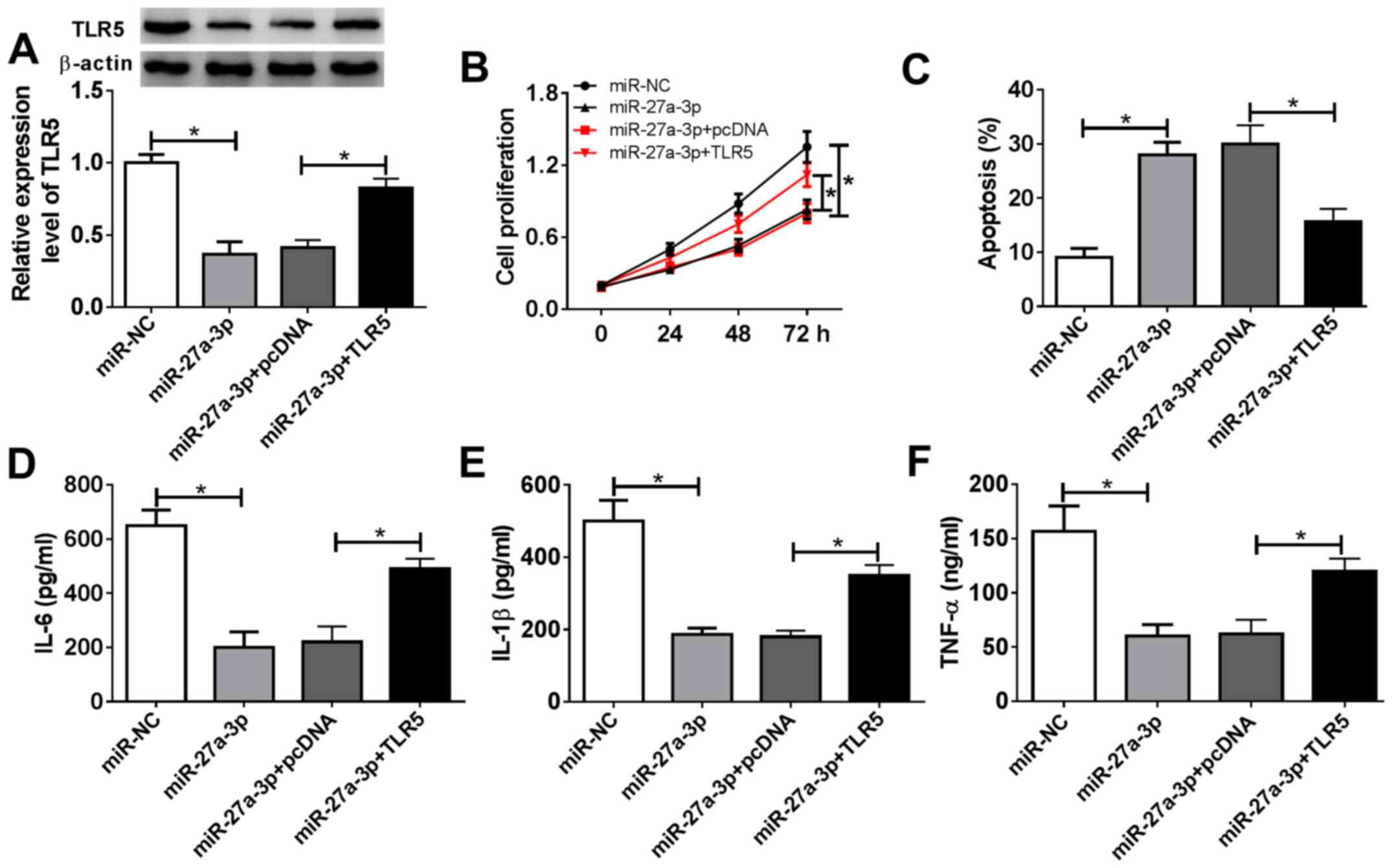
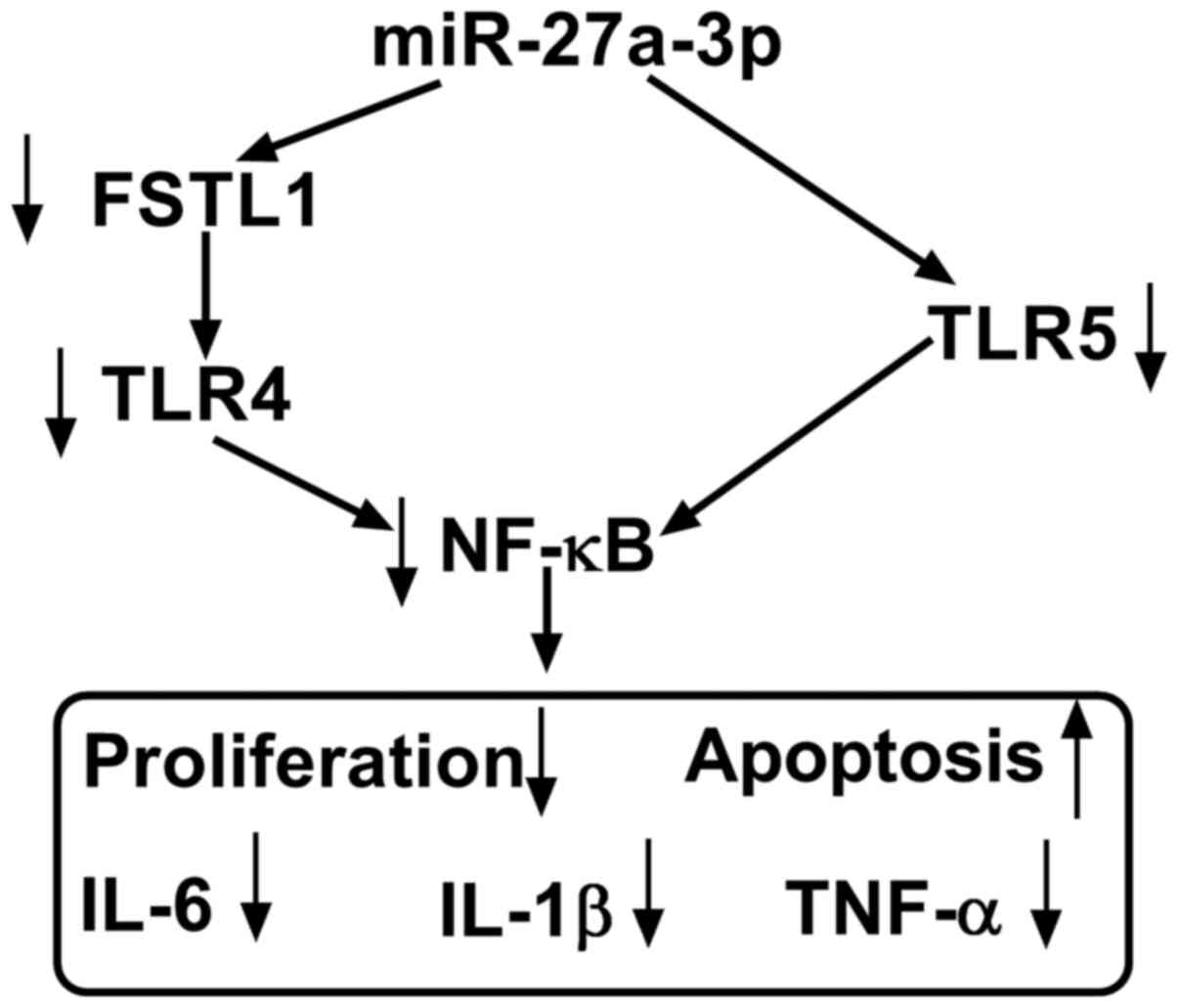
|
1
|
Coras R, Narasimhan R and Guma M: Liquid
biopsies to guide therapeutic decisions in rheumatoid arthritis.
Transl Res. 201:1–12. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Araki Y and Mimura T: The mechanisms
underlying chronic inflammation in rheumatoid arthritis from the
perspective of the epigenetic landscape. J Immunol Res.
2016(6290682)2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Croia C, Bursi R, Sutera D, Petrelli F,
Alunno A and Puxeddu I: One year in review 2019: Pathogenesis of
rheumatoid arthritis. Clin Exp Rheumatol. 37:347–357.
2019.PubMed/NCBI
|
|
4
|
Fang Q, Zhou C and Nandakumar KS:
Molecular and cellular pathways contributing to joint damage in
rheumatoid arthritis. Mediators Inflamm.
2020(3830212)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Alunno A, Carubbi F, Giacomelli R and
Gerli R: Cytokines in the pathogenesis of rheumatoid arthritis: New
players and therapeutic targets. BMC Rheumatol. 1(3)2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Falconer J, Murphy AN, Young SP, Clark AR,
Tiziani S, Guma M and Buckley CD: Review: Synovial cell metabolism
and chronic inflammation in rheumatoid arthritis. Arthritis
Rheumatol. 70:984–999. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Shi J, Ermann J, Weissman BN, Smith SE and
Mandell JC: Thinking beyond pannus: A review of retro-odontoid
pseudotumor due to rheumatoid and non-rheumatoid etiologies.
