Fibroblast growth factor 23 (FGF23) expression is
elevated in early-stage chronic kidney disease (CKD), and continues
to increase as the glomerular filtration rate decreases (1-4).
FGF23 is a hormone derived from osteocytes that regulates the
metabolism of phosphorus and 1,25-dihydroxyvitamin D (1,25[OH]2D).
1,25(OH)2D and high dietary phosphate intake upregulate FGF23
expression, resulting in increased renal excretion of phosphate and
decreased synthesis of 1,25(OH)2D, which decreases FGF23
expression, thus completing a negative feedback loop (5). Recent studies have reported that iron
deficiency (6-9)
and erythropoietin (EPO) (10-13)
also affect FGF23 production. In addition, a study on mice reported
that applying hypoxia inducible factor-proline hydroxylase
inhibitors (HIF-PHIs) increases the serum level of FGF23(8). Several clinical studies on patients
with kidney disease have demonstrated that increased FGF23
expression is associated with poor patient outcomes (14-17).
High-dose EPO treatment also contributes to the high mortality and
morbidity rates of patients diagnosed with CKD (18,19).
Thus, it is important to determine the associations among FGF23,
1,25(OH)2D, EPO and HIFs to identify novel therapeutic targets for
the treatment of CKD-mineral bone disorder (MBD) and renal anemia,
and improve the prognosis of these patients. The present review
aimed to discuss the associations among FGF23, iron, EPO and HIFs
in CKD-MBD.
Iron deficiency, induced by overproduction of EPO or
activation of HIFs, upregulates FGF23 expression and is associated
with adverse events in patients with CKD (8). Thus, it is important to identify
interventions that downregulate FGF23 expression to correct iron
deficiency and improve patient outcomes.
A total of 79 single nucleotide polymorphisms (SNPs)
in 29 genes are associated with CKD-MBD (20). Among these, five SNPs (rs1126616,
rs35068180, rs1800247, rs4236 and rs2248359) are in proteins
involved in mineral metabolism (osteocalcin, osteopontin, Gla
protein matrix metalloprotease 3, and 24 hydroxylase), which may be
responsible for upregulating FGF23 expression (20). A Previous study has demonstrated
that FGF23 expression closely parallels plasma EPO expression,
either from administration of exogenous EPO or increased production
of endogenous EPO, induced by HIF-PHIs (12).
The present review compares the induction of FGF23
expression by EPO administration and HIF-PHIs, and discusses
optimal therapeutic regimens for CKD-related anemia and MBD. EPO is
associated with an increase in the inactive form of FGS23,
C-terminal (c)FGF23, which can result in adverse events (21). Thus, the biological activity of
cFGF23 requires further investigation.
Iron functions as a cofactor in several enzymatic
reactions and a critical component of hemoglobin, which is required
for normal oxygen transport (22).
Several factors can cause iron deficiency and anemia, including
pregnancy (23), poor diet
(24-26),
inflammation (27,28), iron malabsorption (29,30)
and CKD (31,32). A study on patients with CKD reported
an association between low iron levels and high cFGF23 expression
levels (33). Recently, Eisenga
et al (34) confirmed that
iron deficiency is associated with high serum FGF23 levels in
patients with CKD and kidney transplant recipients (9,34).
Molecular studies have reported that iron deficiency upregulates
furin by stabilizing HIF1-α, which subsequently cleaves FGF23 into
cFGF23 fragments (6,35,36).
Furthermore, Eisenga et al (37) demonstrated that increased FGF23
levels mediate the association between iron deficiency and
mortality. Iron deficiency, as a crucial determinant of FGF23
expression, can also occur during blood loss from trauma, surgery
and bowel dysfunction (38,39). Rabadi et al (11) observed that acute bleeding elevated
cFGF23 expression in healthy mice, and that the number of
transfusions was positively associated with cFGF23 expression in
patients in intensive care. It has also been demonstrated that
GalNac-transferase 3 bone marrow mRNA expression decreases in mice
following acute blood loss, which protects intact (i)FGF23 from
proteolysis by furin, and thus enhances FGF23 cleavage (40). These findings regarding the
regulation of FGF23 via iron deficiency are consistent with
previous studies (35,36). However, a recent study reported that
iFGF23 expression has a significantly negative association with
serum iron parameters in elderly men (41). Presumably, certain age-related
factors may affect the cleavage of FGF23 in individuals with iron
deficiency.
Previous studies have demonstrated that iron
deficiency increases cFGF23 expression, and that iFGF23 expression
may remain normal due to FGF23 cleavage (42). However, the factors that affect
FGF23 cleavage remain unclear. In particular, the functions of
cleaved fragments in patients with CKD, such as cFGF23, remain
largely unknown.
