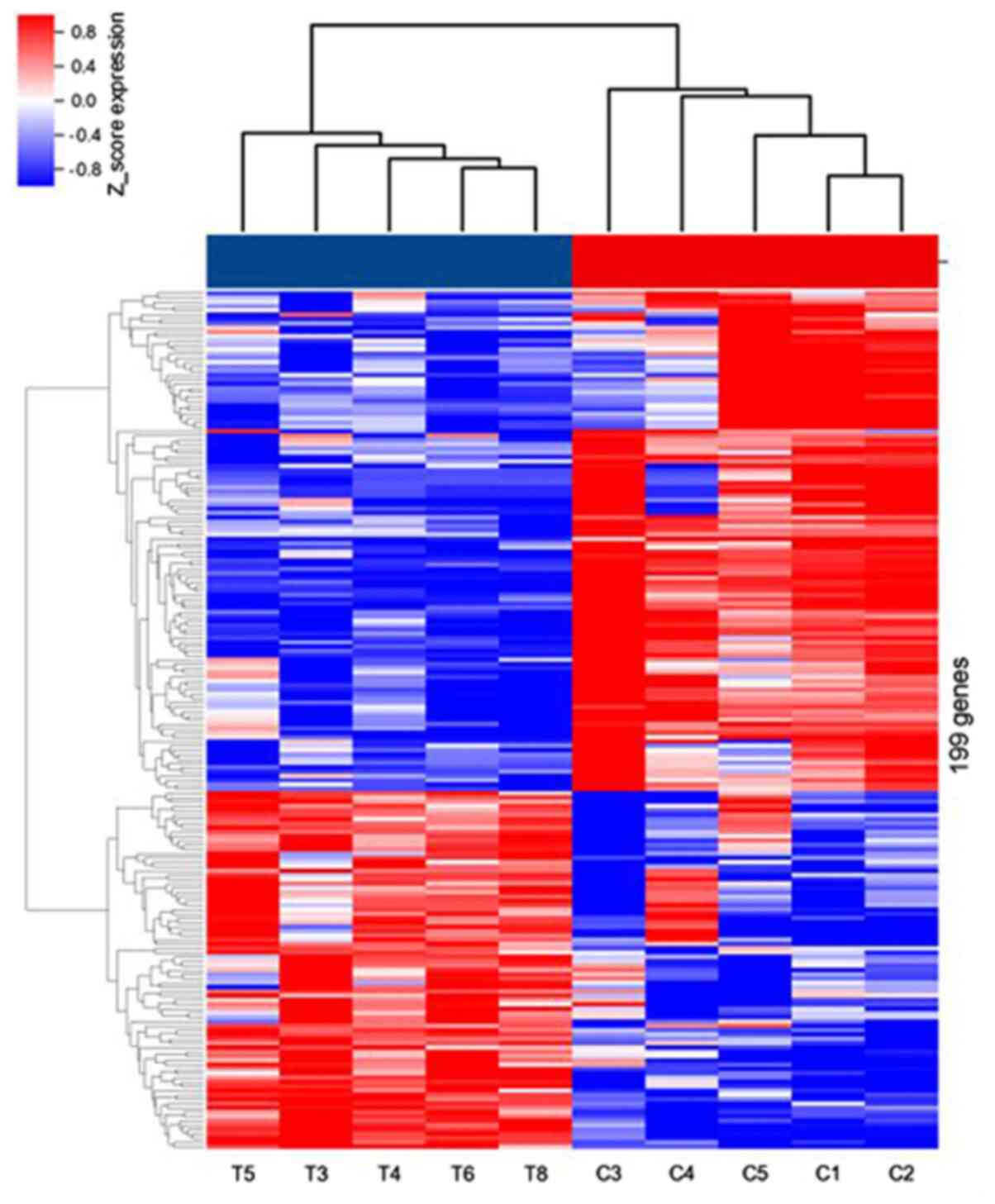
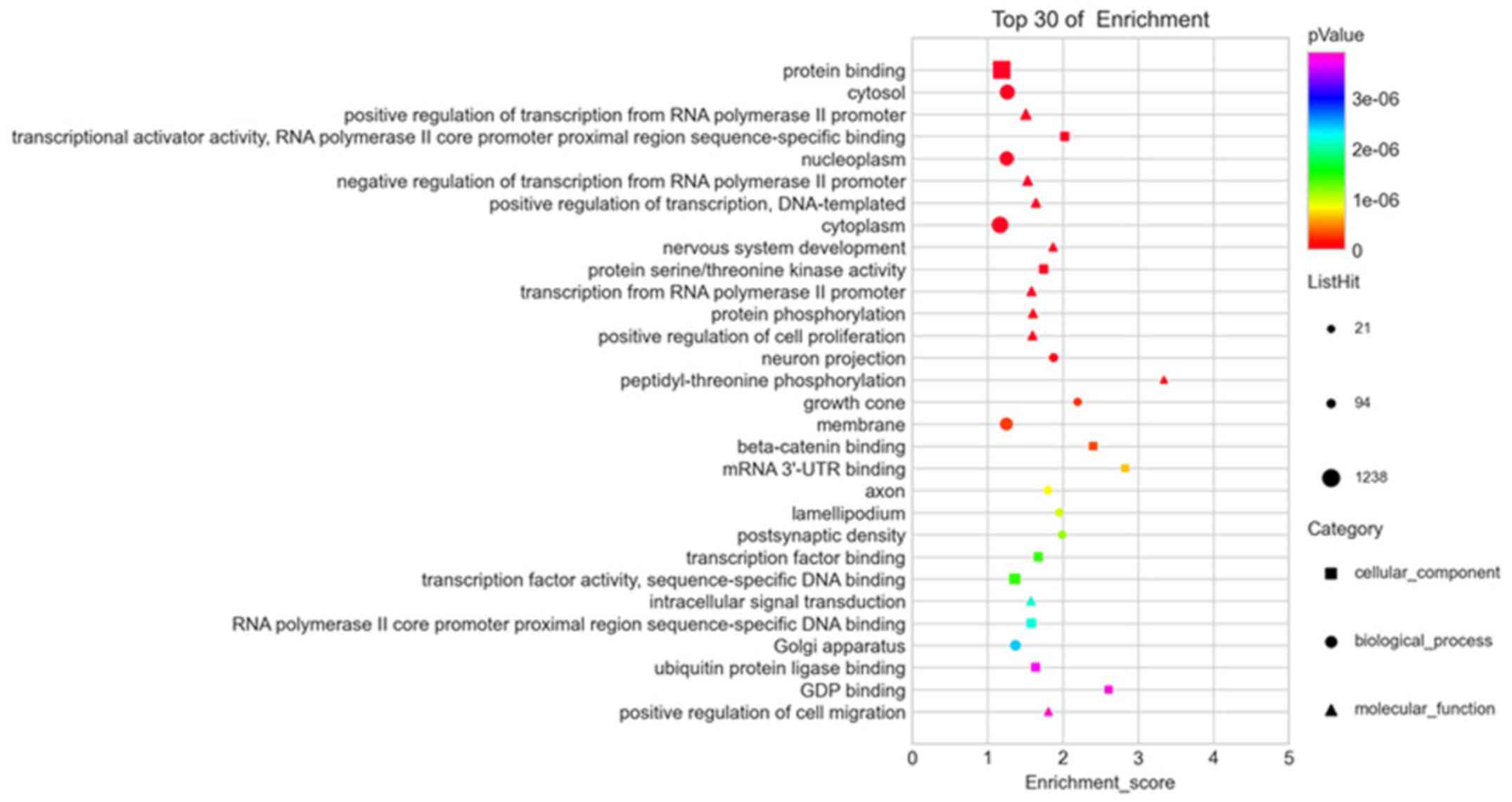
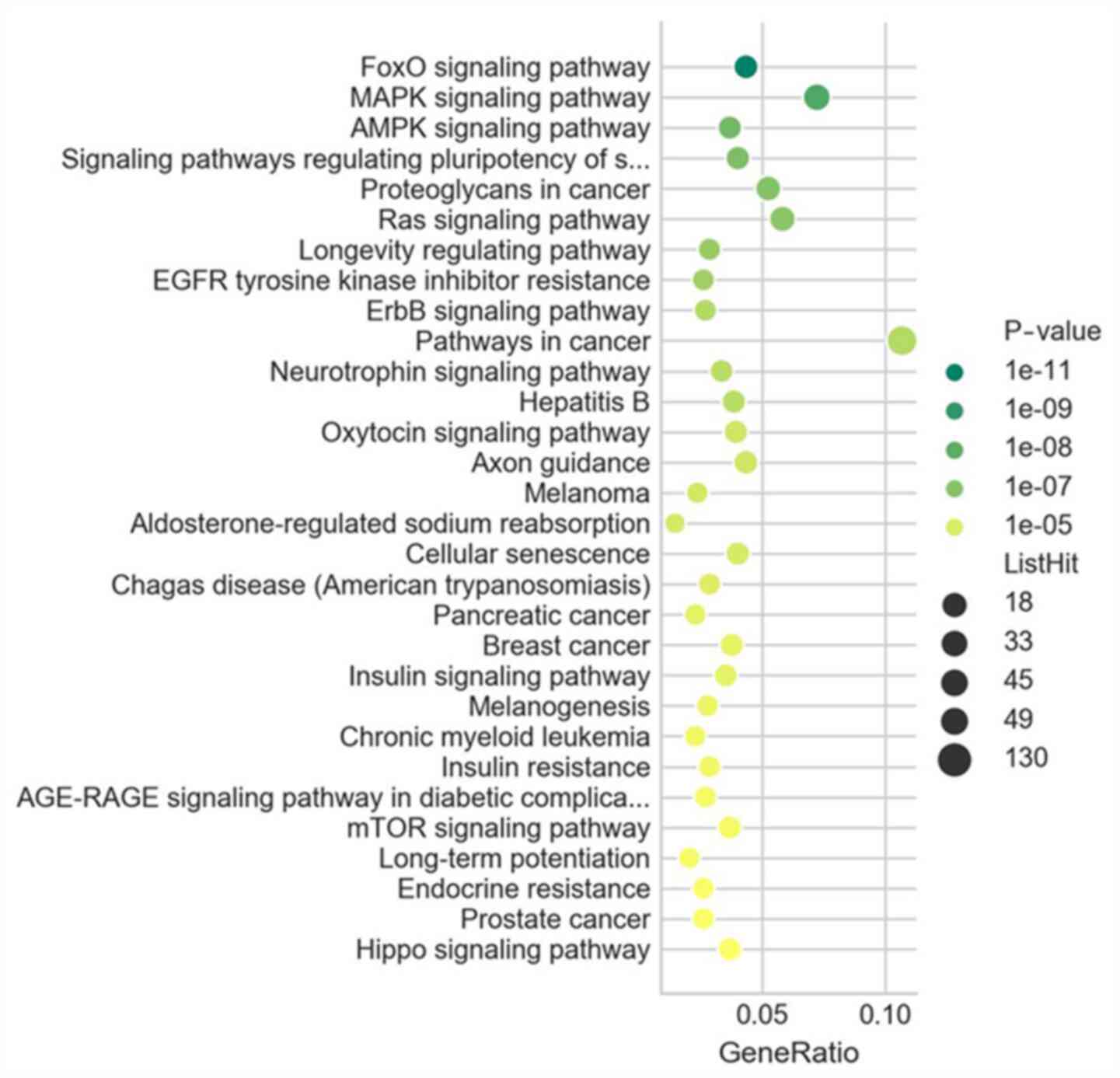
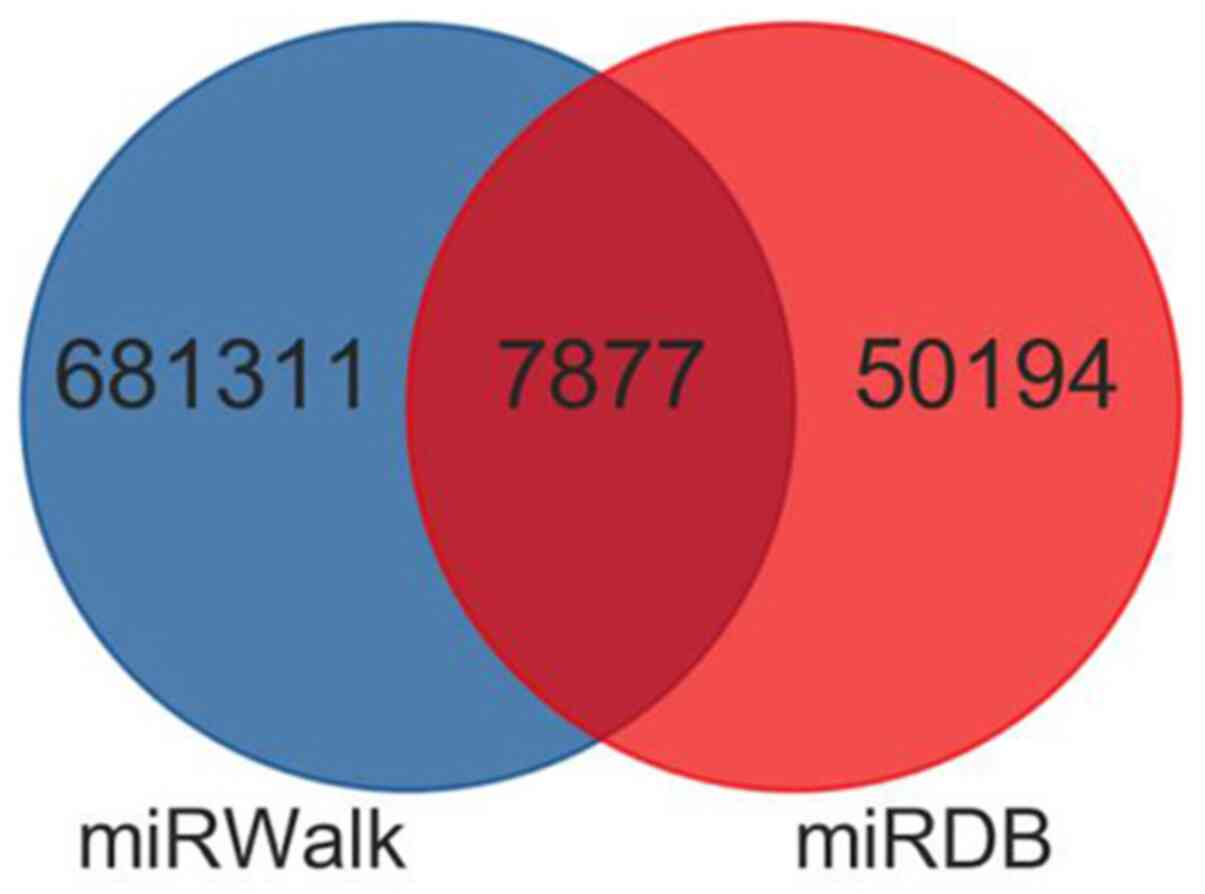
|
1
|
Siersema PD: How to approach a patient
with refractory or recurrent benign esophageal stricture.
Gastroenterology. 156:7–10. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Yan X, Nie D, Zhang Y, Chang H and Huang
Y: Effectiveness of an orally administered steroid gel at
preventing restenosis after endoscopic balloon dilation of benign
esophageal stricture. Medicine (Baltimore).
98(e14565)2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Fuccio L, Hassan C, Frazzoni L, Miglio R
and Repici A: Clinical outcomes following stent placement in
refractory benign esophageal stricture: A systematic review and
meta-analysis. Endoscopy. 48:141–148. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
