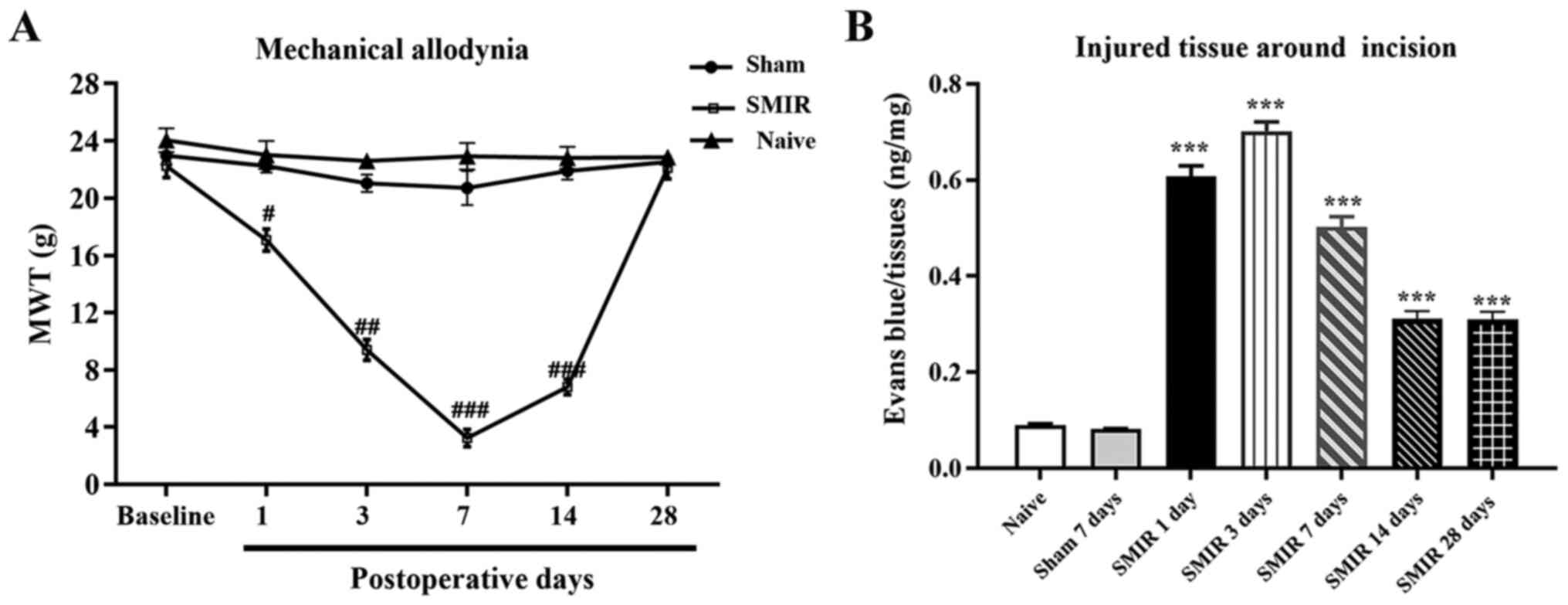
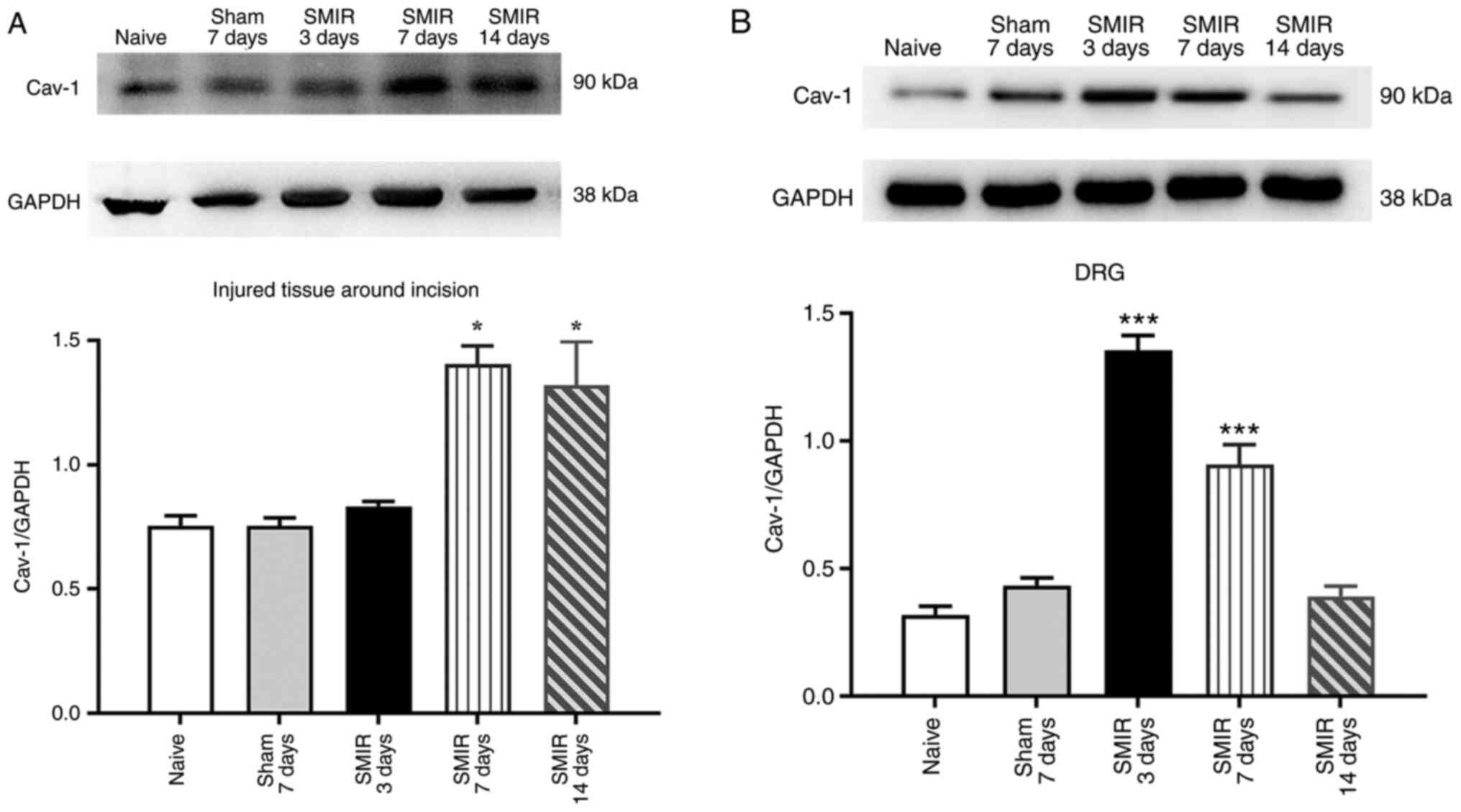
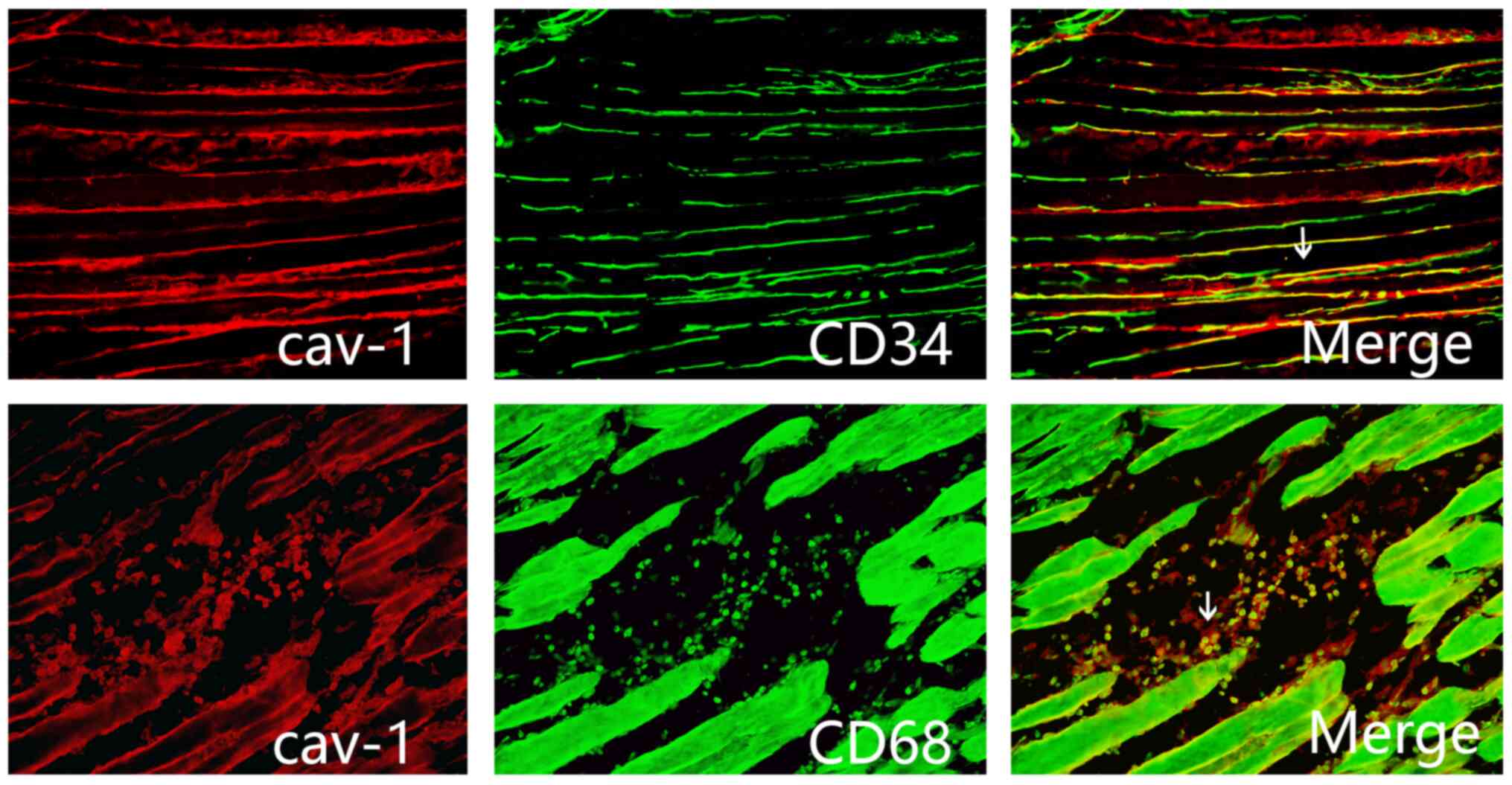
