Introduction
Colorectal cancer (CRC) contributes to 1.2 million
new cases and 700,000 mortalities every year (1). This disease remains the fourth leading
cause of cancer-related mortality worldwide after lung, stomach and
liver cancer (1,2). CRC is considered a primary public
health issue in the majority of industrialized countries (3). Unfortunately, ≥80% of patients with
CRC are diagnosed at advanced stages, leading to low response to
treatment and poor survival (4).
This suggests the necessity for identifying novel molecular targets
to specifically inhibit oncogenic processes.
Based on the recent advances of medical science and
technology, as well as the improved understanding of the
progression and molecular pathogenesis of CRC, additional targeted
therapies have been applied for the treatment of patients with this
disease (5). Despite these
achievements, the clinical prognosis of patients with CRC remains
unsatisfactory (6). 5-Fluorouracil
(5-FU) is an important constituent of the systemic chemotherapy in
the palliative and adjuvant treatments used for patients with CRC
(7). However, its clinical efficacy
in patients with CRC is low due to increased chemoresistance
(8).
MicroRNAs (miRs) are ~22 nucleotides in length and
inhibit protein expression by targeting coding genes (9). Generally, miRs are complementary to
the site of the 3'untranslated region (UTR) in their target mRNAs
(10). miR-based target therapies
that exhibit substantial efficacy in cancer treatment have been
identified in a previous study, including breast cancer, glioma and
lung cancer (11). Numerous miRNAs
have been reported to regulate the chemoresistance in CRC. For
instance, miR-27a promotes CRC resistance to chemotherapy by
promoting aerobic glycolytic metabolism, which results in excessive
proliferation (12). In addition,
exosome-transmitted miR-128-3p has been shown to promote
chemosensitivity of oxaliplatin-resistant CRC by inhibiting
epithelial-mesenchymal transition and inducing intracellular
oxaliplatin accumulation (13).
However, the interaction of miR-3135b with the sensitivity of CRC
tumors to 5-FU remains unknown. The present study aimed to
investigate the potential association between these two
parameters.
Materials and methods
Clinical samples
In total, 40 tumor tissue samples and their
corresponding adjacent tissue samples, which were ≥5 cm distal to
the tumor margins, were extracted from 40 patients (28 males and 12
females) with CRC who underwent surgery in the ChinaJapan Union
Hospital of Jilin University (Changchun, China) between February
2018 and May 2019. These patients had stage III CRC tumors and were
aged between 45 and 85 years (63.5±11.7 years). Among these
patients, 34 received 5-FU treatment. A total of 17 patients who
received 5-FU treatment experienced recurrence during follow-up.
The present study was approved by the Ethics Committee of
China-Japan Union Hospital of Jilin University. Written informed
consent was obtained from all patients with CRC with the following
inclusion criteria: i) Aged >18 years; ii) Confirmed diagnosis
of CRC by surgical pathological diagnosis (14); iii) Are aware they will receive
adjuvant chemotherapy; and iv) Can communicate with the medical
professional team. In the present study, patients with CRC who
received neoadjuvant therapy were excluded from the study.
Cell culture
Normal human colon epithelial NCM460 cells, human
colon adenocarcinoma SW480 cell line and human colon carcinoma
HCT116 cells were purchased from the American Type Culture
Collection. The cells were grown in DMEM (Invitrogen; Thermo Fisher
Scientific, Inc.) supplemented with 10% FBS (Invitrogen; Thermo
Fisher Scientific, Inc.) and 50 U/ml penicillin-streptomycin
(Invitrogen; Thermo Fisher Scientific, Inc.) in a 5% CO2
humidified atmosphere at 37˚C.
Bioinformatics analysis
Clinical and miR microarray expression data from 119
non-recurrent and 73 recurrent patients receiving 5-FU treatment
were retrieved from the Gene Expression Omnibus (https://www.ncbi.nlm.nih.gov/geo/; accession. no.
GSE81653). The differential expression analysis was conducted using
GEO2R (https://www.ncbi.nlm.nih.gov/geo/geo2r/). mRNAs that
could be potential targets of miR-3135b were predicted using
TargetScan software 7.2 (http://www.targetscan.org/vert_72/).
Transfection
The cells were transfected with miR-3135b mimic (30
nM, 5'-GGCUGGAGCGAGUGCAGUGGUG-3'), pcDNA3.1- PIM1 (20 µg),
miR-negative control (NC; 60 nM, 5'-UUCUCCGAACGUGUCACGUTT-3') and
pcDNA3.1, which were obtained from Shanghai GenePharma Co., Ltd.,
using Lipofectamine® 2000 (Thermo Fisher Scientific,
Inc.) according to the manufacturer's instructions. The cells were
seeded in six-well plates (2x105 cells/well) and
incubated in DMEM for 48 h at 37˚C until they reached 60-70%
confluence. Subsequently, the transfection efficiency was assessed
by reverse transcription-quantitative PCR (RT-qPCR) to evaluate the
expression levels of miR-3135b or PIM1 at 48 h
post-transfection.
Treatment with 5-FU
To determine 5-FU (Selleck Chemicals) sensitivity,
the cells were treated for 48 h at 37˚C with various concentrations
(5, 10, 20, 50 and 100 µM) of 5-FU.
Cell Counting Kit-8 (CCK8) assay
The cells were resuspended and seeded in 96-well
plates (5x103 cells/well). CCK-8 (Dojindo Molecular
Technologies, Inc.) was performed according to the manufacturer's
instructions to detect cell viability. Briefly, 10 µl CCK-8 reagent
was added to each well at 0, 24, 48, 72 and 96 h. Following
incubation for 2 h at 37˚C, the absorbance was recorded at 450 nm
in each well using a microplate reader.
Flow cytometric analysis
The induction of cellular apoptosis was quantified
by FITClabeled Annexin V and PI reagents from an Annexin VFITC
Apoptosis Detection Kit (EMD Millipore). The cells were seeded into
12-well plates (3x105 cells/well) and cultured for 48 h
at 37˚C. Subsequently, they were resuspended in 100 µl HEPES
buffer, followed with incubation of 5 µl Annexin V and 5 µl PI at
room temperature for 10 min in the dark according to the
manufacturer's instructions. The samples were analyzed using a BD
FACSCalibur™ flow cytometer (BD Biosciences) within 1 h. The
results were analyzed using FlowJo 10.2 software (FlowJo LLC).