Skeletal Radiol. 48:1511–1523. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Laev SS and Salakhutdinov NF:
Anti-arthritic agents: Progress and potential. Bioorg Med Chem.
23:3059–3080. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Yuasa S, Yamaguchi H, Nakanishi Y,
Kawaminami S, Tabata R, Shimizu N, Kohno M, Shimizu T, Miyata J,
Nakayama M, et al: Treatment responses and their predictors in
patients with rheumatoid arthritis treated with biological agents.
J Med Invest. 60:77–90. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zamanpoor M: The genetic pathogenesis,
diagnosis and therapeutic insight of rheumatoid arthritis. Clin
Genet. 95:547–557. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Moran-Moguel MC, Petarra-Del Rio S,
Mayorquin-Galvan EE and Zavala-Cerna MG: Rheumatoid arthritis and
mirnas: A critical review through a functional view. J Immunol Res.
2018(2474529)2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Dong H, Lei J, Ding L, Wen Y, Ju H and
Zhang X: MicroRNA: Function, detection, and bioanalysis. Chem Rev.
113:6207–6233. 2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Dudics S, Venkatesha SH and Moudgil KD:
The micro-RNA expression profiles of autoimmune arthritis reveal
novel biomarkers of the disease and therapeutic response. Int J Mol
Sci. 19(19)2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Zhang H, Huang X, Ye L, Guo G, Li X, Chen
C, Sun L, Li B, Chen N and Xue X: B cell-related circulating
MicroRNAs with the potential value of biomarkers in the
differential diagnosis, and distinguishment between the disease
activity and lupus nephritis for systemic lupus erythematosus.
Front Immunol. 9(1473)2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Regev K, Paul A, Healy B, von Glenn F,
Diaz-Cruz C, Gholipour T, Mazzola MA, Raheja R, Nejad P, Glanz BI,
et al: Comprehensive evaluation of serum microRNAs as biomarkers in
multiple sclerosis. Neurol Neuroimmunol Neuroinflamm.
3(e267)2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Evangelatos G, Fragoulis GE, Koulouri V
and Lambrou GI: MicroRNAs in rheumatoid arthritis: From
pathogenesis to clinical impact. Autoimmun Rev.
18(102391)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Sharma AR, Sharma G, Lee SS and
Chakraborty C: miRNA-regulated key components of cytokine signaling
pathways and inflammation in rheumatoid arthritis. Med Res Rev.
36:425–439. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Sode J, Krintel SB, Carlsen AL, Hetland
ML, Johansen JS, Hørslev-Petersen K, Stengaard-Pedersen K,
Ellingsen T, Burton M, Junker P, et al: Plasma MicroRNA profiles in
patients with early rheumatoid arthritis responding to adalimumab
plus methotrexate vs, methotrexate alone: A Placebo-controlled
Clinical Trial. J Rheumatol. 45:53–61. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Takamura Y, Aoki W, Satomura A, Shibasaki
S and Ueda M: Small RNAs detected in exosomes derived from the MH7A
synovial fibroblast cell line with TNF-α stimulation. PLoS One.
13(e0201851)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Maeda Y, Farina NH, Matzelle MM, Fanning
PJ, Lian JB and Gravallese EM: Synovium-derived MicroRNAs regulate
bone pathways in rheumatoid arthritis. J Bone Miner Res.
32:461–472. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Shi DL, Shi GR, Xie J, Du XZ and Yang H:
MicroRNA-27a inhibits cell migration and invasion of
fibroblast-like synoviocytes by targeting follistatin-like protein
1 in rheumatoid arthritis. Mol Cells. 39:611–618. 2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Aletaha D, Neogi T, Silman AJ, Funovits J,
Felson DT, Bingham CO III, Birnbaum NS, Burmester GR, Bykerk VP,
Cohen MD, et al: 2010 rheumatoid arthritis classification criteria:
An American College of Rheumatology/European League Against
Rheumatism collaborative initiative. Ann Rheum Dis. 69:1580–1588.
2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Li XJ, Xu M, Zhao XQ, Zhao JN, Chen FF, Yu
W, Gao DY and Luo B: Proteomic analysis of synovial fibroblast-like
synoviocytes from rheumatoid arthritis. Clin Exp Rheumatol.
31:552–558. 2013.PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zou Y, Xu S, Xiao Y, Qiu Q, Shi M, Wang J,
Liang L, Zhan Z, Yang X, Olsen N, et al: Long noncoding RNA LERFS
negatively regulates rheumatoid synovial aggression and
proliferation. J Clin Invest. 128:4510–4524. 2018.PubMed/NCBI View
Article : Google Scholar
|
|
26
|
Alamanos Y and Drosos AA: Epidemiology of
adult rheumatoid arthritis. Autoimmun Rev. 4:130–136.
