mRNAs were examined,
and the results revealed that only CBX5 was downregulated upon
miR-2355-5p overexpression (Fig.
4B). Therefore, CBX5 was demonstrated to be targeted by
miR-2355-5p and selected for the subsequent experiments. In
addition, it was revealed that CBX5 expression was upregulated in
TNBC tissues and cells (Fig. 4C and
D). The binding fragment of
miR-2355-5p on 3'UTR of CBX5 was predicted by TargetScan (Fig. 4E). Additionally, luciferase activity
of CBX5-Wt was decreased with miR-2355-5p upregulation (Fig. 4F). Concurrently, both CBX5 and
miR-2355-5p were enriched in Ago2-conjugated beads compared with
normal IgG (Fig. 4G). In addition,
SNHG11 silencing or miR-2355-5p upregulation suppressed the
expression of CBX5 (Fig. 4H).
Furthermore, CBX5 expression in TNBC was negatively correlated with
miR-2355-5p expression but positively correlated to SNHG11
expression in TNBC tissues (Fig.
4I). To sum up, CBX5 was directly targeted by miR-2355-5p, and
SNHG11 upregulated CBX5 by sponging miR-2355-5p.
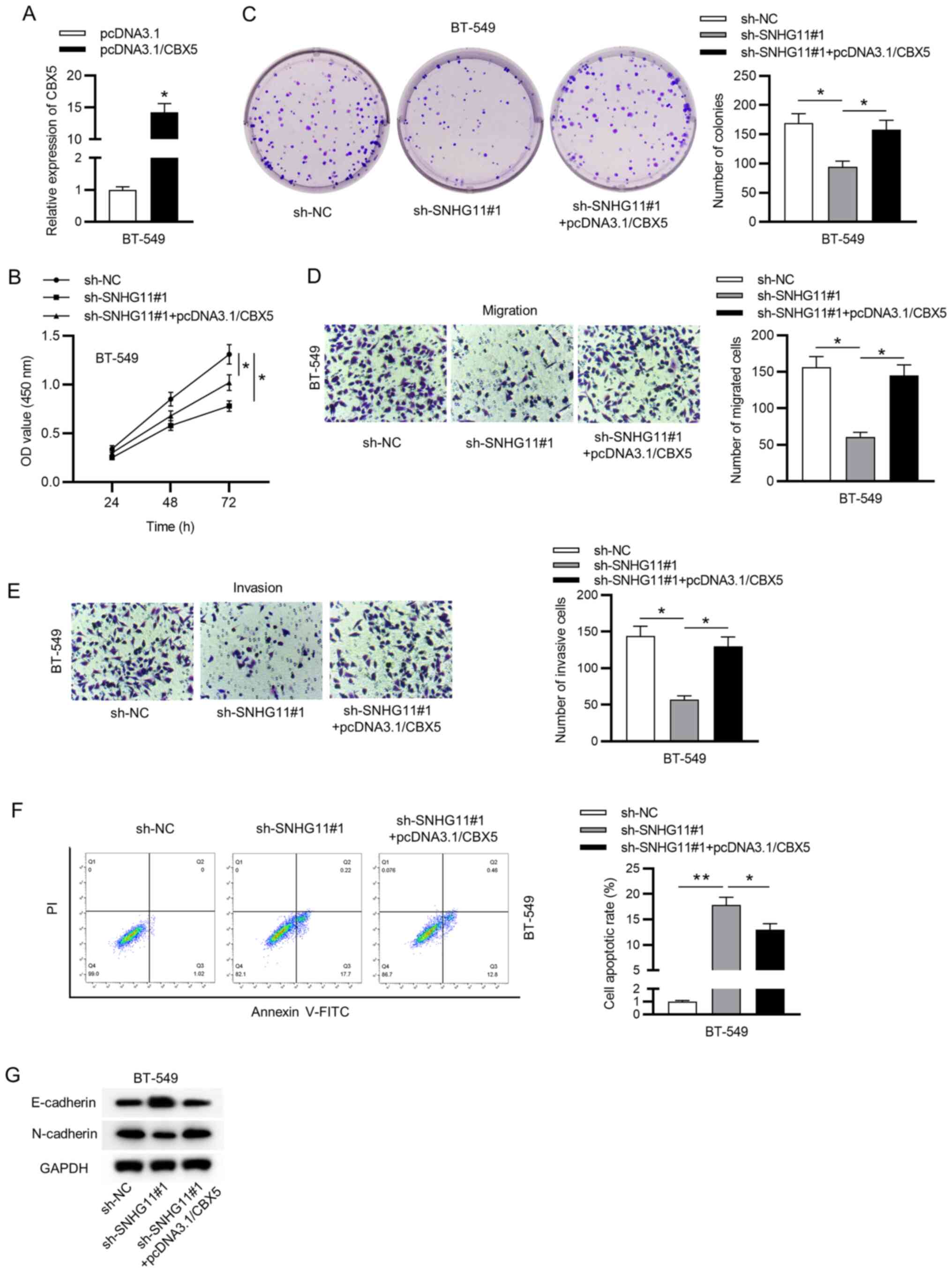
SNHG11 aggravates TNBC proliferation
and migration by sponging miR-2355-5p and regulating CBX5
Finally, to determine the impact of the
SNHG11/miR-2355-5p/CBX5 axis on TNBC, rescue experiments were
implemented in BT-549 cells. Firstly, the overexpressed efficiency
of CBX5 in BT-549 cells was validated by RT-qPCR analysis (Fig. 5A). Furthermore, upregulated CBX5
reversed the inhibitive influence of SNHG11 silencing on cell
viability and proliferation in TNBC (Fig. 5B and C). Additionally, overexpressed CBX5
rescued the migrative and invasive abilities suppressed under
SNHG11 downregulation in TNBC (Fig.
5D and E). Flow cytometric
analysis indicated that knockdown of SNHG11 promoted cell
apoptosis, but this effect could be reversed by overexpressing CBX5