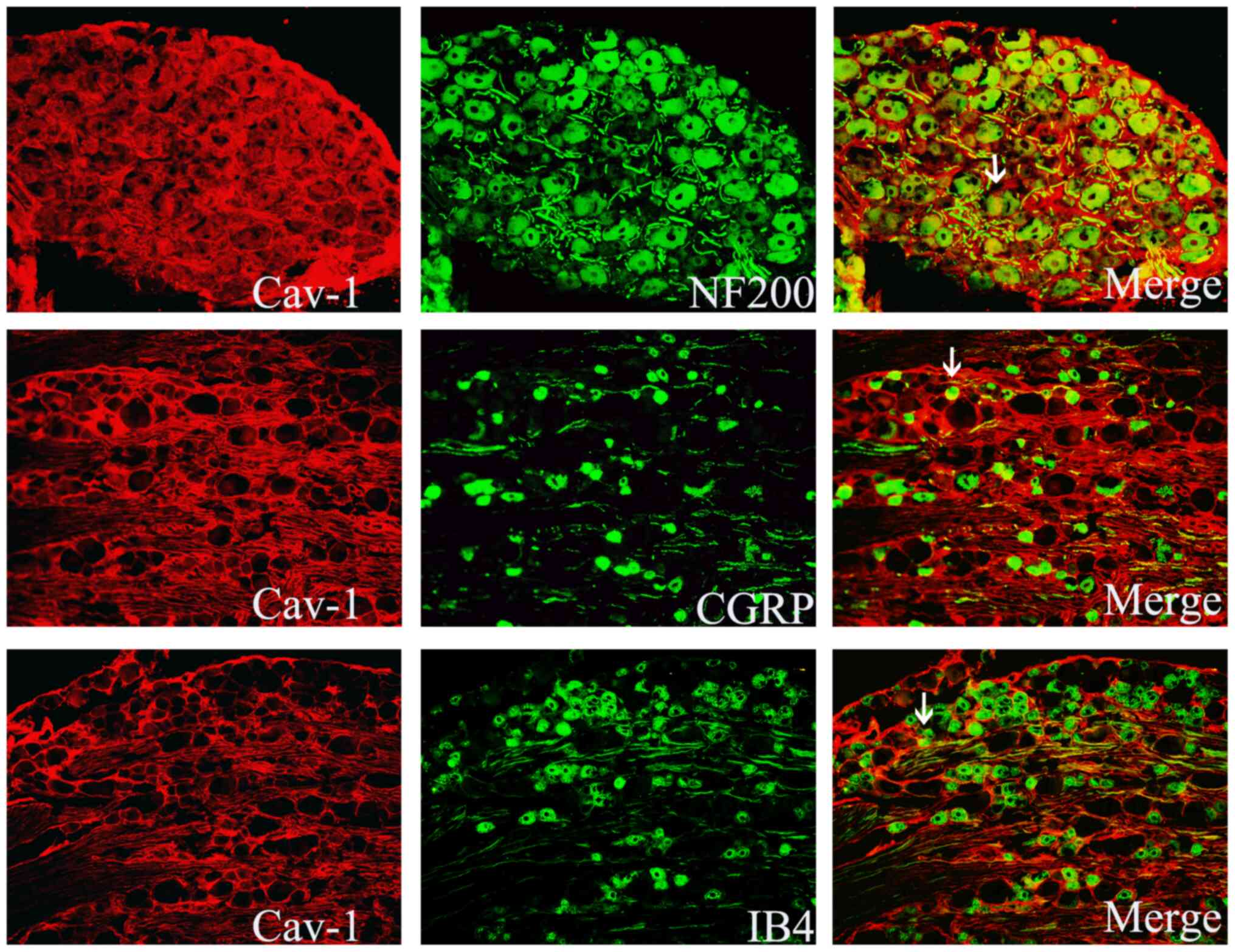
|
1
|
Kim DH, Pearson-Chauhan KM, McCarthy RJ
and Buvanendran A: Predictive factors for developing chronic pain
after total knee arthroplasty. J Arthroplasty. 33:3372–3378.
2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Maguire J, Thibodeau ML and Oliver J: CPSP
2014 results: What have we learned? Paediatr Child Health.