2005.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Liu Y, Pan YF, Xue YQ, Fang LK, Guo XH,
Guo X, Liu M, Mo BY, Yang MR, Liu F, et al: uPAR promotes
tumor-like biologic behaviors of fibroblast-like synoviocytes
through PI3K/Akt signaling pathway in patients with rheumatoid
arthritis. Cell Mol Immunol. 15:171–181. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Cai P, Jiang T, Li B, Qin X, Lu Z, Le Y,
Shen C, Yang Y, Zheng L and Zhao J: Comparison of rheumatoid
arthritis (RA) and osteoarthritis (OA) based on microarray profiles
of human joint fibroblast-like synoviocytes. Cell Biochem Funct.
37:31–41. 2019.PubMed/NCBI View
Article : Google Scholar
|
|
29
|
Ye Y, Gao X and Yang N: LncRNA ZFAS1
promotes cell migration and invasion of fibroblast-like
synoviocytes by suppression of miR-27a in rheumatoid arthritis. Hum
Cell. 31:14–21. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Yang S and Yang Y: Downregulation of
microRNA 221 decreases migration and invasion in fibroblast like
synoviocytes in rheumatoid arthritis. Mol Med Rep. 12:2395–2401.
2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Neumann E, Lefèvre S, Zimmermann B, Gay S
and Müller-Ladner U: Rheumatoid arthritis progression mediated by
activated synovial fibroblasts. Trends Mol Med. 16:458–468.
2010.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Karouzakis E, Gay RE, Gay S and Neidhart
M: Epigenetic control in rheumatoid arthritis synovial fibroblasts.
Nat Rev Rheumatol. 5:266–272. 2009.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Li H, Guan SB, Lu Y and Wang F: MiR-140-5p
inhibits synovial fibroblasts proliferation and inflammatory
cytokines secretion through targeting TLR4. Biomed Pharmacother.
96:208–214. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Fu D, Xiao C, Xie Y, Gao J and Ye S:
MiR-3926 inhibits synovial fibroblasts proliferation and
inflammatory cytokines secretion through targeting toll like
receptor 5. Gene. 687:200–206. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Thunyakitpisal P, Ruangpornvisuti V,
Kengkwasing P, Chokboribal J and Sangvanich P: Acemannan increases
NF-κB/DNA binding and IL-6/-8 expression by selectively binding
Toll-like receptor-5 in human gingival fibroblasts. Carbohydr
Polym. 161:149–157. 2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Sabroe I, Read RC, Whyte MK, Dockrell DH,
Vogel SN and Dower SK: Toll-like receptors in health and disease:
Complex questions remain. J Immunol. 171:1630–1635. 2003.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Elshabrawy HA, Essani AE, Szekanecz Z, Fox
DA and Shahrara S: TLRs, future potential therapeutic targets for
RA. Autoimmun Rev. 16:103–113. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Thwaites RS, Unterberger S, Chamberlain G,
Walker-Bone K, Davies KA and Sacre S: TLR1/2 and 5 induce elevated
cytokine levels from rheumatoid arthritis monocytes independent of
ACPA or RF autoantibody status. Rheumatology (Oxford).
59:3533–3539. 2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Smith KD, Andersen-Nissen E, Hayashi F,
Strobe K, Bergman MA, Barrett SL, Cookson BT and Aderem A:
Toll-like receptor 5 recognizes a conserved site on flagellin
required for protofilament formation and bacterial motility. Nat
Immunol. 4:1247–1253. 2003.PubMed/NCBI View
Article : Google Scholar
|
|
40
|
Wu YW, Tang W and Zuo JP: Toll-like
receptors: Potential targets for lupus treatment. Acta Pharmacol
Sin. 36:1395–1407. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Chamberlain ND, Vila OM, Volin MV, Volin
MV, Volkov S, Pope RM, Swedler W, Mandelin AM 2nd, Shahrara S, et
al: TLR5, a novel and unidentified inflammatory mediator in
rheumatoid arthritis that correlates with disease activity score
and joint TNF-α levels. J Immunol. 189:475–483. 2012.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Kim SJ, Chen Z, Chamberlain ND, Essani AB,
Volin MV, Amin MA, Volkov S, Gravallese EM, Arami S, Swedler W, et
al: Ligation of TLR5 promotes myeloid cell infiltration and
differentiation into mature osteoclasts in rheumatoid arthritis and
experimental arthritis. J Immunol. 193:3902–3913. 2014.PubMed/NCBI View Article : Google Scholar
|