Introduction
Preeclampsia is a hypertensive disorder affecting
3-5% of pregnant women and one of the three most frequent causes of
maternal mortality (1) A total of
~16% of maternal deaths can be attributed to hypertensive disorders
(2). Immune disorders, shallow
placental implantation, vascular endothelial damage, genetic
factors and nutritional deficiencies have been considered as
causative factors for preeclampsia (3). Several studies have shown that the
reduced ability of trophoblasts to invade the spiral artery of the
uterus decreases placental blood supply (4,5) and
increases the apoptosis of migratory trophoblasts in the placental
tissue during the early stage of preeclampsia (6). At present, the pathogenesis of various
preeclampsia phenotypes has not been fully elucidated. A deep
understanding of trophoblast cell apoptosis and migration may
reveal the pathogenesis of preeclampsia (7). Therefore, it is important to identify
key regulatory molecules involved in the preeclampsia-associated
apoptosis and migration of trophoblasts (8).
A number of molecules have been demonstrated to be
associated with the apoptosis and migration of trophoblast cells in
preeclampsia. For instance, let-7a has been reported to increase
the apoptosis of trophoblast cells in preeclampsia by inhibiting
the expression of Yes-associated protein 1 and Bcl-xl (9). In addition, exosome-secreted
microRNA-133b has been demonstrated to suppress apoptosis and boost
the proliferation and invasion of trophoblasts in preeclampsia by
restricting the expression of serum/glucocorticoid regulated kinase
1(10). Also, protein disulfide
isomerase 3 has been shown to regulate trophoblast apoptosis and
proliferation in preeclampsia via the E3 ubiquitin-protein ligase
Mdm2/p53 pathway (11). The Fas/Fas
ligand (FasL) system, which belongs to the tumor necrosis factor
family of receptors and ligands, is one of the most common
apoptotic pathways (12). Fas and
FasL are widely distributed in various tissues where they regulate
apoptosis in pregnancy during mammalian development (13). There is evidence to indicate that
Fas and FasL genes contribute to the development of preeclampsia
(14). Previous studies have shown
that Fas is highly expressed in placental tissues during
preeclampsia (15,16). However, the specific role of Fas in
preeclampsia is unclear.
Nuclear factor κB (NF-κB) is a transcription factor
that regulates the expression of various genes associated with
tumorigenesis, apoptosis, inflammation and immune diseases
(17). NF-κB can be activated
through classical or alternative pathways (17). NF-κB in the placental tissue
promotes the expression of cytokines, which induces immune and
inflammatory reactions and placental cell damage, thereby
accelerating the pathogenesis of preeclampsia (18,19).
The DNA-binding activity and nuclear localization of p65, a NF-κB
subunit, in the fetal membrane and myometrium influences the
pathogenesis of preeclampsia (20).
Therefore, the mechanism through which NF-κB regulates preeclampsia
merits further exploration.
The present study aimed to investigate the
underlying role of Fas and NF-κB in preeclampsia with the
overarching goal of identifying suitable biomarkers for the
screening and treatment of preeclampsia.
Materials and methods
JAR cell culture
JAR choriocarcinoma cells, a neoplastic trophoblast
cell line, were obtained from the Cell Bank of the Chinese Academy
of Sciences (Shanghai, China). The cells were cultured in DMEM (cat
no. A4192101) supplemented with 10% FBS (cat. no. 16140071) and 1%
penicillin/streptomycin (cat. no. 15140122; all Gibco; Thermo
Fisher Scientific, Inc.) under a humidified environment with 5%
CO2 at 37˚C.
Vector construction and lentiviral
infection
A lentiviral overexpression vector was designed and
constructed based on the human full-length coding protein sequence
of Fas (GenBank accession no.: NM_000043) by GeneChem, Inc. A
knockdown lentiviral vector was constructed to express small
interfering RNAs targeting the human Fas sequence, and a negative
control (NC) lentivirus with a non-targeting sequence was also
designed. The short hairpin RNA (shRNA) sequence used for the
knockdown of Fas was 5'-GCGTATGACACATTGATTAAA-3' and the
non-targeting sequence was 5'-TTCTCCGAACGTGTCACGT-3'. The shRNA
sequences were cloned into a GV248 vector (GeneChem, Inc.) to
produce lentiviral vectors. All vectors carried green fluorescent
protein. The vectors (100 nM) and packaging plasmids [psPaX2; third
generation lentivirus packaging system (vector: Packaging vector:
Envelope ratio, 10:3:1) Promega Corporation] were co-transfected
into 1x106 293T cells (Cell Bank of the Chinese Academy
of Sciences) with Lipofectamine® 3000 (Invitrogen;
Thermo Fisher Scientific, Inc.). Following cell culture of the 293T
cells for 4 days, cell supernatant was collected and centrifuged at
251.55 x g for 5 min at 37˚C. The supernatant was subsequently
filtered using a 0.22 µM membrane to collect the Fas overexpression
lentivirus (OE-Fas), empty lentivirus (OE-NC), Fas knockdown
lentivirus (sh-Fas) and knockdown NC lentivirus (KD-NC) vectors.
