Introduction
Ulcerative colitis (UC) is a complex, chronic and
non-specific inflammatory condition of the gastrointestinal tract
(1). It is one of the most common
subtypes of inflammatory bowel disease (2). The etiology of UC remains unclear,
although it is considered to involve genetic, environmental and
immunological factors, all of which contribute to the disease at
least in part by interacting with the gut microbiota (3,4). In
particular, the composition and function of the gut microbiota have
been increasingly reported to influence the immune system and
physiological functions in the colon (5). In fact, treatment options that are
currently available for UC primarily focus on preventing colonic
inflammation by restoring the composition of the gut microbiota
(6,7).
The NOD-, LRR- and pyrin domain-containing protein 3
(NLRP3) inflammasome is a multi-protein complex comprising NLRP3,
PYD and CARD domains that contain pro-caspase-1, which has been
found to serve a role in UC (8).
The NLRP3 inflammasome is stimulated by a diverse series of
agonists (such as ATP and UV radiation, and pattern-associated
molecular patterns derived from bacteria, virus, fungus, and
protozoa) (9), before activates
caspase-1, which results in the maturation of the proinflammatory
cytokine IL-1β (10). It was
previously reported that the activation of NLRP3 inflammasome
served an inflammatory role in dextran sulfate sodium (DSS)-induced
colitis (11).
Sinomenine (CAS: 115-53-7) is a pure alkaloid that
was originally extracted from the Chinese herb, Sinomenium
acutum (Rehder and Wilson) of the Menispermaceae family
(12). Sinomenine hydrochloride
(SIN) is a hydrochloride form of sinomenine that has attracted the
interest of researchers due to its reported safety and potent
anti-inflammatory and immunoregulatory effects both in vitro
and in vivo (13,14). It has been reported that SIN
demonstrated notable therapeutic efficacy for treating
collagen-induced arthritis in a rat model (15). However, little is currently known of
the potential effects of SIN on human UC. To date, several
sinomenine derivatives or Sinomenium acutum extracts have
been applied to treat rheumatoid arthritis in the clinic, such as
Sinomenone hydrochloride enteric-coated tablets (16). In addition, several pharmacokinetic
studies of sinomenine have been performed, which suggested that
sinomenine can be metabolized in humans and rats in vivo
(17,18). In addition, sinomenine can be
metabolized by human microsomal proteins in vitro (17,18).
Liu et al (19) previously
found that oral administration of sinomenine may be applicable for
future clinical studies, because at a dosage of 90 mg/kg in rats,
it achieved ~80% in terms of bioavailability. A previous study also
demonstrated that SIN alleviated dextran sulfate sodium
(DSS)-induced colitis in mice, by decreasing the levels of
oxidative stress in thecolon (20).
However, to the best of our knowledge, whether SIN exerts effects
on the composition of the gut microbiota remains to be
investigated.
UC has been reported to be partially caused by
disruptions in the balance between the immune system and the gut
microbiota (21). Consequently, the
present study aimed to assess the effects of SIN on colonic
inflammation and the composition of gut microbiota in a mouse model
of DSS-induced colitis. The present results may provide novel
insights into the possible application of SIN as a therapeutic
agent for regulating microbial homeostasis in UC.
Materials and methods
Reagents
SIN (cat. no. S105956) was purchased from Shanghai
Aladdin Biochemical Technology Co., Ltd. and was dissolved in 0.9%
NaCl. DSS (cat. no. 0216011080; molecular weight, 36,000-50,000 Da)
was purchased from MP Biomedicals, LLC.
Animals and study design
In total, 15 female C57BL/6 mice (age, 6-8 weeks;
body weight, 18-22 g) were purchased from Cavens Laboratory Animal
Co., Ltd. (Changzhou, China). All animal experiments were approved
by the Ethics Committee of Changzhou No. 2 People's Hospital
[approval no. SCXK (SU) 2016-0010; Changzhou, China] and animal
protocols were strictly conducted in accordance with the Animal
Research: Reporting of in vivo Experiments guidelines
(22). All mice were housed in
pathogen-free animal facilities under a standard 12-h light/dark
cycle at 25˚C and 40-50% humidity. All mice were group-housed in a
standard rodent unit with free access to food and water.
The 15 animals were randomized into the following
three groups (5 mice per group): i) Control; ii) DSS model; and
iii) DSS + SIN (100 mg/kg) groups. Colitis was not induced in the
control group and these mice were fed with standard food and water
during the whole experiment. Mice in the DSS and DSS + SIN groups
were treated with 3% DSS, which was supplemented in drinking water,
for 7 days and then provided with normal drinking water for 3 days
for recovery (23). By contrast,
mice in the DSS + SIN group were provided with 100 mg/kg SIN by
oral gavage from days 1 to 10 daily. On day 11, all the mice were
sacrificed by CO2 suffocation. The air replacement rate
of CO2 was 10-30% of the container volume per min, which
lasted for ~5 min.
During the experiment, body weight, stool
consistency and fecal bleeding scores of each animal were observed
to calculate the disease activity index (DAI) (15,24)
every day. The scoring system for the DAI is presented in Table I. Following sacrifice, the spleens
were obtained from each mice and weighed. Spleen index (%)=Spleen
weight (mg)/body weight on day 11 (g) x10. In addition, after the
mice were sacrificed, the colons were removed from each mouse and
the colon length was measured. The dissected colon tissues were
then washed with cold PBS before one section of the distal colon
was stored at -80˚C for subsequent biochemical examination. The
remaining tissue was fixed in 4% paraformaldehyde at room
temperature for 24 h for further histopathological analysis.
 | Table IDisease activity index scoring
systema. |
Table I
Disease activity index scoring
systema.
| Score | Weight loss
(%)b | Stool
consistency | Blood in stool |
|---|
| 0 | None | Well-formed
stools | Negative |
| 1 | 1-5% | Well-formed
stools | Negative |
| 2 | 6-10% | Pasty stools that
did not adhere to the anus | Hemoccult
positive |
| 3 | 11-15% | Pasty stools that
did not adhere to the anus | Hemoccult
positive |
| 4 | >15% | Liquid stools that
adhered to the anus | Gross bleeding |
Histological analysis
The fixed colon sections were dehydrated with an
ascending ethanol gradient (70-100%) and embedded in paraffin
before 4-µm colon sections were obtained and stained with the
Hematoxylin-Eosin Staining kit (cat. no. G1120; Beijing Solarbio
Science & Technology Co., Ltd.) according to the manufacturer's
protocol (25). Briefly, the
sections were deparaffinized in xylene at room temperature,
rehydrated with a decreasing concentration of ethanol (100-70%) and
washed with distilled water. The sections were incubated with
hematoxylin solution for 10 min and eosin solution for 2 min, both
at room temperature before being washed with water. The stained
sections were dehydrated in an ascending ethanol gradient
(90-100%), cleared with xylene and observed using a light
microscope (magnifications, x40, x100 and x200; Olympus BX51;
Olympus Corporation).