(Fig. 5F). Moreover, upregulated
CBX5 counteracted SNHG11 silencing-mediated attenuation of the EMT
process (Fig. 5G). Collectively,
our data revealed that SNHG11 accelerated the cell growth of TNBC
via modulating the miR-2355-5p/CBX5 axis.
Discussion
Recently, numerous studies have revealed that SNHG11
functions as an oncogene in lung (12), prostate (13) and liver (14). In the present study, initial
evidence was provided of the high expression of SNHG11 in TNBC
tissues and cells. Then loss-of-function assays were conducted to
determine the function of SNHG11 in TNBC. It was revealed that
SNHG11 knockdown inhibited TNBC cell viability, proliferation,
migration, invasion and the EMT process, whereas it contributed to
TNBC cell apoptosis. Therefore, the results also indicated that
SNHG11 acted as a tumor promoter in the cell growth of TNBC.
As is well known, lncRNAs regulate the behaviors of
cancer cells via sponging miRNAs (26). LncRNAs can reduce the regulatory
effect of miRNAs (27). The target
gene of SNHG11 was predicted and it was determined that miR-2355-5p
was a putative target gene of SNHG11. Previous studies have
verified that miR-2355-5p has a suppressive effect in various
cancers, including chondrosarcoma (23) and bladder cancer (24). In our present study, it was
determined that miR-2355-5p expression was downregulated in TNBC.
As reported, lncRNAs contain structural domains that can sense and
bind other RNAs via complementary base pairing (28). Hence, further mechanism experiments
were performed and it was demonstrated that there was a direct
binding sequence between SNHG11 and miR-2355-5p, and that there was
a negative expression correlation between these two genes. In
brief, binding of SNHG11 and miR-2355-5p could be observed in
TNBC.