JAR cells (~1x106) were then infected with the
lentiviral vectors at 37˚C, at a multiplicity of infection
value=25. The transfected cells were collected 96 h after infection
to determine the infection efficiency. The following experiment was
conducted 96 h after transfection.
NF-κB p65 overexpression
An NF-κB p65 sequence (GenBank ID: NM_021975) was
cloned into an LV003 vector (Invitrogen; Thermo Fisher Scientific,
Inc.) to prepare a NF-κB p65 overexpression vector. Empty LV003
vector was used as a control. The JAR cells were cultured to ~80%
confluence, and then transfected with the Fas knockdown lentiviral
vector (sh-Fas) and/or the LV003 vector harboring the NF-κB p65
sequence or empty LV003 control using Lipofectamine 3000 reagent.
After continuous culture for 12 h, the conventional medium without
virus was replaced and cells were cultured at 37˚C. Subsequent
experiments were conducted following 48 h of transfection.
Reverse transcription-quantitative PCR
(RT-qPCR)
RNAiso Plus (Takara Bio, Inc.) was used to isolate
total RNA from the transfected trophoblast cells. Complementary DNA
(cDNA) was produced from the RNA using a PrimeScript™ RT
Reagent kit with gDNA Eraser, according to the manufacturer's
instructions (Takara Bio, Inc.). The cDNA was subjected to qPCR
using SYBRGreen Realtime PCR kit (Takara Bio, Inc.). qPCR was
performed using 30 cycles at 42˚C for 30 min and 95˚C for 5 min.
The following components were used in a total reaction volume of 25
µl: cDNA (2 µl), SYBR Premix Ex Taq™ (2X; 10 µl),
reverse primer (10 µM; 1 µl), forward primer (10 µΜ; 1 µl),
double-distilled water (10 µl) and ROX Reference dye II (1:50 in
PBS; 1 µl). Relative expression of the target mRNA was normalized
to the expression of GAPDH mRNA and calculated using the
2-ΔΔCq method (21). The
sequences of the primers used were as follows: GAPDH (gene
accession no. CH471116) forward, 5'-TGACTTCAACAGCGACACCCA-3' and
reverse, 5'-CACCCTGTTGCTGTAGCCAAA-3'; Fas (gene accession no.
AH006106) forward, 5'-CTTCTTTTGCCAATTCCAC-3' and reverse,
5'-CAGATAAATTTATTGCCACTG-3'; and RELA (also known as p65; gene
accession no. AY455868) forward, 5'-GACTACGACCTGAATGCTGTG-3' and
reverse, 5'-ACCTCAATGTCCTCTTTCTGC-3'.
Cell viability assay
Transfected trophoblast cells were seeded in 96-well
cell culture plates at a density of 2x103 cells/well and
cultured in an incubator for 48 h. Next, Cell Counting Kit-8
(CCK-8) reagent (10 µl/well; MedChemExpress) was added to the cells
and incubated for 2 h at 37˚C. A spectrometer (Thermo Fisher
Scientific, Inc.) was used to measure the absorbance at 450 nm. The
cell viability was calculated from the absorbance values each day
for 5 consecutive days.
Apoptosis analysis
An Annexin V-FITC/7AAD kit (BioLegend, Inc.) was
used to detect cell apoptosis following the manufacturer's
instructions. The trophoblast cells in the logarithmic growth phase
after transfection were harvested and digested with 0.25% trypsin
for 2 min at 37˚C The cells were centrifuged at 400 x g for 3 min
at 37˚C, and subsequently washed twice with cold PBS. The cells
were re-suspended in 500 µl binding buffer to a final concentration
of 1x106 cells/ml. A 100-µl portion of the cell
suspension was transferred to a 5-ml flow tube into which 5 µl
7-AAD and 5 µl Annexin V-PE were added. The contents in the flow
tube were mixed and incubated for 15 min in the dark. A flow
cytometer (CytoFLEX; Beckman Coulter, Inc.) was used to examine the
cells and FlowJo software (Version 10; Tree Star, Inc.) was used to
analyze the data.
Cell migration assay
A Transwell assay was conducted to measure the
changes in trophoblast cell migration after transfection. The
transfected cells were washed three times using serum-free DMEM
(cat no. A4192101; Gibco; Thermo Fisher Scientific, Inc.) medium
and counted. The cells were suspended in the medium at a
concentration of 7,000 cells/well in a 12-well plate (3,500
cells/ml). Then, 150 µl serum-free cell suspension and 600 µl DMEM
supplemented with 10% FBS (cat. no. 16140071; Gibco; Thermo Fisher
Scientific, Inc.) were added to the lower and upper chambers of the
Transwell apparatus, respectively, and the Transwell was incubated
for 48 h at 37˚C in an incubator. The cells on the basolateral
chamber were washed twice using PBS, and stained with 1% crystal
violet for 30 min at 37˚C. The cells were then washed twice using
PBS and images captured under a microscope (magnification, x200;
Olympus cX2; Olympus Corporation).