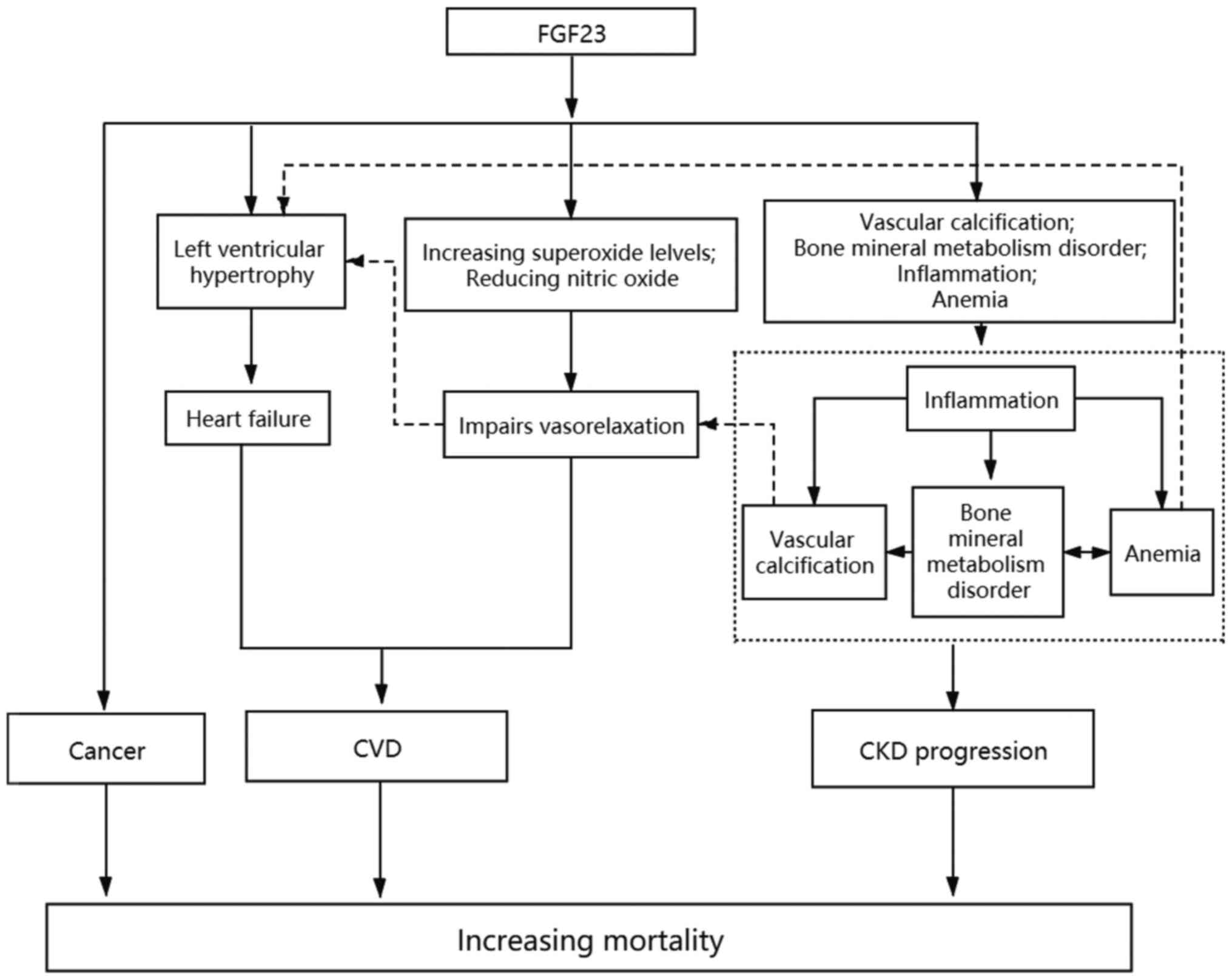
The physiological mechanisms underlying the
association between increased FGF23 expression and mortality are
presented in Fig. 1. FGF23 can
directly induce left ventricular hypertrophy and eventually lead to
heart failure, during which time oxidative stress enhances, the
level of nitric oxide decreases and vasodilatation is impaired.
FGF23 can increase the extent of vascular calcification and risk of
cardiovascular disease. Vascular calcification may promote
dysregulation of bone mineral metabolism, aggravate anemia and
inflammatory response, and contribute to the progression of CKD. In
addition, FGF23 increases the likelihood of cancer and mortality
risk, and these factors may interact with one another. For example,
inflammation may affect vasodilatation by promoting vascular
calcification directly or indirectly through bone and mineral
metabolism disorder. Anemia aggravates heart failure by increasing
left ventricular hypertrophy, and aggravates mineral metabolism
disorder. Conversely, bone mineral metabolism disorder and
inflammation further promote the occurrence of anemia (43). Given the effect of elevated FGF23
expression on mortality and other pathophysiological outcomes
(44-51),
it is important to understand the pathways and molecular mechanisms
underlying elevated FGF23 expression. As a major determinant of
FGF23, iron deficiency can be easily modified to decrease FGF23
expression. Thus, the potential therapeutic benefits of iron
supplementation should be considered. A previous study reported the
effect of iron supplementation on elevated iFGF23 expression in two
patients with osteomalacia (52).
Currently, two major iron formulations are used as supplements,
oral iron and intravenous (IV) iron. Patients with early-stage
(non-dialysis) CKD often receive oral iron supplementation as
treatment for iron deficiency and mild anemia. Clinicians
administer IV iron to circumvent gastrointestinal intolerance and
improve treatment efficacy, particularly in patients with
late-stage CKD who are receiving hemodialysis (53).
A prospective randomized study assessed the use of
oral iron and IV iron for 10 weeks as treatment for patients with
CKD who were receiving hemodialysis and had iron-deficiency anemia.
Serum cFGF23 levels decreased in both groups, while serum iFGF23
levels increased in the IV iron group (54). These findings suggest that oral iron
is superior to IV iron in preventing high iFGF23 expression levels.
However, the majority of patients with CKD also received EPO or
EPO-stimulating agents, the effects of which were not compared
between both groups. Thus, whether EPO administration affects the
iFGF23/cFGF23 ratio remains unclear.
Hyperphosphatemia, due to decreased phosphate
secretion, is significantly associated with CKD progression. Thus,
researchers have developed several novel phosphate binders as
treatment (55). Sucroferric
oxyhydroxide effectively decreases serum phosphorus levels
(56) and simultaneously improves
iron parameters (57). Notably,
serum FGF23 levels markedly decrease following treatment with
sucroferric oxyhydroxide in patients with CKD undergoing
hemodialysis (58). Ferric citrate,
another iron-based phosphate binder, efficiently decreases serum
phosphorus levels (59) and notably
improves iron parameters (60),
while simultaneously decreasing serum FGF23 levels in patients with
CKD undergoing hemodialysis (61).