Measurement of colonic inflammatory
cytokines
The mRNA expression levels of TNF-α, IL-6, IL-10,
inducible nitric oxide synthase (iNOS) and arginase 1 (Arg1) in the
colon tissues of mice were determined using reverse
transcription-quantitative PCR (RT-qPCR). Total RNA was extracted
from colon tissues using a RNAsimple Total RNA kit (cat. no. DP419;
Tiangen Biotech Co., Ltd.) according to manufacturer's protocol.
Total RNA was reverse transcribed into cDNA using a PrimeScript™ RT
reagent kit with gDNA Eraser (Perfect Real-time; cat. no. RR047A;
Takara Bio, Inc.) at 37˚C for 5 min and 85˚C for 5 sec before the
samples were cooled to 4˚C. qPCR was subsequently performed using
an AceQ® qPCR SYBR® Green Master mix (Low ROX
Premixed; cat. no. Q131-02; Vazyme Biotech Co., Ltd.) on an ABI
7500 Real-time PCR system (Applied Biosystems; Thermo Fisher
Scientific, Inc.). The following thermocycling conditions were used
for the qPCR: Initial denaturation at 95˚C for 5 min; followed by
40 cycles at 95˚C for 10 sec and 60˚C for 30 sec. The fold changes
in the expression levels of each gene were calculated using the
2-ΔΔCq method (26). The
mRNA expression levels for each target gene were normalized to the
expression levels of GAPDH. The primers used for the qPCR are
listed in Table II.
 | Table IIPrimer sequences for reverse
transcription-PCR. |
Table II
Primer sequences for reverse
transcription-PCR.
| Gene | Sequences
(5'-3') |
|---|
| TNF-α | F:
CATCTTCTCAAAATTCGAGTGAC |
| | R:
TGGGAGTAGACAAGGTACAACCC |
| IL-6 | F:
GCTGGTGACAACCACGGCCT |
| | R:
AGCCTCGACTTGTGAAGTGGT |
| IL-10 | F:
AGATGATGACCCTTTTGGCTC |
| | R:
AGATGATGACCCTTTTGGCTC |
| iNOS | F:
CCAACCTGCAGGTCTTCGATG |
| | R:
GTCGATGCACAACTGGGTGAAC |
| Arg 1 | F:
GAACCCAACTCTTGGGAAGAC |
| | R:
GGAGAAGGCGTTTGCTTAGTT |
| GAPDH | F:
AAGGTCGGAGTCAACGGATTT |
| | R:
AGATGATGACCCTTTTGGCTC |
Gut microbiota 16S ribosomal (r)DNA
analysis
Murine fecal samples were collected between days 8
and 11 and then stored at -80˚C for use in subsequent experiments.
Bacterial DNA was isolated from the samples using an E.Z.N.A.™
stool DNA kit (cat. no. D5625-01; Omega Bio-Tek, Inc.) according to
the manufacturer's protocol. Determination of DNA quality and 16S
rDNA analysis were conducted by Guangzhou RiboBio Co., Ltd. DNA
integrity was detected by 1% agarose gel electrophoresis, and was
amplified using NEBNext® Ultra™ II Q5 Master Mix (cat.
no. M0544L 250rxn; New England Biolabs, Inc.) for 15 cycles with a
specific primer set containing the RiboBio® barcode
(Guangzhou RiboBio Co., Ltd.). The products were verified using an
Agilent 2200 Bioanalyzer (Agilent Technologies, Inc.). Bacterial
16S rRNA gene sequences (V3-V4 region) were performed using a MiSeq
Reagent Kit v3 (600 cycle) (cat. no. MS-102-3003; Illumina, Inc.)
on a Illumina HiSeq 2500 platform (Illumina Inc.) and 250 bp
paired-end reads were obtained. The DNA loading concentration was
10 pmol, which measured using Qubit 2.0. The paired-end reads were
merged using Fast Length Adjustment of SHort reads (27) and then assigned to each sample based
on their unique barcodes conducted by QIIME software (version 1.7;
QIIME development team; https://qiime.org/). Within-sample microbiota
diversity (α-diversity) was evaluated using Chao1 and Shannon
(28), which measured gut
microbiota richness and evenness. Additional α-diversity assessment
was performed with PD Whole Tree, which measure species phylogeny
(29). All α-diversity indices were
calculated with QIIME (version 1.7; QIIME development team).
Principal component analysis (PCA) and principal coordinate
analysis (PCoA) was measured at the operational taxonomic units
(OTU) level and hierarchical clustering tree on Genus level to
compare the differences among microbial communities (30). The community structure was based on
the weighted UniFrac distance (31). In this study, the OTUs were
clustered with a similarity cutoff value of 97% using UPARSE
software (version 7.0) (32).
Western blotting
Total protein was extracted from colon samples using
Western & IP cell lysis buffer (cat. no. P0013; Beyotime
Institute of Biotechnology) supplemented with the PMSF protease
inhibitor (Beyotime Institute of Biotechnology) according to the
manufacturer's protocol. Protein concentration was determined using
an Enhanced BCA Protein Assay kit (cat. no. P0010; Beyotime
Institute of Biotechnology). Protein extracts were stored at -70˚C
until required for further experimentation. Proteins (40 µg/lane)
were separated via 12% SDS-PAGE, transferred onto PVDF membranes
(cat. no. IPVH00010; EMD Millipore) and blocked using blocking
buffer (cat. no. P0023B-100 ml; Beyotime Institute of
Biotechnology) for 2 h at 37˚C. The membranes were then incubated
with the following primary antibodies overnight at 4˚C: Anti-NLRP3
(1:1,000; cat. no. ab210491; rabbit monoclonal; Abcam),
anti-caspase-1 (1:1,000; cat. no. ab207802; rabbit monoclonal;
Abcam) and anti-β-actin (1:1,000; cat. no. 4970T; rabbit
monoclonal; Cell Signaling Technology, Inc.). Following primary
antibody incubation, the membranes were washed with TBS-Tween 20
(0.05%) and incubated with an HRP-conjugated Affini-pure goat
anti-rabbit IgG (H + L) secondary antibody (1:7,500; cat. no.
