1. Introduction
Chronic obstructive pulmonary disease (COPD) and
asthma are two common respiratory diseases with distinct
pathophysiology that share some clinical features such as cough,
shortness of breath, and wheezing, making differential diagnosis an
essential step in their management (1-3).
Despite great progress in understanding the molecular mechanisms
governing the development and evolution of these conditions, there
is still room for improvement in setting an early diagnosis and
providing effective therapy.
COPD is one of the most common causes of death, an
important chronic morbidity, and is characterized by persistent
respiratory symptoms and airflow limitation due to anomalies of the
airways and/or alveolae caused by exposure to toxic particles or
gases (1). Asthma is a treatable
and common disease that causes symptoms such as shortness of
breath, chest tightness, and wheezing (2). Even though the two diseases are
characterized by an obstructive syndrome, there are many
differences between the two entities, the most representative
consisting of the fact that COPD has less variability and is never
completely cured, while asthma shows reversibility. Some patients
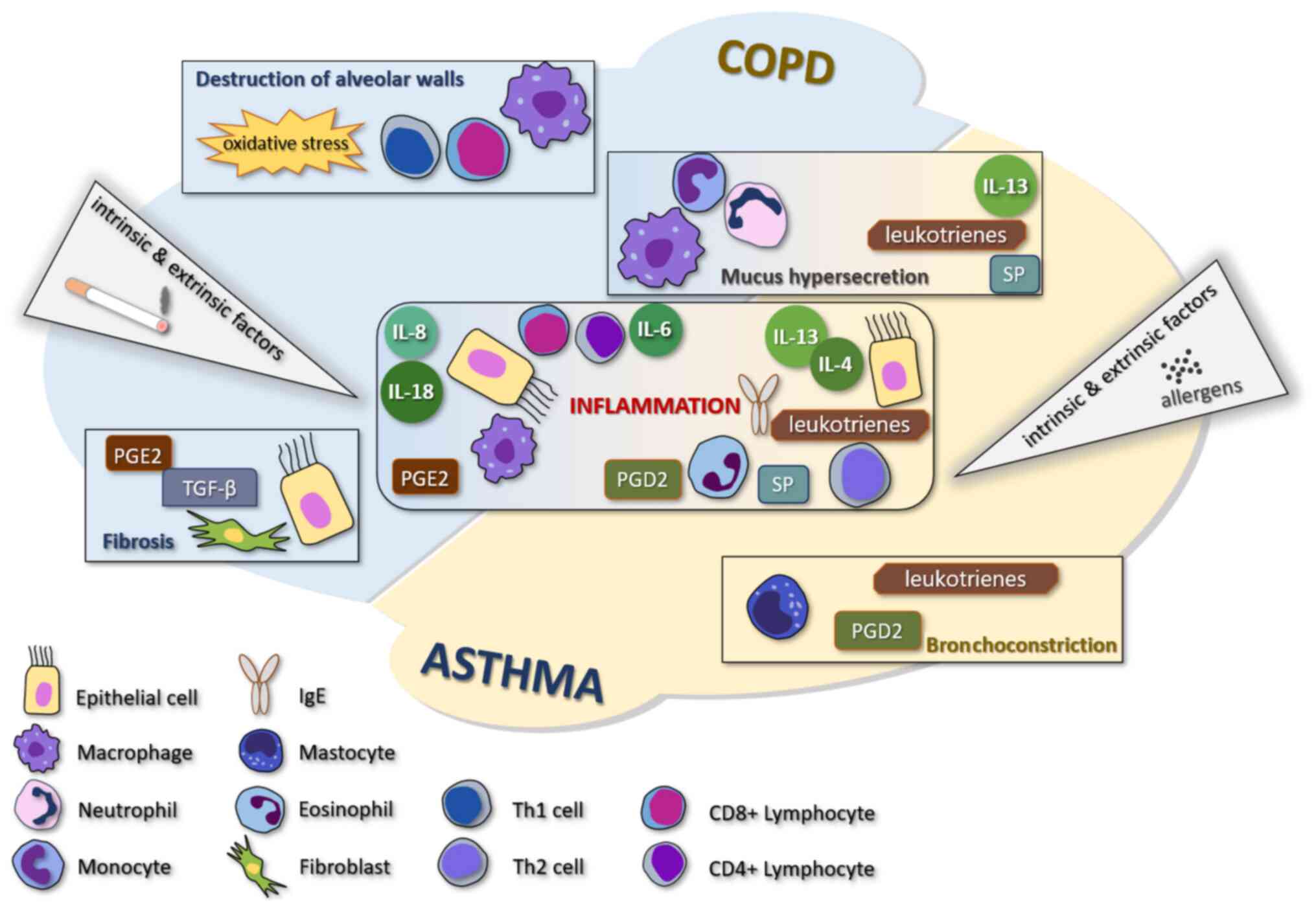
may be affected by both diseases simultaneously (3). The comparative pathogenesis of COPD
and asthma is shown in Fig. 1.
Capsaicin, the most pungent substance in chilli
peppers, is an intensely studied molecule, with many applications
in various diseases due to its anti-inflammatory and antitumoral
properties (4-7).
In the pulmonary system, capsaicin is used as an index of bronchial
hypersensitivity, being able to produce cough and sustained
bronchoconstriction, in a dose-dependent manner when inhaled
(8-10).
Transient receptor potential cation channel subfamily V member 1
(TRPV1) is the receptor for capsaicin in the human body. Capsaicin
cough challenge shows a good correlation with the presence or
absence of pathological cough (11). Capsaicin is used in many studies as
a chemical agent in the diagnosis or treatment of various
disorders, including respiratory conditions (12-15).
A better understanding of the effects of capsaicin in COPD and
asthma may reveal new ways to diagnose and differentiate these
diseases and potentially new directions of treatment.
2. Capsaicin and its receptor in the
pulmonary system
Intensely studied in various conditions and on
different experimental models, in the respiratory system, capsaicin
has demonstrated great pleomorphism in its actions and is closely
involved in triggering an abundance of signaling pathways, at times
showing converse effects in pathological situations (16). Prolonged exposure to capsaicin
aerosols such as those dispersed for crowd control may be toxic,
irritating the respiratory tract and causing nerve damage (17,18).
In extreme doses and under certain conditions, capsaicin may cause
significant respiratory symptoms such as sneezing, cough, excessive
mucus secretion, pain, and severe complications, and was
demonstrated to be lethal in certain concentrations on test animals
(18,19). Moreover, in murine models, it was
shown that the intravenous administration of capsaicin instantly
induces apnea, followed by an increase in the respiratory rate
(20). These acute effects could be
reduced by vagotomy, but not in all situations (20,21).
However, in a clinical setting, when studying the beneficial
effects of capsaicin in respiratory conditions, the doses of
inhaled capsaicin are far too low to trigger significant adverse
effects (22-24).
While capable of inducing direct effects, most of
capsaicin's actions are mediated through its receptor, TRPV1. TRPV1
is a non-selective receptor that structurally belongs to the TRP
family of ion channels. Besides capsaicin, it may be activated by
different factors such as high temperature, acidity (pH <6.0),
endocannabinoids, endogenous lipids, and other potential
activators, such as numerous mediators of inflammation or various
neurotransmitters (25,26). The receptor activation sends
impulses to the spinal cord and brain producing a variety of
effects, such as sensations of burning, stinging, itching, warming,
or tingling. The terminations of the capsaicin-sensitive nerves
include numerous neuropeptides, for example, substance P (SP) or
calcitonin gene-related peptide (CGRP). Their activation is
followed by a temporary inflammatory process known as neurogenic
inflammation because of the local release of pro-inflammatory
peptides (27-30).
Even though the number of TRPV1 receptors in the respiratory tract
is not as high as in the other regions of the body (31), they can be found in all organs and
structures of the respiratory system (32). Various pathogenic processes may
influence the distribution of receptors, as was revealed in
patients with emphysema which show higher levels of TRPV1 receptors
in the respiratory system compared with healthy subjects (33,34).
TRPV1 receptors are mainly expressed in lung C-fiber afferents
(35) generally recognized as
fibers with polymodal sensitivity, which originate from nociceptive
neurons (36). Most C-fibers are
receptive to capsaicin, which acts as an important respiratory
irritant (37). TRPV1 was also
identified in bronchial epithelial cells (28). Alongside TRPV1, the Transient
Receptor Potential Ankyrin 1 (TRPA1) receptor was revealed as being
co-expressed in the airways in a population of C-fibers, and it was
shown to be permeable to calcium ions (38). Although not directly stimulated by
capsaicin, TRPA1 may be activated by various natural products
(39), but also may be sensitized
through inflammatory signaling pathways that also involve TRPV1,
potentially contributing to increased chemical sensitivity
(38,40,41).
TRPV1 may be activated by various ligands, including
derivates of ployunsaturated fatty acids, oxytocin,
neurotransmitters, chalcone derivatives, and cannabinoids (42-46).
Cannabinoids are of particular interest, as they have demonstrated
some similarities to capsaicin in regard to their anti-inflammatory
and anti-tumoral effects in various organs, albeit some of these
are mediated by specific receptors (47). Cannabinoids are not able to induce
similar channel states as capsaicin on TRPV1 but they manage to
target the receptor and can interact with other receptors from the
TRP family, as well, which emphasises the potential interaction and
synergic effects of these substances (48). Cannabinoids exert a series of TRPV1
effects and the modulation of the endocannabinoid system has proven
extremely important in managing a variety of disorders affecting
the central nervous system as well as conditions with intestinal,
pulmonary, and cutaneous locations, a virtual structure termed
‘gut-lung-skin axis’ (49-52).
The activation of TRPV1 has demonstrated a variety
of effects (53). Several studies
have shown that TRPV1 agonists may cause apoptosis of human lung
cells in alveolar epithelial cells (54,55).
The inhalation of capsaicinoids for 30 min in rats causes an
inflammatory reaction of the airways, destruction of epithelial
cells of the trachea and nasal cavity, and injury to the
bronchiolar and alveolar cells (54). Furthermore, an in vivo murine
study has shown that a TRPV1 antagonism reduces the destruction of
epithelial cells, preventing apoptosis (56). One of the frequently studied TRPV1
antagonists is capsazepine. Capsazepine is a specific antagonist of
capsaicin-induced C-fiber activation and has been used to uncover
additional roles of the TRPV1 receptor, specifically, its
involvement in the onset of clinical respiratory symptoms (57-59).
TRPV1 may mediate cough (60) and bronchoconstriction, and the use
of capsazepine reduces these symptoms in vivo (61). Furthermore, two additional TRPV1
antagonists demonstrated similar effects in inhibiting acid-induced
cough in guinea pigs. These antagonists have similar effects and
efficacy to that of codeine (62).
