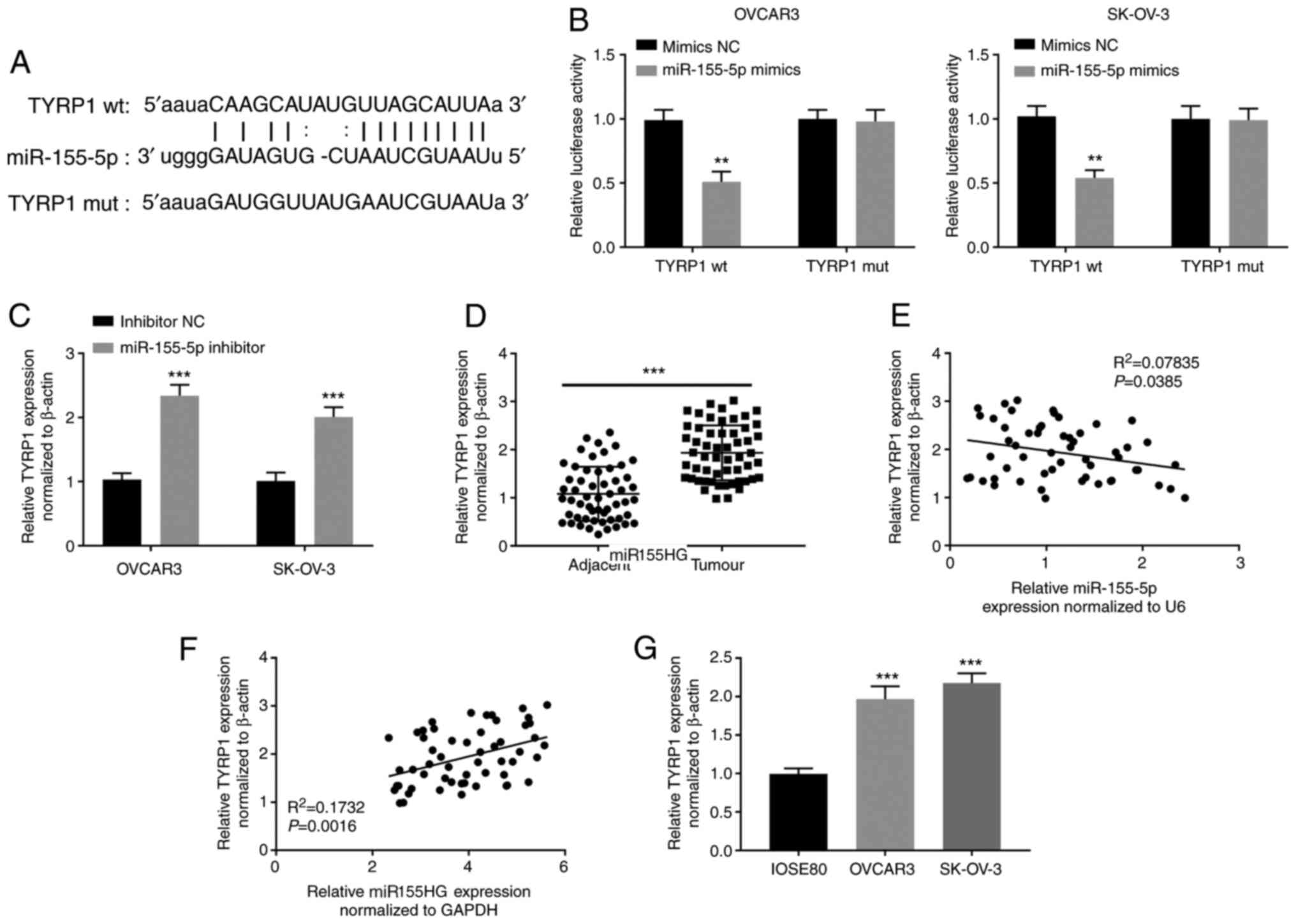
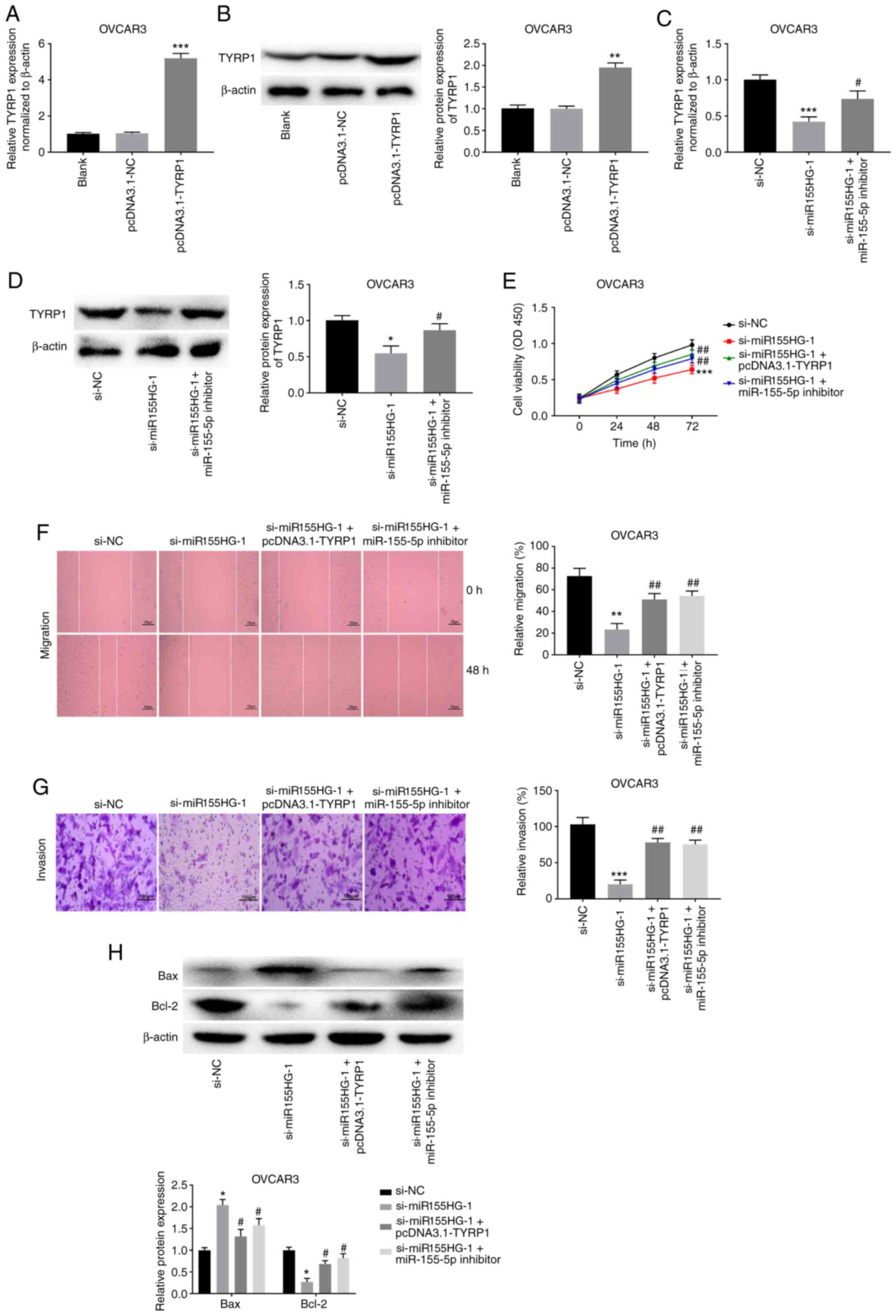
|
1
|
Nash Z and Menon U: Ovarian cancer
screening: Current status and future directions. Best Pract Res
Clin Obstet Gynaecol. 65:32–45. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Nimmagadda S and Penet MF: Ovarian cancer
targeted theranostics. Front Oncol. 9(1537)2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Chandra A, Pius C, Nabeel M, Nair M,
Vishwanatha JK, Ahmad S and Basha R: Ovarian cancer: Current status
and strategies for improving therapeutic outcomes. Cancer Med.