Tumor necrosis factor α (TNF-a) and
interleukin-2 (IL-2) measurement
The levels of TNF-α and IL-2 in the supernatant of
the trophoblast cells were detected using enzyme-linked
immunosorbent assay kits (cat. nos. RAB0480 and RAB0286,
respectively; Merck KGaA). The calibration curves were plotted on
semi-log papers, and the concentrations of TNF-α and IL-2 were
determined based on a comparison of the optical density values of
the samples with the standard curve.
Western blot analysis
Transfected cells were washed three times with cold
PBS and proteins were extracted from the cells using RIPA lysis
buffer (cat. no. HY-K1001; MedChemExpress). The protein
concentration was quantified using a BCA Protein assay kit (cat.
no. 23225; Thermo Fisher Scientific, Inc.). The protein samples (30
µg/lane) were resolved using 20% Precise Protein Gels (Pierce;
Thermo Fisher Scientific, Inc.) and transferred to polypropylene
fluoride membranes (Amersham; Cytiva). The membranes were blocked
for 1 h at room temperature with 3% skimmed milk in Tris-buffered
saline (TBS) containing 0.1% Tween 20. The membranes were then
probed using primary antibodies against Fas (cat. no. 8023), Bax
(cat. no. 14796), NF-κB p65 (cat. no. 8242), Bcl-2 (cat. no. 15071)
and b-actin (cat. no. 3700). All antibodies were bought from Cell
Signaling Technology, Inc. The blots were then washed in TBS
containing 0.1% Tween-20 prior to labelling with horseradish
peroxidase-conjugated secondary anti-rabbit antibody for 2 h at
room temperature (cat. no. 4414S; Cell Signaling Technology, Inc.).
The primary antibodies were used at a dilution of 1:1,000 and the
secondary antibody was used at a dilution of 1:5,000 according to
the manufacturer's instructions. The blots were visualized using an
ECL plus Western Blotting Detection System (Amersham; Cytiva).
Relative protein expression levels were normalized to the
expression of b-actin. Image J (version 1.48, National Institutes
of Health) was used for densitometric analysis.
Statistical analysis
Statistical analysis was performed using GraphPad
Prism software (8.0; GraphPad Software, Inc.). Each experiment was
repeated three times, Data are presented as the mean ± standard