BA1054; Boster Biological Technology) for 1.5 h at room
temperature. All antibodies were diluted in the primary antibody
dilution buffer (cat. no. P0023A-100 ml; Beyotime Institute of
Biotechnology). Protein bands were visualized using SuperSignal
West Pico Chemiluminescent substrate (cat. no. 34077; Thermo Fisher
Scientific, Inc.) and immunoblots were quantified using ImageJ
version 1.8 software (National Institutes of Health).
Statistical analysis
All data are presented as the mean ± SEM.
Statistical differences among groups were determined using
Kruskal-Wallis followed by Dunn's test, one-way ANOVA and Sidak's
multiple comparison test with GraphPad Prism 5 software (GraphPad
Software, Inc.). P≤0.05 was considered to indicate a statistically
significant difference.
Results
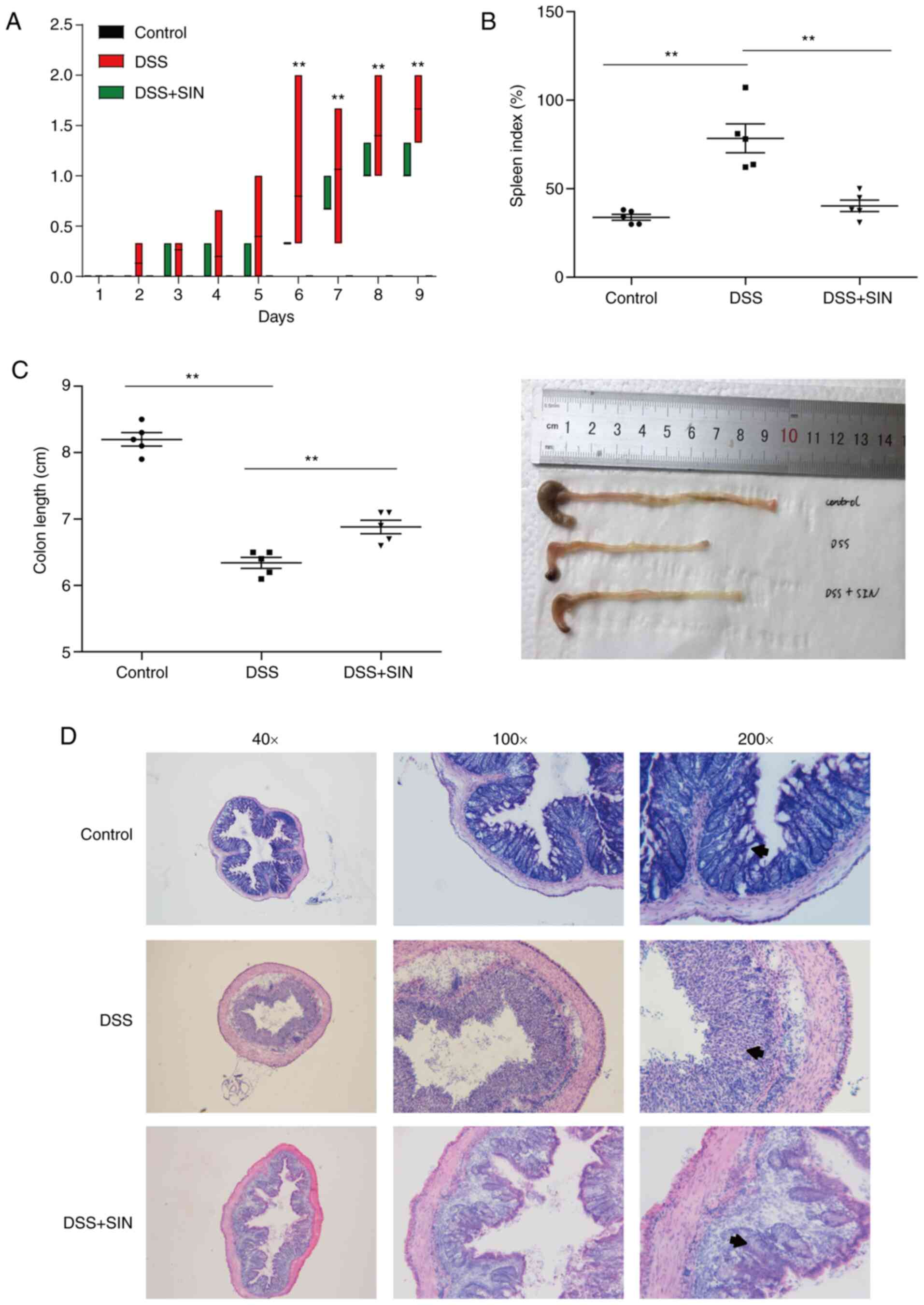
SIN alleviates DSS-induced colitis in
mice
Mice treated with 3% DSS developed severe colitis,
which exhibited significantly higher DAI scores compared with those
in control mice from day 6 (P<0.01; Fig. 1A). Notably, the DAI score of DSS +
SIN group was decreased compared with that in the DSS group from
day 4, though there was no statistical difference (Fig. 1A). SIN supplementation significantly
reduced the DAI scores spleen indices compared with those in the
untreated DSS-induced mice (Fig.
1B). DSS-induced colonic shortening was also significantly
improved by SIN administration (Fig.
1C). Pathologically, the colons of mice in the control group
had an intact structure (Fig. 1D).
As the arrows indicated, gland defects, mucosal ulcerations and
inflammatory cell infiltration were observed in the colons of
DSS-induced mice, whilst SIN supplementation attenuated this
histological damage (Fig. 1D).