8:7018–7031. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Stewart C, Ralyea C and Lockwood S:
Ovarian cancer: An integrated review. Semin Oncol Nurs. 35:151–156.
2019.PubMed/NCBI View Article : Google Scholar
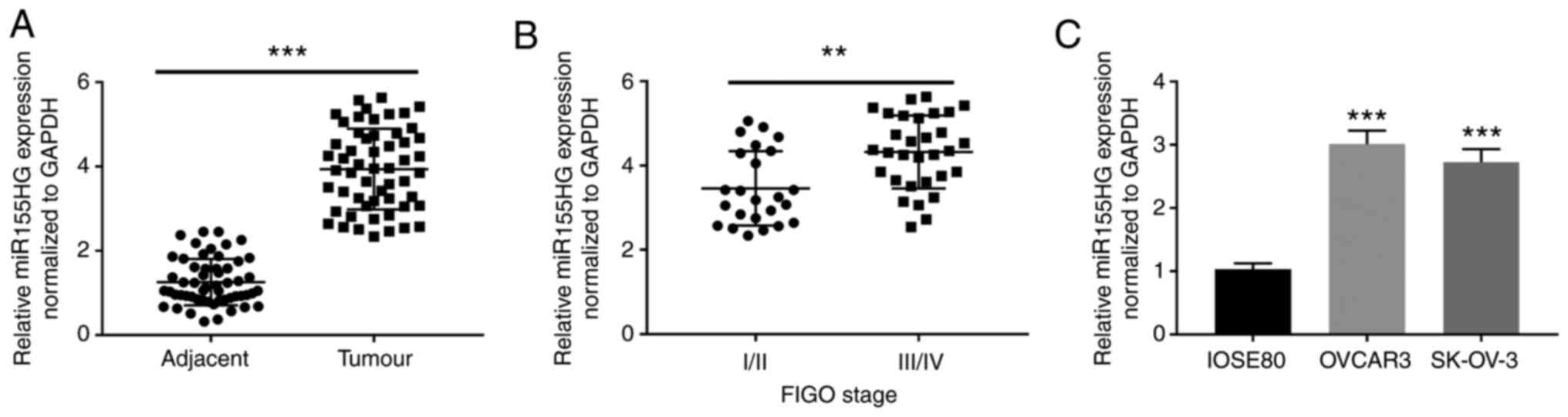
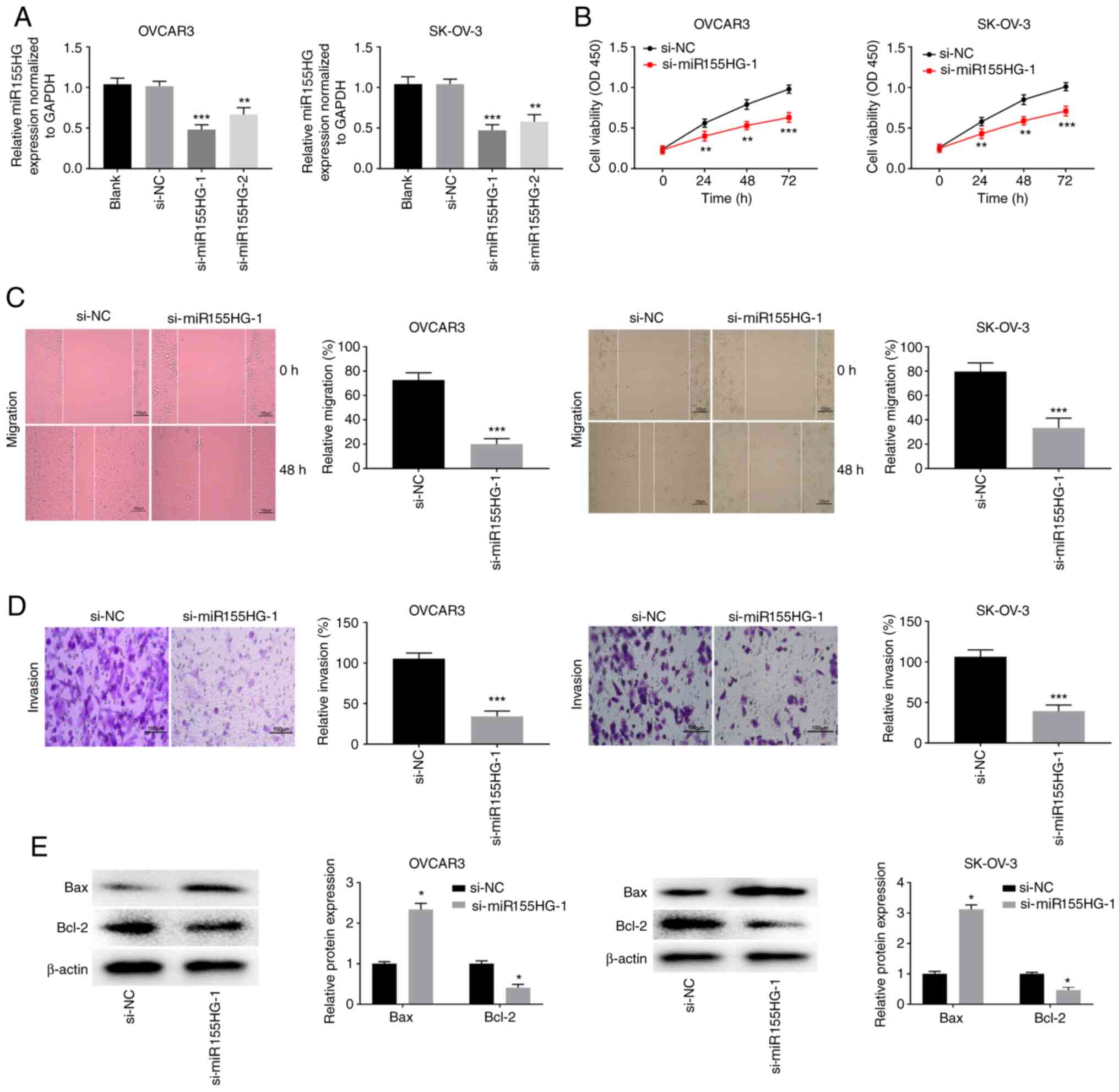
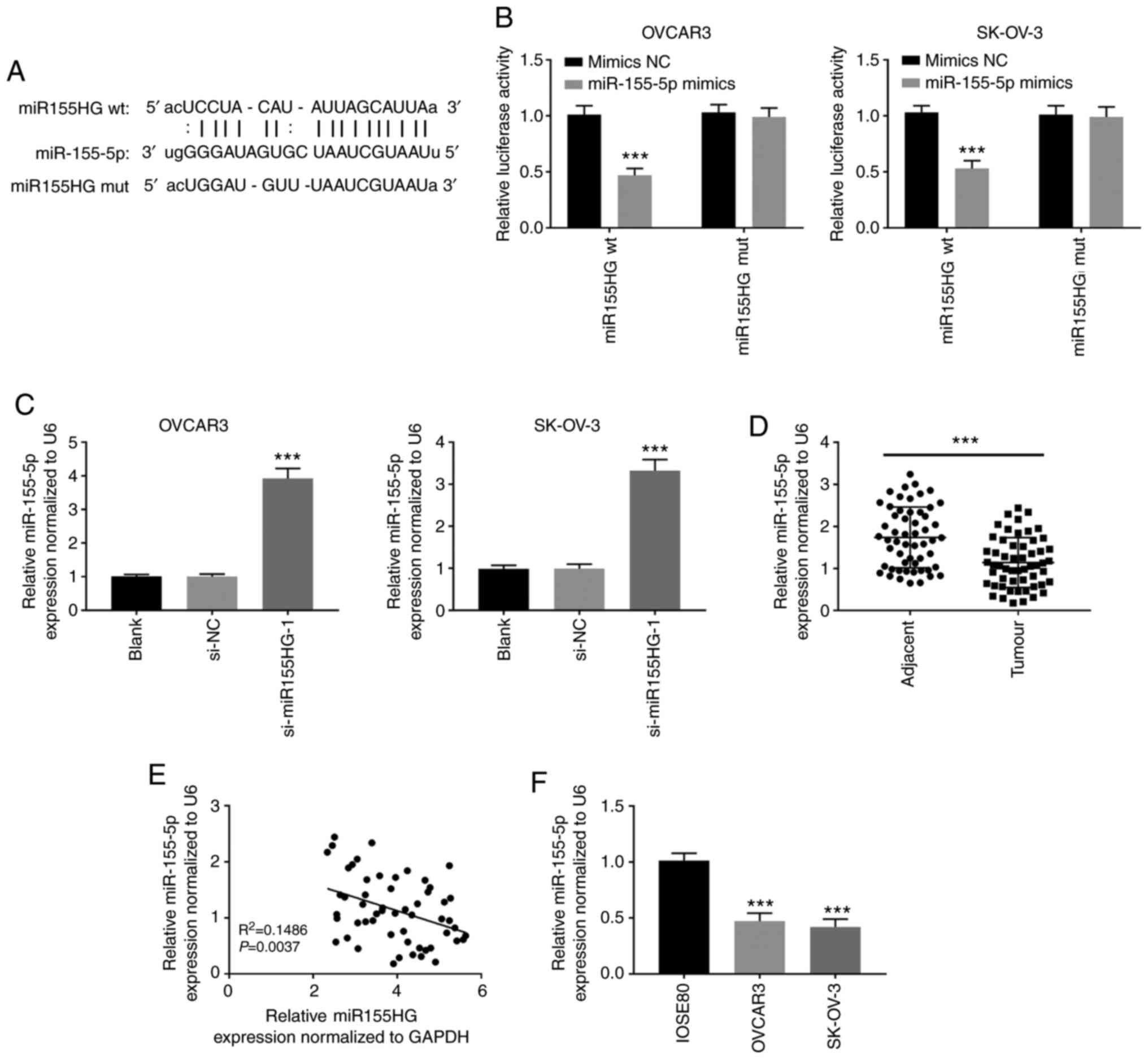
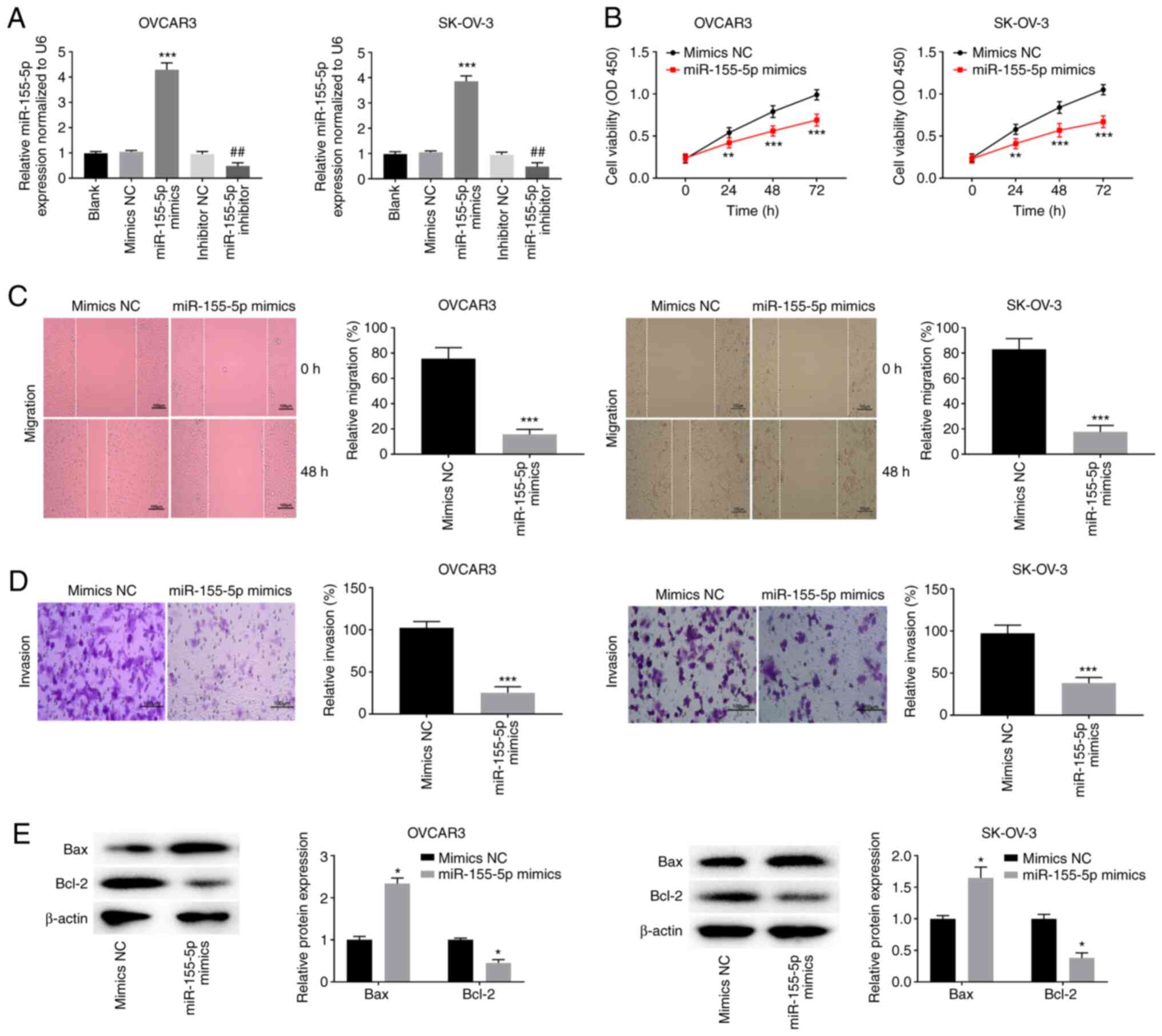
|
|
5
|
Chen Y, Du H, Bao L and Liu W: LncRNA PVT1
promotes ovarian cancer progression by silencing miR-214. Cancer
Biol Med. 15:238–250. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Du W, Feng Z and Sun Q: LncRNA LINC00319