deviation. Differences between groups were compared using one-way
ANOVA analysis followed by Tukey's post hoc test. P<0.05 was
considered to indicate a statistically significant difference.
Results
Transfection effectively modulates Fas
mRNA and protein expression
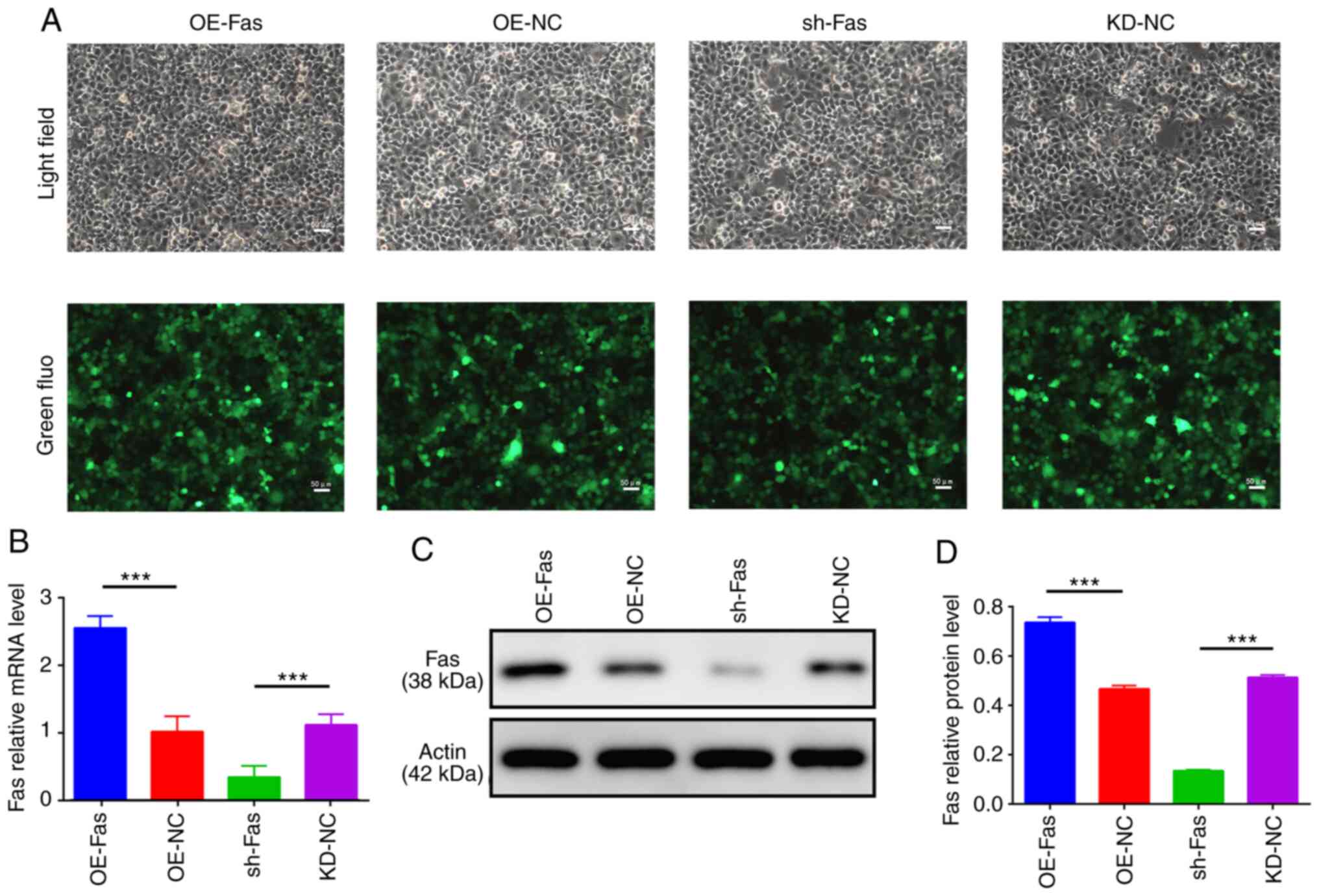
Fluorescence imaging indicated that the transfection
efficiency in each group of trophoblast cells was >80% (Fig. 1A). The RT-qPCR analysis revealed
that the Fas mRNA level of the trophoblast cells in the OE-Fas
group was 251% that of the OE-NC group, whereas the Fas mRNA level
in the sh-Fas group was 26.04% that of the KD-NC group (Fig. 1B). Western blotting results
indicated that the protein expression of Fas in trophoblast cells
in the OE-Fas group was significantly higher compared with that in
the OE-NC group, while the protein expression of Fas in the sh-Fas
group was significantly lower compared with that in the KD-NC group
(Fig. 1C and D).
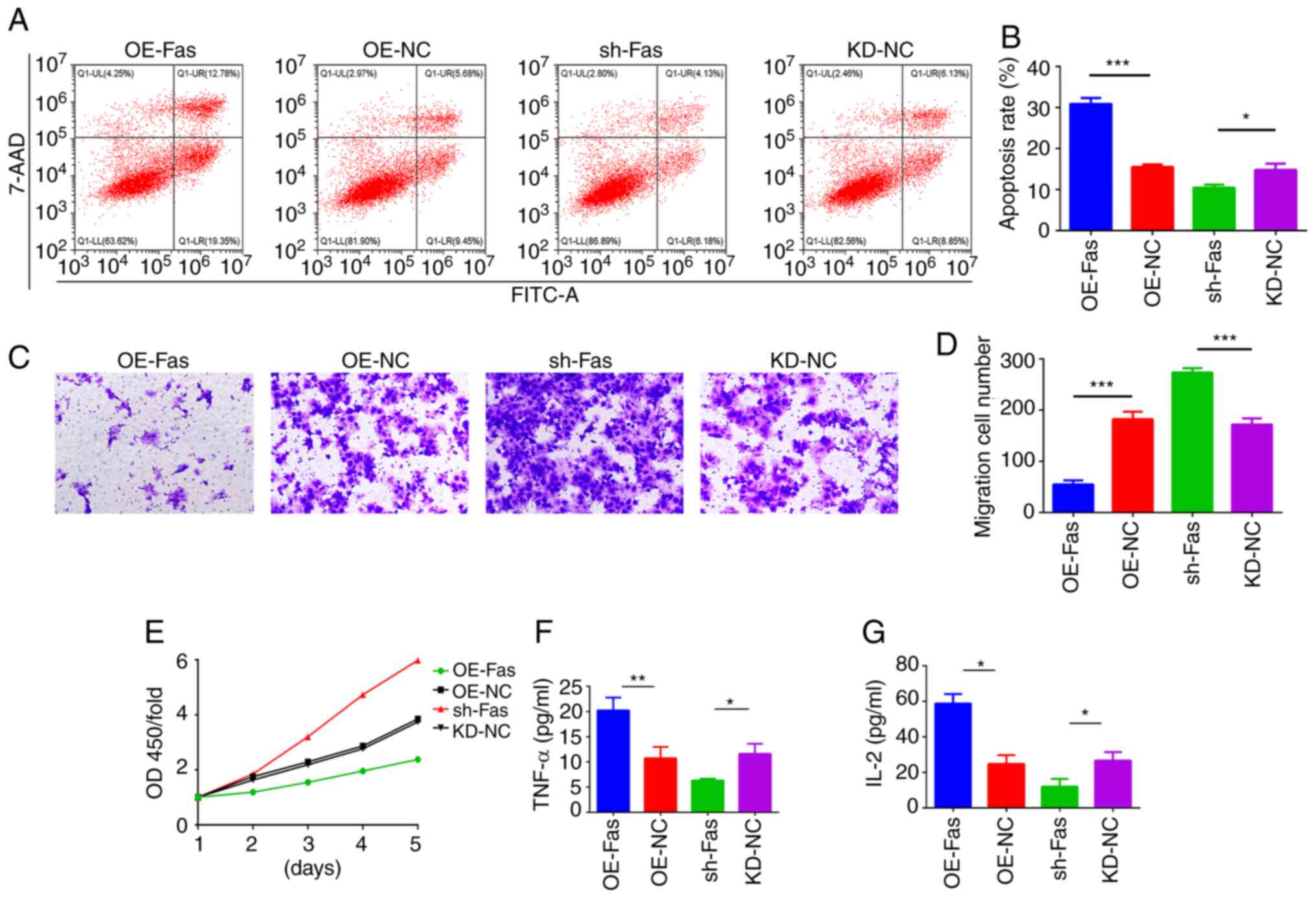
Fas regulates the apoptosis and
migration of trophoblast cells
Flow cytometry results indicated that the apoptosis
of the trophoblast cells was significantly promoted by the
overexpression of Fas and suppressed by the knockdown of Fas
(Fig. 2A and B). In addition, the results of the
Transwell assay indicated that the overexpression of Fas inhibited
trophoblast cell migration, while the knockdown of Fas
significantly promoted trophoblast cell migration (Fig. 2C and D). Moreover, the CCK-8 assay results
revealed that the viability of the trophoblast cells was reduced by
the overexpression of Fas and increased by the knockdown of Fas
(Fig. 2E). Furthermore, the
overexpression of Fas in trophoblast cells upregulated the
secretion of TNF-α and IL-2 while the knockdown of Fas decreased
this secretion (Fig. 2F and
G).
Fas may function via NF-κB
Western blot analysis indicated that the
overexpression of Fas in trophoblast cells significantly
upregulated the expression of NF-κB and Bax and significantly
decreased the expression of Bcl-2 (Fig.
3A-D). By contrast, the knockdown of Fas in trophoblast cells
significantly decreased the expression of NF-κB and Bax and
significantly upregulated the expression of Bcl-2 (Fig. 3A-D). To further investigate the