RTqPCR
Total RNA from CRC tumor tissue samples and cell
lines was extracted using TRIzol® (Invitrogen; Thermo
Fisher Scientific, Inc.) according to the manufacturer's
instructions. Subsequently, the RNA was reverse transcribed into
cDNA using a PrimeScript™ RT Reagent Kit (Takara Bio, Inc.)
according to the following thermal cycling protocols: A total three
cycles of 37˚C at 15 min, termination at 85˚C for 5 sec, and
maintenance at 4˚C. qPCR was performed with a SYBR®
Premix Ex Taq™ kit (Takara Bio, Inc.) on a CFX-96 Real-Time PCR
Detection System (Bio-Rad Laboratories, Inc.) according to the
manufacturer's instructions. The thermal cycling protocols were as
follows: Denaturation at 95˚C for 30 sec; annealing at 95˚C for 5
sec; elongation at 60˚C for 30 sec for a total of 40 amplification
cycles. The primer sequences used were as follows: Forward,
5'-CCCGACAGTTTCGTCCTGAT-3' and reverse, 5'-ACCCGAAGTCGATGAGCTTG-3'
for PIM1; forward, 5'-ACAGAGCCTCGCCTTTGCCGAT-3' and reverse,
5'-CTTGCACATGCCGGAGCCGTT-3' for β-actin; forward,
5'-GGCTGGAGCGAGTGCAGTGGTG-3' and reverse,
5'-CACCACTGCACTCGCTCCAGCC-3' for miR-3135b; forward,
5'-CTCGCTTCGGCAGCACA-3' and reverse, 5'-AACGCTTCACGAATTTGCGT-3' for
U6. β-actin and U6 served as internal controls to normalize PIM1
mRNA and miR-3135b expression levels, respectively. The relative
PIM1 mRNA and miR-3135b expression levels were calculated using the
comparative 2-ΔΔCq method (15).
Western blot analysis
Total protein from the CRC cell lines was isolated
using Mammalian Protein Extraction solution (Thermo Fisher
Scientific, Inc.) supplemented with a protease inhibitor cocktail
(Merck KGaA). The protein concentration was analyzed using the
Bio-Rad DC Protein Assay (Bio-Rad Laboratories, Inc.) according to
the manufacturer's instructions. A total of 20 µg protein was
separated by 8% polyacrylamide gels and transferred to 0.2-µm
nitrocellulose membranes (Bio-Rad Laboratories, Inc.).
Subsequently, the membranes were blocked with 5% non-fat milk for
60 min at 37˚C and incubated overnight at 4˚C with primary
antibodies against PIM1 (cat. no. ab245417; dilution, 1:2,000) and
β-actin (cat. no. ab8227; dilution, 1:2,000), which were obtained
from Abcam. The following day, the membranes were incubated for 60
min at 37˚C with HRP goat anti-rabbit secondary antibody IgG
H&L preadsorbed (cat. no. ab7090; dilution, 1:5,000) obtained
from Abcam. The bound antibodies were visualized by enhanced
chemiluminescence (Thermo Fisher Scientific, Inc.). Densitometry
was used for protein semi-quantification using ImageJ Software
version 1.8.0 (National Institutes of Health) and β-actin served as
the internal control.
Luciferase reporter gene assay
The sequence of PIM1 3'-UTR with the miR-3135b seed
region was amplified from the HCT116 cell cDNA and cloned into the
pGL2-control vector (Promega Corporation) and termed the
PIM1-wild-type (WT). The sequence of PIM13'-UTR with the mutated
miR-3135b seed region was obtained by introducing two-point
mutations into the pGL2-PIM1 plasmid using a Quick Site-Directed
Mutation Kit (Agilent Technologies Deutchland GmbH), which was
defined as PIM1-mutant (MUT). Subsequently, HCT116 cells were
seeded into a 24-well plate (3x104 cells/well) and
co-transfected with the corresponding vector (100 ng pGL2-PIM1-WT
or pGL2-PIM1-MUT) and miR-NC (100 ng) or miR-3135b mimic. (100 ng)
Following 48 h of incubation at 37˚C, luciferase activity was
determined using the Dual-Luciferase® Reporter Assay
(Promega Corporation) on an FB12 Luminometer (Titertek-Berthold).
Renilla luciferase activity served as the internal control
for normalizing the luciferase activity levels.
Statistical analysis
Statistical analysis was performed using Student's
t-test or one-way ANOVA followed by Tukey's multiple comparison
post hoc test. Tumor and adjacent non-tumor samples were analyzed
using paired Student's t-test. Analysis was conducted using
GraphPad Prism 6.0 (GraphPad Software, Inc.). The association
between miR-3135b and PIM1 mRNA levels was evaluated using the
Pearson correlation coefficient. P<0.05 was considered to
indicated a statistically significant difference and all values are
presented as means ± SD. For each assay, there were three
independent experimental repeats.