van Halsema EE, Noordzij IC, van Berge
Henegouwen MI, Fockens P, Bergman JJ and van Hooft JE: Endoscopic
dilation of benign esophageal anastomotic strictures over 16 mm has
a longer lasting effect. Surg Endosc. 31:1871–1881. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Haverkamp L, van der Sluis PC, Verhage RJ,
Siersema PD, Ruurda JP and van Hillegersberg R: End-to-end cervical
esophagogastric anastomoses are associated with a higher number of
strictures compared with end-to-side anastomoses. J Gastrointest
Surg. 17:872–876. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Liao Z, Liao G, Yang X, Peng X, Zhang X,
Xie X, Zhao X, Yang S, Fan C and Bai J: Transplantation of
autologous esophageal mucosa to prevent stricture after
circumferential endoscopic submucosal dissection of early
esophageal cancer (with video). Gastrointest Endosc. 88:543–546.
2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rockey DC, Bell PD and Hill JA: Fibrosis -
a common pathway to organ injury and failure. N Engl J Med.
372:1138–1149. 2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Honda M, Nakamura T, Hori Y, Shionoya Y,
Nakada A, Sato T, Yamamoto K, Kobayashi T, Shimada H, Kida N, et
al: Process of healing of mucosal defects in the esophagus after
endoscopic mucosal resection: Histological evaluation in a dog
model. Endoscopy. 42:1092–1095. 2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Yates LA, Norbury CJ and Gilbert RJ: The
long and short of microRNA. Cell. 153:516–519. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Blaya D, Coll M, Rodrigo-Torres D,
Vila-Casadesús M, Altamirano J, Llopis M, Graupera I, Perea L,
Aguilar-Bravo B, Díaz A, et al: Integrative microRNA profiling in
alcoholic hepatitis reveals a role for microRNA-182 in liver injury
and inflammation. Gut. 65:1535–1545. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sun Y, Ge Y, Fu Y, Yan L, Cai J, Shi K,
Cao X and Lu C: Mitomycin C induces fibroblasts apoptosis and
reduces epidural fibrosis by regulating miR-200b and its targeting
of RhoE. Eur J Pharmacol. 765:198–208. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wang BB, Xie H, Wu T, Xie N, Wu J, Gu Y,
Tang F and Liu J: Controlled-release mitomycin C-polylactic acid
film prevents epidural scar hyperplasia after laminectomy by
inducing fibroblast autophagy and regulating the expression of
miRNAs. Eur Rev Med Pharmacol Sci. 21:2526–2537. 2017.PubMed/NCBI
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wu L, Li H, Jia CY, Cheng W, Yu M, Peng M,
Zhu Y, Zhao Q, Dong YW, Shao K, et al: MicroRNA-223 regulates FOXO1
expression and cell proliferation. FEBS Lett. 586:1038–1043.