Thus, concurrent reduction of serum phosphorus levels and
correction of iron deficiency may be used to effectively decrease
iFGF23 expression.
Taken together, these findings suggest that elevated
serum FGF23 levels contribute to high mortality in patients with
CKD. The correction of iron deficiency, a major determinant of
elevated FGF23 expression, can reverse high FGF23 expression
levels. Clinical studies have indicated that oral iron is superior
to IV iron. Iron-based phosphate treatment, which simultaneously
decreases phosphorus levels and corrects iron deficiency, may be
used to decrease serum FGF23 levels and improve the long-term
outcomes of these patients.
EPO is a hematopoietic hormone that is primarily
produced by the kidneys as a physiological response to iron
deficiency (62). Iron deficiency
and anemia are co-morbidities in CKD (63) that promote renal function decline
(64). There are several causes of
iron deficiency in CKD, but reduced EPO synthesis is the main
factor (65). Thus, clinicians
often administer rhEPO to correct iron deficiency and anemia in
these patients (66). However, the
effect of rhEPO administration on serum FGF23 levels remains
unclear. Previous studies have reported that acute injections of
rhEPO significantly increase the expression levels of
cFGF23(11) and iFGF23 (10,67) in
mice. Conversely, it has been demonstrated that rhEPO injections
only increase cFGF23 expression in mice with CKD (68). Notably, these observed effects were
independent of iron status. Studies on patients with chronic heart
failure and CKD have reported that exogenous EPO injections
markedly increase cFGF23 expression (69,70).
The potential reasons for these differences may be attributed to
the differences in renal functions of patients and/or the dose of
rhEPO used. A previous study demonstrated that high cFGF23
expression competes with iFGF23 by binding to the FGF receptor,
thereby decreasing iFGF23 expression (71). Another study on adult rats reported
that in vitro treatment with cFGF23 increases the surface
area of ventricular myocytes (72).
Thus, it is important to identify which specific fragments of FGF23
are responsible for its effect on mortality. Prospective studies
are required to assess the biological activity of different FGF23
fragments, and the effects of rhEPO replacement treatment on FGF23
expression in patients with CKD.
HIFs are proteins that regulate transcription in
response to low cellular oxygen levels. HIF prolyl-hydroxylase
(PHD) regulates HIFs in an oxygen-dependent manner (73). HIFs are heterodimers composed of an
α subunit and β subunit; HIF-α is oxygen-sensitive and stabilized
due to decreased PHD-dependent hydroxylation during hypoxia
(74-76).
HIF-PHI stabilizes HIFs by stimulating hypoxia, thereby activating
HIF signaling, inducing the transcription of endogenous EPO
(77), promoting iron uptake and
availability (78), and further
influencing FGF23 expression (8,79,80).
The HIF-PH inhibitor, roxadustat, has been used as an oral drug for
the treatment of anemia (81). This
drug increases FGF23 production, suggesting that HIF-PHI may affect
FGF23 expression and cleavage by inducing the transcription of
endogenous EPO (80). Generally,
FGF23 produced by osteocytes is affected by the level of phosphate,
parathyroid hormone and 1,25(OH)2D (82). However, a recent study reported that
other factors may regulate FGF23 production and cleavage, including
iron, HIF and EPO signaling (83).
Recent data have demonstrated that FGF23 expression may be directly
regulated by HIF signaling in osteogenic cells, or indirectly
regulated by EPO (12,82). Another study reported that treatment
with HIF-PHI increases FGF23 expression (8). Examinations of the underlying
molecular mechanism have demonstrated that inflammation or iron
deficiency induce HIF-1α (8), and
that binding of HIF-1α to the FGF23 promoter increases its
synthesis in osteogenic cells (84). In addition, HIF-1α may also
indirectly increase FGF23 expression by inducing EPO, thus
promoting FGF23 transcription and cleavage (10-12,68,85).
It has been reported that interleukin-6 and tumor necrosis factor-α
levels are induced during CKD, indicating a general
microinflammatory state in patients with CKD (86). FGF23 bioactivity and
anti-inflammatory cytokine expression are abnormal in patients with
CKD, which may be associated with the widespread microinflammatory
state (87).
Low 1,25(OH)2D expression is associated with
nutritional deficiency or endogenous resistance of EPO in patients
with CKD, and often contributes to anemia (88). However, these associations are not
affected by inflammatory status or secondary hyperparathyroidism
(89). 1,25(OH)2D3 upregulates HIF
in PMA-differentiated U937 cells, which is inhibited by rapamycin,
suggesting that mTOR signaling is also involved in this process
(90).
Administrating an anti-EPO antibody abrogates the
effect of EPO on upregulation of FGF23(91). This confirms that HIF-PHI increases
FGF23 expression by induction of EPO (8). Notably, the majority of FGF23 induced
via endogenous EPO following treatment with HIF-PHI is cFGF23,
instead of iFGF23 (8,12). However, the pathological
significance of increased expression levels of cFGF23 fragments
remains unclear. Normal plasma iFGF23 levels are maintained in
spite of increased FGF23 expression due to increased cleavage of
iFGF23(35). HIF-PHI efficiently
induces erythropoiesis due to upregulation of endogenous EPO, but
because the EPO level remains near the normal physiological range
this treatment is superior to traditional rhEPO administration for
treatment of renal anemia (92).