Results
Increased miR-3135b expression levels
enhance the sensitivity of CRC cells to 5-FU treatment
To investigate the key miRs involved in the
mechanism of CRC resistance to 5-FU, the microarray data of miR
expression levels were processed (accession. no. GSE81653). The
analysis involved 192 patients with CRC who received 5-FU treatment
and comprised two main groups defined as 5-FU non-recurrence
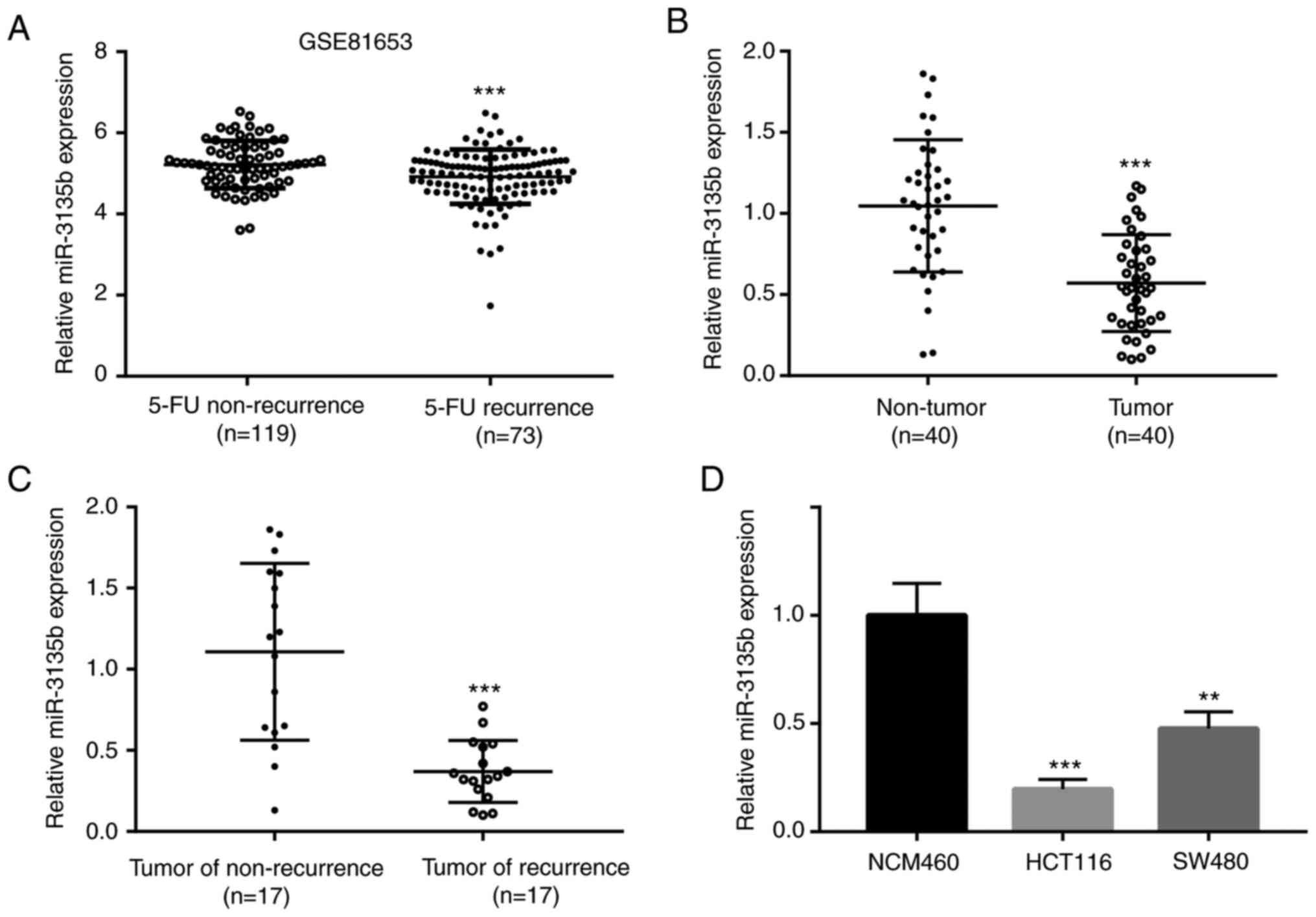
(n=119) and 5-FU recurrence (n=73). miR-3135b was identified as one
of the differentially expressed miRs between these two groups. The
level of miR-3135b expression was lower in recurrent patients with
5-FU (n=73) when compared with that of those who were non-recurrent
(n=119) as determined by the microarray data (Fig. 1A). In addition, miR-3135b expression
levels were lower in tumor tissue samples (n=40) when compared with
non-tumor tissue samples (n=40; Fig.
1B and C). The level of
miR-3135b expression was not associated with age, sex, tumor site
and lymph node metastasis of patients with CRC (Table I). Similar findings were noted in
the tumor recurrent (n=17) and non-recurrent (n=17) groups
(Fig. 1B and C). Subsequently, the miR-3135b expression
levels were lower in CRC cell lines, including HCT116 and SW480
when compared with those observed in the NCM460 cells (Fig. 1D). The results indicated the tumor
suppressive function of miR-3135b levels in CRC and the recurrence
of CRC in patients that were administered with 5-FU.
 | Table IAssociation between miR-3135b
expression levels and clinical factors of patients with colorectal
cancer. |
Table I
Association between miR-3135b
expression levels and clinical factors of patients with colorectal
cancer.
| | Relative miR-3135b
expression | |
|---|
| Variable | Total | High | Low | P-value |
|---|
| Age (years) | | | | 0.527 |
|
<60 | 19 | 8 | 11 | |
|
≥60 | 21 | 12 | 9 | |
| Sex | | | | 0.082 |
|
Male | 28 | 11 | 17 | |
|
Female | 12 | 9 | 3 | |
| Tumor site | | | | 0.999 |
|
Colon | 23 | 11 | 12 | |
|
Rectum | 17 | 9 | 8 | |
| Lymph node
metastasis | | | | 0.515 |
|
Yes | 15 | 6 | 9 | |
|
No | 25 | 14 | 11 | |
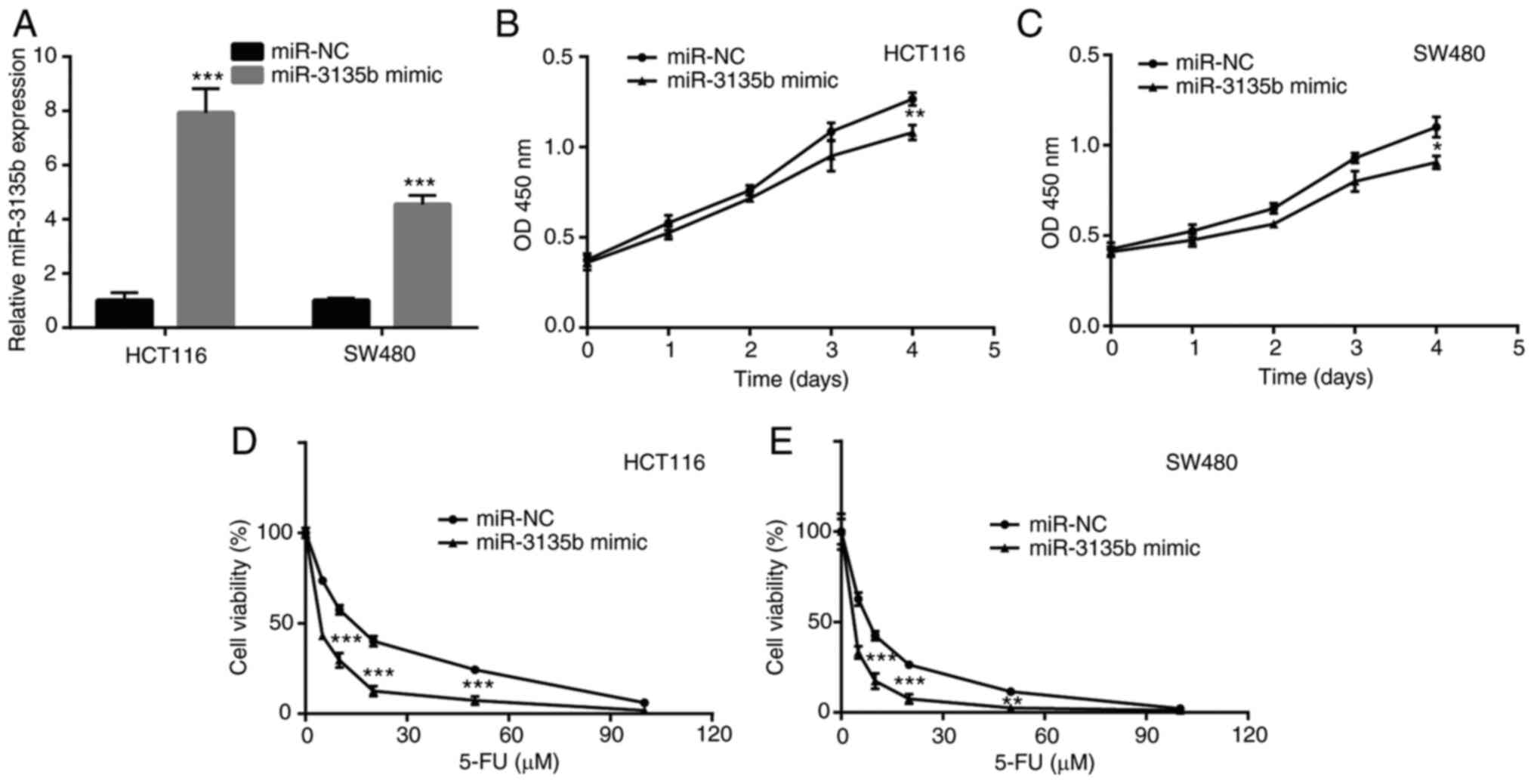
The function of miR-3135b in CRC cell proliferation
and the sensitivity of CRC cells to 5-FU were also investigated.