Within the respiratory system, identical signaling
pathways regulate the onset of cough, bronchoconstriction, and
airway narrowing, while also enhancing the sensation of irritation
as well as fluid secretion. Stimulation of airway neurons may have
favorable or unfavorable effects. It was reported that it might
contribute to airway protection, disposing of chemical irritants
and pathogens that cause infections, while preserving and
initiating tissue recovery and favoring the immune responses in
murine models (63,64). However, the stimulation of airway
neurons may cause inflammation in the respiratory airways that
complicates underlying diseases, as was demonstrated on TRPV1
neurons in mouse models of asthma (65). A recent study by Baral et al
indicates that pulmonary TRPV1 neurons are involved in cross-talk
with immune cells via CGRP, SP, glutamate, and other signaling
molecules, showing that these neurons may cause neutrophil
depletion as well as cytokine and T-cell release impairment
(66). These converse findings
reinforce the need to further study the cascade of intricate
effects triggered by TRPV1 activation and to develop novel models
capable of properly translating the in vivo actions of
capsaicin. In some respiratory diseases, a variety of
pro-inflammatory mediators and peptides are involved, such as
histamine, prostaglandins, cysteinyl leukotrienes, proteases,
growth factors, and bradykinin (64,67).
Bradykinin is a pro-inflammatory molecule, acting through B1 and B2
receptors found in the respiratory system, which can also be
involved in neuroinflammation associated with an increase in SP and
CGRP (64). Bradykinin causes cough
and bronchoconstriction (67,68)
and is involved in airway chronic inflammation, responsiveness, and
remodeling through activation of a variety of cells that cause
these unfavorable effects (69).
Capsaicin and the major clinical
respiratory symptoms
Inhaled capsaicin is the main agent for the
measurement of cough reflex sensitivity because of a lack of side
effects when properly administered, low price, and good correlation
with the presence or absence of pathological cough. A review from
2005 that contained 122 published studies (1984-2005) on 4.833
subjects, including healthy subjects, patients with COPD, asthma,
and other diagnoses, did not manage to isolate a single serious
adverse effect of inhaled capsaicin in controlled conditions when
using regulated concentrations (11). The usual symptoms reported during
capsaicin cough challenge are increased cough, rhinorrhea, and
throat and eye irritation (70).
Asthmatics without cough could not be differentiated
from healthy individuals after the capsaicin cough challenge.
Moreover, it was demonstrated that hyperresponsiveness of airways
and cough were mediated through different neural pathways (71).
In vitro research using fiberoptic
bronchoscopy in order to obtain mucosal biopsies from 29 patients
with chronic cough showed an increase in the number of TRPV1
receptors in these subjects compared to 16 controls. Those data
demonstrate a correlation between chronic cough and TRPV1
receptors. The cause of the increase was not determined. The
subjects also received aerosols of a capsaicin solution dissolved
in 0.9% sodium chloride until cough was produced five or more
times. Results suggest an increased frequency of cough when
capsaicin was inhaled by patients with chronic cough (72). In addition, cold air seems to
increase the sensitivity of TRPV1 to capsaicin and increase cough
sensitivity (73).
In an in vivo study on guinea pigs, the
delivery of capsaicin by aerosol to the airways induced cough,
while the administration of a capsaicin antagonist caused a
decrease in the induced cough (74). The antagonist for capsaicin used in
that study completely blocked the receptor for capsaicin and
prevented its response to the variation of pH. Additionally, it
inhibited the influx of Ca2+ that blocks the effects of
capsaicin. This is paramount evidence of the major role of
capsaicin in cough and is an important finding for basing future
human trials. In terms of the action mechanism, it appears that the
effects of capsaicin were carried on by direct TRPV1 effects but
also mediated by tachykinins such as SP and neurokinin A (NKA)
(74).
Long-term respiratory effects after exposure to
capsaicin aerosols were analyzed in several major studies. Two
studies showed no difference between hot pepper workers and healthy
individuals in regard to their pulmonary function (75,76).
In vitro and in vivo studies suggested
that capsaicin can be mutagenic; conversely, multiple studies
revealed that topical, dietary, or injected capsaicin may
demonstrate a chemoprotective effect (77-80).
Two decades of experience with capsaicin
demonstrated that the capsaicin cough challenge is a safe
investigation, and this procedure may prove to be an extremely
important tool for future research.
The effects of capsaicin on mucus secretion in COPD
and asthma were also investigated. In vitro, findings of
several studies showed that SP stimulates mucus secretion in the
respiratory system (81-83)
and an increase of SP appears after stimulation of sensory nerves
by capsaicin (84). In an in
vivo study by Karmouty-Quintana et al increased mucus
production caused by the activation of airway sensory nerves with
intratracheal administered capsaicin was observed, and the results
were confirmed showing a reduced level of mucin concentration after
administration of capsazepine, a TRPV-1 antagonist. These effects
seem to be mostly determined by SP, CGRP, and NKA released as a
response to sensory nerve stimulation by capsaicin (85).
Dyspnea is a common respiratory symptom in both COPD
and asthma, however, it has different attributes. In COPD, dyspnea
is progressive and proportional to the airflow obstruction, while
in asthma it appears simultaneously with the transitory
bronchoconstriction (86). Dyspnea
is a symptom that appears after the stimulation of adenosine
receptors, and capsaicin shows no interference with these receptors
(87). When investigated in
clinical applications, no evidence that capsaicin may cause dyspnea
was found, neither inhaled nor administered intravenously, in
tolerable doses (88,89).
Smoking is a major causative and aggravating factor
in lung inflammation. However, acute and chronic infection, whether
viral, bacterial, or fungal, may influence the prognosis of these
patients and their response to treatment (90). Some of the infections trigger
exacerbations and cause a decline in lung function, while the
patient does not benefit from effective therapeutical strategies,
which poses significant problems in the management of these
patients (91). The lung microbiome
may experience changes related to the exacerbations and may
influence biomarkers such as sputum neutrophils percentage and
IL-8, as well as serum IL-10 and MMP-7(92). Capsaicin has demonstrated
antimicrobial properties and may provide an added benefit in COPD
patients with concurring infections, a research direction which
needs to be further explored (93).
3. Capsaicin in chronic obstructive
pulmonary disease
COPD is one of the leading causes of death and an
important chronic morbidity featuring limitation of airflow, cough,
mucus hypersecretion, and dyspnea. It is caused by long-term
exposure to toxic particles or gases, usually tobacco smoke, and
may sometimes affect patients with various genetic abnormalities or
concurring respiratory diseases (94). COPD demonstrates a steady increase
in mortality and morbidity and is estimated to maintain this trend
(95). Besides the clinical
context, spirometry is necessary for the diagnosis by confirming
the airflow limitation that is established when forced expiratory
volume in one second (FEV1) to forced vital capacity (FVC) ratio is
under 70% of the normal limits after the use of a bronchodilator
(96). In their clinical evolution,
patients with COPD can have periods of time when they show no
symptoms, and periods of exacerbations (1).
Capsaicin, smoking and inflammation in
COPD
Inflammation is one of the fundamental
characteristics of COPD. It accelerates the disease progression and
it is not reversible. Inflammation in COPD is usually a consequence
of smoking, which is a major factor in the pathogenesis of COPD.
Most cigarette smokers have a chronic cough, which is usually
present prior to the onset of airflow obstruction. Smoking induces
airway inflammation causing an increase in the number of
neutrophils, macrophages, and T lymphocytes (CD8+ and
CD4+) (97). These cells
release a large number of cytokines and mediators that initiate and
maintain the inflammatory process (98). The mediators with increased
concentrations in COPD are leukotriene B4, neutrophil and T-cell
chemoattractant, chemotactic factors such as interleukin (IL)-8 and
growth-related oncogene α, pro-inflammatory cytokine (TNF)-α,
IL-1β, IL-6), and transforming growth factor-β (98,99).
Alongside the inflammatory process, an imbalance between protease
and antiprotease activity can be identified in COPD. This results
from the intensified production and activity of proteases and
decreased production and activity of antiprotease, caused by
cigarette smoke and inflammation. Neutrophils release elastase,
cathepsin G, and protease 3, while macrophages produce cysteine
protease, cathepsins E, A, L, S, and matrix metalloproteinase-8, -9
and -12. α1 antitrypsin, secretory leucoprotease inhibitor, and
tissue inhibitors of metalloproteases are the major antiproteases
that participate in emphysema in COPD (31,98,100).
The alteration of parasympathetic afferent and efferent fibers may
contribute to the onset of bronchospasm, cough, and dyspnea
(101).
In vivo, in a study on mice after exposure to
cigarette smoke, the levels of leukocyte infiltration and the high
level of inflammatory mediators caused the progression of COPD and
the decline of lung function. These processes increased the
production of IL-1β and IL-18, two cytokines released in
association with the stimulating action of TRPV1 agonists,
including capsaicin, in a cell-based model using primary human
cells (33). Moreover, TRPV1 is
found in CD4- T cells in mice. These receptors are
activated after stimulation of the T-cell antigen receptor, which
contributes to the influx of Ca2+. After this influx,
the T cells are activated playing an important role in the
development of inflammation. This process indicates that TRPV1 may
have a fundamental function in the inflammatory process,
particularly after smoke exposure, which is the main cause of COPD
(102).
Recent studies also revealed the role of TRPV1 in
mediating the effects of cigarette smoke on the alveolar epithelial
cells through the increase of inflammation, oxidative stress, and
mitochondrial damage (103,104).
In patients with COPD, TRPV1 mRNA expression is increased in
comparison with non-smokers (33).
The expression of TRPV1 is related to the intensity
of the inflammatory process induced by cigarette smoking (105). In a mouse model, Jian et al
have shown that the decreased expression of TRPV1 by using total
flavonoids is followed by a subsequent decrease in the inflammation
and oxidative stress in the lung parenchyma (106). A 2020 article has shown that
single nucleotide polymorphisms of TRPV1 are associated with a
higher risk of developing COPD in smokers (107).
In another study, human cells were exposed to
cigarette smoking, and the expression of TRPV1 in pulmonary tissue
was increased, as was the concentration of pro-inflammatory
cytokines (108).
Stimulation of TRPV1 in COPD releases inflammatory
neuropeptides which increase vascular permeability, cause
extravasation of plasma proteins, bronchoconstriction, and amplify
the concentration of mucus (109,110). Mucus hypersecretion causes
increased sputum production and seems to correlate with the
severity of COPD (111).