involvement of NF-κB in the Fas-mediated apoptosis and migration of
trophoblast cells, NF-κB p65 was overexpressed in the JAR cells
(Fig. 3E). Notably, the
overexpression of NF-κB reversed the Fas knockdown-induced
inhibitory effects on apoptosis and stimulatory effects on
migration in trophoblast cells (Fig.
3F and G). In addition, the
overexpression of NF-κB in trophoblast cells attenuated the
reduction in Bax expression and increase in Bcl-2 expression
induced by Fas knockdown (Fig.
3H-K). These results indicate that Fas regulates the apoptosis
and migration of trophoblast cells via NF-κB.
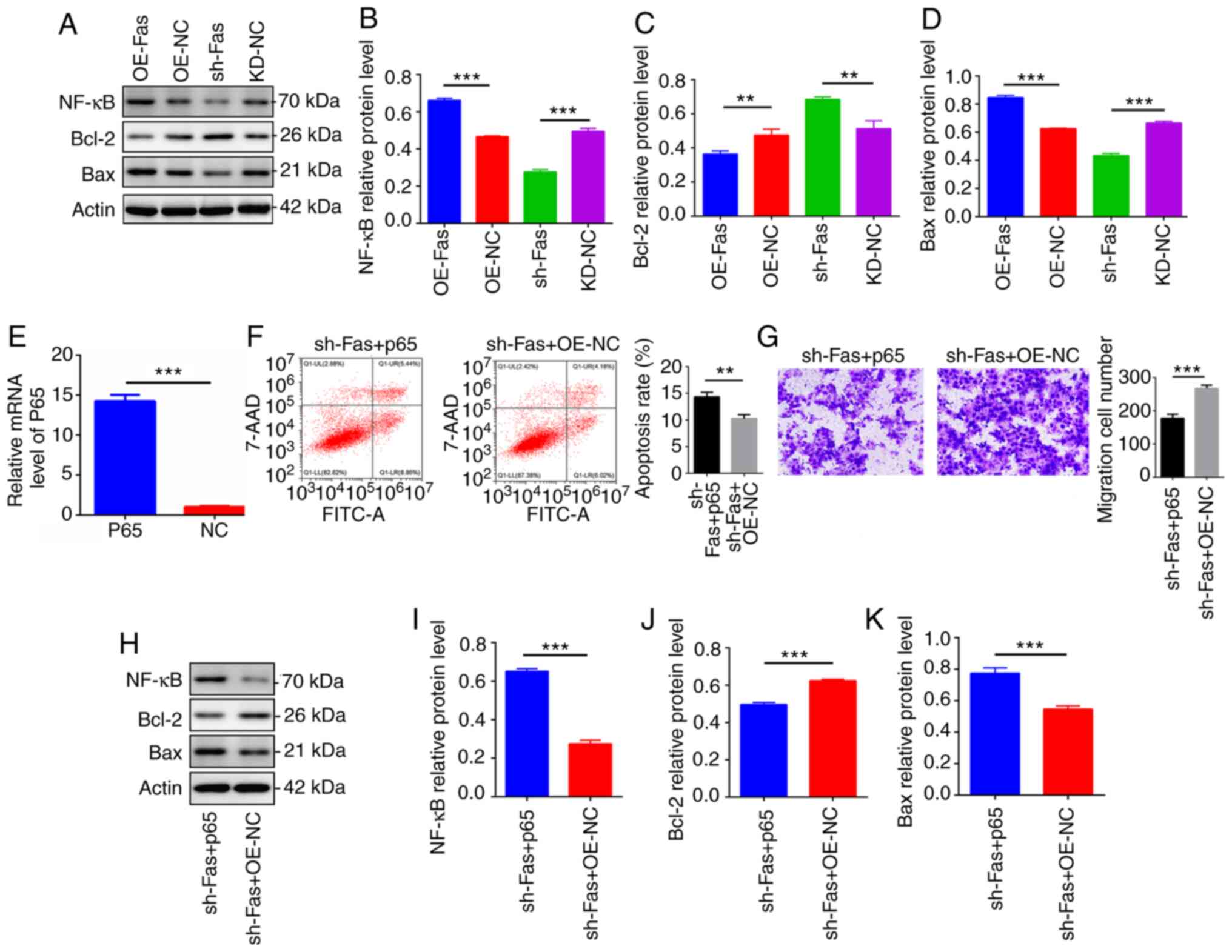 | Figure 3Overexpression of NF-κB reversed the
Fas knockdown-mediated promotion of apoptosis and inhibition of
cell migration. (A) Protein levels of NF-κB, Bcl-2 and Bax in
trophoblast cells transfected with OE-Fas, OE-NC, shRNA-Fas and
KD-NC examined using western blot analysis. The protein levels of
(B) NF-κB, (C) Bcl-2 and (D) Bax were quantified by densitometry
and normalized to the expression of β-actin. (E) Measurement of the
mRNA level of NF-κB p65 in the trophoblast cells confirmed the
transfection efficiency of the NF-κB p65 overexpression vector. (F)
Trophoblast cell apoptosis in the sh-Fas + p65 and sh-Fas + OE-NC
groups was evaluated using flow cytometry and the percentage of
apoptotic cells in each group is shown. (G) The migration ability
of trophoblast cells in the sh-Fas + p65 and sh-Fas + OE-NC groups
was examined using a Transwell assay (magnification, x200).
Quantification of the migration rate in three separate experiments
is shown. (H) Representative western blots showing the protein
levels of NF-κB, Bcl-2 and Bax in the sh-Fas + p65 and sh-Fas +
OE-NC groups of trophoblast cells. Protein levels of (I) NF-κB, (J)
Bcl-2 and (K) Bax were quantified by densitometry and normalized to
the expression of β-actin. Results are expressed as the mean ± SD
(n=3). **P<0.01 and ***P<0.001. OE,
overexpression; NC, negative control; KD, knockdown; sh, short
hairpin; NF-κB, nuclear factor κB. |
Discussion
Women with preeclampsia are more likely than those
without preeclampsia to have pre-term births (22). Preeclampsia may lead to