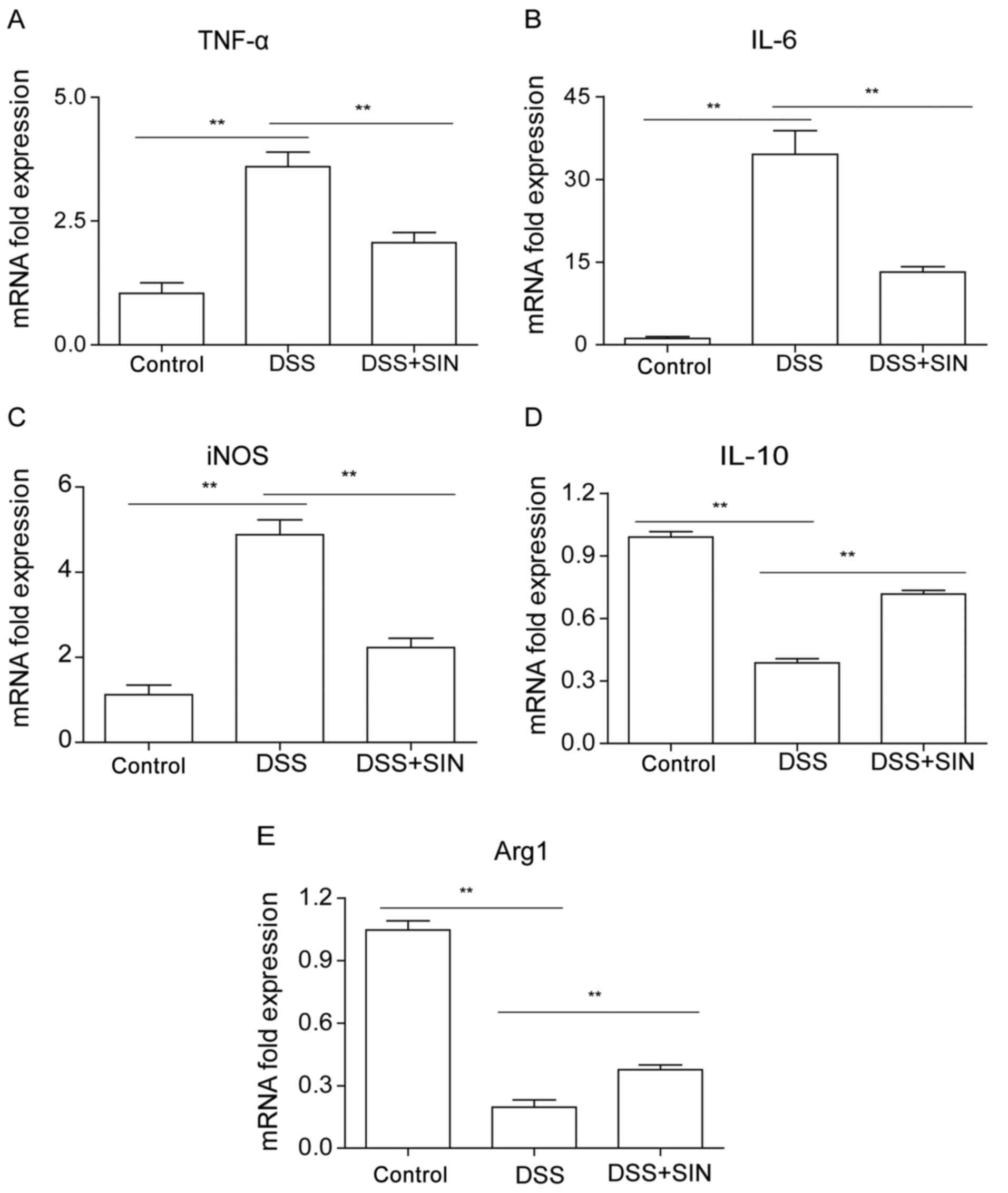
SIN regulates the expression levels of
inflammatory cytokines in the colon
As presented in Fig.
2A-C, the mRNA expression levels of TNF-α, IL-6 and iNOS were
found to be significantly upregulated in the colon tissues of mice
treated with DSS, whilst the combined treatment of DSS with SIN
significantly suppressed the increased expression. DSS treatment
significantly downregulated IL-10 and Arg1 mRNA expression levels
in the colon compared with those in the control group, which were
significantly reversed by SIN treatment (Fig. 2D and E).
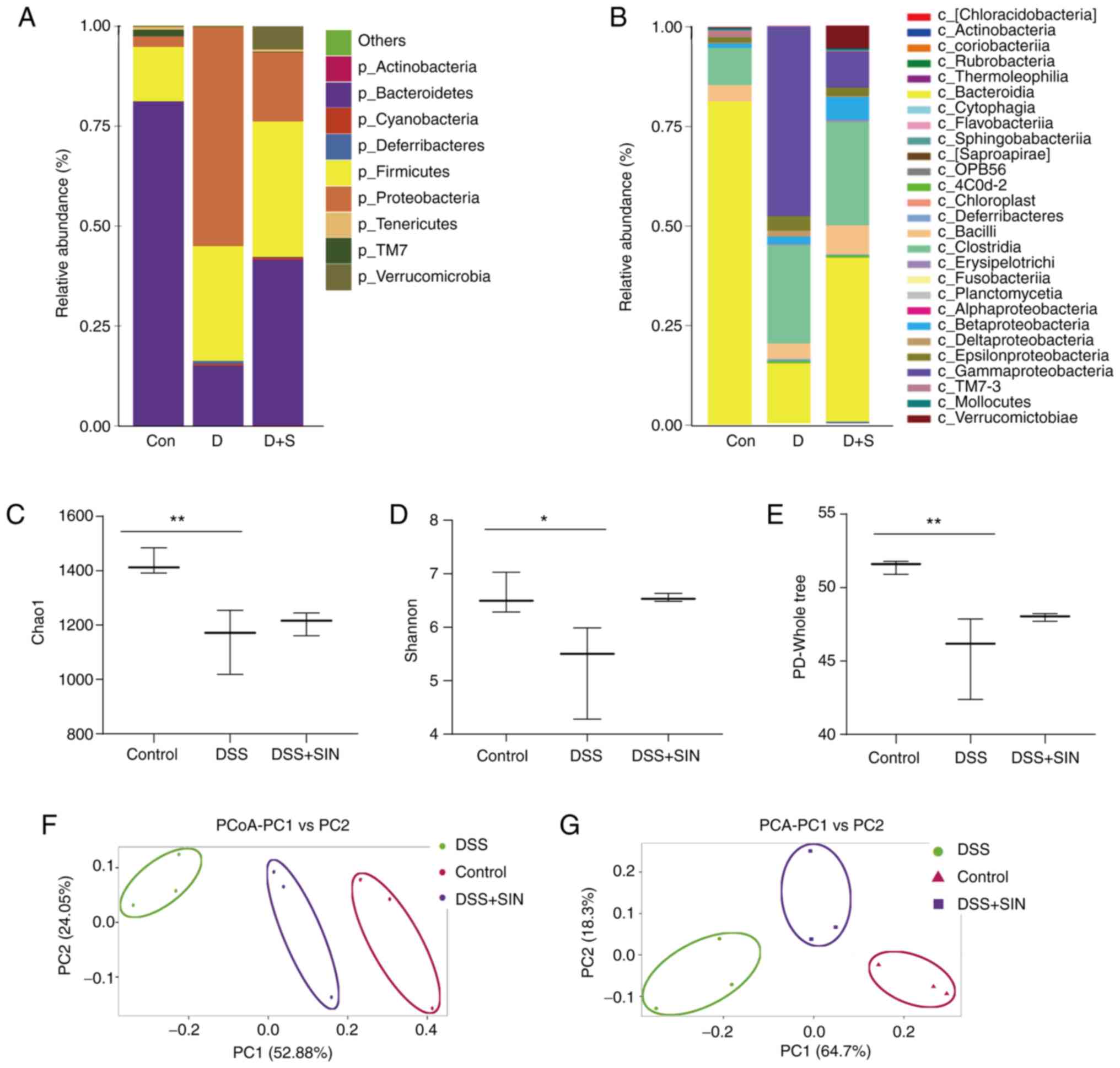
SIN moderates the gut microbiota
composition in the colons of mice
The species abundance and community composition of
the gut microbiota in mice from the different treatment groups were
analyzed using 16S rDNA sequencing. On the phylum level, the
relative abundance of Bacteroidetes in mice was markedly
reduced following DSS treatment, which was partially reversed
following SIN treatment (Fig. 3A).