accelerates ovarian cancer progression through miR-423-5p/NACC1
pathway. Biochem Biophys Res Commun. 507:198–202. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kong FR, Lv YH, Yao HM, Zhang HY, Zhou Y
and Liu SE: LncRNA PCAT6 promotes occurrence and development of
ovarian cancer by inhibiting PTEN. Eur Rev Med Pharmacol Sci.
23:8230–8238. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Qin Y, Liu X, Pan L, Zhou R and Zhang X:
Long noncoding RNA MIR155HG facilitates pancreatic cancer
progression through negative regulation of miR-802. J Cell Biochem.
120:17926–17934. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Wu W, Yu T, Wu Y, Tian W, Zhang J and Wang
Y: The miR155HG/miR-185/ANXA2 loop contributes to glioblastoma
growth and progression. J Exp Clin Cancer Res.
38(133)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Li N and Zhan X: Identification of
clinical trait-related lncRNA and mRNA biomarkers with weighted
gene co-expression network analysis as useful tool for personalized
medicine in ovarian cancer. EPMA J. 10:273–290. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Xiang G and Cheng Y: miR-126-3p inhibits
ovarian cancer proliferation and invasion via targeting PLXNB2.
Reprod Biol. 18:218–224. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Lu J, Wang L, Chen W, Wang Y, Zhen S, Chen
H, Cheng J, Zhou Y, Li X and Zhao L: miR-603 targeted hexokinase-2
to inhibit the malignancy of ovarian cancer cells. Arch Biochem
Biophys. 661:1–9. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Duan Y, Dong Y, Dang R, Hu Z, Yang Y, Hu Y
and Cheng J: MiR-122 inhibits epithelial mesenchymal transition by
regulating P4HA1 in ovarian cancer cells. Cell Biol Int.