eclampsia-associated complications, including shaking, coma, heart
problems, placental abruption and renal impedance (23). Studies have reported that
trophoblast migration is associated with the onset of preeclampsia
(4,24).
Trophoblast apoptosis has been confirmed to occur in
the placental tissue during pregnancy (25). The excessive apoptosis of placental
trophoblasts leads to shallow implantation and the failure of
uterine spiral artery reconstruction. Eventually, this decreases
placental blood perfusion, leading to preeclampsia. Therefore, the
apoptosis and migration of placental trophoblasts are important
processes contributing to the development of preeclampsia (23,26).
FasL is a natural ligand of Fas that is encoded by a
gene with five exons located on chromosome 1q23 region. It belongs
to the TNF family and is a type I membrane protein. The FasL cDNA
contains 1,623 nucleotides that encode a peptide composed of 278
amino acids linking with a signal sequence at the N-terminus
(24). Several studies have
reported that Fas and FasL gene polymorphisms play important roles
in the development of preeclampsia (27-29).
However, the involvement of Fas in the apoptosis, viability and
migration of placental trophoblasts remains unclear. The present
study demonstrated that the overexpression of Fas in trophoblast
cells induced apoptosis and decreased the viability and migration
of the cells. Moreover, it increased the expression of TNF-α, IL-2
and Bax, and decreased the expression of Bcl-2. These results
indicate that increased Fas expression stimulated an inflammatory
response and cell apoptosis. By contrast, the knockdown of Fas
suppressed the apoptosis of trophoblast cells, and significantly
increased trophoblast viability and migration. These results are
consistent with the role served by Fas in osteosarcoma (30), cervical cancer (31), lung cancer (32) and breast cancer (33). The knockdown of Fas decreased the
expression of TNF-α, IL-2 and Bax, and increased the expression of
Bcl-2. These results indicate that the knockdown of Fas suppressed
the inflammatory response and apoptosis of the trophoblast
cells.
NF-κB has been studied as an inflammatory activator
in preeclampsia (34-36).