Conversely, the relative abundance of Firmicutes and
Proteobacteria was increased in DSS group compared with that
in the control group (Fig. 3A). In
the DSS + SIN group, the abundance of Proteobacteria was
markedly decreased compared with that in the DSS-only group
(Fig. 3A). On a class level,
Bacteroidia, Clostridia and γ-proteobacteria were the dominant
species present (Fig. 3B).
Following DSS treatment, the abundance of Bacteroidia was
decreased, whereas the abundance of Clostridia and γ-proteobacteria
was increased compared with that in the control group (Fig. 3B). SIN treatment markedly reduced
the abundance of γ-proteobacteria whilst increasing the abundance
of Bacteroidia compared with that in the DSS-only group. However,
no differences were observed in the abundance of Clostridia between
the DSS and DSS + SIN groups (Fig.
3B).
Subsequently, comparisons of the α-diversity of
different groups were performed using the Chao1(29), Shannon (33) and PD-Whole tree indices (30). DSS treatment significantly decreased
the α-diversity of gut microbiota community compared with that in
the control group (Fig. 3C-E).
Although those in the DSS + SIN group exhibited markedly increased
Shannon and PD-Whole tree indices, the differences were not
significant (Fig. 3D and E). A marked difference was identified in
the microbial β-diversity based on principal coordinate and
principal component analyzes between the DSS and control groups,
whilst the microbial β-diversity in the DSS + SIN group appeared to
be closer to that of the control group (Fig. 3F and G).
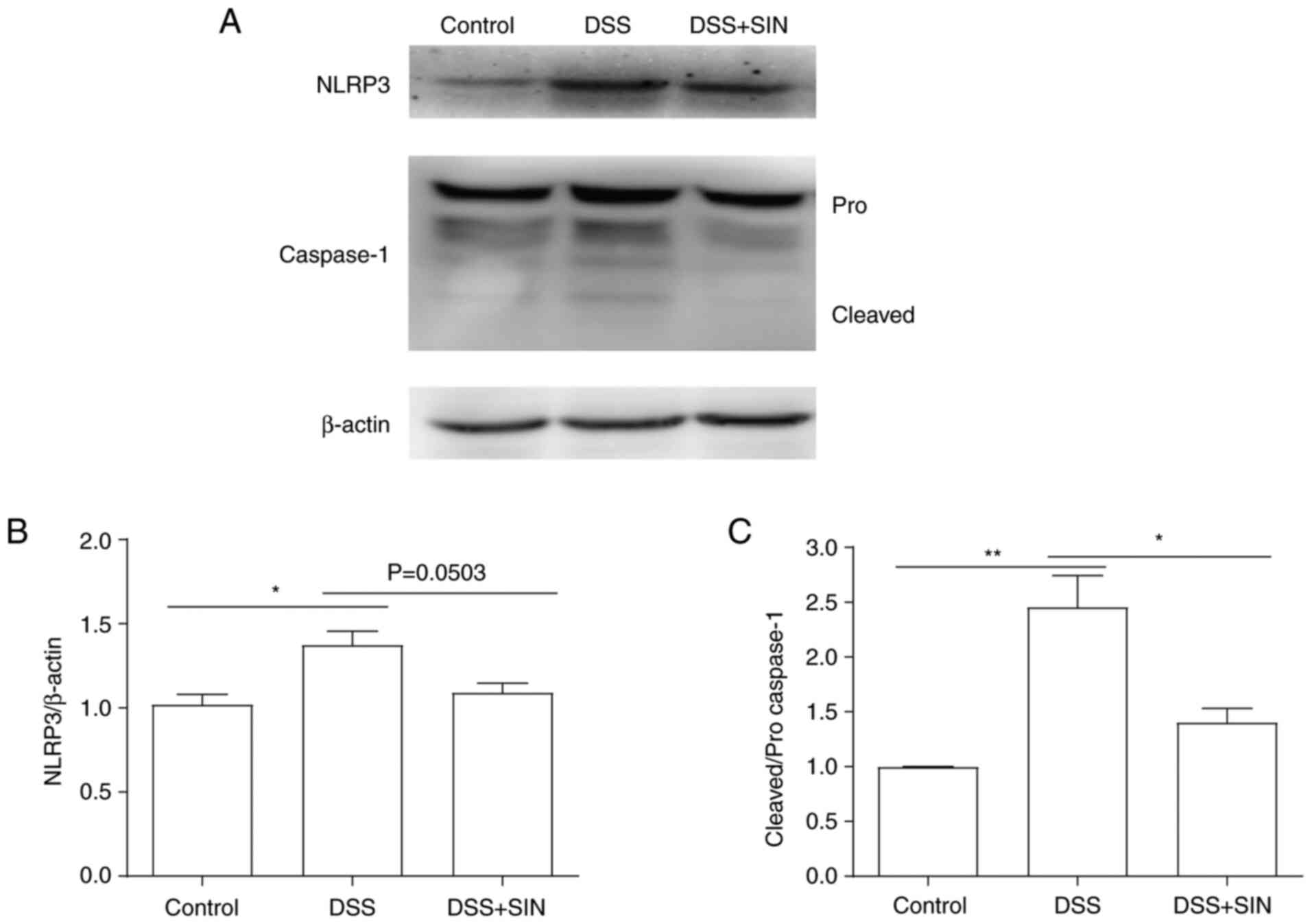
SIN inhibits the activation of the
NLRP3 inflammasome
The results in Fig.
4 revealed that activation of the NLRP3 inflammasome and
cleaved caspase-1 expression levels were significantly increased
following DSS treatment, whilst treatment with SIN markedly
decreased both the activation of the NLRP3 inflammasome and
downregulated the expression levels of cleaved caspase-1. However,
no significance was observed in NLRP3 expression between the DSS
and DSS + SIN groups. The expression levels of pro-caspase-1
remained unchanged following the different treatments (Fig. 4).