Initially, HCT116 and SW480 cells were transiently transfected with
miR-3135b mimic and miR-NC. The increase in miR-3135b expression
indicated successful transfection (Fig.
2A). The miR-3135b mimic significantly reduced HCT116 and SW480
cell viability (Fig. 2B and
C). In addition, the miR-3135b
mimic caused a significant increase in the sensitivity of HCT116
and SW480 cells to 5-FU (Fig. 2D
and E).
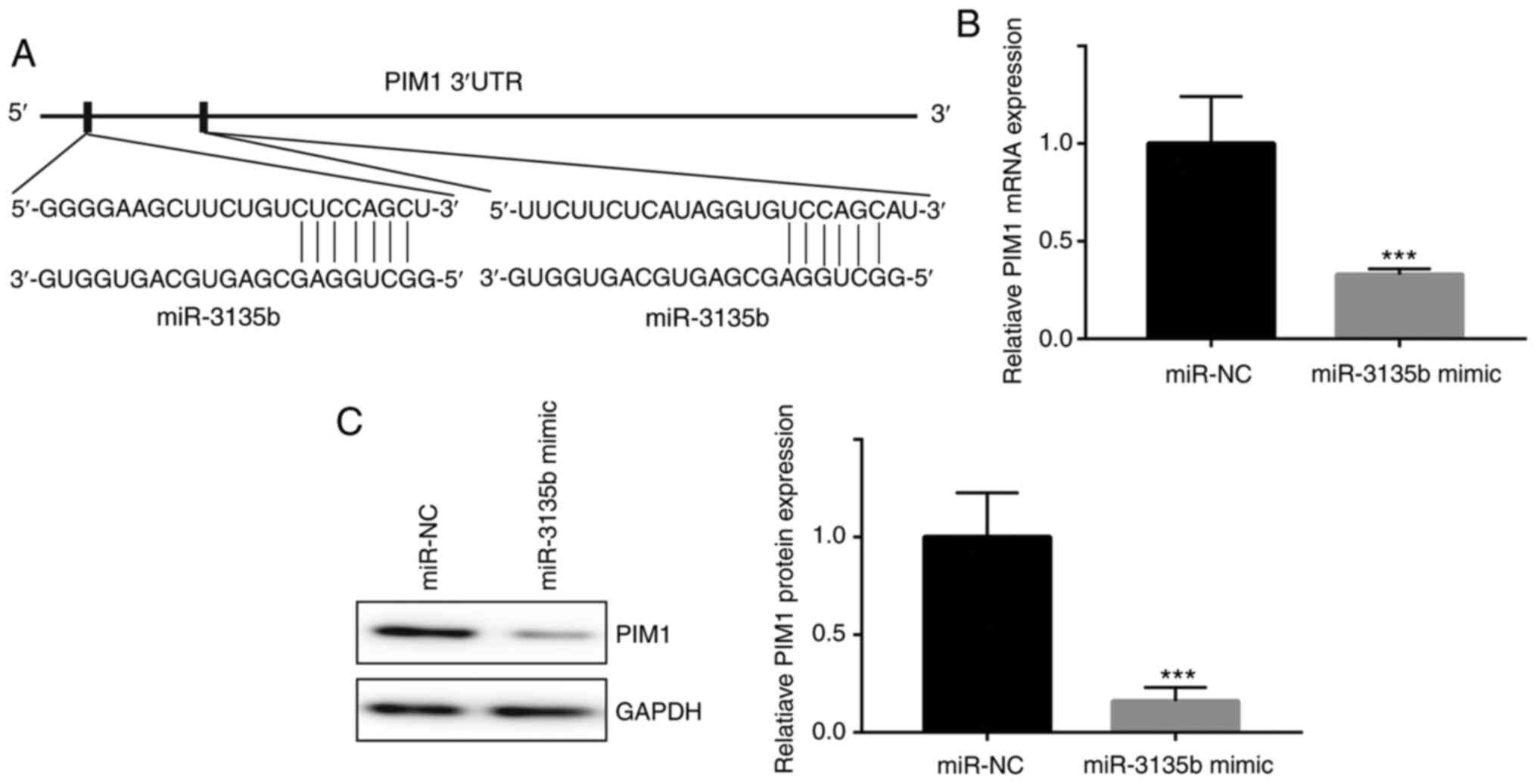
PIM1 is a target gene of
miR-3135b
Bioinformatics analysis identified two different
complementary sites of PIM1 that could bind to miR-3135b (Fig. 3A). miR-3135b mimic significantly
inhibited the relative mRNA expression levels of PIM1 when compared
with those of the miR-NC group (Fig.
3B). In addition, similar effects were induced by miR-3135b
mimic on the expression levels of PIM1 (Fig. 3C).