Interestingly, in vitro studies showed that
cigarette smoke can cause neuropeptide release by stimulating TRPA1
and acetylcholine receptors, contributing to the inflammatory
process, with decreased or lack of TRPV1 involvement (112). Conversely, in vivo murine
models suggest that the mediation of inflammation is exclusively
performed by activation TRPV1 and 4, and not by TRPA1(33). These contradictory findings require
the need for future studies, ideally on human subjects.
Capsaicin stimulates TRPV1 with further release of
pro-inflammatory cytokines in the airways. Activation of TRPV1 by
capsaicin in patients with COPD stimulates the secretion of ILs,
TNF-α, and prostaglandin E2 (PGE2) (113). Special attention was paid to the
capsaicin-induced stimulation of IL-6 production in human
respiratory epithelial cells (114). IL-6 has a very important role in
the transition from acute to chronic inflammation because it
stimulates T-cells and B-cells. This stimulation favors a chronic
inflammatory response due to the activation of endothelial cells
that release IL-8 and monocyte chemoattractant protein 1 and
activate the expression of adhesion molecules (115). Nassini et al used mouse
models and in vitro studies on human small airway epithelial
cells, fibroblasts and smooth muscle cells exposed to cigarette
smoke to demonstrate that capsaicin inhalation stimulates TRPV1
receptors in sensitive nerve fibers promoting neurogenic
inflammation and favoring the release of IL-8, most likely through
coactivation of TRPA1 receptors in non-neuronal cells (116). This mechanism may maintain and
even increase inflammation in patients with COPD, enhancing its
negative effects. Furthermore, bronchoconstriction may exacerbate
the airflow limitation and intensify the dyspnea of patients.
Studying the in vivo response of the exposure
of guinea pigs to cigarette smoke revealed that nebulized capsaicin
enhances cough production in smoke-exposed animals, through a
non-cyclo-oxygenase-mediated mechanism. The increased
responsiveness to capsaicin appears to depend on sensory nerves
containing CGRP-like substances (117). Moreover, while increasing
sensitivity to capsaicin, exposure to smoke seems to decrease the
response to PGE2, promoting the concept that sensory nerves are
affected in COPD in a disease-specific manner (118).
A consensus was not yet reached regarding the
overall effects of capsaicin in patients with airflow obstruction.
A large cross-sectional study by Blanc et al (119) compared the effects of inhaled
capsaicin on non-smokers and smokers with and without airflow
obstruction. An increase of responsiveness in all groups of
patients was demonstrated, more significantly in patients with
COPD. Asymptomatic smokers registered no complaint, despite their
hyperresponsiveness to capsaicin compared to non-smokers. In the
same study, women were more sensitive than men in all three groups
(119). However, no correlation
was identified between the cough response intensity and the degree
of airflow obstruction in COPD patients appreciated by FEV1 values,
and these findings were confirmed in another study, by Doherty
et al (120). Conversely,
research data published in 1999 showed no significant difference in
cough sensitivity to capsaicin between patients with COPD and
airflow obstruction compared to healthy controls (121).
Capsaicin and cough in COPD
TRP channels have a protective role in physiological
situations when the airways are not affected by pathological
changes. In a disorder such as COPD, this role can be altered, and
TRP channels may be responsible for the symptoms of COPD,
especially cough and they may also participate in the inflammatory
process identified in COPD (32).
Cough is usually the first symptom in patients with
COPD. Cough may be sporadic and sometimes unproductive (1), it can affect the quality of life in
patients with COPD and this is an important reason to research it
and potentially identify new therapies (122). C and Aδ fibers are expressed in
the mechanism of pathological cough, so TRP ion channels are an
important component of this process (38). Capsaicin is the most common and
usable agonist of TRPV1, used in a variety of studies on patients
with chronic cough, a category that includes patients suffering
from COPD (123,124).
Several clinical studies using capsaicin aerosols
have been developed for patients with cough and COPD. Capsaicin
responsiveness and cough in COPD was researched in a study by
Doherty et al (120). The
presented data suggest that inhaled capsaicin caused an increase in
cough in patients with COPD and no relationship between cough and
airflow limitation after exposure to capsaicin was observed
(120). Another study, by Terada
et al (125), showed an
increase in the number and frequency of exacerbations after
capsaicin inhalation in patients with COPD compared to controls,
demonstrated by lower concentrations of capsaicin needed to produce
five or more coughs; furthermore, bronchial hypersensitivity
correlated with the frequency of exacerbations and the serum
C-reactive protein, indicating that ongoing airway inflammation is
associated with hypersensitivity of the cough reflex to capsaicin
and may precipitate the exacerbations (125). Capsaicin cough challenge may be an
important aid in assessing, managing COPD and its complications,
and advancing the development of a new antitussive therapy. It does
not yield serious adverse effects as it was demonstrated in a paper
reviewing 20 years of practicing capsaicin cough challenge
(11). A cough challenge test
performed on 20 patients with exacerbated COPD revealed that their
sensitivity to capsaicin was increased compared to the repeated
test after recovery, and if hypersensitivity was maintained during
recovery this announced future exacerbations (126).
4. Capsaicin in asthma
Asthma is a chronic, frequent, and treatable
pulmonary disease characterized by respiratory symptoms, limitation
of activity, and exacerbations that occasionally need urgent
medical care, and can be a potentially lethal condition. The most
common respiratory symptoms in asthma are wheezing, shortness of
breath, cough, chest tightness, and variable expiratory airflow.
The main risk factors that may aggravate asthma are viral
infections, allergens, tobacco smoke, pollens, food, drugs, or
exercise. Spirometry is required to set the diagnosis: FEV1
increases by 12% and a minimum of 200 ml of the baseline values
post-bronchodilator (2).
Asthma is regarded as a typical Th2 disease, with
increased immunoglobulin E (IgE) levels, airway inflammation, and
the presence of numerous eosinophils. Usually, patients begin
suffering from asthma in childhood. The allergens are inhaled and
stimulate Th2-helper cell proliferation and the increase of IL-4,
IL-5, and IL-13 levels (127). A
fundamental characteristic of these patients is long-term airway
inflammation. Consequently, chronicity and disease evolution
disease may occur. The roles of IL-4 are to support B-cell isotype
swapping, increase the response of stimulus of adhesion molecules,
eotaxin creation, and improvement of airway hyperresponsiveness and
goblet cell metaplasia (128-130).
IL-13 partly shares its receptor with IL-4 and plays a critical
role in the pathophysiology of asthma by increasing mucus secretion
and modulating the functions of epithelial cells (131). Eosinophils and IgE are also of
great importance in asthma and act via distinctive pathways which
do not interfere with the mechanisms of IL-13 (132,133).
TRPV1 and allergens in asthma
TRPV1 may play important roles in the modulation of
the pathogenic changes occurring in asthma (105). The expression of TRPV1 and Th2
levels seems to correlate with the asthmatic debut in the pediatric
population (134). Recent data
showed that TRPV1 can mediate the response of epithelial cells to
allergens, increasing IL-33 secretion and the activation of dual
oxidase 1 and epidermal growth factor receptor (135). Furthermore, an in vivo
study on mice published in 2020 has shown that TRPV1 stimulates the
production of mucus and cytokines in asthma by regulating the
expression of MUC5AC and nuclear factor kappa-light-chain-enhancer
of activated B-cell pathway, with probable involvement of
neuropeptides SP and CGRP (136).
TRPV1 also mediates the appearance of cough via a neuronal
mechanism and shows increased expression after exposure to
allergens (137,138). Although expressed on airway smooth
muscle cells, TRPV1 activation does not significantly contribute to
the initiation of bronchoconstriction (139). In vitro studies have shown
that coal fly ash causes TRPV1 activation and worsens asthma
symptom control (140). A study on
ovalbumin (OVA)-induced asthmatic mice showed that exposure to
nanoparticles causes neuroinflammation mediated through TRPV1 and
TRPV4, and is accompanied by an increase in SP, CGRP, and
bradykinin (64). A similar study
showed that a pollutant known as trimellitic anhydride can increase
TRPV1 expression as well as amplify the levels of IL-13, SP,
prostaglandin D2, and nerve growth factor in the lungs of OVA
asthmatic mice (141). In the same
experimental model, Li et al identified ozone as an
environmental pollutant with similar effects on TRPV1 and the
inflammation pattern in asthma as the allergens mentioned above
(142). Small particulate matter
can also inflict bronchial mucosal damage and thickening of
bronchial smooth muscles in asthmatic mice (143). Combining pollutants builds a model
closer to real-life situations (144), and, by doing so on allergic Balb/c
mice, activation of TRPV1 signaling and increases of CGRP and SP
levels were observed contributing to the neurogenic inflammation of
asthma (145). Allergen exposure
may lead to pathological changes outside the respiratory tract. In
an in vivo study, Spaziano et al (146) showed that sensitization of the
nucleus solitary tract (NST) occurs following exposure to
allergens, and this is a basis for increased airway sensitivity.
When capsaicin was inhaled, an increase in the neural firings of
the NST were identified. However, TRPV1 may play a complex role in
modulating excitation as its activation by endocannabinoids may
stimulate glutamatergic signaling and alter the bronchoconstrictive
reflex (146).
These observations were demonstrated by studies on
the same animal model showing that inhibition of the TRPV1 mRNA and
protein expression using various antagonists including capsazepine
caused an improvement in pulmonary function, decreased airway
hyperresponsiveness, and reduced cytokine concentrations in
aggravated asthma (145,147-149).
In addition, the use of allergens to induce bronchoconstriction
seems to increase the TRPV1 response to capsaicin, increasing cough
reflex sensitivity, as demonstrated in a recent clinical trial
(150). The effects of stimulating
TRPV1 receptors with capsaicin are increased in mice with atopic
dermatitis to the extent that asthmatic-like inflammation of the
airways is produced while compliance of the lungs is decreased
(151).
In an in vitro study, by McGarvey et
al, the TRPV1 protein was found in a culture with primary
bronchial epithelial cells through patch-clamp experiments. That
study confirmed that capsaicin induces the release of IL-8
especially in patients with chronic airway inflammation (152).
Capsaicin and inflammation in
asthma
In asthma, chronic inflammation is one of the
fundamental features of the disease. Inflammation progresses when
inflammatory cells interact with local cells to create a cascade of
events that triggers and maintains chronic inflammation and causes
clinical symptoms. The consequences of inflammation in asthma are
bronchospasm, airways mucus secretion and edema,
bronchoconstriction, and bronchial epithelial damage (153).
The role of capsaicin in the process of inflammation
in asthma is unclear, as some studies cite pro-inflammatory
properties of capsaicin, while other recent studies revealed its
anti-inflammatory effects (154).