Discussion
Anti-inflammatory and immunosuppressive drugs are
commonly used for treating UC in the clinic, with examples
including glucocorticoids, azathioprine, biological agents, janus
kinase inhibitors (TNF antibodies, ustekinumab, vedolizumab, and
tofacitinib) and calcineurin inhibitors (34). However, these drugs can cause
numerous side effects, such as Sweet's syndrome (35). Traditional Chinese herbal medicine
has attracted interest due to its reported effects in preventing
and controlling digestive diseases (36,37).
Park et al (36) reported
that pristimerin attenuated tumorigenesis in a mouse model of
experimental colitis-associated colon cancer. In another study,
Zheng et al (37) revealed
that silibinin administration ameliorated colitis and inhibited
colitis-associated tumorigenesis in a mouse model. Sinomenine can
be extracted from the herb Sinomenium acutum, such that SIN
is its form of hydrochloride (12).
Given that there were few reports about the effects of SIN on
diseases in the digestive system (20,38),
the present study aimed to determine the possible effects and
underlying mechanism of SIN on a mouse model of DSS-induced
colitis, which has been previously found to successfully mimic the
clinical symptoms of UC (39).
Consistent with the findings of previous studie (20,38),
the results of the present study revealed that the DAI and spleen
indices were markedly increased, and colon length was shortened in
DSS-treated mice compared with those in the control group. Notably,
SIN treatment significantly attenuated the DSS-induced effects on
the DAI, spleen index and colon length. Histological analysis
revealed that SIN prevented crypt destruction and inflammatory cell
infiltration caused by DSS treatment. These findings suggest that
SIN may exert protective effects against UC in the colon of
mice.
Recent studies suggested that inflammatory cytokines
and mediators serve important roles in facilitating mucosal immune
responses (40,41). High levels of inflammatory
cytokines, including TNF, IFN-γ and IL-6 can cause intestinal
dysfunction, which is associated with the severity of UC (42). Macrophages are abundant within the
intestine, where its polarization occurs during the development of
UC (43). M1 macrophages
are characterized by the high expression levels of iNOS, which
produce a wide range of proinflammatory cytokines, including TNF-α
and IL-6, to aggravate UC (32,33).
By contrast, M2 macrophages release anti-inflammatory
mediators, including Arg1 and IL-10, to reduce UC symptoms
(44,45). The present results revealed that SIN
treatment significantly downregulated the DSS-induced upregulation
of TNF-α, IL-6 and iNOS expression. Moreover, SIN treatment
increased the production of IL-10 and Arg1, which were initially
decreased by DSS.
Changes in the gut microbiota profile is considered
to be one of the key factors associated with UC (46). The colonic immune system can be
activated by gut microbiota (47).
Loss of diversity in the species of flora in the gut can affect the
host's gut immune responses, leading to increased risk of
inflammation and pathological damage (48-50).
The results of the present study demonstrated that SIN may exert an
anti-colitis effect by modulating the gut microbiota composition.
The loss of phylum Bacteroidetes (class Bacteroidia) species
diversity in patients with UC was previously found, which was in
accordance with the endoscopic index of severity (51,52).
In the present study, DSS treatment decreased the phylum
Bacteroidetes (class Bacteroidia) abundance, whilst SIN
treatment increased the abundance compared with that in the
DSS-only group. During gut inflammation, Enterobacteriaceae
family (phylum Proteobacteria) has previously exhibited a dysbiotic
expansion (53). The present study
also observed that the phylum Proteobacteria and class
γ-proteobacteria were expanded after DSS treatment, whilst SIN
administration suppressed their abundance, compared with that in
the DSS-only group. However, the mechanism through which SIN
regulated the composition of the gut microbiota in this mouse model
of colitis requires further investigation.
The NLRP3 inflammasome is a large multimeric protein
complex (8). Activation of the
NLRP3 inflammasome has been found to promote colon inflammation and
DSS-induced UC by activating caspase-1(54). In the present study, the expression
levels of NLRP3 and caspase-1 in colonic tissues were analyzed, and
the results demonstrated that SIN treatment inhibited the
activation of the NLRP3 inflammasome in DSS-induced colitis.
In conclusion, the present results demonstrated that
SIN treatment alleviated DSS-induced colitis not only by regulating
the gut microbiota composition, but also by inhibiting the
activation of the NLRP3 inflammasome. These beneficial effects may
provide novel preclinical evidence for the potential application of
SIN for treating UC.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by grants from the
National Natural Science Foundation of China (grant no. 81803498),
China Postdoctoral Science Foundation (grant no. 2020M670012ZX),
Jiangsu Natural Science Foundation (grant no. BK20181155), Jiangsu
Planned Projects for Postdoctoral Research Funds (grant no.
2019K157), Changzhou Sci&Tech Program (grant no. CJ20200090),
Young Science & Technology Project of Changzhou Health
Commission (grant no. QN202032) and Funding from Young Talent
Development Plan of Changzhou Health Commission (grant nos.
CZQM2020071 and CZQM2020063).
Availability of data and materials
The datasets generated and/or analyzed during the
current study are available in the Sequence Read Archive
repository; https://www.ncbi.nlm.nih.gov/bioproject/739479.
Authors' contributions
YZ designed the study and wrote the manuscript. YZ,
SC, XS and WG conducted the experiments and performed the
statistical analysis. YZ, LW and LT supervised the experiments,
analyzed the data and revised the manuscript. YZ and LW confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent for
participation
Research involving animals was approved by the
Ethics Committee of the Affiliated Changzhou No. 2 People's
Hospital of Nanjing Medical University (Changzhou, China). All
animal protocols performed in this study were conducted strictly
based on the guidelines of the Jiangsu Committee on Animal
Care.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Null KD, Xu Y, Pasquale MK, Su C, Marren
A, Harnett J, Mardekian J, Manuchehri A and Healey P: Ulcerative
colitis treatment patterns and cost of care. Value Health.
20:752–761. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
van den Brink G, van Gaalen MAC, de Ridder
L, van der Woude CJ and Escher JC: Health care transition outcomes
in inflammatory bowel disease: A multinational delphi study. J
Crohns Colitis. 13:1163–1172. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Munyaka PM, Rabbi MF, Khafipour E and Ghia
JE: Acute dextran sulfate sodium (DSS)-induced colitis promotes gut
microbial dysbiosis in mice. J Basic Microbiol. 56:986–998.