20:435–436. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Uemoto Y, Uchida M, Kondo N,
Wanifuchi-Endo Y, Fujita T, Asano T, Hisada T, Nishikawa S,
Katagiri Y, Terada M, et al: Predictive factors for patients who
need treatment for chronic post-surgical pain (CPSP) after breast
cancer surgery. Breast Cancer 22: doi: 10.1007, 2021.
|
|
4
|
Blichfeldt-Eckhardt MR: From acute to
chronic postsurgical pain: The significance of the acute pain
response. Dan Med J. 65(B5326)2018.PubMed/NCBI
|
|
5
|
Fregoso G, Wang A, Tseng K and Wang J:
Transition from acute to chronic pain: Evaluating risk for chronic
postsurgical pain. Pain Physician. 22:479–488. 2019.PubMed/NCBI
|
|
6
|
Pan P, Huang SS, Shen SR, Lu CE, Qin YB,
Zhang JL and Cao S: Role of p120 catenin in Epac1-induced chronic
postsurgical pain in rats. Pain Res Manag.
2019(9017931)2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Parton RG, McMahon KA and Wu Y: Caveolae:
Formation, dynamics, and function. Curr Opin Cell Biol. 65:8–16.
2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tiruppathi C, Regmi SC, Wang DM, Mo GCH,
Toth PT, Vogel SM, Stan RV, Henkemeyer M, Minshall RD, Rehman J and
Malik AB: EphB1 interaction with caveolin-1 in endothelial cells
modulates caveolae biogenesis. Mol Biol Cell. 31:1167–1182.
2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Huang Q, Zhong W, Hu Z and Tang X: A
review of the role of cav-1 in neuropathology and neural recovery
after ischemic stroke. J Neuroinflammation. 15(348)2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
de Souza GM, de Albuquerque Borborema ME,
de Lucena TMC, da Silva Santos AF, de Lima BR, de Oliveira DC and
de Azevêdo Silva J: Caveolin-1 (CAV-1) up regulation in metabolic
syndrome: All roads leading to the same end. Mol Biol Rep.
47:9245–9250. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Patel HH, Murray F and Insel PA: Caveolae
as organizers of pharmacologically relevant signal transduction
molecules. Annu Rev Pharmacol Toxicol. 48:359–391. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Codrici E, Albulescu L, Popescu ID, Mihai
S, Enciu AM, Albulescu R, Tanase C and Hinescu ME:
Caveolin-1-knockout mouse as a model of inflammatory diseases. J
Immunol Res. 29(2498576)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhang Y, Luo H, Lv X, Liu J, Chen X, Li Y,
Liu A and Jiang Y: Axin-1 binds to caveolin-1 to regulate the
LPS-induced inflammatory response in AT-I cells. Biochem Biophys
Res Commun. 513:261–268. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yang JX, Hua L, Li YQ, Jiang YY, Han D,
Liu H, Tang QQ, Yang XN, Yin C, Hao LY, et al: Caveolin-1 in the
anterior cingulate cortex modulates chronic neuropathic pain via
regulation of NMDA receptor 2B subunit. J Neurosci. 35:36–52.
2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chen JL, Lu JH, Xie CS, Shen YJ, Wang JW,
Ye XY, Zhang MB, Jia GL, Tao YX, Li J and Cao H: Caveolin-1 in
spinal cord modulates type-2 diabetic neuropathic pain through the
Rac1/NOX2/NR2B signaling pathway. Am J Transl Res. 12:1714–1727.
2020.PubMed/NCBI
|
|
16
|
Jia GL, Huang Q, Cao YN, Xie CS, Shen YJ,
Chen JL, Lu JH, Zhang MB, Li J, Tao YX and Cao H: Cav-1
participates in the development of diabetic neuropathy pain through
the TLR4 signaling pathway. J Cell Physiol. 235:2060–2070.
2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wang Y, Gao Y, Tian Q, Deng Q, Wang Y,
Zhou T, Liu Q, Mei K, Wang Y, Liu H, et al: TRPV1 SUMOylation
regulates nociceptive signaling in models of inflammatory pain. Nat
Commun. 9(1529)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bang S, Xie YK, Zhang ZJ, Wang Z, Xu ZZ
and Ji RR: GPR37 regulates macrophage phagocytosis and resolution
of inflammatory pain. J Clin Invest. 128:3568–3582. 2018.PubMed/NCBI View
Article : Google Scholar
|
|
19
|
Xu Q, Du F, Zhang Y, Teng Y, Tao M, Chen
AF and Jiang R: Preeclampsia serum induces human glomerular
vascular endothelial cell hyperpermeability via the
HMGB1-Caveolin-1 pathway. J Reprod Immunol. 129:1–8.