A previous study has revealed that functional
interactions in competing endogenous RNA (ceRNA) networks aid in
coordinating a number of biologic processes and, when perturbed,
contribute to disease pathogenesis (29). Therefore, to complete the ceRNA
mode, starBase 2.0 was utilized to predict the downstream target
genes. Among mRNA candidates, CBX5 has previously been reported to
aggravate gastric cancer progression (30). Moreover, CBX5 acted as an oncogene
and could be positively regulated by BRD4 in ovarian cancer
(31). In the present study, it was
confirmed that miR-2355-5p could bind to CBX5, and their expression
levels were negatively correlated in TNBC tissues. Such ceRNAs thus
alter their abundance by competing for binding to miRNAs (32). In addition, as reported, lncRNAs
play vital roles in the regulation of cancers via modulating mRNA
or the epigenetic landscape of their target genes (33). Hence, subsequent rescue assays
verified that overexpression of CBX5 could rescue the inhibitive
influence of SNHG11 silencing on TNBC. In summary, lncRNA SNHG11
regulated CBX5 expression via sponging miR-2355-5p in TNBC.
To sum up, the present data initially demonstrated
the upregulated expression of SNHG11 in TNBC. SNHG11 upregulated
the expression of CBX5 to play a tumor-promoting role in TNBC via
sponging miR-2355-5p, which indicated a novel regulatory axis in
TNBC. These findings revealed that SNHG11 may be a potential
biomarker for TNBC treatment.
Acknowledgements
Not applicable.
Funding
Funding: The current work was supported by Jiangsu Taizhou
People's Hospital 2019 Hospital-level Scientific Research Fund
Project (grant no. ZL201917).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LY and WZ conceived and designed the experiments.
LY, WZ, PW, QZ, AC, XY and KS carried out the experiments. LY, WZ
and KS analyzed the data. LY, WZ and KS drafted the manuscript. All
authors agreed to be accountable for all aspects of the work. LY,
WZ and KS confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved from the Ethics
Committee of Jiangsu Taizhou People's Hospital (Taizhou, China) and
informed consent was obtained from the participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
DeSantis CE, Miller KD, Goding Sauer A,
Jemal A and Siegel RL: Cancer statistics for African Americans,
2019. CA Cancer J Clin. 69:211–233. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bergin ART and Loi S: Triple-negative
breast cancer: Recent treatment advances. F1000Res.
8(1342)2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Lyons TG and Traina TA: Emerging novel
therapeutics in triple-negative breast cancer. Adv Exp Med Biol.
1152:377–399. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Torre LA, Siegel RL, Ward EM and Jemal A:
Global cancer incidence and mortality rates and trends-an update.
Cancer Epidemiol Biomarkers Prev. 25:16–27. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hombach S and Kretz M: Non-coding RNAs:
Classification, biology and functioning. Adv Exp Med Biol.
937:3–17. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Riva P, Ratti A and Venturin M: The long
non-coding RNAs in neurodegenerative diseases: Novel mechanisms of
pathogenesis. Curr Alzheimer Res. 13:1219–1231. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Chu C, Quinn J and Chang HY: Chromatin
isolation by RNA purification (ChIRP). J Vis Exp.
61(e3912)2012.PubMed/NCBI View
Article : Google Scholar
|
|
9
|
Mercer TR and Mattick JS: Structure and
function of long noncoding RNAs in epigenetic regulation. Nat
Struct Mol Biol. 20:300–307. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Ju C, Liu R, Zhang YW, Zhang Y, Zhou R,
Sun J, Lv XB and Zhang Z: Mesenchymal stem cell-associated lncRNA
in osteogenic differentiation. Biomed Pharmacother.
115(108912)2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Bhan A, Soleimani M and Mandal SS: Long
noncoding RNA and cancer: A new paradigm. Cancer Res. 77:3965–3981.
2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zheng D, Chen D, Lin F, Wang X, Lu L, Luo
S, Chen J and Xu X: LncRNA NNT-AS1 promote glioma cell
proliferation and metastases through miR-494-3p/PRMT1 axis. Cell
Cycle. 19:1621–1631. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Li Y and Jiang A: ST8SIA6-AS1 promotes
hepatocellular carcinoma by absorbing miR-5195-3p to regulate
HOXB6. Cancer Biol Ther. 21:647–655. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Liu T, Wang X, Zhai J, Wang Q and Zhang B:
Long noncoding RNA UCA1 facilitates endometrial cancer development
by regulating KLF5 and RXFP1 gene expressions. Cancer Biother
Radiopharm: May 15, 2020 (Epub ahead of print).
|
|
15
|
Liu S, Yang N, Wang L, Wei B, Chen J and
Gao Y: lncRNA SNHG11 promotes lung cancer cell proliferation and
migration via activation of Wnt/β-catenin signaling pathway. J Cell
Physiol. 235:7541–7553. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bicak M, Wang X, Gao X, Xu X, Väänänen RM,
Taimen P, Lilja H, Pettersson K and Klein RJ: Prostate cancer risk
SNP rs10993994 is a trans-eQTL for SNHG11 mediated through MSMB.