2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Alavala S, Sangaraju R, Nalban N, Sahu BD,
Jerald MK, Kilari EK and Sistla R: Stevioside, a diterpenoid
glycoside, shows anti-inflammatory property against dextran
sulphate sodium-induced ulcerative colitis in mice. Eur J
Pharmacol. 855:192–201. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Klimesova K, Kverka M, Zakostelska Z,
Hudcovic T, Hrncir T, Stepankova R, Rossmann P, Ridl J, Kostovcik
M, Mrazek J, et al: Altered gut microbiota promotes
colitis-associated cancer in IL-1 receptor-associated kinase
M-deficient mice. Inflamm Bowel Dis. 19:1266–1277. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wang K, Jin X, You M, Tian W, Le Leu RK,
Topping DL, Conlon MA, Wu L and Hu F: Dietary propolis ameliorates
dextran sulfate sodium-induced colitis and modulates the gut
microbiota in rats fed a western diet. Nutrients.
9(875)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Yeom Y, Kim BS, Kim SJ and Kim Y: Sasa
quelpaertensis leaf extract regulates microbial dysbiosis by
modulating the composition and diversity of the microbiota in
dextran sulfate sodium-induced colitis mice. BMC Complement Altern
Med. 16(481)2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
He R, Li Y, Han C, Lin R, Qian W and Hou
X: L-Fucose ameliorates DSS-induced acute colitis via inhibiting
macrophage M1 polarization and inhibiting NLRP3 inflammasome and
NF-kB activation. Int Immunopharmacol. 73:379–388. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Davis BK, Wen H and Ting JP: The
inflammasome NLRs in immunity, inflammation, and associated
diseases. Annu Rev Immunol. 29:707–735. 2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lian D, Lai J, Wu Y, Wang L and Chen Y,
Zhang Y, Boini KM, Huang Y and Chen Y: Cathepsin B-mediated NLRP3
inflammasome formation and activation in angiotensin II-induced
hypertensive mice: Role of macrophage digestion dysfunction. Cell
Physiol Biochem. 50:1585–1600. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
He X, Wei Z, Wang J, Kou J, Liu W, Fu Y
and Yang Z: Alpinetin attenuates inflammatory responses by
suppressing TLR4 and NLRP3 signaling pathways in DSS-induced acute
colitis. Sci Rep. 6(28370)2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chan K, Liu ZQ, Jiang ZH, Zhou H, Wong YF,
Xu HX and Liu L: The effects of sinomenine on intestinal absorption
of paeoniflorin by the everted rat gut sac model. J Ethnopharmacol.
103:425–432. 2006.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chen H, Wang Y, Jiao FZ, Yang F, Li X and
Wang LW: Sinomenine attenuates acetaminophen-induced acute liver
injury by decreasing oxidative stress and inflammatory response via
regulating TGF-β/smad pathway in vitro and in vivo. Drug Des Devel
Ther. 14:2393–2403. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Li X, Wang K, Ren Y, Zhang L, Tang XJ,
Zhang HM, Zhao CQ, Liu PJ, Zhang JM and He JJ: MAPK signaling
mediates sinomenine hydrochloride-induced human breast cancer cell
death via both reactive oxygen species-dependent and -independent
pathways: An in vitro and in vivo study. Cell Death Dis.
5(e1356)2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Tong B, Yu J, Wang T, Dou Y, Wu X, Kong L,
Dai Y and Xia Y: Sinomenine suppresses collagen-induced arthritis
by reciprocal modulation of regulatory T cells and Th17 cells in
gut-associated lymphoid tissues. Mol Immunol. 65:94–103.
2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Huang RY, Pan HD, Wu JQ, Zhou H, Li ZG,
Qiu P, Zhou YY, Chen XM, Xie ZX, Xiao Y, et al: Comparison of
combination therapy with methotrexate and sinomenine or leflunomide
for active rheumatoid arthritis: A randomized controlled clinical
trial. Phytomedicine. 57:403–410. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yao YM, Tan ZR, Hu ZY, Guo X, Cheng ZN,
Wang LS and Zhou HH: Determination of sinomenine in human plasma by
HPLC/ESI/ion trap mass spectrum. Clin Chim Acta. 356:212–217.
2005.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Long LH, Wu PF, Chen XL, Zhang Z, Chen Y,
Li YY, Jin Y, Chen JG and Wang F: HPLC and LC-MS analysis of
sinomenine and its application in pharmacokinetic studies in rats.
Acta Pharmacol Sin. 31:1508–1514. 2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Liu ZQ, Chan K, Zhou H, Jiang ZH, Wong YF,
Xu HX and Liu L: The pharmacokinetics and tissue distribution of
sinomenine in rats and its protein binding ability in vitro. Life
Sci. 77:3197–3209. 2005.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhou Y, Liu H, Song J, Cao L, Tang L and
Qi C: Sinomenine alleviates dextran sulfate sodium-nduced colitis
via the Nrf2/NQO-1 signaling pathway. Mol Med Rep. 18:3691–3698.
2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Gkouskou KK, Deligianni C, Tsatsanis C and
Eliopoulos AG: The gut microbiota in mouse models of inflammatory
bowel disease. Front Cell Infect Microbiol. 4(28)2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kilkenny C, Browne WJ, Cuthill IC, Emerson
M and Altman DG: Improving bioscience research reporting: The
ARRIVE guidelines for reporting animal research. J Pharmacol
Pharmacother. 1:94–99. 2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Bang B and Lichtenberger LM: Methods of
inducing inflammatory bowel disease in mice. Curr Protoc Pharmacol.
72:5.58.1–5.58.42. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Vong LB, Tomita T, Yoshitomi T, Matsui H
and Nagasaki Y: An orally administered redox nanoparticle that
accumulates in the colonic mucosa and reduces colitis in mice.
Gastroenterology. 143:1027–1036.e3. 2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Feldman AT and Wolfe D: Tissue processing
and hematoxylin and eosin staining. Methods Mol Biol. 1180:31–43.
2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Magoč T and Salzberg SL: FLASH: Fast
length adjustment of short reads to improve genome assemblies.