However, TRPV1 activation seems to play an important
role in the inflammatory cascade of asthma, and pharmacological
inhibition of TRPV1 leads to a reduction in IgE levels as well as
an attenuation of airway inflammation in mice (155).
In vitro, after using a TRPV1 antagonist,
inflammation in the airway tissues of patients with chronic asthma
was attenuated. These results may suggest that blocking TRPV1 may
be a new direction for the anti-inflammatory treatment in asthma
(156).
A study by Rehman et al showed in vivo
that blocking TRPV1 in a murine model attenuates the symptomatology
of asthma, probably by alleviating the inflammation of the airways.
TRPV1 inhibition reduced the concentration of IL-13 and its effects
on inflammation in the airways. Consequently, hyperresponsiveness
and inflammation were reduced (157). Conversely, a different murine
study revealed that inhibition of the TRPV1 gene may increase
airway inflammation. The levels of the IgE, eosinophils, and IL-4
may be increased in the bronchoalveolar lavage fluid in this case.
The authors revealed that the effects achieved by TRPV1 employ
multiple mechanisms, both direct and mediated by SP, CGRP, NkA, and
somatostatin (158).
Capsaicin and cough in asthma
Cough is a frequent and important symptom that
influences the quality of life in patients with asthma (159) and is regulated by sensory nerves
in the airways (60,160). In the previously mentioned in
vitro study by McGarvey et al, the expression of TRPV1
in bronchial biopsies from asthmatics refractory to corticotherapy
was found to be higher than that in patients without asthma or in
those with asthma that were responsive to corticoids (152). Those findings were supported by
Chen et al by analyzing TRPV1 mRNA in the peripheral blood
of asthmatics and concluding that TRPV1 expression levels are major
factors for bronchial asthma in children (161). As mentioned before, cold air seems
to increase the effects of capsaicin on TRPV1, but humified warm
air has been shown to trigger cough and bronchoconstriction in mild
asthmatic patients via increased activation of C-fibers (162). While spirometry is useful in
investigating the response of the large airways to capsaicin,
impulse oscillometry system has proven more sensitive in detecting
peripheral airway function in asthmatics and the changes induced by
capsaicin (163).
In a study performed in vivo on asthmatic
mice with cough, the inhalation of capsaicin caused a more frequent
cough and was accompanied by eosinophil infiltration detected in
the bronchoalveolar lavage fluid (164). There are also data showing
neutrophil infiltration in the submucosal layer in asthmatic rats
after the capsaicin cough challenge, an effect of both direct TRPV1
action as well as due to the release of neuropeptides (SP and CGRP)
inducing neurogenic inflammation (165). A study on guinea pigs sensitized
with capsaicin showed that the rate of coughs was notably
increased, and the proposed mechanism was associated with airway
tract eosinophilic inflammation (166).
Previous findings showed an increase in the
frequency of cough in patients with asthma after inhalation of
capsaicin. This is an effect of the hyperresponsiveness that
characterizes patients with asthma. The mechanism probably involves
neuronal dysfunction. When capsaicin stimulates the TRPV1 receptor,
inflammatory mediators are released with further increased
stimulation of the nerve fibers. This process determines membrane
depolarization and release of the inflammatory mediators which are
in high concentration in asthmatic patients (167). This is a possible explanation of
why increased sensitivity to capsaicin has been identified as a
risk factor for severe forms of asthma (168).
A study from 2019 comparing asthmatics and healthy
controls showed no difference in the cough threshold after inhaled
capsaicin between the two groups (163). However, in patients with asthma,
the frequency of cough is higher than in healthy subjects. In
addition, a higher sensitivity to capsaicin was identified in women
and older patients (169). In
asthmatic children, there is a decreased sensitivity to capsaicin
compared to controls, which seems to be mediated by
neurotransmitters released from parasympathetic neurons (170). This finding is strengthened by
another recent study showing that some nervous phenotypes may
induce excessive coughing in asthmatic patients due to a neuronal
dysfunction (137,171). Capsaicin cough challenge is more
sensitive in patients with cough-variant asthma even if
bronchodilators were used in these patients (120). These patients have a lower quality
of life because of their frequent exposure to irritants in daily
life and due to their permanent discomfort. Research data have
shown a direct correlation between the quality of life and
sensitivity to capsaicin, as asthmatic patients with hyperactivity
to inhaled capsaicin have a significantly poorer quality of life
than controls (172,173).
A study published in 2020 tested the effects of
inhaled capsaicin on 385 chronic cough patients, revealing that the
capsaicin cough challenge is a proper method for investigating
patients with variable clinical factors in asthma (174). Additionally, the test is a safe
method to employ in severe asthma (175).
However, in regard to therapeutic prognostic, cough
sensitivity to capsaicin may hold an important role in predicting
the response to bronchial thermoplasty when used for treating
patients with severe asthma (176). Alongside the different diagnostic
benefits cited in asthma, capsaicin is a molecule gaining attention
and is increasingly studied in animal models and human trials.
The intended finality of these findings is to
improve the management of asthmatic cough. The use of the
antimuscarinic bronchodilator Tiotropium has proved effective in
controlling asthmatic cough in patients unresponsive to
corticosteroids and long-acting β2 agonists, and it improved
capsaicin cough reflex sensitivity, leading to the conclusion that
its effects are mediated through sensory nerves, rather than
effective bronchoconstrictors (177).
In summary, capsaicin demonstrates complex effects
on cough and inflammation in COPD and asthma, either through direct
TRPV1 activity or mediated by released factors, and these findings
were summarized in Table I.
 | Table IComparison of capsaicin effects on
cough and inflammation in COPD and asthma. |
Table I
Comparison of capsaicin effects on
cough and inflammation in COPD and asthma.
| Component | Effect | Study type | (Refs.) |
|---|
| COPD | | | |
| Cough | Increase in
frequency | In vitro
(mucosal cells) | (60) |
| | Increase in
frequency | In vivo
(guinea pigs) | (62) |
| | Increase in
frequency | Trial | (104,109) |
| | Rise of
exacerbation incidence | | (109) |
| Inflammation | Release of IL-1α,
TNF-α and PGE2 | In vitro
(human primary bronchial fibroblasts) | (97) |
| | Release IL-8 and
pro-inflammatory cytokines | In vitro
(primary bronchial epithelia cells) | (92,136) |
| | Release IL-1β and
IL-18 | | |
| | Maintain
inflammation | In vivo
(mice) | (32) |
| Asthma | | | |
| Cough | Increase in
frequency | In vitro
(bronchial cells) | (136) |
| | Increase in
frequency | In vivo
(guinea pigs) | (149,150) |
| | Increase in
frequency | Trial | (151,153) |
| Inflammation |
Pro-inflammatory | In vitro
(bronchial cells) | (140) |
| | Eosinophil
infiltration | In vivo
(guinea pigs) | (150) |
| | Pro- and
anti-inflammatory | In vivo
(mice) | (141,142) |
5. Conclusions
Capsaicin may exhibit a variety of clinical and
paraclinical effects in COPD and asthma. Some are similar in both
diseases, while others may be significantly different or opposite.
In many cited studies, the frequency and intensity of cough are
increased after capsaicin inhalation in COPD, while other authors
report only an increase in the frequency of cough in asthmatic
patients. The effects of capsaicin on inflammation in these two
diseases are different. In COPD, several studies showed that
capsaicin has pro-inflammatory effects, while, in asthma, the role
of capsaicin in inflammation is unclear, as various studies showed
conflicting results, citing pro-inflammatory as well as
anti-inflammatory effects. Most authors revealed that the
hyperresponsiveness to capsaicin is higher in smokers with airflow
obstruction than non-smokers and smokers without airflow
obstruction. Capsaicin appears to be a safe product as we failed to
identify any studies showing an increase of dyspnea in COPD or
asthma after capsaicin administration, when used in tolerable
doses. Capsaicin may be a very promising, cost-effective, natural,
and safe tool in expediting the diagnosis of COPD and asthma in the
future, with increased accuracy in selected cases, especially due
to its effects on cough and inflammation.
Acknowledgements
Not applicable.
Funding
Funding: This research and APC were funded by the following
grants: PN.19.29.01.01/2019, PN-III-P1-1.2-PCCDI-2017-0341 and
PN-III-P1-1.2-PCCDI-2017-0782.
Availability of data and materials
Not applicable.
Authors' contributions
MDD, CS, and CCa conceived and designed the review.
CS and CCa have developed the methodology and scientific approach.
Preliminary documentation, data selection and analysis, writing and
editing of the original draft were performed by MDD, ASJ, CS, IAB,
GDAP, AC, DOC, RSC, CCo, MN, and CCa. Content review and editing
were performed by CS, AC, and CCa. Supervision was conducted by
IAB, CCo, MN, and CCa. All authors have read and agreed to the
published version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare no conflict of interest.
References
|
1
|
Global Initiative for Chronic Obstructive
Lung Disease: Pocket Guide to COPD Diagnosis, Management and
Prevention: A Guide for Health Care Professionals. Independently
published, November 20, 2019.
|
|
2
|
Global Initiative for Asthma: Pocket Guide
for Asthma Management: For Adults and Children over 5 years.
Independently published, April 1, 2020.
|
|
3
|
Collard CD: Harrison's Pulmonary and
Critical Care Medicine. Tex Heart Inst J. 37(736)2010.
|
|
4
|
Popescu GD, Scheau C, Badarau IA,
Dumitrache MD, Caruntu A, Scheau AE, Costache DO, Costache RS,
Constantin C, Neagu M, et al: The effects of capsaicin on
gastrointestinal cancers. Molecules. 26(26)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Scheau C, Mihai LG, Bădărău IA and Caruntu
C: Emerging applications of some important natural compounds in the
field of oncology. Farmacia. 68:984–991. 2020.
|
|
6
|
Căruntu C and Boda D: Evaluation through
in vivo reflectance confocal microscopy of the cutaneous neurogenic
inflammatory reaction induced by capsaicin in human subjects. J
Biomed Opt. 17(085003)2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Frydas S, Varvara G, Murmura G, Saggini A,
Caraffa A, Antinolfi P, Tete' S, Tripodi D, Conti F, Cianchetti E,
et al: Impact of capsaicin on mast cell inflammation. Int J
Immunopathol Pharmacol. 26:597–600. 2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Fuller RW, Dixon CM and Barnes PJ:
Bronchoconstrictor response to inhaled capsaicin in humans. J Appl
Physiol (1985). 58:1080–1084. 1985.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Midgren B, Hansson L, Karlsson JA,
Simonsson BG and Persson CG: Capsaicin-induced cough in humans. Am
Rev Respir Dis. 146:347–351. 1992.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Forsberg K, Karlsson JA, Theodorsson E,
Lundberg JM and Persson CG: Cough and bronchoconstriction mediated
by capsaicin-sensitive sensory neurons in the guinea-pig. Pulm
Pharmacol. 1:33–39. 1988.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dicpinigaitis PV and Alva RV: Safety of
capsaicin cough challenge testing. Chest. 128:196–202.