Hum Mol Genet. 29:1581–1591. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Huang W, Huang F, Lei Z and Luo H: LncRNA
SNHG11 promotes proliferation, migration, apoptosis, and autophagy
by regulating hsa-miR-184/AGO2 in HCC. Onco Targets Ther.
13:413–421. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Simonson B and Das S: MicroRNA
therapeutics: The next magic bullet? Mini Rev Med Chem. 15:467–474.
2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Morales S, Monzo M and Navarro A:
Epigenetic regulation mechanisms of microRNA expression. Biomol
Concepts. 8:203–212. 2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jámbor I, Szabó K, Zeher M and Papp G: The
importance of microRNAs in the development of systemic autoimmune
disorders. Orv Hetil. 160:563–572. 2019.PubMed/NCBI View Article : Google Scholar : (In Hu).
|
|
21
|
Yan K, Hou L, Liu T, Jiao W, Ma Q, Fang Z,
Zhang S, Song D, Liu J, Gao X and Fan Y: lncRNA OGFRP1 functions as
a ceRNA to promote the progression of prostate cancer by regulating
SARM1 level via miR-124-3p. Aging (Albany NY). 12:8880–8892.
2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Li Y, Liang M, Zhang Y, Yuan B, Gao W, Shi
Z and Bai J: miR-93, miR-373, and miR-17-5p negatively regulate the
expression of TBP2 in lung cancer. Front Oncol.
10(526)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cheng C, Zhang Z, Cheng F and Shao Z:
Exosomal lncRNA RAMP2-AS1 derived from chondrosarcoma cells
promotes angiogenesis through miR-2355-5p/VEGFR2 axis. Onco Targets
Ther. 13:3291–3301. 2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Chen D, Chen J, Gao J, Zhang Y, Ma Y, Wei
W and Wei Y: LncRNA DDX11-AS1 promotes bladder cancer occurrence
via protecting LAMB3 from downregulation by sponging miR-2355-5p.
Cancer Biother Radiopharm. 35:319–328. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Soudyab M, Iranpour M and Ghafouri-Fard S:
The role of long non-coding RNAs in breast cancer. Arch Iran Med.
19:508–517. 2016.PubMed/NCBI
|
|
27
|
Paraskevopoulou MD and Hatzigeorgiou AG:
Analyzing miRNA-lncRNA interactions. Methods Mol Biol.
1402:271–286. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Guil S and Esteller M: RNA-RNA
interactions in gene regulation: The coding and noncoding players.
Trends Biochem Sci. 40:248–256. 2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Karreth FA and Pandolfi PP: ceRNA
cross-talk in cancer: When ce-bling rivalries go awry. Cancer
Discov. 3:1113–1121. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Guo J, Zhang Z, Pan L and Zhou Y:
Identification of miR-758-3p as potential modulator of CBX5
expression in gastric cancer. Technol Cancer Res Treat.
17(1533033818816061)2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Pongas G, Kim MK, Min DJ, House CD, Jordan
E, Caplen N, Chakka S, Ohiri J, Kruhlak MJ and Annunziata CM: BRD4
facilitates DNA damage response and represses CBX5/Heterochromatin
protein 1 (HP1). Oncotarget. 8:51402–51415. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Smillie CL, Sirey T and Ponting CP:
Complexities of post-transcriptional regulation and the modeling of
ceRNA crosstalk. Crit Rev Biochem Mol Biol. 53:231–245.
2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Romero-Barrios N, Legascue MF, Benhamed M,
Ariel F and Crespi M: Splicing regulation by long noncoding RNAs.
Nucleic Acids Res. 46:2169–2184. 2018.PubMed/NCBI View Article : Google Scholar
|