To verify the direct interaction between miR-3135b
and PIM1, luciferase reporter assays were performed in HCT116
cells. Three sequences were constructed including one full-length
(FL) PIM1 3'-UTR and two truncated forms, namely the truncate 1
(Trun1) form of PIM1 3'-UTR and Trun2 (Fig. 4A). HCT116 cells were co-transfected
with FL, Trun1 or Trun2 of PIM1 3'-UTR and the miR-NC or miR-3135b
mimic. The luciferase activity was assessed and the data indicated
that the lowest activity levels were observed in FL, followed by
Trun1 (Fig. 4B). The activity
levels were not decreased in the Trun2 group (Fig. 4B), suggesting that the first binding
site between PIM1 3'-UTR and miR-3135b was more important. The
combining sites between miR-3135b and PIM1, as well as the Mut
sequences of PIM1, are presented in Fig. 4C. The miR-3135b mimic significantly
inhibited the relative luciferase activity of HCT116 cells
transfected with PIM1 3'-UTR-WT when compared with miR-NC, whereas
it did not have a significant effect on HCT116 cells transfected
with PIM1 3'UTR-Mut (Fig. 4D).
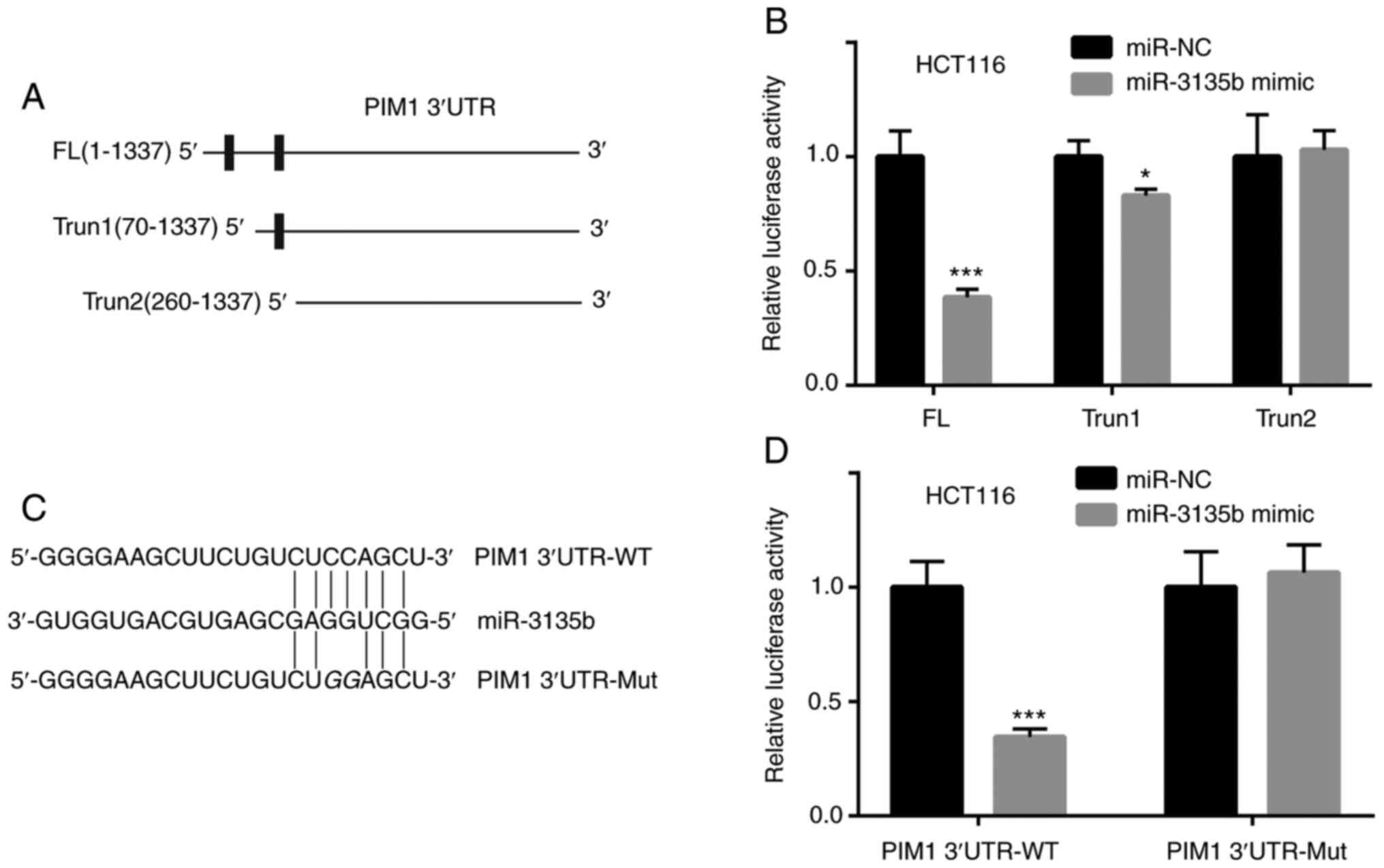 | Figure 4miR-3135b targeted the PIM1 3'UTR. (A)
FL, Trun1 and Trun2 of PIM1 3'UTR are presented. (B) Compared with
miR-NC, the relative luciferase activity of HCT116 cells
co-transfected with FL was significantly decreased by the miR-3135b
mimic, whereas in the HCT116 cells co-transfected with miR-3135b
mimic and Trun1 the decrease was significant, but the difference
was less. The HCT116 cells co-transfected with Trun2 were not
affected by the miR-3135b mimic. (C) The combining sites between
miR-3135b and PIM1, as well as the Mut sequences of PIM1 are
presented. (D) The miR-3135b mimic significantly inhibited the
relative luciferase activity of HCT116 cells transfected with PIM1
3'UTR-WT, but not PIM1 3'UTR-Mut when compared with miR-NC.