2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhang X, Ramírez CM, Aryal B,
Madrigal-Matute J, Liu X, Diaz A, Torrecilla-Parra M, Suárez Y,
Cuervo AM, Sessa WC and Fernández-Hernando C: Cav-1 (Caveolin-1)
deficiency increases autophagy in the endothelium and attenuates
vascular inflammation and atherosclerosis. Arterioscler Thromb Vasc
Biol. 40:1510–1522. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Chung JW, Kim DH, Oh MJ, Cho YH, Kim EH,
Moon GJ, Ki CS, Cha J, Kim KH, Jeon P, et al: Cav-1 (Caveolin-1)
and arterial remodeling in adult moyamoya disease. Stroke.
49:2597–2604. 2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Flatters SJ: Characterization of a model
of persistent postoperative pain evoked by skin/muscle incision and
retraction (SMIR). Pain. 135:119–130. 2008.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zahn PK, Pogatzki EM and Brennan TJ:
Mechanisms for pain caused by incisions. Reg Anesth Pain Med.
27:514–516. 2002.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mirra A, Spadavecchia C, Bruckmaier R,
Gutzwiller A and Casoni D: Acute pain and peripheral sensitization
following cautery disbudding in 1- and 4-week-old calves. Physiol
Behav. 84:248–260. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Zhang Y and Wang Y: TRPV1: An important
molecule involved in the peripheral sensitization during chronic
pain and central pain modulation. Sheng Li Xue Bao. 25:677–684.
2017.PubMed/NCBI(In Chinese).
|
|
27
|
Filippini A and D'Alessio A: Caveolae and
lipid rafts in endothelium: Valuable organelles for multiple
functions. Biomolecules. 10(1218)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Chow BW, Nuñez V, Kaplan L, Granger AJ,
Bistrong K, Zucker HL, Kumar P, Sabatini BL and Gu C: Caveolae in
CNS arterioles mediate neurovascular coupling. Nature. 579:106–110.
2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Jin H, Xu Y, Shi F and Hu S: Vaccination
at different anatomic sites induces different levels of the immune
responses. Res Vet Sci. 122:50–55. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Parton RG, Tillu VA and Collins BM:
Caveolae. Curr Biol. 23:R402–R405. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Oliveira SDS, Chen J, Castellon M, Mao M,
Raj JU, Comhair S, Erzurum S, Silva CLM, Machado RF, Bonini MG and
Minshall RD: Injury-induced shedding of extracellular vesicles
depletes endothelial cells of Cav-1 (Caveolin-1) and enables TGF-β
(Transforming Growth Factor-β)-dependent pulmonary arterial
hypertension. Arterioscler Thromb Vasc Biol. 39:1191–1202.
2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Luo M, Xu C, Luo Y, Wang G, Wu J and Wan
Q: Circulating miR-103 family as potential biomarkers for type 2
diabetes through targeting CAV-1 and SFRP4. Acta Diabetol.
57:309–322. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wang DX, Pan YQ, Liu B and Dai L: Cav-1
promotes atherosclerosis by activating JNK-associated signaling.
Biochem Biophys Res Commun. 503:513–520. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Konrad FM, Meichssner N, Bury A, Ngamsri
KC and Reutershan J: Inhibition of SDF-1 receptors CXCR4 and CXCR7
attenuates acute pulmonary inflammation via the adenosine
A2B-receptor on blood cells. Cell Death Dis.