2005.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Caruntu C, Negrei C, Ghita MA, Caruntu A
and Ba AI: Capsaicin, a hot topic in skin pharmacology and
physiology. Farmacia. 63:487–491. 2015.
|
|
13
|
Cavaliere C, Masieri S and Cavaliere F:
Therapeutic applications of capsaicin in upper airways. Curr Drug
Targets. 19:1166–1176. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ternesten-Hasséus E, Johansson EL and
Millqvist E: Cough reduction using capsaicin. Respir Med.
109:27–37. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Fattori V, Hohmann MS, Rossaneis AC,
Pinho-Ribeiro FA and Verri WA: Capsaicin: Current understanding of
its mechanisms and therapy of pain and other pre-clinical and
clinical uses. Molecules. 21(21)2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ilie MA, Caruntu C, Tampa M, Georgescu SR,
Matei C, Negrei C, Ion RM, Constantin C, Neagu M and Boda D:
Capsaicin: Physicochemical properties, cutaneous reactions and
potential applications in painful and inflammatory conditions. Exp
Ther Med. 18:916–925. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Copeland S and Nugent K: Persistent
respiratory symptoms following prolonged capsaicin exposure. Int J
Occup Environ Med. 4:211–215. 2013.PubMed/NCBI
|
|
18
|
Satpute RM, Kushwaha PK, Nagar DP and Rao
PV: Comparative safety evaluation of riot control agents of
synthetic and natural origin. Inhal Toxicol. 30:89–97.
2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Steffee CH, Lantz PE, Flannagan LM,
Thompson RL and Jason DR: Oleoresin capsicum (pepper) spray and
‘in-custody deaths’. Am J Forensic Med Pathol. 16:185–192.
1995.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Hedner J, Hedner T and Jonason J:
Capsaicin and regulation of respiration: Interaction with central
substance P. mechanisms. J Neural Transm (Vienna). 61:239–252.
1985.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kaczyńska K and Szereda-Przestaszewska M:
Respiratory effects of capsaicin occur beyond the lung vagi in
anaesthetized rats. Acta Neurobiol Exp (Warsz). 60:159–165.
2000.PubMed/NCBI
|
|
22
|
Kumar P, Deb U and Kaushik MP: Evaluation
of oleoresin capsicum of Capsicum frutescenes var. Nagahari
containing various percentages of capsaicinoids following
inhalation as an active ingredient for tear gas munitions. Inhal
Toxicol. 24:659–666. 2012.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Dicpinigaitis PV: Review: Effect of drugs
on human cough reflex sensitivity to inhaled capsaicin. Cough.
8(10)2012.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Costanzo MT, Yost RA and Davenport PW:
Standardized method for solubility and storage of capsaicin-based
solutions for cough induction. Cough. 10(6)2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tominaga M and Tominaga T: Structure and
function of TRPV1. Pflugers Arch. 451:143–150. 2005.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Filippi A, Caruntu C, Gheorghe RO, Deftu
A, Amuzescu B and Ristoiu V: Catecholamines reduce transient
receptor potential vanilloid type 1 desensitization in cultured
dorsal root ganglia neurons. J Physiol Pharmacol. 67:843–850.
2016.PubMed/NCBI
|
|
27
|
Holzer P: Local effector functions of
capsaicin-sensitive sensory nerve endings: Involvement of
tachykinins, calcitonin gene-related peptide and other
neuropeptides. Neuroscience. 24:739–768. 1988.PubMed/NCBI View Article : Google Scholar
|
|
28
|
De Logu F, Patacchini R, Fontana G and
Geppetti P: TRP functions in the broncho-pulmonary system. Semin
Immunopathol. 38:321–329. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Grigore O, Mihailescu AI, Solomon I, Boda
D and Caruntu C: Role of stress in modulation of skin neurogenic
inflammation. Exp Ther Med. 17:997–1003. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ghiţă MA, Căruntu C, Rosca AE, Căruntu A,
Moraru L, Constantin C, Neagu M and Boda D: Real-time investigation
of skin blood flow changes induced by topical capsaicin. Acta
Dermatovenerol Croat. 25:223–227. 2017.PubMed/NCBI
|
|
31
|
Kunert-Keil C, Bisping F, Krüger J and
Brinkmeier H: Tissue-specific expression of TRP channel genes in
the mouse and its variation in three different mouse strains. BMC
Genomics. 7(159)2006.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Grace MS, Baxter M, Dubuis E, Birrell MA
and Belvisi MG: Transient receptor potential (TRP) channels in the
airway: Role in airway disease. Br J Pharmacol. 171:2593–2607.
2014.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Baxter M, Eltom S, Dekkak B, Yew-Booth L,
Dubuis ED, Maher SA, Belvisi MG and Birrell MA: Role of transient
receptor potential and pannexin channels in cigarette
smoke-triggered ATP release in the lung. Thorax. 69:1080–1089.
2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Belvisi MG and Birrell MA: The emerging
role of transient receptor potential channels in chronic lung
disease. Eur Respir J. 50(1601357)2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Fisher JT: The TRPV1 ion channel:
Implications for respiratory sensation and dyspnea. Respir Physiol
Neurobiol. 167:45–52. 2009.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Gu QD, Joe DS and Gilbert CA: Activation
of bitter taste receptors in pulmonary nociceptors sensitizes TRPV1
channels through the PLC and PKC signaling pathway. Am J Physiol
Lung Cell Mol Physiol. 312:L326–L333. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Alamri A, Bron R, Brock JA and Ivanusic
JJ: Transient receptor potential cation channel subfamily V member
1 expressing corneal sensory neurons can be subdivided into at
least three subpopulations. Front Neuroanat. 9(71)2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Bessac BF and Jordt SE: Breathtaking TRP
channels: TRPA1 and TRPV1 in airway chemosensation and reflex
control. Physiology (Bethesda). 23:360–370. 2008.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Bandell M, Story GM, Hwang SW, Viswanath
V, Eid SR, Petrus MJ, Earley TJ and Patapoutian A: Noxious cold ion
channel TRPA1 is activated by pungent compounds and bradykinin.
Neuron. 41:849–857. 2004.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Millqvist E, Bende M and Löwhagen O:
Sensory hyperreactivity--a possible mechanism underlying cough and
asthma-like symptoms. Allergy. 53:1208–1212. 1998.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Brooks SM: Irritant-induced chronic cough:
Irritant-induced TRPpathy. Lung. 186 (Suppl 1):S88–S93.
2008.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Benso B, Bustos D, Zarraga MO, Gonzalez W,
Caballero J and Brauchi S: Chalcone derivatives as non-canonical
ligands of TRPV1. Int J Biochem Cell Biol. 112:18–23.
2019.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Benítez-Angeles M, Morales-Lázaro SL,
Juárez-González E and Rosenbaum T: TRPV1: Structure, endogenous
agonists, and mechanisms. Int J Mol Sci. 21(21)2020.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Szallasi A and Sheta M: Targeting TRPV1
for pain relief: Limits, losers and laurels. Expert Opin Investig
Drugs. 21:1351–1369. 2012.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Muller C, Morales P and Reggio PH:
Cannabinoid ligands targeting TRP channels. Front Mol Neurosci.
11(487)2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Salehi B, Quispe C, Chamkhi I, El Omari N,
Balahbib A, Sharifi-Rad J, Bouyahya A, Akram M, Iqbal M, Docea AO,
et al: Pharmacological properties of chalcones: A review of
preclinical including molecular mechanisms and clinical evidence.
Front Pharmacol. 11(592654)2021.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Scheau C, Badarau IA, Mihai LG, Scheau AE,
Costache DO, Constantin C, Calina D, Caruntu C, Costache RS and
Caruntu A: Cannabinoids in the pathophysiology of skin
inflammation. Molecules. 25(25)2020.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Starkus J, Jansen C, Shimoda LMN, Stokes
AJ, Small-Howard AL and Turner H: Diverse TRPV1 responses to
cannabinoids. Channels (Austin). 13:172–191. 2019.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Calina D, Buga AM, Mitroi M, Buha A,
Caruntu C, Scheau C, Bouyahya A, El Omari N, El Menyiy N and Docea
AO: The treatment of cognitive, behavioural and motor impairments
from brain injury and neurodegenerative diseases through
cannabinoid system modulation-evidence from in vivo studies. J Clin
Med. 9(9)2020.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Karoly HC, Mueller RL, Bidwell LC and
Hutchison KE: Cannabinoids and the microbiota-gut-brain axis:
Emerging effects of cannabidiol and potential applications to
alcohol use disorders. Alcohol Clin Exp Res. 44:340–353.
2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Ribeiro A, Almeida VI, Costola-de-Souza C,
Ferraz-de-Paula V, Pinheiro ML, Vitoretti LB, Gimenes-Junior JA,
Akamine AT, Crippa JA, Tavares-de-Lima W, et al: Cannabidiol
improves lung function and inflammation in mice submitted to
LPS-induced acute lung injury. Immunopharmacol Immunotoxicol.
37:35–41. 2015.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Cintosun A, Lara-Corrales I and Pope E:
Mechanisms of cannabinoids and potential applicability to skin
diseases. Clin Drug Investig. 40:293–304. 2020.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Zholos AV: TRP channels in respiratory
pathophysiology: the role of oxidative, chemical irritant and
temperature stimuli. Curr Neuropharmacol. 13:279–291.
2015.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Reilly CA, Taylor JL, Lanza DL, Carr BA,
Crouch DJ and Yost GS: Capsaicinoids cause inflammation and
epithelial cell death through activation of vanilloid receptors.
Toxicol Sci. 73:170–181. 2003.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Thomas KC, Sabnis AS, Johansen ME, Lanza
DL, Moos PJ, Yost GS and Reilly CA: Transient receptor potential
vanilloid 1 agonists cause endoplasmic reticulum stress and cell
death in human lung cells. J Pharmacol Exp Ther. 321:830–838.
2007.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Agopyan N, Head J, Yu S and Simon SA:
TRPV1 receptors mediate particulate matter-induced apoptosis. Am J
Physiol Lung Cell Mol Physiol. 286:L563–L572. 2004.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Cabral LD and Giusti-Paiva A: The
transient receptor potential vanilloid 1 antagonist capsazepine
improves the impaired lung mechanics during endotoxemia. Basic Clin
Pharmacol Toxicol. 119:421–427. 2016.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Tani M, Kotani S, Hayakawa C, Lin ST, Irie
S, Ikeda K, Kawakami K and Onimaru H: Effects of a TRPV1 agonist
capsaicin on respiratory rhythm generation in brainstem-spinal cord
preparation from newborn rats. Pflugers Arch. 469:327–338.