*P<0.05 and ***P<0.001 vs. miR-NC. miR,
microRNA; PIM1, proviral integration site for Moloney murine
leukemia virus 1; UTR, untranslated region; FL, full-length; Trun,
truncated; Mut, mutant; WT, wild-type; NC, negative control. |
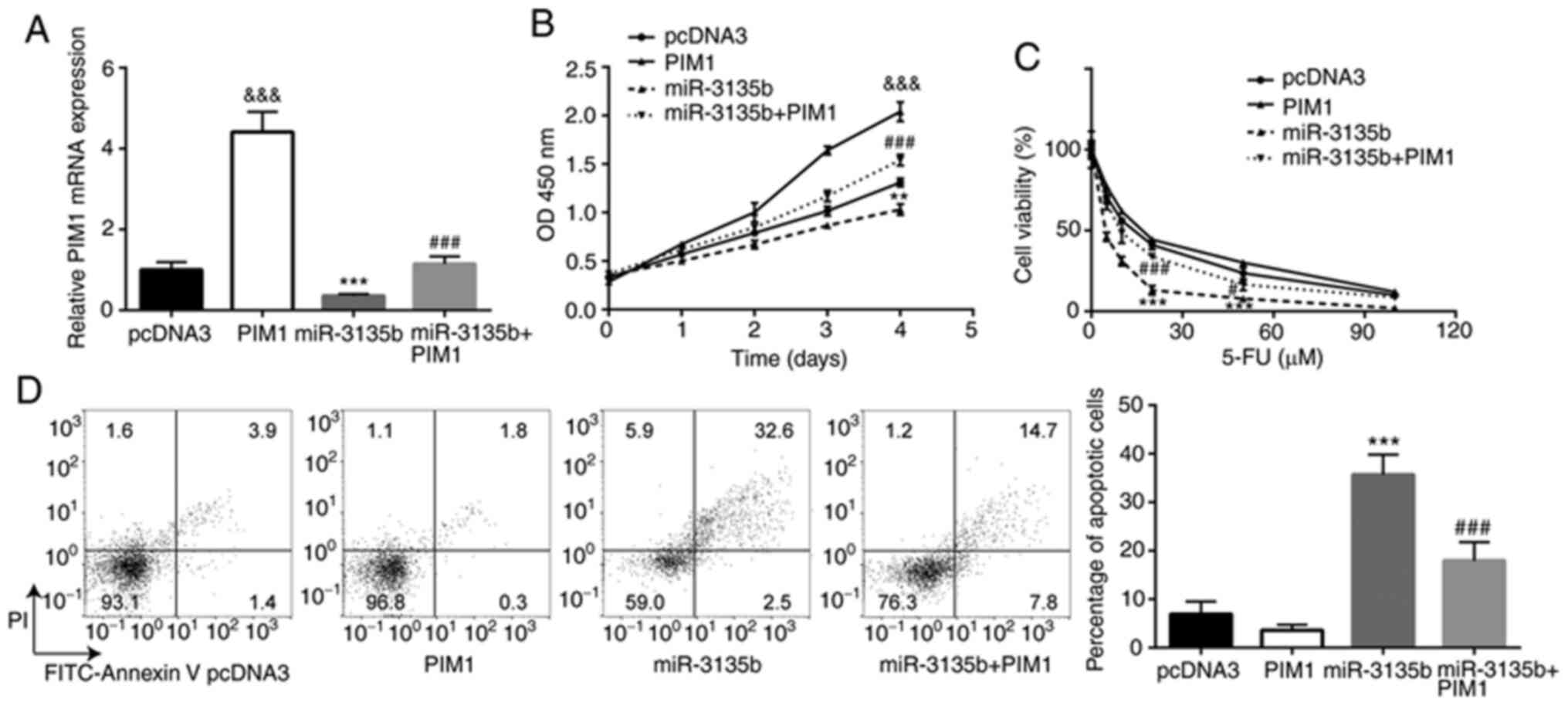
PIM1 overexpression compensates the
effects of miR-3135b in CRC cells
The interaction between miR-3135b and PIM1 was
assessed with regard to the sensitivity of HCT116 cells to 5-FU.
HCT116 cells were transiently transfected with pcDNA3-PIM1 and
pcDNA3. The higher PIM1 mRNA expression suggested that successful
transfection was achieved (Fig.
5A). pcDNA3-PIM1 also reversed the downregulation of PIM1
expression in cells treated with miR-3135b mimic (Fig. 5A). PIM1 overexpression promoted cell
proliferation in HCT116 cells, and miR-3135b overexpression
significantly reduced the cell viability of HCT116 cells when
compared with the pcDNA3 group, which was significantly compensated
by PIM1 overexpression (Fig. 5B).
In addition, while PIM1 overexpression showed little effect on 5-FU
sensitivity, miR-3135 overexpression significantly increased the
sensitivity of HCT116 cells to 5-FU, which was compensated by PIM1
overexpression (Fig. 5C). PIM1
overexpression showed little effect on cell apoptosis, whereas
miR-3135b overexpression caused a significant increase in cell
apoptosis of HCT116 cells when compared to the pcDNA3 group, which
was compensated by PIM1 overexpression (Fig. 5D).
Correlation analysis between PIM1 mRNA
and miR-3135b expression levels in CRC tumor samples
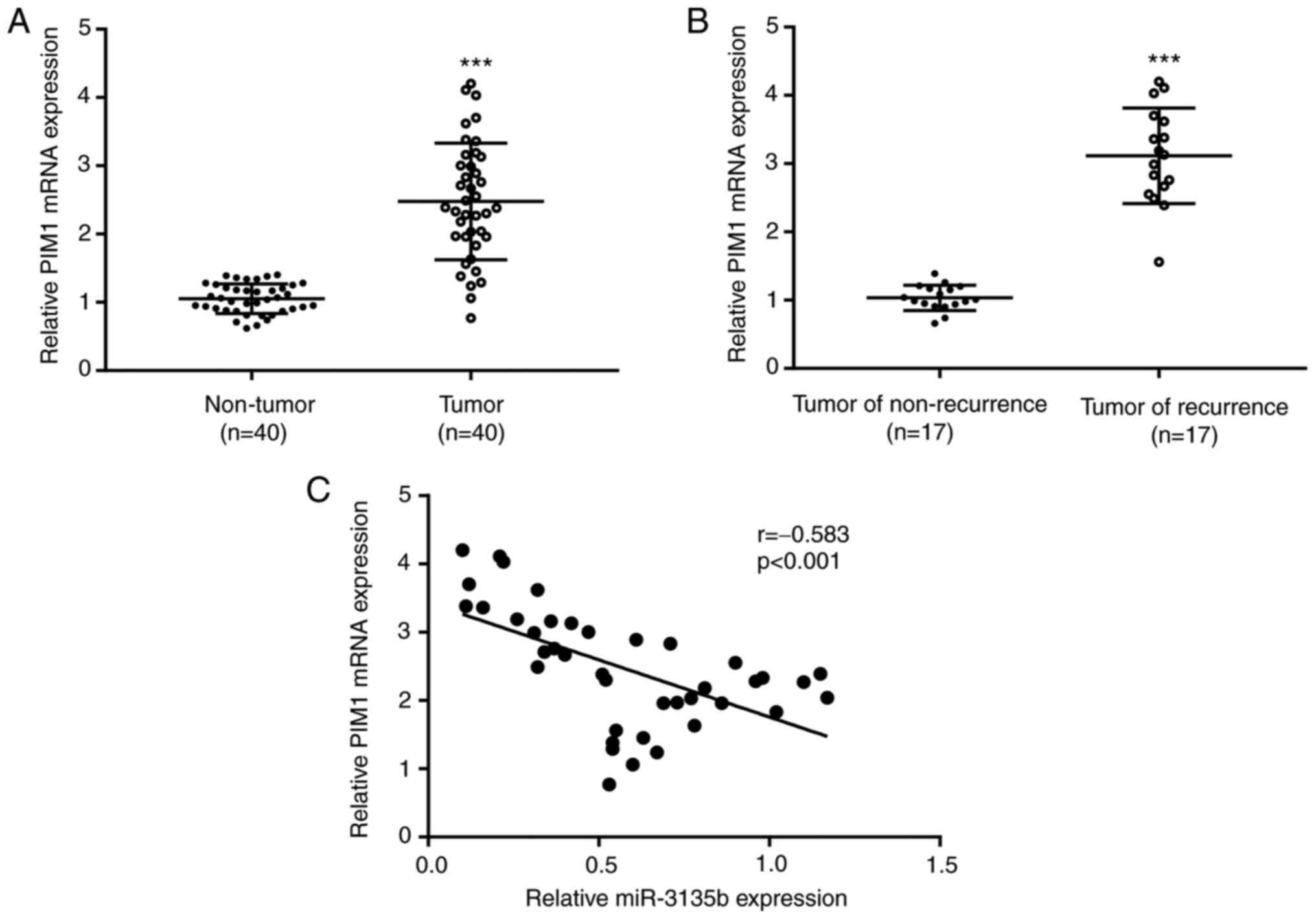
PIM1 expression levels were higher in tumor tissue
samples (n=40) when compared with those in non-tumor tissue samples
(n=40; Fig. 6A). Similar findings
were observed in PIM1 expression when comparing tumor recurrent
cases (n=17) with non-recurrent cases (n=17; Fig. 6B). In addition, an inverse
correlation (r=-0.583) was identified between PIM1 mRNA levels and
miR-3135b expression in CRC tumor tissue samples (Fig. 6C).