2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Han LL, Zhou XJ, Li FJ, Hao XW, Jiang Z,
Dong Q and Chen X: MiR-223-3p promotes the growth and invasion of
neuroblastoma cell via targeting FOXO1. Eur Rev Med Pharmacol Sci.
23:8984–8990. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Cheng N, Liu C, Li Y, Gao S, Han YC, Wang
X, Du J and Zhang C: MicroRNA-223-3p promotes skeletal muscle
regeneration by regulating inflammation in mice. J Biol Chem.
295:10212–10223. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Liu X, Xu Y, Deng Y and Li H: MicroRNA-223
regulates cardiac fibrosis after myocardial infarction by targeting
RASA1. Cell Physiol Biochem. 46:1439–1454. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Chen YT, Chen FY, Vijmasi T, Stephens DN,
Gallup M and McNamara NA: Pax6 downregulation mediates abnormal
lineage commitment of the ocular surface epithelium in
aqueous-deficient dry eye disease. PLoS One.
8(e77286)2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Feng Y, Li M, Wang S, Cong W, Hu G, Song
Y, Xiao H, Dong E and Zhang Y: Paired box 6 inhibits cardiac
fibroblast differentiation. Biochem Biophys Res Commun.
528:561–566. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Li C, Tan YH, Sun J, Deng FM and Liu YL:
PAX6 contributes to the activation and proliferation of hepatic
stellate cells via activating Hedgehog/GLI1 pathway. Biochem
Biophys Res Commun. 526:314–320. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kiriazis H, Wang K, Xu Q, Gao XM, Ming Z,
Su Y, Moore XL, Lambert G, Gibbs ME, Dart AM, et al: Knockout of
beta(1)- and beta(2)-adrenoceptors attenuates pressure
overload-induced cardiac hypertrophy and fibrosis. Br J Pharmacol.
153:684–692. 2008.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ito A, Ohnuki Y, Suita K, Ishikawa M,
Mototani Y, Shiozawa K, Kawamura N, Yagisawa Y, Nariyama M, Umeki
D, et al: Role of β-adrenergic signaling in masseter muscle. PLoS
One. 14(e0215539)2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Steiner JE, Cottrell CE, Streicher JL,
Jensen JN, King DM, Burrows PE, Siegel DH, Tollefson MM, Drolet BA
and Püttgen KB: Scarring in patients With PIK3CA-related overgrowth
syndromes. JAMA Dermatol. 154:452–455. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Yang X, Li X, Lin Q and Xu Q:
Up-regulation of microRNA-203 inhibits myocardial fibrosis and
oxidative stress in mice with diabetic cardiomyopathy through the
inhibition of PI3K/Akt signaling pathway via PIK3CA. Gene.
715(143995)2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wegner KA, Mueller BR, Unterberger CJ,
Avila EJ, Ruetten H, Turco AE, Oakes SR, Girardi NM, Halberg RB,
Swanson SM, et al: Prostate epithelial-specific expression of
activated PI3K drives stromal collagen production and accumulation.
J Pathol. 250:231–242. 2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Zhu J, Zhou L, Wei B, Qian Z, Wang J, Hui
H and Sun Y: miR-142-5p inhibits pancreatic cancer cell migration
and invasion by targeting PIK3CA. Mol Med Rep. 22:2085–2092.
2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Cai L, Chao G, Li W, Zhu J, Li F, Qi B,
Wei Y, Chen S, Zhou G, Lu X, et al: Activated CD4+ T
cells-derived exosomal miR-142-3p boosts post-ischemic ventricular
remodeling by activating myofibroblast. Aging (Albany NY).