Bioinformatics. 27:2957–2963. 2011.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Wang FG, Bai RX, Yan WM, Yan M, Dong LY
and Song MM: Differential composition of gut microbiota among
healthy volunteers, morbidly obese patients and post-bariatric
surgery patients. Exp Ther Med. 17:2268–2278. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Carter SJ, Hunter GR, Blackston JW, Liu N,
Lefkowitz EJ, Van Der Pol WJ, Morrow CD, Paulsen JA and Rogers LQ:
Gut microbiota diversity is associated with cardiorespiratory
fitness in post-primary treatment breast cancer survivors. Exp
Physiol. 104:529–539. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lozupone C and Knight R: UniFrac: A new
phylogenetic method for comparing microbial communities. Appl
Environ Microbiol. 71:8228–8235. 2005.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lozupone C, Lladser ME, Knights D,
Stombaugh J and Knight R: UniFrac: An effective distance metric for
microbial community comparison. ISME J. 5:169–172. 2011.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Edgar RC: UPARSE: Highly accurate OTU
sequences from microbial amplicon reads. Nat Methods. 10:996–998.
2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Dong T, Feng Q, Liu F, Chang LK, Zhou X,
Han M, Tian X, Zhong N and Liu S: Alteration of stomach microbiota
compositions in the progression of gastritis induces nitric oxide
in gastric cell. Exp Ther Med. 13:2793–2800. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Kucharzik T, Koletzko S, Kannengiesser K
and Dignass A: Ulcerative colitis-diagnostic and therapeutic
algorithms. Dtsch Arztebl Int. 117:564–574. 2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Moreno Márquez C, Maldonado Pérez B and
Castro Laria L: Infliximab as rescue treatment in Sweet's syndrome
related to corticodependent ulcerative colitis. J Crohns Colitis.
12:755–756. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Park JH and Kim JK: Pristimerin, a
naturally occurring triterpenoid, attenuates tumorigenesis in
experimental colitis-associated colon cancer. Phytomedicine.
42:164–171. 2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Zheng R, Ma J, Wang D, Dong W, Wang S, Liu
T, Xie R, Liu L, Wang B and Cao H: Chemopreventive effects of
silibinin on colitis-associated tumorigenesis by inhibiting
IL-6/STAT3 signaling pathway. Mediators Inflamm.
2018(1562010)2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xiong H, Tian L, Zhao Z, Chen S, Zhao Q,
Hong J, Xie Y, Zhou N and Fu Y: The sinomenine enteric-coated
microspheres suppressed the TLR/NF-κB signaling in DSS-induced
experimental colitis. Int Immunopharmacol. 50:251–262.
2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Wang K, Jin X, Li Q, Sawaya ACHF, Le Leu
RK, Conlon MA, Wu L and Hu F: Propolis from different geographic
origins decreases intestinal inflammation and bacteroides spp.
populations in a model of DSS-induced colitis. Mol Nutr Food Res.
62(e1800080)2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Wang W, Li X, Zheng D, Zhang D, Peng X,
Zhang X, Ai F, Wang X, Ma J, Xiong W, et al: Dynamic changes and
functions of macrophages and M1/M2 subpopulations during ulcerative
colitis-associated carcinogenesis in an AOM/DSS mouse model. Mol
Med Rep. 11:2397–2406. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Yao D, Dong M, Dai C and Wu S:
Inflammation and inflammatory cytokine contribute to the initiation
and development of ulcerative colitis and its associated cancer.
Inflamm Bowel Dis. 25:1595–1602. 2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Guan Q and Zhang J: Recent advances: The
imbalance of cytokines in the pathogenesis of inflammatory bowel
disease. Mediators Inflamm. 2017(4810258)2017.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Zhou X, Li W, Wang S, Zhang P, Wang Q,
Xiao J, Zhang C, Zheng X, Xu X, Xue S, et al: YAP aggravates
inflammatory bowel disease by regulating M1/M2 macrophage
polarization and gut microbial homeostasis. Cell Rep.
27:1176–1189.e5. 2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Aass HCD, Hellum M, Trøseid AS, Brandtzaeg
P, Berg JP, Øvstebø R and Henriksson CE: Whole-blood incubation
with the Neisseria meningitidis lpxL1 mutant induces less
pro-inflammatory cytokines than the wild type, and IL-10 reduces
the MyD88-dependent cytokines. Innate Immun. 24:101–111.
2018.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Liu L, Liang L, Liang H, Wang M, Lu B, Xue
M, Deng J and Chen Y: Fusobacterium nucleatum aggravates the
progression of colitis by regulating M1 macrophage polarization via
AKT2 pathway. Front Immunol. 10(1324)2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Cui H, Cai Y, Wang L, Jia B, Li J, Zhao S,
Chu X, Lin J, Zhang X, Bian Y and Zhuang P: Berberine regulates
Treg/Th17 balance to treat ulcerative colitis through modulating
the gut microbiota in the colon. Front Pharmacol.
9(571)2018.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Ilott NE, Bollrath J, Danne C, Schiering
C, Shale M, Adelmann K, Krausgruber T, Heger A, Sims D and Powrie
F: Defining the microbial transcriptional response to colitis
through integrated host and microbiome profiling. ISME J.
10:2389–2404. 2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Forbes JD, Van Domselaar G and Bernstein
CN: The gut microbiota in immune-mediated inflammatory diseases.
Front Microbiol. 7(1081)2016.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Gao X, Cao Q, Cheng Y, Zhao D, Wang Z,
Yang H, Wu Q, You L, Wang Y, Lin Y, et al: Chronic stress promotes
colitis by disturbing the gut microbiota and triggering immune
system response. Proc Natl Acad Sci USA. 115:E2960–E2969.
2018.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Zhang M, Sun K, Wu Y, Yang Y, Tso P and Wu
Z: Interactions between intestinal microbiota and host immune
response in inflammatory bowel disease. Front Immunol.
8(942)2017.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Ishikawa D, Sasaki T, Osada T,
Kuwahara-Arai K, Haga K, Shibuya T, Hiramatsu K and Watanabe S:
Changes in intestinal microbiota following combination therapy with
fecal microbial transplantation and antibiotics for ulcerative
colitis. Inflamm Bowel Dis. 23:116–125. 2017.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Ishikawa D, Sasaki T, Takahashi M,
Kuwahara-Arai K, Haga K, Ito S, Okahara K, Nakajima A, Shibuya T,
Osada T, et al: The microbial composition of bacteroidetes species
in ulcerative colitis is effectively improved by combination
therapy with fecal microbiota transplantation and antibiotics.
Inflamm Bowel Dis. 24:2590–2598. 2018.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Zhu W, Winter MG, Byndloss MX, Spiga L,
Duerkop BA, Hughes ER, Büttner L, de Lima Romão E, Behrendt CL,
Lopez CA, et al: Precision editing of the gut microbiota
ameliorates colitis. Nature. 553:208–211. 2018.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Rathinam VAK and Chan FK: Inflammasome,
inflammation, and tissue homeostasis. Trends Mol Med. 24:304–318.
2018.PubMed/NCBI View Article : Google Scholar
|