Thus, clinicians should consider the use of HIF-PHI as a novel
treatment strategy for renal anemia. A recent study reported that
iFGF23 expression is not associated with rhEPO dose in hemodialysis
patients (93). However, some
unrecognized confounders may affect the measurement of FGF23 during
CKD, which require further investigation in future studies.
In conclusion, iron deficiency, mediated by HIF1α
and EPO, independently increases FGF23 expression and promotes the
cleavage of FGF23, which is dependent on age and renal function.
HIF-PHI induces the transcription of endogenous EPO, although EPO
remains within its normal physiological range, and it also affects
FGF23 expression and cleavage (12). Further studies are required to
determine how endogenous or exogenous EPO increases FGF23
expression and affects FGF23 cleavage. Generally, endogenous EPO
levels are elevated during early-stage CKD (94), exogenous EPO is typically
administered to patients with late-stage CKD due to the presence of
renal anemia, and FGF23 cleavage may decrease as renal function
declines (95,96). iFGF23 is considered the bioactive
form, and high cFGF23 expression is associated with poor prognosis
in patients with CKD (17,69). Future studies are required to
determine the biological activities of the C-terminal fragments of
FGF23. The compensatory increase in FGF23 expression during the
early stages of CKD may enhance urinary phosphate excretion, which
may be beneficial to patients with CKD. The induction of FGF23
expression by EPO administration or HIF-PHIs may provide
therapeutic regimens for the treatment of patients with CKD. In
addition, the potential therapeutic benefits of iron
supplementation should be considered, particularly in early-stage
CKD with iron deficiency and mild anemia. However, the continuous
increase in FGF23 expression during late stages of CKD may
aggravate CKD-MBD and promote CKD-related anemia (97). A recent study suggested that
blockade of FGF23 signaling prevents renal anemia in a murine model
of CKD (21). Future studies must
consider the complex molecular and physiological interactions that
occur during renal anemia and CKD-MBD to develop novel therapeutic
interventions.
Not applicable.
Funding: The present review was supported by Jilin Province
Technology and Science Department (grant no. 20200201366JC).
Not applicable.
JBZ designed the present study and performed the
literature review. RZ drafted the initial manuscript. SYW and FY
analyzed the physiopathological mechanisms . SM, XL and CK
contributed to the interpretation of the molecular mechanisms. All
authors have read and approved the final manuscript. Data sharing
is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Isakova T, Wahl P, Vargas GS, Gutiérrez
OM, Scialla J, Xie H, Appleby D, Nessel L, Bellovich K, Chen J, et
al: Fibroblast growth factor 23 is elevated before parathyroid
hormone and phosphate in chronic kidney disease. Kidney Int.
79:1370–1378. 2011.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Larsson T, Nisbeth U, Ljunggren O, Juppner
H and Jonsson KB: Circulating concentration of FGF-23 increases as
renal function declines in patients with chronic kidney disease,
but does not change in response to variation in phosphate intake in
healthy volunteers. Kidney Int. 64:2272–2279. 2003.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Gutierrez O, Isakova T, Rhee E, Shah A,
Holmes J, Collerone G, Juppner H and Wolf M: Fibroblast growth
factor-23 mitigates hyperphosphatemia but accentuates calcitriol
deficiency in chronic kidney disease. J Am Soc Nephrol.
16:2205–2215. 2005.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Portale AA, Wolf M, Jüppner H, Messinger
S, Kumar J, Wesseling-Perry K, Schwartz GJ, Furth SL, Warady BA and
Salusky IB: Disordered FGF23 and mineral metabolism in children
with CKD. Clin J Am Soc Nephrol. 9:344–353. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Han X and Quarles LD: Multiple faces of
fibroblast growth factor-23. Curr Opin Nephrol Hypertens.
25:333–342. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Farrow EG, Yu X, Summers LJ, Davis SI,
Fleet JC, Allen MR, Robling AG, Stayrook KR, Jideonwo V, Magers MJ,
et al: Iron deficiency drives an autosomal dominant
hypophosphatemic rickets (ADHR) phenotype in fibroblast growth
factor-23 (Fgf23) knock-in mice. Proc Natl Acad Sci USA.
108:E1146–E1155. 2011.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Clinkenbeard EL, Farrow EG, Summers LJ,
Cass TA, Roberts JL, Bayt CA, Lahm T, Albrecht M, Allen MR, Peacock
M and White KE: Neonatal iron deficiency causes abnormal phosphate
metabolism by elevating FGF23 in normal and ADHR mice. J Bone Miner
Res. 29:361–369. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
David V, Martin A, Isakova T, Spaulding C,
Qi L, Ramirez V, Zumbrennen-Bullough KB, Sun CC, Lin HY, Babitt JL
and Wolf M: Inflammation and functional iron deficiency regulate
fibroblast growth factor 23 production. Kidney Int. 89:135–146.
2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hanudel MR, Chua K, Rappaport M, Gabayan
V, Valore E, Goltzman D, Ganz T, Nemeth E and Salusky IB: Effects
of dietary iron intake and chronic kidney disease on fibroblast
growth factor 23 metabolism in wild-type and hepcidin knockout
mice. Am J Physiol Renal Physiol. 311:F1369–F1377. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Clinkenbeard EL, Hanudel MR, Stayrook KR,
Appaiah HN, Farrow EG, Cass TA, Summers LJ, Ip CS, Hum JM, Thomas
JC, et al: Erythropoietin stimulates murine and human fibroblast
growth factor-23, revealing novel roles for bone and bone marrow.
Haematologica. 102:e427–e430. 2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Rabadi S, Udo I, Leaf DE, Waikar SS and
Christov M: Acute blood loss stimulates fibroblast growth factor 23
production. Am J Physiol Renal Physiol. 314:F132–F139.