2017.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Yang J, Yu HM, Zhou XD, Kolosov VP and
Perelman JM: Study on TRPV1-mediated mechanism for the
hypersecretion of mucus in respiratory inflammation. Mol Immunol.
53:161–171. 2013.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Bonvini SJ and Belvisi MG: Cough and
airway disease: The role of ion channels. Pulm Pharmacol Ther.
47:21–28. 2017.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Belvisi MG, Miura M, Stretton D and Barnes
PJ: Capsazepine as a selective antagonist of capsaicin-induced
activation of C-fibres in guinea-pig bronchi. Eur J Pharmacol.
215:341–344. 1992.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Leung SY, Niimi A, Williams AS, Nath P,
Blanc FX, Dinh QT and Chung KF: Inhibition of citric acid- and
capsaicin-induced cough by novel TRPV-1 antagonist, V112220, in
guinea-pig. Cough. 3(10)2007.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Talbot S, Abdulnour RE, Burkett PR, Lee S,
Cronin SJ, Pascal MA, Laedermann C, Foster SL, Tran JV, Lai N, et
al: Silencing nociceptor neurons reduces allergic airway
inflammation. Neuron. 87:341–354. 2015.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Kim BG, Park MK, Lee PH, Lee SH, Hong J,
Aung MM, Moe KT, Han NY and Jang AS: Effects of nanoparticles on
neuroinflammation in a mouse model of asthma. Respir Physiol
Neurobiol. 271(103292)2020.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Tränkner D, Hahne N, Sugino K, Hoon MA and
Zuker C: Population of sensory neurons essential for asthmatic
hyperreactivity of inflamed airways. Proc Natl Acad Sci USA.
111:11515–11520. 2014.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Baral P, Umans BD, Li L, Wallrapp A, Bist
M, Kirschbaum T, Wei Y, Zhou Y, Kuchroo VK, Burkett PR, et al:
Nociceptor sensory neurons suppress neutrophil and γδ T cell
responses in bacterial lung infections and lethal pneumonia. Nat
Med. 24:417–426. 2018.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Abe K, Watanabe N, Kumagai N, Mouri T,
Seki T and Yoshinaga K: Circulating kinin in patients with
bronchial asthma. Experientia. 23:626–627. 1967.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Dolovich J, Back N and Arbesman CE:
Kinin-like activity in nasal secretions of allergic patients. Int
Arch Allergy Appl Immunol. 38:337–344. 1970.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Ricciardolo FLM, Folkerts G, Folino A and
Mognetti B: Bradykinin in asthma: Modulation of airway inflammation
and remodelling. Eur J Pharmacol. 827:181–188. 2018.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Ternesten-Hasséus E, Johansson K, Löwhagen
O and Millqvist E: Inhalation method determines outcome of
capsaicin inhalation in patients with chronic cough due to sensory
hyperreactivity. Pulm Pharmacol Ther. 19:172–178. 2006.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Dicpinigaitis PV, Dobkin JB and Reichel J:
Antitussive effect of the leukotriene receptor antagonist
zafirlukast in subjects with cough-variant asthma. J Asthma.
39:291–297. 2002.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Groneberg DA, Niimi A, Dinh QT, Cosio B,
Hew M, Fischer A and Chung KF: Increased expression of transient
receptor potential vanilloid-1 in airway nerves of chronic cough.
Am J Respir Crit Care Med. 170:1276–1280. 2004.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Millqvist E: TRP channels and temperature
in airway disease-clinical significance. Temperature. 2:172–177.
2015.PubMed/NCBI View Article : Google Scholar
|
|
74
|
McLeod RL, Fernandez X, Correll CC, Phelps
TP, Jia Y, Wang X and Hey JA: TRPV1 antagonists attenuate
antigen-provoked cough in ovalbumin sensitized guinea pigs. Cough.
2(10)2006.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Blanc P, Liu D, Juarez C and Boushey HA:
Cough in hot pepper workers. Chest. 99:27–32. 1991.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Lankatilake KN and Uragoda CG: Respiratory
function in chilli grinders. Occup Med (Lond). 43:139–142.
1993.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Muralidhara and Narasimhamurthy K:
Non-mutagenicity of capsaicin in albino mice. Food Chem Toxicol.
26:955–958. 1988.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Surh YJ and Lee SS: Capsaicin in hot chili
pepper: Carcinogen, co-carcinogen or anticarcinogen? Food Chem
Toxicol. 34:313–316. 1996.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Park KK and Surh YJ: Effects of capsaicin
on chemically-induced two-stage mouse skin carcinogenesis. Cancer
Lett. 114:183–184. 1997.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Georgescu SR, Sârbu MI, Matei C, Ilie MA,
Caruntu C, Constantin C, Neagu M and Tampa M: Capsaicin: Friend or
foe in skin cancer and other related malignancies? Nutrients.
9(1365)2017.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Rogers DF, Aursudkij B and Barnes PJ:
Effects of tachykinins on mucus secretion in human bronchi in
vitro. Eur J Pharmacol. 174:283–286. 1989.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Meini S, Mak JCW, Rohde JAL and Rogers DF:
Tachykinin control of ferret airways: Mucus secretion,
bronchoconstriction and receptor mapping. Neuropeptides. 24:81–89.
1993.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Ramnarine SI, Hirayama Y, Barnes PJ and
Rogers DF: ‘Sensory-efferent’ neural control of mucus secretion:
Characterization using tachykinin receptor antagonists in ferret
trachea in vitro. Br J Pharmacol. 113:1183–1190. 1994.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Szallasi A and Blumberg PM: Vanilloid
(Capsaicin) receptors and mechanisms. Pharmacol Rev. 51:159–212.
1999.PubMed/NCBI
|
|
85
|
Karmouty-Quintana H, Cannet C, Sugar R,
Fozard JR, Page CP and Beckmann N: Capsaicin-induced mucus
secretion in rat airways assessed in vivo and non-invasively by
magnetic resonance imaging. Br J Pharmacol. 150:1022–1030.
2007.PubMed/NCBI View Article : Google Scholar
|
|
86
|
O'Donnell DE, Elbehairy AF, Berton DC,
Domnik NJ and Neder JA: Advances in the evaluation of respiratory
pathophysiology during exercise in chronic lung diseases. Front
Physiol. 8(82)2017.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Smutzer G and Devassy RK: Integrating
TRPV1 receptor function with capsaicin psychophysics. Adv Pharmacol
Sci. 2016(1512457)2016.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Burki NK and Lee LY: Mechanisms of
dyspnea. Chest. 138:1196–1201. 2010.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Winning AJ, Hamilton RD, Shea SA and Guz
A: Respiratory and cardiovascular effects of central and peripheral
intravenous injections of capsaicin in man: evidence for pulmonary
chemosensitivity. Clin Sci (Lond). 71:519–526. 1986.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Leung JM, Tiew PY, Mac Aogáin M, Budden
KF, Yong VF, Thomas SS, Pethe K, Hansbro PM and Chotirmall SH: The
role of acute and chronic respiratory colonization and infections
in the pathogenesis of COPD. Respirology. 22:634–650.
2017.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Linden D, Guo-Parke H, Coyle PV, Fairley
D, McAuley DF, Taggart CC and Kidney J: Respiratory viral
infection: A potential “missing link” in the pathogenesis of COPD.
Eur Respir Rev. 28(28)2019.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Wang Z, Bafadhel M, Haldar K, Spivak A,
Mayhew D, Miller BE, Tal-Singer R, Johnston SL, Ramsheh MY, Barer
MR, et al: Lung microbiome dynamics in COPD exacerbations. Eur
Respir J. 47:1082–1092. 2016.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Adaszek Ł, Gadomska D, Mazurek Ł, Łyp P,
Madany J and Winiarczyk S: Properties of capsaicin and its utility
in veterinary and human medicine. Res Vet Sci. 123:14–19.
2019.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Rabe KF, Hurd S, Anzueto A, Barnes PJ,
Buist SA, Calverley P, Fukuchi Y, Jenkins C, Rodriguez-Roisin R,
van Weel C, et al: Global Initiative for Chronic Obstructive Lung
Disease: Global strategy for the diagnosis, management, and
prevention of chronic obstructive pulmonary disease: GOLD executive
summary. Am J Respir Crit Care Med. 176:532–555. 2007.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Hillas G, Perlikos F, Tsiligianni I and
Tzanakis N: Managing comorbidities in COPD. Int J Chron Obstruct
Pulmon Dis. 10:95–109. 2015.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Eschenbacher WL: Defining Airflow
Obstruction. Chronic Obstr Pulm Dis (Miami). 3:515–518.
2016.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Tetley TD: Inflammatory cells and chronic
obstructive pulmonary disease. Curr Drug Targets Inflamm Allergy.
4:607–618. 2005.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Wang Y, Xu J, Meng Y, Adcock IM and Yao X:
Role of inflammatory cells in airway remodeling in COPD. Int J
Chron Obstruct Pulmon Dis. 13:3341–3348. 2018.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Sethi S, Mahler DA, Marcus P, Owen CA,
Yawn B and Rennard S: Inflammation in COPD: Implications for
management. Am J Med. 125:1162–1170. 2012.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Barbu C, Iordache M and Man MG:
Inflammation in COPD: Pathogenesis, local and systemic effects. Rom
J Morphol Embryol. 52:21–27. 2011.PubMed/NCBI
|
|
101
|
Audrit KJ, Delventhal L, Aydin Ö and
Nassenstein C: The nervous system of airways and its remodeling in
inflammatory lung diseases. Cell Tissue Res. 367:571–590.