12:7380–7396. 2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Guiot J, Cambier M, Boeckx A, Henket M,
Nivelles O, Gester F, Louis E, Malaise M, Dequiedt F, Louis R, et
al: Macrophage-derived exosomes attenuate fibrosis in airway
epithelial cells through delivery of antifibrotic miR-142-3p.
Thorax. 75:870–881. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Wang Z, Liu Z, Fang X and Yang H:
MiR-142-5p suppresses tumorigenesis by targeting PIK3CA in
non-small cell lung cancer. Cell Physiol Biochem. 43:2505–2515.
2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Mei J, Zhang Y, Lu S and Wang J: Long
non-coding RNA NNT-AS1 regulates proliferation, apoptosis,
inflammation and airway remodeling of chronic obstructive pulmonary
disease via targeting miR-582-5p/FBXO11 axis. Biomed Pharmacother.
129(110326)2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Vuppalanchi R, Liang T, Goswami CP,
Nalamasu R, Li L, Jones D, Wei R, Liu W, Sarasani V, Janga SC, et
al: Relationship between differential hepatic microRNA expression
and decreased hepatic cytochrome P450 3A activity in cirrhosis.
PLoS One. 8(e74471)2013.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Yang F, Luo L, Zhu ZD, Zhou X, Wang Y, Xue
J, Zhang J, Cai X, Chen ZL, Ma Q, et al: Chlorogenic acid inhibits
liver fibrosis by blocking the miR-21-regulated TGF-β1/Smad7
signaling pathway in vitro and in vivo. Front Pharmacol.
8(929)2017.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhou XL, Xu H, Liu ZB, Wu QC, Zhu RR and
Liu JC: miR-21 promotes cardiac fibroblast-to-myofibroblast
transformation and myocardial fibrosis by targeting Jagged1. J Cell
Mol Med. 22:3816–3824. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Sun C, Tian J, Liu X and Guan G: MiR-21
promotes fibrosis and hypertrophy of ligamentum flavum in lumbar
spinal canal stenosis by activating IL-6 expression. Biochem
Biophys Res Commun. 490:1106–1111. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Conickx G, Mestdagh P, Avila Cobos F,
Verhamme FM, Maes T, Vanaudenaerde BM, Seys LJ, Lahousse L, Kim RY,
Hsu AC, et al: MicroRNA profiling reveals a role for
MicroRNA-218-5p in the pathogenesis of chronic obstructive
pulmonary disease. Am J Respir Crit Care Med. 195:43–56.
2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Li Z, Qian R, Zhang J and Shi X:
MiR-218-5p targets LHFPL3 to regulate proliferation, migration, and
epithelial-mesenchymal transitions of human glioma cells. Biosci
Rep. 39(BSR20180879)2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Brkić J, Dunk C, O'Brien J, Fu G, Nadeem
L, Wang YL, Rosman D, Salem M, Shynlova O, Yougbaré I, et al:
MicroRNA-218-5p promotes endovascular trophoblast differentiation
and spiral artery remodeling. Mol Ther. 26:2189–2205.
2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Han S, Chen Y, Gao Y, Sun B and Kong Y:
MicroRNA-218-5p inhibit the migration and proliferation of
pterygium epithelial cells by targeting EGFR via PI3K/Akt/mTOR
signaling pathway. Exp Eye Res. 178:37–45. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Baker Frost D, Savchenko A, Ogunleye A,
Armstrong M and Feghali-Bostwick C: Elucidating the cellular
mechanism for E2-induced dermal fibrosis. Arthritis Res Ther.
23(68)2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Zhang Z, Zhu D, Zhang X, Liu Y, Wang J and
Yan L: Tanshinone IIA regulates fibroblast proliferation and
migration and post-surgery arthrofibrosis through the
autophagy-mediated PI3K and AMPK-mTOR signaling pathway. Am J
Transl Res. 13:565–584. 2021.PubMed/NCBI
|
|
41
|
Qi L, He W, Yang J, Gao Y and Chen J:
Endoscopic balloon dilation and submucosal injection of
triamcinolone acetonide in the treatment of esophageal stricture: A
single-center retrospective study. Exp Ther Med. 16:5248–5252.
2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Zhang Y, Wang X, Liu L, Chen JP and Fan
ZN: Intramuscular injection of mitomycin C combined with endoscopic
dilation for benign esophageal strictures. J Dig Dis. 16:370–376.
2015.PubMed/NCBI View Article : Google Scholar
|