However, the specific mechanism underlying the effects of NF-κB in
preeclampsia has not been elucidated. The present study has
demonstrated that Fas is able to regulate the expression of NF-κB.
Notably, the overexpression of NF-κB reversed the Fas
knockdown-mediated suppression of cell apoptosis and increase in
the migration of trophoblast cells. These results suggest that
NF-κB may be regulated by Fas in preeclampsia.
The present study has certain limitations. The
potential interaction between Fas and NF-κB was not verified. In
addition, only in vitro experiments were performed, and it
is necessary to establish an animal model to validate the role of
Fas in preeclampsia. Another limitation of the present study is
that only one cell line, JAR, was used. Therefore, the findings of
the present study require validation in other cell lines and animal
models in the future.
In conclusion, the present study suggests that the
Fas gene regulates the apoptosis and migration of trophoblast cells
by targeting NF-κB. These findings indicate the importance of Fas
in the induction of apoptosis and inhibition of migration of normal
trophoblast cells. As the present study demonstrates that Fas
inhibits the viability and migration of trophoblast cells by
targeting NF-κB, the silencing of Fas may be a promising
therapeutic strategy for the treatment of preeclampsia.
Acknowledgements
Not applicable.
Funding
Funding: This study was supported by the Natural Science
Foundation of Hainan Province (grant no. 818MS123).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
RL carried out cell detection and writing, YY
carried out cell detection and writing, JS completed the
construction of the vector, LW analyzed the data and HG provided
funding and experimental design. RL and YY confirm the authenticity
of all the raw data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
ORCID for Humin Gong: https://orcid.org/0000-0003-1739-5988.
References
|
1
|
Broumand F, Lak SS, Nemati F and Mazidi A:
A study of the diagnostic value of inhibin a tests for occurrence
of preeclampsia in pregnant women. Electronic Physician.
10:6186–6192. 2018.PubMed/NCBI View
Article : Google Scholar
|
|
2
|
Gestational hypertension and preeclampsia.
ACOG practice bulletin, number 222. Obstet Gynecol. 135:e237–e260.
2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Phipps EA, Thadhani R, Benzing T and
Karumanchi SA: Author correction: Pre-eclampsia: Pathogenesis,
novel diagnostics and therapies. Nat Rev Nephrol.
15(386)2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
James-Allan LB, Whitley GS, Leslie K,
Wallace AE and Cartwright JE: Decidual regulation of trophoblast is
altered in pregnancies at risk of pre-eclampsia. J Mol Endocrinol.
60:239–246. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ngene NC and Moodley J: Role of angiogenic
factors in the pathogenesis and management of pre-eclampsia. Int J
Gynaecol Obstet. 141:5–13. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Possomato-Vieira JS and Khalil RA:
Mechanisms of endothelial dysfunction in hypertensive pregnancy and
preeclampsia. Adv Pharmacol. 77:361–431. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kaufmann P, Black S and Huppertz B:
Endovascular trophoblast invasion: Implications for the
pathogenesis of intrauterine growth retardation and preeclampsia.
Biol Reprod. 1:1–7. 2003.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mol BWJ, Roberts CT, Thangaratinam S,
Magee LA, de Groot CJM and Hofmeyr GJ: Pre-eclampsia. Lancet.
387:999–1011. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zha W, Guan S, Liu N, Li Y, Tian Y, Chen
Y, Wang Y and Wu F: Let-7a inhibits Bcl-xl and YAP1 expression to
induce apoptosis of trophoblast cells in early-onset severe
preeclampsia. Sci Total Environ. 745(139919)2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wang D, Na Q, Song GY and Wang L: Human
umbilical cord mesenchymal stem cell-derived exosome-mediated
transfer of microRNA-133b boosts trophoblast cell proliferation,
migration and invasion in preeclampsia by restricting SGK1. Cell
Cycle. 19:1869–1883. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mo HQ, Tian FJ, Ma XL, Zhang YC, Zhang CX,
Zeng WH, Zhang Y and Lin Y: PDIA3 regulates trophoblast apoptosis
and proliferation in preeclampsia via the MDM2/p53 pathway.
Reproduction. 160:293–305. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Cai G, Si M, Li X, Zou H, Gu J, Yuan Y,
Liu X, Liu Z and Bian J: Zearalenone induces apoptosis of rat
Sertoli cells through Fas-Fas ligand and mitochondrial pathway.
Environ Toxicol. 34:424–433. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Mor G, Straszewski S and Kamsteeg M: Role
of the Fas/Fas ligand system in female reproductive organs:
Survival and apoptosis. Biochem Pharmacol. 64:1305–1315.
2002.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wang T and Lian Y: The relationship
between Fas and Fas ligand gene polymorphism and preeclampsia risk.
Biosci Rep. 39(BSR20181901)2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Shi L, Gong HM and Ru MY: The relationship
between Fas & Bcl-2 and cell apoptosis in placenta and their
significance in pathogenesis of preeclampsia. Mod Prev Med.