Discussion
In the past decade, significant advances have been
made with regard to investigating the molecular mechanisms of tumor
resistance to 5-FU. Substantial evidence has been derived from
pre-clinical models and clinical trials (16). Considerable attention has been paid
to miR dysregulation and the molecular events associated with this
process (17). For example, miR-552
downregulation promotes 5-FU resistance by targeting SMAD2 in CRC
(18). miR-543 deficiency enhances
5-FU chemosensitivity of CRC cells by targeting PTEN (19). miR-200c can be used as a predictive
biomarker for 5-FU sensitivity in CRC (20). In the present study, microarray data
analysis indicated that the level of miR-3135b expression was
decreased in patients with CRC who experienced disease recurrence
following 5-FU treatment compared with those who were
non-recurrent. However, to date, to the best of our knowledge, the
role of miR-3135b in the sensitivity of CRC to 5-FU has not been
investigated.
In the present study, a decrease was observed in the
expression levels of miR-3135b in patients with recurrent CRC who
were treated with 5-FU when compared with those who were
non-recurrent to the disease. Moreover, overexpression of miR-3135b
was shown to increase the sensitivity of CRC cells to 5-FU. Taken
together, the aforementioned findings demonstrated for the first
time, to the best of our knowledge, the tumor suppressive role of
miR-3135b in CRC and the effects of its increased expression on the
sensitivity of CRC to 5-FU.
miRs are complementary to their target mRNA 3'-UTR
(10). The present study identified
PIM1 as a novel target gene of miR-3135b in CRC cells. As a member
of the constitutively activated serine/threonine kinases, PIM1 was
initially identified as a proviral integration site for the Moloney
murine leukemia virus in 1989(21).
Overexpression of PIM1 is observed in multiple cancer cell types,
including glioblastoma (22), lung
cancer (23) and CRC (24), and is associated with their
development and progression. In addition, PIM1 is associated with
cell apoptosis (25), cell
proliferation (26), tumor growth
(26) and chemotherapy response
(27) in various cancer types.
Furthermore, PIM1 inhibits cancer cell sensitivity to 5-FU. Human
antigen R induces hypoxia-mediated chemoresistance by upregulation
of PIM1 protein in pancreatic cancer (28). Based on this evidence, the
development of combinatorial therapies for CRC treatment can be
mediated by increasing 5-FU sensitization following PIM1 knockdown,
which is targeted by miR-15b (29).
However, to the best of our knowledge, the interaction between PIM1
and miR-3135b in CRC and its effects on the sensitivity of CRC
cells to 5-FU have not previously been reported.
The present study demonstrated that PIM1
overexpression compensated the effect of miR-3135b in CRC cells. An
inverse correlation between PIM1 mRNA expression and miR-3135b
expression was also observed. The downregulation of miR-3135b in
CRC cell lines and clinical tumor samples has recently been
reported; ectopic expression of miR-3135b sensitized cells to 5-FU
by targeting Golgi phosphoprotein-3(30). These observations are consistent
with the present findings on the effects of miR-3135b on the
sensitivity of CRC cells to 5-FU.
Therefore, the present study identified miR-3135b as
a novel regulator of 5-FU sensitivity in CRC by targeting PIM1,
implicated a potential new therapeutic target for the treatment of
patients with CRC. However, there were certain limitations of the
present study. Further investigation into whether miR-3135b
regulates 5-FU resistance in established 5-FU resistance cell lines
is required. Furthermore, the function and mechanism of miR-3135b
in animals, such as nude mice, will be investigated in future
work.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the International
Cooperation of Jilin Provincial Science & Technology Department
(grant. nos. 20150101175JC and 20190201221JC) and the National
Natural Science Foundation of China (grant. nos. 81172000 and
30772488).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YW, XC and SM performed the experiments, analyzed
the data, and confirm the authenticity of all the raw data. HZ
designed the experiments. All authors read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of ChinaJapan Union Hospital of Jilin University
(Changchun, China). Written informed consent was provided by all
patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Brody H: Colorectal cancer. Nature.
521(S1)2015.PubMed/NCBI View
Article : Google Scholar
|
|
3
|
Pourhoseingholi MA: Increased burden of
colorectal cancer in Asia. World J Gastrointest Oncol. 4:68–70.
2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
García-Osogobio S, Téllez-Ávila FI, Méndez
N and Uribe-Esquivel M: Results of the first program of colorectal
cancer screening in Mexico. Endoscopia. 27:59–63. 2015.
|
|
5
|
Arnold D, Prager GW, Quintela A, Stein A,
Vera SM, Mounedji N and Taieb J: Beyond second-line therapy in
patients with metastatic colorectal cancer: A systematic review.