2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Flamme I, Ellinghaus P, Urrego D and
Krüger T: FGF23 expression in rodents is directly induced via
erythropoietin after inhibition of hypoxia inducible factor proline
hydroxylase. PLoS One. 12(e0186979)2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Toro L, Barrientos V, León P, Rojas M,
Gonzalez M, González-Ibáñez A, Illanes S, Sugikawa K, Abarzua N,
Bascunan C, et al: Erythropoietin induces bone marrow and plasma
fibroblast growth factor 23 during acute kidney injury. Kidney Int.
93:1131–1141. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Souma N, Isakova T, Lipiszko D, Sacco RL,
Elkind MS, DeRosa JT, Silverberg SJ, Mendez AJ, Dong C, Wright CB,
et al: Fibroblast growth factor 23 and cause-specific mortality in
the general population: The Northern Manhattan Study. J Clin
Endocrinol Metab. 101:3779–3786. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Gutiérrez OM, Mannstadt M, Isakova T,
Rauh-Hain JA, Tamez H, Shah A, Smith K, Lee H, Thadhani R, Jüppner
H and Wolf M: Fibroblast growth factor 23 and mortality among
patients undergoing hemodialysis. N Engl J Med. 359:584–592.
2008.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Baia LC, Humalda JK, Vervloet MG, Navis G,
Bakker SJ and de Borst MH: NIGRAM Consortium. Fibroblast growth
factor 23 and cardiovascular mortality after kidney
transplantation. Clin J Am Soc Nephrol. 8:1968–1978.
2013.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Isakova T, Xie H, Yang W, Xie D, Anderson
AH, Scialla J, Wahl P, Gutierrez OM, Steigerwalt S, He J, et al:
Fibroblast growth factor 23 and risks of mortality and end-stage
renal disease in patients with chronic kidney disease. Jama.
305:2432–2439. 2011.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Szczech LA, Barnhart HX, Inrig JK, Reddan
DN, Sapp S, Califf RM, Patel UD and Singh AK: Secondary analysis of
the CHOIR trial epoetin-alpha dose and achieved hemoglobin
outcomes. Kidney Int. 74:791–798. 2008.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lestz RM, Fivush BA and Atkinson MA:
Association of higher erythropoiesis stimulating agent dose and
mortality in children on dialysis. Pediatr Nephrol. 29:2021–2028.
2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Valls J, Cambray S, Pérez-Guallar C, Bozic
M, Bermúdez-López M, Fernández E, Betriu À, Rodríguez I and
Valdivielso JM: Association of candidate gene polymorphisms with
chronic kidney disease: Results of a case-control analysis in the
Nefrona Cohort. Front Genet. 10(118)2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Agoro R, Montagna A, Goetz R, Aligbe O,
Singh G, Coe LM, Mohammadi M, Rivella S and Sitara D: Inhibition of
fibroblast growth factor 23 (FGF23) signaling rescues renal anemia.
FASEB J. 32:3752–3764. 2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Geissler C and Singh M: Iron, meat and
health. Nutrients. 3:283–316. 2011.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Brannon PM and Taylor CL: Iron
supplementation during pregnancy and infancy: Uncertainties and
implications for research and policy. Nutrients.
9(1327)2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Müller O and Krawinkel M: Malnutrition and
health in developing countries. CMAJ. 173:279–286. 2005.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Skalicky A, Meyers AF, Adams WG, Yang Z,
Cook JT and Frank DA: Child food insecurity and iron deficiency
anemia in low-income infants and toddlers in the United States.
Matern Child Health J. 10:177–185. 2006.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Díaz-Castro J, López-Frías MR, Campos MS,
López-Frías M, Alférez MJ, Nestares T, Ojeda ML and López-Aliaga I:
Severe nutritional iron-deficiency anaemia has a negative effect on
some bone turnover biomarkers in rats. Eur J Nutr. 51:241–247.
2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Cartwright GE, Lauritsen MA, Humphreys S,
Jones PJ, Merrill IM and Wintrobe MM: The anemia associated with
chronic infection. Science. 103:72–73. 1946.PubMed/NCBI
|
|
28
|
Cartwright GE, Lauritsen MA, Jones PJ,
Merrill IM and Wintrobe MM: The anemia of infection; hypoferremia,
hypercupremia, and alterations in porphyrin metabolism in patients.
J Clin Invest. 25:65–80. 1946.PubMed/NCBI
|
|
29
|
Qamar K, Saboor M, Qudsia F, Khosa SM and
Moinuddin Usman M: Malabsorption of iron as a cause of iron
deficiency anemia in postmenopausal women. Pak J Med Sci.
31:304–308. 2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Filmann N, Rey J, Schneeweiss S, Ardizzone
S, Bager P, Bergamaschi G, Koutroubakis I, Lindgren S, Morena Fde
L, Moum B, et al: Prevalence of anemia in inflammatory bowel
diseases in european countries: A systematic review and individual
patient data meta-analysis. Inflamm Bowel Dis. 20:936–945.
2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Gotloib L, Silverberg D, Fudin R and
Shostak A: Iron deficiency is a common cause of anemia in chronic
kidney disease and can often be corrected with intravenous iron. J
Nephrol. 19:161–167. 2006.PubMed/NCBI
|
|
32
|
Lankhorst CE and Wish JB: Anemia in renal
disease: Diagnosis and management. Blood Rev. 24:39–47.
2010.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Mehta R, Cai X, Hodakowski A, Lee J,
Leonard M, Ricardo A, Chen J, Hamm L, Sondheimer J, Dobre M, et al:
Fibroblast growth factor 23 and anemia in the chronic renal
insufficiency cohort study. Clin J Am Soc Nephrol. 12:1795–1803.