42:1564–1574. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Li S, Zhang T, Zhou X, Du Z, Chen F, Luo J
and Liu Q: The tumor suppressor role of miR-155-5p in gastric
cancer. Oncol Lett. 16:2709–2714. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Luo X, Dong J, He X, Shen L, Long C, Liu
F, Liu X, Lin T, He D and Wei G: MiR-155-5p exerts
tumor-suppressing functions in wilms tumor by targeting IGF2 via
the PI3K signaling pathway. Biomed Pharmacother.
125(109880)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ysrafil Y, Astuti I, Anwar SL, Martien R,
Sumadi FAN, Wardhana T and Haryana SM: MicroRNA-155-5p diminishes
in vitro ovarian cancer cell viability by targeting HIF1α
expression. Adv Pharm Bull. 10:630–637. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Lai X, Wichers HJ, Soler-Lopez M and
Dijkstra BW: Structure and function of human tyrosinase and
tyrosinase-related proteins. Chemistry. 24:47–55. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wang-Rodriguez J, Urquidi V, Rivard A and
Goodison S: Elevated osteopontin and thrombospondin expression
identifies malignant human breast carcinoma but is not indicative
of metastatic status. Breast Cancer Res. 5(9)2003.PubMed/NCBI View
Article : Google Scholar
|
|
19
|
Udono T, Takahashi K, Yasumoto K,
Yoshizawa M, Takeda K, Abe T, Tamai M and Shibahara S: Expression
of tyrosinase-related protein 2/DOPAchrome tautomerase in the
retinoblastoma. Exp Eye Res. 72:225–234. 2001.PubMed/NCBI View Article : Google Scholar
|
|
20
|
El Hajj P, Journe F, Wiedig M, Laios I,
Salès F, Galibert MD, Van Kempen LC, Spatz A, Badran B, Larsimont
D, et al: Tyrosinase-related protein 1 mRNA expression in lymph
node metastases predicts overall survival in high-risk melanoma
patients. Br J Cancer. 108:1641–1647. 2013.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hsu YL, Chen YJ, Chang WA, Jian SF, Fan
HL, Wang JY and Kuo PL: Interaction between tumor-associated
dendritic cells and colon cancer cells contributes to tumor
progression via CXCL1. Int J Mol Sci. 19(2427)2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
El Hajj P, Gilot D, Migault M, Theunis A,
van Kempen LC, Salés F, Fayyad-Kazan H, Badran B, Larsimont D,
Awada A, et al: SNPs at miR-155 binding sites of TYRP1 explain
discrepancy between mRNA and protein and refine TYRP1 prognostic
value in melanoma. Br J Cancer. 113:91–98. 2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Choi JH and Ro JY: The 2020 WHO
classification of tumors of soft tissue: Selected changes and new
entities. Adv Anat Pathol. 28:44–58. 2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Cui W, Meng W, Zhao L, Cao H, Chi W and
Wang B: TGF-β-induced long non-coding RNA MIR155HG promotes the
progression and EMT of laryngeal squamous cell carcinoma by
regulating the miR-155-5p/SOX10 axis. Int J Oncol. 54:2005–2018.
2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Li N, Liu Y and Cai J: LncRNA miR155HG
regulates M1/M2 macrophage polarization in chronic obstructive
pulmonary disease. Biomed Pharmacother. 117(109015)2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Javadi S, Ganeshan DM, Qayyum A, Iyer RB
and Bhosale P: Ovarian cancer, the revised FIGO staging system, and
the role of imaging. AJR AM J Roentgenol. 206:1351–1360.
2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Liang H, Yu T, Han Y, Jiang H, Wang C, You
T, Zhao X, Shan H, Yang R, Yang L, et al: LncRNA PTAR promotes EMT
and invasion-metastasis in serous ovarian cancer by competitively
binding miR-101-3p to regulate ZEB1 expression. Mol Cancer.