40:2421–2425. 2013.(In Chinese).
|
|
16
|
Shi L, Gong HM and Ru MY: Expressions and
significances of Fas/FasL and Bcl-2 in placentae of cases with
preeclampsia. Maternal and Child Health Care of China.
26:3291–3294. 2011.PubMed/NCBI(In Chinese).
|
|
17
|
Mitchell S, Vargas J and Hoffmann A:
Signaling via the NFκB system. Wiley Interdiscip Rev Syst Biol Med.
8:227–241. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zheng L, Shi L, Zhou Z, Chen X, Wang L, Lu
Z and Tang R: Placental expression of AChE, α7nAChR and NF-κB in
patients with preeclampsia. Ginekol Pol. 89:249–255.
2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Vaughan JE and Walsh SW: Activation of
NF-κB in placentas of women with preeclampsia. Hypertens Pregnancy.
31:243–251. 2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Vora S, Abbas A, Kim CJ, Summerfield TL,
Kusanovic JP, Iams JD, Romero R, Kniss DA and Ackerman WE IV:
Nuclear factor-kappa B localization and function within
intrauterine tissues from term and preterm labor and cultured fetal
membranes. Reprod Biol Endocrinol. 8(8)2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Davies EL, Bell JS and Bhattacharya S:
Preeclampsia and preterm delivery: A population-based case-control
study. Hypertens Pregnancy. 35:510–519. 2016.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Sones JL and Davisson RL: Preeclampsia, of
mice and women. Physiol Genomics. 48:565–572. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Namdev S, Bhat V, Adhisivam B and
Zachariah B: Oxidative stress and antioxidant status among neonates
born to mothers with pre-eclampsia and their early outcome. J
Matern Fetal Neonatal Med. 27:1481–1484. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Khovhaeva PA, Krasniy AM, Tyutyunnik NV,
Sergunina OA, Ganichkina MB, Amiraslanov EY, Kan NE and Tyutyunnik
VL: Placental apoptosis in pre-eclampsia. Med Counc. 102–104.
2016.(In Russian).
|
|
26
|
Ma XP, Liu CD, Cao GM and Zhang ZY:
Transthyretin increases migration and invasion of rat placental
trophoblast cells. FEBS Open Bio. 10:1568–1576. 2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Tang R, Mei X, Wang YC, Cui XB, Zhang G,
Li W and Chen SY: LncRNA GAS5 regulates vascular smooth muscle cell
cycle arrest and apoptosis via p53 pathway. Biochim Biophys Acta
Mol Basis Dis. 1865:2516–2525. 2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Han AR, Choi YM, Hong MA, Kim JJ, Lee SK,
Yang KM, Paik EC, Jeong HJ and Jun JK: Fas and FasL genetic
polymorphisms in women with recurrent pregnancy loss: A
case-control study. Hum Fertil (Camb). 22:198–203. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Darmochwal-Kolarz D, Leszczynska-Gorzelak
B, Rolinski J and Oleszczuk J: The expression and concentrations of
Fas/APO-1 (CD95) antigen in patients with severe pre-eclampsia. J
Reprod Immunol. 49:153–164. 2001.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Hollomon MG, Patterson L,
Santiago-O'Farrill J, Kleinerman ES and Gordon N: Knock down of
Fas-associated protein with death domain (FADD) sensitizes
osteosarcoma to TNFα-induced cell death. J Cancer. 11:1657–1667.
2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Tan SC, Ismail MP, Duski DR, Othman NH and
Ankathil R: FAS c.-671A>G polymorphism and cervical cancer risk:
A case-control study and meta-analysis. Cancer Genet. 211:18–25.
2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kiany S, Harrison D and Gordon N: The
histone deacetylase inhibitor entinostat/syndax 275 in
osteosarcoma. Adv Exp Med Biol. 1257:75–83. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kolben T, Jeschke U, Reimer T, Karsten N,
Schmoeckel E, Semmlinger A, Mahner S, Harbeck N and Kolben TM:
Induction of apoptosis in breast cancer cells in vitro by Fas
ligand reverse signaling. J Cancer Res Clin Oncol. 144:249–256.
2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Xue P, Fan W, Diao Z, Li Y, Kong C, Dai X,
Peng Y, Chen L, Wang H, Hu Y and Hu Z: Up-regulation of PTEN via
LPS/AP-1/NF-κB pathway inhibits trophoblast invasion contributing
to preeclampsia. Mol Immunol. 118:182–190. 2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Eddy AC, Howell JA, Chapman H, Taylor E,
Mahdi F, George EM and Bidwell GL III: Biopolymer-delivered,
maternally sequestered NF-κB (nuclear factor-κB) inhibitory peptide
for treatment of preeclampsia. Hypertension. 75:193–201.
2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kim S, Lee KS, Choi S, Kim J, Lee DK, Park
M, Park W, Kim TH, Hwang JY, Won MH, et al: NF-κB-responsive
miRNA-31-5p elicits endothelial dysfunction associated with
preeclampsia via down-regulation of endothelial nitric-oxide
synthase. J Biol Chem. 293:18989–19000. 2018.PubMed/NCBI View Article : Google Scholar
|