2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Eisenga MF, van Londen M, Leaf DE, Nolte
IM, Navis G, Bakker SJL, de Borst MH and Gaillard C: C-terminal
fibroblast growth factor 23, iron deficiency, and mortality in
renal transplant recipients. J Am Soc Nephrol. 28:3639–3646.
2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wolf M and White KE: Coupling fibroblast
growth factor 23 production and cleavage: Iron deficiency, rickets,
and kidney disease. Curr Opin Nephrol Hypertens. 23:411–419.
2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
McMahon S, Grondin F, McDonald PP, Richard
DE and Dubois CM: Hypoxia-enhanced expression of the proprotein
convertase furin is mediated by hypoxia-inducible factor-1: Impact
on the bioactivation of proproteins. J Biol Chem. 280:6561–6569.
2005.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Eisenga MF, De Jong MA, Van der Meer P,
Leaf DE, Huls G, Nolte IM, Gaillard C, Bakker SJL and De Borst MH:
Iron deficiency, elevated erythropoietin, fibroblast growth factor
23, and mortality in the general population of the Netherlands: A
cohort study. PLoS Med. 16(e1002818)2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Vieth JT and Lane DR: Anemia. Emerg Med
Clin North Am. 32:613–628. 2014.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Bryan LJ and Zakai NA: Why is my patient
anemic? Hematol Oncol Clin North Am. 26:205–230, vii.
2012.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Kido S, Fujihara M, Nomura K, Sasaki S,
Mukai R, Ohnishi R, Kaneko I, Segawa H, Tatsumi S, Izumi H, et al:
Molecular mechanisms of cadmium-induced fibroblast growth factor 23
upregulation in osteoblast-like cells. Toxicol Sci. 139:301–316.
2014.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Lewerin C, Ljunggren O, Nilsson-Ehle H,
Karlsson MK, Herlitz H, Lorentzon M, Ohlsson C and Mellstrom D: Low
serum iron is associated with high serum intact FGF23 in elderly
men: The Swedish MrOS study. Bone. 98:1–8. 2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
David V, Francis C and Babitt JL: Ironing
out the cross talk between FGF23 and inflammation. Am J Physiol
Renal Physiol. 312:F1–F8. 2017.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Ärnlöv J, Carlsson AC, Sundström J,
Ingelsson E, Larsson A, Lind L and Larsson TE: Serum FGF23 and risk
of cardiovascular events in relation to mineral metabolism and
cardiovascular pathology. Clin J Am Soc Nephrol. 8:781–786.
2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Faul C, Amaral AP, Oskouei B, Hu MC, Sloan
A, Isakova T, Gutierrez OM, Aguillon-Prada R, Lincoln J, Hare JM,
et al: FGF23 induces left ventricular hypertrophy. J Clin Invest.
121:4393–4408. 2011.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Silswal N, Touchberry CD, Daniel DR,
McCarthy DL, Zhang S, Andresen J, Stubbs JR and Wacker MJ: FGF23
directly impairs endothelium-dependent vasorelaxation by increasing
superoxide levels and reducing nitric oxide bioavailability. Am J
Physiol Endocrinol Metab. 307:E426–E436. 2014.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Smith ER, Tan SJ, Holt SG and Hewitson TD:
FGF23 is synthesised locally by renal tubules and activates
injury-primed fibroblasts. Sci Rep. 7(3345)2017.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Singh S, Grabner A, Yanucil C, Schramm K,
Czaya B, Krick S, Czaja MJ, Bartz R, Abraham R, Di Marco GS, et al:
Fibroblast growth factor 23 directly targets hepatocytes to promote
inflammation in chronic kidney disease. Kidney Int. 90:985–996.
2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Rossaint J, Oehmichen J, Van Aken H,
Reuter S, Pavenstadt HJ, Meersch M, Unruh M and Zarbock A: FGF23
signaling impairs neutrophil recruitment and host defense during
CKD. J Clin Invest. 126:962–974. 2016.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Corn PG, Wang F, McKeehan WL and Navone N:
Targeting fibroblast growth factor pathways in prostate cancer.
Clin Cancer Res. 19:5856–5866. 2013.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Turner N and Grose R: Fibroblast growth
factor signalling: From development to cancer. Nat Rev Cancer.
10:116–129. 2010.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Feng S, Wang J, Zhang Y, Creighton CJ and
Ittmann M: FGF23 promotes prostate cancer progression. Oncotarget.
6:17291–17301. 2015.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Okada M, Imamura K, Fuchigami T, Omae T,
Iida M, Nanishi F, Murakami M, Ohgushi H, Yao T, Fujita K and Ogawa
K: 2 cases of nonspecific multiple ulcers of the small intestine
associated with osteomalacia caused by long-term intravenous
administration of saccharated ferric oxide. Nihon Naika Gakkai
Zasshi. 71:1566–1572. 1982.PubMed/NCBI(In Japanese).
|
|
53
|
Shepshelovich D, Rozen-Zvi B, Avni T,
Gafter U and Gafter-Gvili A: Intravenous versus oral iron
supplementation for the treatment of anemia in CKD: An updated
systematic review and meta-analysis. Am J Kidney Dis. 68:677–690.
2016.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Fukao W, Hasuike Y, Yamakawa T, Toyoda K,
Aichi M, Masachika S, Kantou M, Takahishi SI, Iwasaki T, Yahiro M,
et al: Oral versus intravenous iron supplementation for the
treatment of iron deficiency anemia in patients on maintenance
hemodialysis-effect on fibroblast growth factor-23 metabolism. J
Ren Nutr. 28:270–277. 2018.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Taniguchi K and Kakuta H: Bixalomer, a
novel phosphate binder with a small swelling index, improves
hyperphosphatemia in chronic kidney disease rat. Eur J Pharmacol.