17(119)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Wang X, Yang B, She Y and Ye Y: The lncRNA
TP73-AS1 promotes ovarian cancer cell proliferation and metastasis
via modulation of MMP2 and MMP9. J Cell Biochem. 119:7790–7799.
2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lai XJ and Cheng HF: LncRNA colon
cancer-associated transcript 1 (CCAT1) promotes proliferation and
metastasis of ovarian cancer via miR-1290. Eur Rev Med Pharmacol
Sci. 22:322–328. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zou T, Wang PL, Gao Y and Liang WT: Long
noncoding RNA HOTTIP is a significant indicator of ovarian cancer
prognosis and enhances cell proliferation and invasion. Cancer
Biomark. 25:133–139. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
He X, Sheng J, Yu W, Wang K, Zhu S and Liu
Q: LncRNA MIR155HG promotes temozolomide resistance by activating
the wnt/β-catenin pathway via binding to PTBP1 in glioma. Cell Mol
Neurobiol. 11:1271–1284. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ren XY, Han YD and Lin Q: Long non-coding
RNA MIR155HG knockdown suppresses cell proliferation, migration and
invasion in NSCLC by upregulating TP53INP1 directly targeted by
miR-155-3p and miR-155-5p. Eur Rev Med Pharmacol Sci. 24:4822–4835.
2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Fu X, Zhang L, Dan L, Wang K and Xu Y:
LncRNA EWSAT1 promotes ovarian cancer progression through targeting
miR-330-5p expression. Am J Transl Res. 9:4094–4103.
2017.PubMed/NCBI
|
|
35
|
Mu Y, Li N and Cui YL: The lncRNA CCAT1
upregulates TGFβR1 via sponging miR-490-3p to promote TGFβ1-induced
EMT of ovarian cancer cells. Cancer Cell Int.
18(145)2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Tong L, Ao Y, Zhang H, Wang K, Wang Y and
Ma Q: Long noncoding RNA NORAD is upregulated in epithelial ovarian
cancer and its downregulation suppressed cancer cell functions by
competing with miR-155-5p. Cancer Med. 8:4782–4791. 2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Wu X, Wang Y, Yu T, Nie E, Hu Q, Wu W, Zhi
T, Jiang K, Wang X, Lu X, et al: Blocking MIR155HG/miR-155 axis
inhibits mesenchymal transition in glioma. Neuro Oncol.
19:1195–1205. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Tao M, Zhou Y, Jin Y and Pu J: Blocking
lncRNA MIR155HG/miR-155-5p/-3p inhibits proliferation, invasion and
migration of clear cell renal cell carcinoma. Pathol Res Pract.
216(152803)2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Zhao XS, Han B, Zhao JX, Tao N and Dong
CY: miR-155-5p affects Wilms' tumor cell proliferation and
apoptosis via targeting CREB1. Eur Rev Med Pharmacol Sci.
23:1030–1037. 2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Chen L, Yang X, Zhao J, Xiong M,
Almaraihah R, Chen Z and Hou T: Circ_0008532 promotes bladder
cancer progression by regulation of the miR-155-5p/miR-330-5p/MTGR1
axis. J Exp Clin Cancer Res. 39(94)2020.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Journe F, Id Boufker H, Van Kempen L,
Galibert MD, Wiedig M, Salès F, Theunis A, Nonclercq D, Frau A,
Laurent G, et al: TYRP1 mRNA expression in melanoma metastases
correlates with clinical outcome. Br J Cancer. 105:1726–1732.
2011.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Liu G, Khong HT, Wheeler CJ, Yu JS, Black
KL and Ying H: Molecular and functional analysis of
tyrosinase-related protein (TRP)-2 as a cytotoxic T lymphocyte
target in patients with malignant glioma. J Immunother. 26:301–312.
2003.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Gilot D, Migault M, Bachelot L, Journé F,
Rogiers A, Donnou-Fournet E, Mogha A, Mouchet N, Pinel-Marie ML,
Mari B, et al: A non-coding function of TYRP1 mRNA promotes
melanoma growth. Nat Cell Biol. 19:1348–1357. 2017.PubMed/NCBI View Article : Google Scholar
|