2017.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Tóth BI, Benko S, Szöllosi AG, Kovács L,
Rajnavölgyi E and Bíró T: Transient receptor potential vanilloid-1
signaling inhibits differentiation and activation of human
dendritic cells. FEBS Lett. 583:1619–1624. 2009.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Wang M, Zhang Y, Xu M, Zhang H, Chen Y,
Chung KF, Adcock IM and Li F: Roles of TRPA1 and TRPV1 in cigarette
smoke -induced airway epithelial cell injury model. Free Radic Biol
Med. 134:229–238. 2019.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Bose P, Bathri R, Kumar L, Vijayan VK and
Maudar KK: Role of oxidative stress and transient receptor
potential in chronic obstructive pulmonary disease. Indian J Med
Res. 142:245–260. 2015.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Dietrich A: Modulators of transient
receptor potential (TRP) channels as therapeutic options in lung
disease. Pharmaceuticals (Basel). 12(23)2019.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Jian T, Chen J, Ding X, Lv H, Li J, Wu Y,
Ren B, Tong B, Zuo Y, Su K, et al: Flavonoids isolated from loquat
(Eriobotrya japonica) leaves inhibit oxidative stress and
inflammation induced by cigarette smoke in COPD mice: The role of
TRPV1 signaling pathways. Food Funct. 11:3516–3526. 2020.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Xiong M, Guo M, Huang D, Li J and Zhou Y:
TRPV1 genetic polymorphisms and risk of COPD or COPD combined with
PH in the Han Chinese population. Cell Cycle. 19:3066–3073.
2020.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Deering-Rice CE, Johansen ME, Roberts JK,
Thomas KC, Romero EG, Lee J, Yost GS, Veranth JM and Reilly CA:
Transient receptor potential vanilloid-1 (TRPV1) is a mediator of
lung toxicity for coal fly ash particulate material. Mol Pharmacol.
81:411–419. 2012.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Barnes PJ: Neurogenic inflammation in the
airways. Respir Physiol. 125:145–154. 2001.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Wortley MA, Birrell MA and Belvisi MG:
Drugs Affecting TRP Channels. Handb Exp Pharmacol. 237:213–241.
2017.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Tagaya E, Yagi O, Sato A, Arimura K,
Takeyama K, Kondo M and Tamaoki J: Effect of tiotropium on mucus
hypersecretion and airway clearance in patients with COPD. Pulm
Pharmacol Ther. 39:81–84. 2016.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Kichko TI, Kobal G and Reeh PW: Cigarette
smoke has sensory effects through nicotinic and TRPA1 but not TRPV1
receptors on the isolated mouse trachea and larynx. Am J Physiol
Lung Cell Mol Physiol. 309:L812–L820. 2015.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Sadofsky LR, Ramachandran R, Crow C, Cowen
M, Compton SJ and Morice AH: Inflammatory stimuli up-regulate
transient receptor potential vanilloid-1 expression in human
bronchial fibroblasts. Exp Lung Res. 38:75–81. 2012.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Seki N, Shirasaki H, Kikuchi M and Himi T:
Capsaicin induces the production of IL-6 in human upper respiratory
epithelial cells. Life Sci. 80:1592–1597. 2007.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Gabay C: Interleukin-6 and chronic
inflammation. Arthritis Res Ther. 8 (Suppl 2)(S3)2006.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Nassini R, Pedretti P, Moretto N, Fusi C,
Carnini C, Facchinetti F, Viscomi AR, Pisano AR, Stokesberry S,
Brunmark C, et al: Transient receptor potential ankyrin 1 channel
localized to non-neuronal airway cells promotes non-neurogenic
inflammation. PLoS One. 7(e42454)2012.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Karlsson JA, Zackrisson C and Lundberg JM:
Hyperresponsiveness to tussive stimuli in cigarette smoke-exposed
guinea-pigs: A role for capsaicin-sensitive, calcitonin
gene-related peptide-containing nerves. Acta Physiol Scand.
141:445–454. 1991.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Belvisi MG, Birrell MA, Khalid S, Wortley
MA, Dockry R, Coote J, Holt K, Dubuis E, Kelsall A, Maher SA, et
al: Neurophenotypes in airway diseases. Insights from translational
cough studies. Am J Respir Crit Care Med. 193:1364–1372.
2016.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Blanc FX, Macedo P, Hew M and Chung KF:
Capsaicin cough sensitivity in smokers with and without airflow
obstruction. Respir Med. 103:786–790. 2009.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Doherty MJ, Mister R, Pearson MG and
Calverley PM: Capsaicin responsiveness and cough in asthma and
chronic obstructive pulmonary disease. Thorax. 55:643–649.
2000.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Wong CH and Morice AH: Cough threshold in
patients with chronic obstructive pulmonary disease. Thorax.
54:62–64. 1999.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Deslee G, Burgel PR, Escamilla R, Chanez
P, Court-Fortune I, Nesme-Meyer P, Brinchault-Rabin G, Perez T,
Jebrak G, Caillaud D, et al: Impact of current cough on
health-related quality of life in patients with COPD. Int J Chron
Obstruct Pulmon Dis. 11:2091–2097. 2016.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Morice AH, Millqvist E, Bieksiene K,
Birring SS, Dicpinigaitis P, Domingo Ribas C, Hilton Boon M, Kantar
A, Lai K, McGarvey L, et al: ERS guidelines on the diagnosis and
treatment of chronic cough in adults and children. Eur Respir J.
55(55)2020.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Ternesten-Hasséus E, Larsson C, Larsson S
and Millqvist E: Capsaicin sensitivity in patients with chronic
cough- results from a cross-sectional study. Cough.
9(5)2013.PubMed/NCBI View Article : Google Scholar
|
|
125
|
Terada K, Muro S, Ohara T, Haruna A,
Marumo S, Kudo M, Ogawa E, Hoshino Y, Hirai T, Niimi A, et al:
Cough-reflex sensitivity to inhaled capsaicin in COPD associated
with increased exacerbation frequency. Respirology. 14:1151–1155.
2009.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Cho PSP, Fletcher HV, Turner RD, Patel IS,
Jolley CJ and Birring SS: The relationship between cough reflex
sensitivity and exacerbation frequency in chronic obstructive
pulmonary disease. Lung. 198:617–628. 2020.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Fahy JV: Type 2 inflammation in asthma -
present in most, absent in many. Nat Rev Immunol. 15:57–65.
2015.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Mishra V, Banga J and Silveyra P:
Oxidative stress and cellular pathways of asthma and inflammation:
Therapeutic strategies and pharmacological targets. Pharmacol Ther.
181:169–182. 2018.PubMed/NCBI View Article : Google Scholar
|
|
129
|
Vieira Braga FA, Kar G, Berg M, Carpaij
OA, Polanski K, Simon LM, Brouwer S, Gomes T, Hesse L, Jiang J, et
al: A cellular census of human lungs identifies novel cell states
in health and in asthma. Nat Med. 25:1153–1163. 2019.PubMed/NCBI View Article : Google Scholar
|
|
130
|
Danahay H, Pessotti AD, Coote J,
Montgomery BE, Xia D, Wilson A, Yang H, Wang Z, Bevan L, Thomas C,
et al: Notch2 is required for inflammatory cytokine-driven goblet
cell metaplasia in the lung. Cell Rep. 10:239–252. 2015.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Gour N and Wills-Karp M: IL-4 and IL-13
signaling in allergic airway disease. Cytokine. 75:68–78.
2015.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Wills-Karp M, Luyimbazi J, Xu X, Schofield
B, Neben TY, Karp CL and Donaldson DD: Interleukin-13: Central
mediator of allergic asthma. Science. 282:2258–2261.
1998.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Webb DC, McKenzie AN, Koskinen AM, Yang M,
Mattes J and Foster PS: Integrated signals between IL-13, IL-4, and
IL-5 regulate airways hyperreactivity. J Immunol. 165:108–113.
2000.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Ren YF, Li H, Xing XH, Guan HS, Zhang BA,
Chen CL and Zhang JH: Preliminary study on pathogenesis of
bronchial asthma in children. Pediatr Res. 77:506–510.
2015.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Schiffers C, Hristova M, Habibovic A,
Dustin CM, Danyal K, Reynaert NL, Wouters EF and van der Vliet A:
The transient receptor potential channel vanilloid 1 is critical in
innate airway epithelial responses to protease allergens. Am J
Respir Cell Mol Biol. 63:198–208. 2020.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Jing X, Yan W, Zeng H and Cheng W: Qingfei
oral liquid alleviates airway hyperresponsiveness and mucus
hypersecretion via TRPV1 signaling in RSV-infected asthmatic mice.
Biomed Pharmacother. 128(110340)2020.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Satia I, Nagashima A and Usmani OS:
Exploring the role of nerves in asthma; insights from the study of
cough. Biochem Pharmacol. 179(113901)2020.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Zhang G, Lin R-L, Wiggers M, Snow DM and
Lee L-Y: Altered expression of TRPV1 and sensitivity to capsaicin
in pulmonary myelinated afferents following chronic airway
inflammation in the rat. J Physiol. 586:5771–5786. 2008.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Yocum GT, Chen J, Choi CH, Townsend EA,
Zhang Y, Xu D, Fu XW, Sanderson MJ and Emala CW: Role of transient
receptor potential vanilloid 1 in the modulation of airway smooth
muscle tone and calcium handling. Am J Physiol Lung Cell Mol
Physiol. 312:L812–L821. 2017.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Deering-Rice CE, Stockmann C, Romero EG,
Lu Z, Shapiro D, Stone BL, Fassl B, Nkoy F, Uchida DA, Ward RM, et
al: Characterization of transient receptor potential vanilloid-1
(TRPV1) variant activation by coal fly ash particles and
associations with altered transient receptor potential ankyrin-1
(TRPA1) expression and asthma. J Biol Chem. 291:24866–24879.
2016.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Li M, Fan X, Ji L, Fan Y and Xu L:
Exacerbating effects of trimellitic anhydride in ovalbumin-induced
asthmatic mice and the gene and protein expressions of TRPA1,
TRPV1, TRPV2 in lung tissue. Int Immunopharmacol. 69:159–168.
2019.PubMed/NCBI View Article : Google Scholar
|
|
142
|
Li J, Chen Y, Chen QY, Liu D, Xu L, Cheng
G, Yang X, Guo Z and Zeng Y: Role of transient receptor potential
cation channel subfamily V member 1 (TRPV1) on ozone-exacerbated
allergic asthma in mice. Environ Pollut. 247:586–594.
2019.PubMed/NCBI View Article : Google Scholar
|
|
143
|
Liu H, Fan X, Wang N, Zhang Y and Yu J:
Exacerbating effects of PM2.5 in OVA-sensitized and challenged mice
and the expression of TRPA1 and TRPV1 proteins in lungs. J Asthma.
54:807–817. 2017.PubMed/NCBI View Article : Google Scholar
|
|
144
|
Akopian AN, Fanick ER and Brooks EG: TRP
channels and traffic-related environmental pollution-induced
pulmonary disease. Semin Immunopathol. 38:331–338. 2016.PubMed/NCBI View Article : Google Scholar
|
|
145
|
Song J, Kang J, Lin B, Li J, Zhu Y, Du J,
Yang X, Xi Z and Li R: Mediating role of TRPV1 Ion channels in the
co-exposure to PM2.5 and formaldehyde of Balb/c mice asthma model.