766:129–134. 2015.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Koiwa F, Yokoyama K, Fukagawa M, Terao A
and Akizawa T: Efficacy and safety of sucroferric oxyhydroxide
compared with sevelamer hydrochloride in Japanese haemodialysis
patients with hyperphosphataemia: A randomized, open-label,
multicentre, 12-week phase III study. Nephrology (Carlton).
22:293–300. 2017.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Covic AC, Floege J, Ketteler M, Sprague
SM, Lisk L, Rakov V and Rastogi A: Iron-related parameters in
dialysis patients treated with sucroferric oxyhydroxide. Nephrol
Dial Transplant. 32:1330–1338. 2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Shima H, Miya K, Okada K, Minakuchi J and
Kawashima S: Sucroferric oxyhydroxide decreases serum phosphorus
level and fibroblast growth factor 23 and improves renal anemia in
hemodialysis patients. BMC Res Notes. 11(363)2018.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Yang WC, Yang CS, Hou CC, Wu TH, Young EW
and Hsu CH: An open-label, crossover study of a new
phosphate-binding agent in haemodialysis patients: Ferric citrate.
Nephrol Dial Transplant. 17:265–270. 2002.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Lee CT, Wu IW, Chiang SS, Peng YS, Shu KH,
Wu MJ and Wu MS: Effect of oral ferric citrate on serum phosphorus
in hemodialysis patients: Multicenter, randomized, double-blind,
placebo-controlled study. J Nephrol. 28:105–113. 2015.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Maruyama N, Otsuki T, Yoshida Y, Nagura C,
Kitai M, Shibahara N, Tomita H, Maruyama T and Abe M: Ferric
citrate decreases fibroblast growth factor 23 and improves
erythropoietin responsiveness in hemodialysis patients. Am J
Nephrol. 47:406–414. 2018.PubMed/NCBI View Article : Google Scholar
|
|
62
|
National Clinical Guideline Centre (UK):
Anaemia Management in Chronic Kidney Disease: Partial Update 2015
(Internet). London: Royal College of Physicians (UK), Jun 2015.
https://www.ncbi.nlm.nih.gov/books/NBK299242/.
|
|
63
|
Babitt JL and Lin HY: Mechanisms of anemia
in CKD. J Am Soc Nephrol. 23:1631–1634. 2012.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Hinata A, Iijima M, Nakano Y, Sakamoto T
and Tomita M: Chemical characterization of rabbit alpha
2-macroglobulin. Chem Pharm Bull (Tokyo). 35:271–276.
1987.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Landau D, London L, Bandach I and Segev Y:
The hypoxia inducible factor/erythropoietin (EPO)/EPO receptor
pathway is disturbed in a rat model of chronic kidney disease
related anemia. PLoS One. 13(e0196684)2018.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Thomas S and Rampersad M: Anaemia in
diabetes. Acta diabetologica. 41 (Suppl 1):S13–S17. 2004.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Daryadel A, Bettoni C, Haider T, Imenez
Silva PH, Schnitzbauer U, Pastor-Arroyo EM, Wenger RH, Gassmann M
and Wagner CA: Erythropoietin stimulates fibroblast growth factor
23 (FGF23) in mice and men. Pflugers Arch. 470:1569–1582.
2018.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Hanudel MR, Eisenga MF, Rappaport M, Chua
K, Qiao B, Jung G, Gabayan V, Gales B, Ramos G, de Jong MA, et al:
Effects of erythropoietin on fibroblast growth factor 23 in mice
and humans. Nephrol Dial Transplant. 34:2057–2065. 2019.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Eisenga MF, Emans ME, van der Putten K,
Cramer MJ, Diepenbroek A, Velthuis BK, Doevendans PA, Verhaar MC,
Joles JA, Bakker SJL, et al: Epoetin Beta and C-terminal fibroblast
growth factor 23 in patients with chronic heart failure and chronic
kidney disease. J Am Heart Assoc. 8(e011130)2019.PubMed/NCBI View Article : Google Scholar
|
|
70
|
van der Putten K, Braam B, Jie KE and
Gaillard CA: Mechanisms of disease: Erythropoietin resistance in
patients with both heart and kidney failure. Nat Clin Pract
Nephrol. 4:47–57. 2008.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Goetz R, Nakada Y, Hu MC, Kurosu H, Wang
L, Nakatani T, Shi M, Eliseenkova AV, Razzaque MS, Moe OW, et al:
Isolated C-terminal tail of FGF23 alleviates hypophosphatemia by
inhibiting FGF23-FGFR-Klotho complex formation. Proc Natl Acad Sci
USA. 107:407–412. 2010.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Courbebaisse M, Mehel H, Petit-Hoang C,
Ribeil JA, Sabbah L, Tuloup-Minguez V, Bergerat D, Arlet JB,
Stanislas A, Souberbielle JC, et al: Carboxy-terminal fragment of
fibroblast growth factor 23 induces heart hypertrophy in sickle
cell disease. Haematologica. 102:e33–e35. 2017.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Miikkulainen P, Högel H, Rantanen K, Suomi
T, Kouvonen P, Elo LL and Jaakkola PM: HIF prolyl hydroxylase PHD3
regulates translational machinery and glucose metabolism in clear
cell renal cell carcinoma. Cancer Metab. 5(5)2017.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Ivan M and Kaelin WG Jr: The EGLN-HIF
O2-sensing system: Multiple inputs and feedbacks. Mol
Cell. 66:772–779. 2017.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Pugh CW and Ratcliffe PJ: New horizons in
hypoxia signaling pathways. Exp Cell Res. 356:116–121.