Ann Oncol. 29:835–856. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Stintzing S: Management of colorectal
cancer. F1000Prime Rep. 6(108)2014.PubMed/NCBI View
Article : Google Scholar
|
|
7
|
Vodenkova S, Buchler T, Cervena K,
Veskrnova V, Vodicka P and Vymetalkova V: 5-fluorouracil and other
fluoropyrimidines in colorectal cancer: Past, present and future.
Pharmacol Ther. 206(107447)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Liu C, Zhao Y, Wang J, Yang Y, Zhang Y, Qu
X, Peng S, Yao Z, Zhao S, He B, et al: FoxO3 reverses
5-fluorouracil resistance in human colorectal cancer cells by
inhibiting the Nrf2/TR1 signaling pathway. Cancer Lett. 470:29–42.
2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297.
2004.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wang XJ, Reyes JL, Chua NH and Gaasterland
T: Prediction and identification of arabidopsis thaliana microRNAs
and their mRNA targets. Genome Biol. 5(R65)2004.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Trang P, Weidhaas JB and Slack FJ:
MicroRNAs as potential cancer therapeutics. Oncogene. 27 (Suppl
2):S52–S57. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Barisciano G, Colangelo T, Rosato V,
Muccillo L, Taddei ML, Ippolito L, Chiarugi P, Galgani M,
Bruzzaniti S, Matarese G, et al: miR-27a is a master regulator of
metabolic reprogramming and chemoresistance in colorectal cancer.
Br J Cancer. 122:1354–1366. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Liu T, Zhang X, Du L, Wang Y, Liu X, Tian
H, Wang L, Li P, Zhao Y, Duan W, et al: Exosome-transmitted
miR-128-3p increase chemosensitivity of oxaliplatin-resistant
colorectal cancer. Mol Cancer. 18(43)2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Choi Y, Choi HS, Jeon WK, Kim BI, Park DI,
Cho YK, Kim HJ, Park JH and Sohn CI: Optimal number of endoscopic
biopsies in diagnosis of advanced gastric and colorectal cancer. J
Korean Med Sci. 27:36–39. 2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Xie P, Mo JL, Liu JH, Li X, Tan LM, Zhang
W, Zhou HH and Liu ZQ: Pharmacogenomics of 5-fluorouracil in
colorectal cancer: Review and update. Cell Oncol (Dordr).
43:989–1001. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Blondy S, David V, Verdier M, Mathonnet M,
Perraud A and Christou N: 5-Fluorouracil resistance mechanisms in
colorectal cancer: From classical pathways to promising processes.
Cancer Sci. 111:3142–3154. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zhao P, Ma YG, Zhao Y, Liu D, Dai ZJ, Yan
CY and Guan HT: MicroRNA-552 deficiency mediates 5-fluorouracil
resistance by targeting SMAD2 signaling in
DNA-mismatch-repair-deficient colorectal cancer. Cancer Chemother
Pharmacol. 84:427–439. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Liu G, Zhou J and Dong M: Down-regulation
of miR-543 expression increases the sensitivity of colorectal
cancer cells to 5-Fluorouracil through the PTEN/PI3K/AKT pathway.
Biosci Rep. 39(BSR20190249)2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Dermani FK and Najafi R: miR-200c as a
predictive biomarker for 5-Fluorouracil chemosensitivity in
colorectal cancer. J Gastrointest Cancer. 49:102–103.
2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
van Lohuizen M, Verbeek S, Krimpenfort P,
Domen J, Saris C, Radaszkiewicz T and Berns A: Predisposition to
lymphomagenesis in pim-1 transgenic mice: Cooperation with c-myc
and N-myc in murine leukemia virus-induced tumors. Cell.
56:673–682. 1989.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Herzog S, Fink MA, Weitmann K, Friedel C,
Hadlich S, Langner S, Kindermann K, Holm T, Böhm A, Eskilsson E, et
al: Pim1 kinase is upregulated in glioblastoma multiforme and
mediates tumor cell survival. Neuro Oncol. 17:223–242.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Jiang R, Wang X, Jin Z and Li K:
Association of nuclear PIM1 expression with lymph node metastasis
and poor prognosis in patients with lung adenocarcinoma and
squamous cell carcinoma. J Cancer. 7:324–334. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhang M, Liu T, Sun H, Weng W, Zhang Q,
Liu C, Han Y and Sheng W: Pim1 supports human colorectal cancer
growth during glucose deprivation by enhancing the warburg effect.
Cancer Sci. 109:1468–1479. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Gu JJ, Wang Z, Reeves R and Magnuson NS:
PIM1 phosphorylates and negatively regulates ASK1-mediated
apoptosis. Oncogene. 28:4261–4271. 2009.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Li Q, Chen L, Luo C, ChenYan Ge J, Zhu Z,
Wang K, Yu X, Lei J, Liu T, et al: TAB3 upregulates PIM1 expression
by directly activating the TAK1-STAT3 complex to promote colorectal
cancer growth. Exp Cell Res. 391(111975)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Braso-Maristany F, Filosto S, Catchpole S,
Marlow R, Quist J, Francesch-Domenech E, Plumb DA, Zakka L,
Gazinska P, Liccardi G, et al: PIM1 kinase regulates cell death,
tumor growth and chemotherapy response in triple-negative breast
cancer. Nat Med. 22:1303–1313. 2016.PubMed/NCBI View
Article : Google Scholar
|
|
28
|
Blanco FF, Jimbo M, Wulfkuhle J, Gallagher
I, Deng J, Enyenihi L, Meisner-Kober N, Londin E, Rigoutsos I,
Sawicki JA, et al: The mRNA-binding protein HuR promotes
hypoxia-induced chemoresistance through posttranscriptional
regulation of the proto-oncogene PIM1 in pancreatic cancer cells.
Oncogene. 35:2529–2541. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Weirauch U, Beckmann N, Thomas M,
Grünweller A, Huber K, Bracher F, Hartmann RK and Aigner A:
Functional role and therapeutic potential of the pim-1 kinase in
colon carcinoma. Neoplasia. 15:783–794. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Núñez-Olvera SI, Chávez-Munguía B, Del
Rocío Terrones-Gurrola MC, Marchat LA, Puente-Rivera J, Ruíz-García
E, Campos-Parra AD, Vázquez-Calzada C, Lizárraga-Verdugo ER,
Ramos-Payán R, et al: A novel protective role for microRNA-3135b in
golgi apparatus fragmentation induced by chemotherapy via
GOLPH3/AKT1/mTOR axis in colorectal cancer cells. Sci Rep.
10(10555)2020.PubMed/NCBI View Article : Google Scholar
|