Sci Rep. 7(11926)2017.PubMed/NCBI View Article : Google Scholar
|
|
146
|
Spaziano G, Luongo L, Guida F, Petrosino
S, Matteis M, Palazzo E, Sullo N, de Novellis V, Di Marzo V, Rossi
F, et al: Exposure to allergen causes changes in NTS neural
activities after intratracheal capsaicin application, in
endocannabinoid levels and in the GLIA morphology of NTS. BioMed
Res Int. 2015(980983)2015.PubMed/NCBI View Article : Google Scholar
|
|
147
|
Wang P, Liu H, Fan X, Zhu Z and Zhu Y:
Effect of San'ao decoction on aggravated asthma mice model induced
by PM2.5 and TRPA1/TRPV1 expressions. J Ethnopharmacol. 236:82–90.
2019.PubMed/NCBI View Article : Google Scholar
|
|
148
|
Choi JY, Lee HY, Hur J, Kim KH, Kang JY,
Rhee CK and Lee SY: TRPV1 blocking alleviates airway inflammation
and remodeling in a chronic asthma murine model. Allergy Asthma
Immunol Res. 10:216–224. 2018.PubMed/NCBI View Article : Google Scholar
|
|
149
|
Zhao L, Zhang X, Kuang H, Wu J, Guo Y and
Ma L: Effect of TRPV1 channel on the proliferation and apoptosis in
asthmatic rat airway smooth muscle cells. Exp Lung Res. 39:283–294.
2013.PubMed/NCBI View Article : Google Scholar
|
|
150
|
Satia I, Watson R, Scime T, Dockry RJ, Sen
S, Ford JW, Mitchell PD, Fowler SJ, Gauvreau GM, O'Byrne PM, et al:
Allergen challenge increases capsaicin-evoked cough responses in
patients with allergic asthma. J Allergy Clin Immunol.
144:788–795.e1. 2019.PubMed/NCBI View Article : Google Scholar
|
|
151
|
Han RT, Kim S, Choi K, Jwa H, Lee J, Kim
HY, Kim HJ, Kim HR, Back SK and Na HS: Asthma-like airway
inflammation and responses in a rat model of atopic dermatitis
induced by neonatal capsaicin treatment. J Asthma Allergy.
10:181–189. 2017.PubMed/NCBI View Article : Google Scholar
|
|
152
|
McGarvey LP, Butler CA, Stokesberry S,
Polley L, McQuaid S, Abdullah H, Ashraf S, McGahon MK, Curtis TM,
Arron J, et al: Increased expression of bronchial epithelial
transient receptor potential vanilloid 1 channels in patients with
severe asthma. J Allergy Clin Immunol. 133:704–12.e4.
2014.PubMed/NCBI View Article : Google Scholar
|
|
153
|
Lemanske RF Jr: Inflammatory events in
asthma: An expanding equation. J Allergy Clin Immunol.
105:S633–S636. 2000.PubMed/NCBI View Article : Google Scholar
|
|
154
|
Bujak JK, Kosmala D, Szopa IM, Majchrzak K
and Bednarczyk P: Inflammation, cancer and immunity-implication of
TRPV1 Channel. Front Oncol. 9(1087)2019.PubMed/NCBI View Article : Google Scholar
|
|
155
|
Baker K, Raemdonck K, Dekkak B, Snelgrove
RJ, Ford J, Shala F, Belvisi MG and Birrell MA: Role of the ion
channel, transient receptor potential cation channel subfamily V
member 1 (TRPV1), in allergic asthma. Respir Res.
17(67)2016.PubMed/NCBI View Article : Google Scholar
|
|
156
|
Kim JH: The Emerging role of TRPV1 in
airway inflammation. Allergy Asthma Immunol Res. 10:187–188.
2018.PubMed/NCBI View Article : Google Scholar
|
|
157
|
Rehman R, Bhat YA, Panda L and Mabalirajan
U: TRPV1 inhibition attenuates IL-13 mediated asthma features in
mice by reducing airway epithelial injury. Int Immunopharmacol.
15:597–605. 2013.PubMed/NCBI View Article : Google Scholar
|
|
158
|
Mori T, Saito K, Ohki Y, Arakawa H,
Tominaga M and Tokuyama K: Lack of transient receptor potential
vanilloid-1 enhances Th2-biased immune response of the airways in
mice receiving intranasal, but not intraperitoneal, sensitization.
Int Arch Allergy Immunol. 156:305–312. 2011.PubMed/NCBI View Article : Google Scholar
|
|
159
|
Purokivi M, Koskela H and Kontra K:
Determinants of asthma control and quality of life in stable
asthma: Evaluation of two new cough provocation tests. Clin Respir
J. 7:253–260. 2013.PubMed/NCBI View Article : Google Scholar
|
|
160
|
Puebla-Neira D and Calhoun WJ: Why do
asthma patients cough? New insights into cough in allergic asthma.
J Allergy Clin Immunol. 144:656–657. 2019.PubMed/NCBI View Article : Google Scholar
|
|
161
|
Chen CL, Li H, Xing XH, Guan HS, Zhang JH
and Zhao JW: Effect of TRPV1 gene mutation on bronchial asthma in
children before and after treatment. Allergy Asthma Proc.
36:e29–e36. 2015.PubMed/NCBI View Article : Google Scholar
|
|
162
|
Lin YJ, Lin RL, Khosravi M and Lee LY:
Hypersensitivity of vagal pulmonary C-fibers induced by increasing
airway temperature in ovalbumin-sensitized rats. Am J Physiol Regul
Integr Comp Physiol. 309:R1285–R1291. 2015.PubMed/NCBI View Article : Google Scholar
|
|
163
|
Johansson EL, Gustafsson P, Millqvist E
and Ternesten-Hasséus E: Small and large airways' reactions to
inhaled capsaicin in patients with chronic idiopathic cough, or
asthma and in healthy control subjects. Exp Lung Res. 45:55–64.
2019.PubMed/NCBI View Article : Google Scholar
|
|
164
|
Chen L, Li C, Peng M, Xie J, Lai K and
Zhong N: Establishment of a mouse model with all four clinical
features of eosinophilic bronchitis. Sci Rep.
10(10557)2020.PubMed/NCBI View Article : Google Scholar
|
|
165
|
Xu X, Chen Q, Qiu Z, Shi C, Ding H, Wang
L, Lv H and Yu L: Association of cough hypersensitivity with
tracheal TRPV1 activation and neurogenic inflammation in a novel
guinea pig model of citric acid-induced chronic cough. J Int Med
Res. 46:2913–2924. 2018.PubMed/NCBI View Article : Google Scholar
|
|
166
|
Liu Q, Fujimura M, Tachibana H, Myou S,
Kasahara K and Yasui M: Characterization of increased cough
sensitivity after antigen challenge in guinea pigs. Clin Exp
Allergy. 31:474–484. 2001.PubMed/NCBI View Article : Google Scholar
|
|
167
|
Satia I, Tsamandouras N, Holt K, Badri H,
Woodhead M, Ogungbenro K, Felton TW, O'Byrne PM, Fowler SJ and
Smith JA: Capsaicin-evoked cough responses in asthmatic patients:
Evidence for airway neuronal dysfunction. J Allergy Clin Immunol.
139:771–779.e10. 2017.PubMed/NCBI View Article : Google Scholar
|
|
168
|
Kanemitsu Y, Fukumitsu K, Kurokawa R,
Takeda N, Suzuki M, Yap J, Nishiyama H, Tajiri T, Fukuda S, Uemura
T, et al: Increased capsaicin sensitivity in patients with severe
asthma is associated with worse clinical outcome. Am J Respir Crit
Care Med. 201:1068–1077. 2020.PubMed/NCBI View Article : Google Scholar
|
|
169
|
Lai K, Long L, Yi F, Tang J, Chen Z, Chen
F, Zhou J, Peng W, Zhang L, Li H, et al: Age and sex distribution
of chinese chronic cough patients and their relationship with
capsaicin cough sensitivity. Allergy Asthma Immunol Res.
11:871–884. 2019.PubMed/NCBI View Article : Google Scholar
|
|
170
|
Kunc P, Fabry J, Lucanska M, Zatko T,
Grendar M and Pecova R: Cough reflex sensitivity in asthmatic
children. Physiol Res. 69 (Suppl 1):S147–S150. 2020.PubMed/NCBI View Article : Google Scholar
|
|
171
|
Satia I and O'Byrne PM: Identifying a
neurophenotype in severe asthma. Am J Respir Crit Care Med.
201:1024–1025. 2020.PubMed/NCBI View Article : Google Scholar
|
|
172
|
Nakajima T, Nishimura Y, Nishiuma T,
Kotani Y, Nakata H and Yokoyama M: Cough sensitivity in pure cough
variant asthma elicited using continuous capsaicin inhalation.
Allergol Int. 55:149–155. 2006.PubMed/NCBI View Article : Google Scholar
|
|
173
|
Millqvist E, Löwhagen O and Bende M:
Quality of life and capsaicin sensitivity in patients with sensory
airway hyperreactivity. Allergy. 55:540–545. 2000.PubMed/NCBI View Article : Google Scholar
|
|
174
|
Park KH, Kim BG, Lee PH, Hong J, Lee J,
Park SW, Kim DJ and Jang AS: Impact of capsaicin concentration
evoking coughs on clinical variables in patients with asthma. Exp
Lung Res. 47:1–8. 2021.PubMed/NCBI View Article : Google Scholar
|
|
175
|
Wang R, Fowler SJ, Niven R, Ryan D, Holt
K, Mitchell J, Dockry R, Al-Sheklly B, Satia I and Smith JA:
Investigating the safety of capsaicin cough challenge in severe
asthma. Clin Exp Allergy. 49:932–934. 2019.PubMed/NCBI View Article : Google Scholar
|
|
176
|
Yamamura K, Hara J, Ohkura N, Abo M, Sone
T, Kimura H and Kasahara K: Increased cough receptor sensitivity to
capsaicin predicts a positive bronchial thermoplasty response: A
single-center retrospective study. J Bronchology Interv Pulmonol.
26:137–141. 2019.PubMed/NCBI View Article : Google Scholar
|
|
177
|
Fukumitsu K, Kanemitsu Y, Asano T, Takeda
N, Ichikawa H, Yap JM, Fukuda S, Uemura T, Takakuwa O, Ohkubo H, et
al: Tiotropium attenuates refractory cough and capsaicin cough
reflex sensitivity in patients with asthma. J Allergy Clin Immunol
Pract. 6:1613–1620.e1612. 2018.PubMed/NCBI View Article : Google Scholar
|