2017.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Prabhakar NR and Semenza GL: Oxygen
sensing and homeostasis. Physiology (Bethesda). 30:340–348.
2015.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Maxwell PH and Eckardt KU: HIF prolyl
hydroxylase inhibitors for the treatment of renal anaemia and
beyond. Nat Rev Nephrol. 12:157–168. 2016.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Wyatt CM and Drüeke TB: HIF stabilization
by prolyl hydroxylase inhibitors for the treatment of anemia in
chronic kidney disease. Kidney Int. 90:923–925. 2016.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Kanbay M, Vervloet M, Cozzolino M,
Siriopol D, Covic A, Goldsmith D and Solak Y: Novel faces of
fibroblast growth factor 23 (FGF23): Iron deficiency, inflammation,
insulin resistance, left ventricular hypertrophy, proteinuria and
acute kidney injury. Calcif Tissue Int. 100:217–228.
2017.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Agoro R, Ni P, Noonan ML and White KE:
Osteocytic FGF23 and its kidney function. Front Endocrinol
(Lausanne). 11(592)2020.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Provenzano R, Besarab A, Sun CH, Diamond
SA, Durham JH, Cangiano JL, Aiello JR, Novak JE, Lee T, Leong R, et
al: Oral hypoxia-inducible factor Prolyl hydroxylase inhibitor
roxadustat (FG-4592) for the treatment of anemia in patients with
CKD. Clin J Am Soc Nephrol. 11:982–991. 2016.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Hanudel MR, Laster M and Salusky IB:
Non-renal-Related Mechanisms of FGF23 Pathophysiology. Curr
Osteoporos Rep. 16:724–729. 2018.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Wheeler JA and Clinkenbeard EL: Regulation
of fibroblast growth factor 23 by iron, EPO, and HIF. Curr Mol Biol
Rep. 5:8–17. 2019.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Zhang Q, Doucet M, Tomlinson RE, Han X,
Quarles LD, Collins MT and Clemens TL: The hypoxia-inducible
factor-1α activates ectopic production of fibroblast growth factor
23 in tumor-induced osteomalacia. Bone Res. 4(16011)2016.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Babitt JL and Sitara D: Crosstalk between
fibroblast growth factor 23, iron, erythropoietin, and inflammation
in kidney disease. Curr Opin Nephrol Hypertens. 28:304–310.
2019.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Khozeymeh F, Mortazavi M, Khalighinejad N,
Akhavankhaleghi M and Alikhani M: Salivary levels of interleukin-6
and tumor necrosis factor-α in patients undergoing hemodialysis.
Dent Res J (Isfahan). 13:69–73. 2016.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Wu J, Guo N, Chen X and Xing C:
Coexistence of micro-inflammatory and macrophage phenotype
abnormalities in chronic kidney disease. Int J Clin Exp Pathol.
13:317–323. 2020.PubMed/NCBI
|
|
88
|
Kim IY, Kim JH, Kim MJ, Lee DW, Hwang CG,
Han M, Rhee H, Song SH, Seong EY and Lee SB: Low
1,25-dihydroxyvitamin D level is associated with erythropoietin
deficiency and endogenous erythropoietin resistance in patients
with chronic kidney disease. Int Urol Nephrol. 50:2255–2260.
2018.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Icardi A, Paoletti E, De Nicola L,
Mazzaferro S, Russo R and Cozzolino M: Renal anaemia and EPO
hyporesponsiveness associated with vitamin D deficiency: The
potential role of inflammation. Nephrol Dial Transplant.
28:1672–1679. 2013.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Lee B, Kwon E, Kim Y, Kim JH, Son SW, Lee
JK, Kim DW, Sohn J, Kim TH and Ji JD: 1α,25-Dihydroxyvitamin D3
upregulates HIF-1 and TREM-1 via mTOR signaling. Immunol Lett.
163:14–21. 2015.PubMed/NCBI View Article : Google Scholar
|
|
91
|
van Vuren AJ, Gaillard C, Eisenga MF, van
Wijk R and van Beers EJ: The EPO-FGF23 signaling pathway in
erythroid progenitor cells: Opening a new area of research. Front
Physiol. 10(304)2019.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Yousaf F and Spinowitz B:
Hypoxia-inducible factor stabilizers: A new avenue for reducing BP
while helping hemoglobin? Curr Hypertens Rep. 18(23)2016.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Honda H, Michihata T, Shishido K,
Takahashi K, Takahashi G, Hosaka N, Ikeda M, Sanada D and Shibata
T: High fibroblast growth factor 23 levels are associated with
decreased ferritin levels and increased intravenous iron doses in
hemodialysis patients. PLoS One. 12(e0176984)2017.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Artunc F and Risler T: Serum
erythropoietin concentrations and responses to anaemia in patients
with or without chronic kidney disease. Nephrol Dial Transplant.
22:2900–2908. 2007.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Smith ER, Cai MM, McMahon LP and Holt SG:
Biological variability of plasma intact and C-terminal FGF23
measurements. J Clin Endocrinol Metab. 97:3357–3365.
2012.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Shimada T, Urakawa I, Isakova T, Yamazaki
Y, Epstein M, Wesseling-Perry K, Wolf M, Salusky IB and Jüppner H:
Circulating fibroblast growth factor 23 in patients with end-stage
renal disease treated by peritoneal dialysis is intact and
biologically active. J Clin Endocrinol Metab. 95:578–585.
2010.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Kalyanasundaram A and Fedorov VV:
Fibroblast growth factor 23: A novel key to find hidden substrates
of atrial fibrillation? Circulation. 130:295–297. 2014.PubMed/NCBI View Article : Google Scholar
|