8(e2832)2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wang N, Zhang D, Sun G, Zhang H, You Q,
Shao M and Yue Y: Lipopolysaccharide-induced caveolin-1
phosphorylation-dependent increase in transcellular permeability
precedes the increase in paracellular permeability. Drug Des Devel
Ther. 9:4965–4977. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Andreone BJ, Chow BW, Tata A, Lacoste B,
Ben-Zvi A, Bullock K, Deik AA, Ginty DD, Clish CB and Gu C:
Blood-brain barrier permeability is regulated by lipid
transport-dependent suppression of caveolae-mediated transcytosis.
Neuron. 94:581–594. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Bravo-Caparrós I, Ruiz-Cantero MC,
Perazzoli G, Cronin SJF, Vela JM, Hamed MF, Penninger JM, Baeyens
JM, Cobos EJ and Nieto FR: Sigma-1 receptors control neuropathic
pain and macrophage infiltration into the dorsal root ganglion
after peripheral nerve injury. FASEB J. 34:5951–5966.
2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Mekhail N, Deer TR, Poree L, Staats PS,
Burton AW, Connolly AT, Karst E, Mehanny DS, Saweris Y and Levy RM:
Cost-effectiveness of dorsal root ganglion stimulation or spinal
cord stimulation for complex regional pain syndrome.
Neuromodulation. 9:708–714. 2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
He DD, Gao Y, Wang S, Xie Z and Song XJ:
Systematic administration of B vitamins alleviates diabetic pain
and inhibits associated expression of P2X3 and TRPV1 in dorsal root
ganglion neurons and proinflammatory cytokines in spinal cord in
rats. Pain Res Manag. 10(3740162)2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Li QY, Xu HY and Yang HJ: Effect of
proinflammatory factors TNF-α,IL-1β, IL-6 on neuropathic pain.
Zhongguo Zhong Yao Za Zhi. 42:3709–3712. 2017.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
41
|
Ding HH, Zhang SB, Lv YY, Ma C, Liu M,
Zhang KB, Ruan XC, Wei JY, Xin WJ and Wu SL: TNF-α/STAT3 pathway
epigenetically upregulates Nav1.6 expression in DRG and contributes
to neuropathic pain induced by L5-VRT. J Neuroinflammation.
16(29)2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Zhang ZJ, Jiang BC and Gao YJ: Chemokines
in neuron-glial cell interaction and pathogenesis of neuropathic
pain. Cell Mol Life Sci. 74:3275–3291. 2017.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Li CD, Zhao JY, Chen JL, Lu JH, Zhang MB,
Huang Q, Cao YN, Jia GL, Tao YX, Li J and Cao H: Mechanism of the
JAK2/STAT3-CAV-1-NR2B signaling pathway in painful diabetic
neuropathy. Endocrine. 64:55–66. 2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kassan A, Egawa J, Zhang Z,
Almenar-Queralt A, Nguyen QM, Lajevardi Y, Kim K, Posadas E, Jeste
DV, Roth DM, et al: Caveolin-1 regulation of
disrupted-in-schizophrenia-1 as a potential therapeutic target for
schizophrenia. J Neurophysiol. 117:436–444. 2017.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Sheikh NK and Dua A: Neuroanatomy,
substantia gelatinosa. In: StatPearls. StatPearls Publishing,
Treasure Island, FL, 2020.
|
|
46
|
Roh J, Hwang SM, Lee SH, Lee K, Kim YH and
Park CK: Functional expression of piezo1 in dorsal root ganglion
(DRG) neurons. Int J Mol Sci. 21(3834)2020.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Zhang J, Harada Y and Hayashi Y: A
TLR-CXCL1 pathway in DRG neurons induces neutrophil accumulation in
the DRG and mechanical allodynia in EAE mice. Sci Rep.
9(12003)2019.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Huang H, Wang M and Hong Y: Intrathecal
administration of adrenomedullin induces mechanical allodynia and
neurochemical changes in spinal cord and DRG. Neurosci Lett.
690:196–201. 2019.PubMed/NCBI View Article : Google Scholar
|