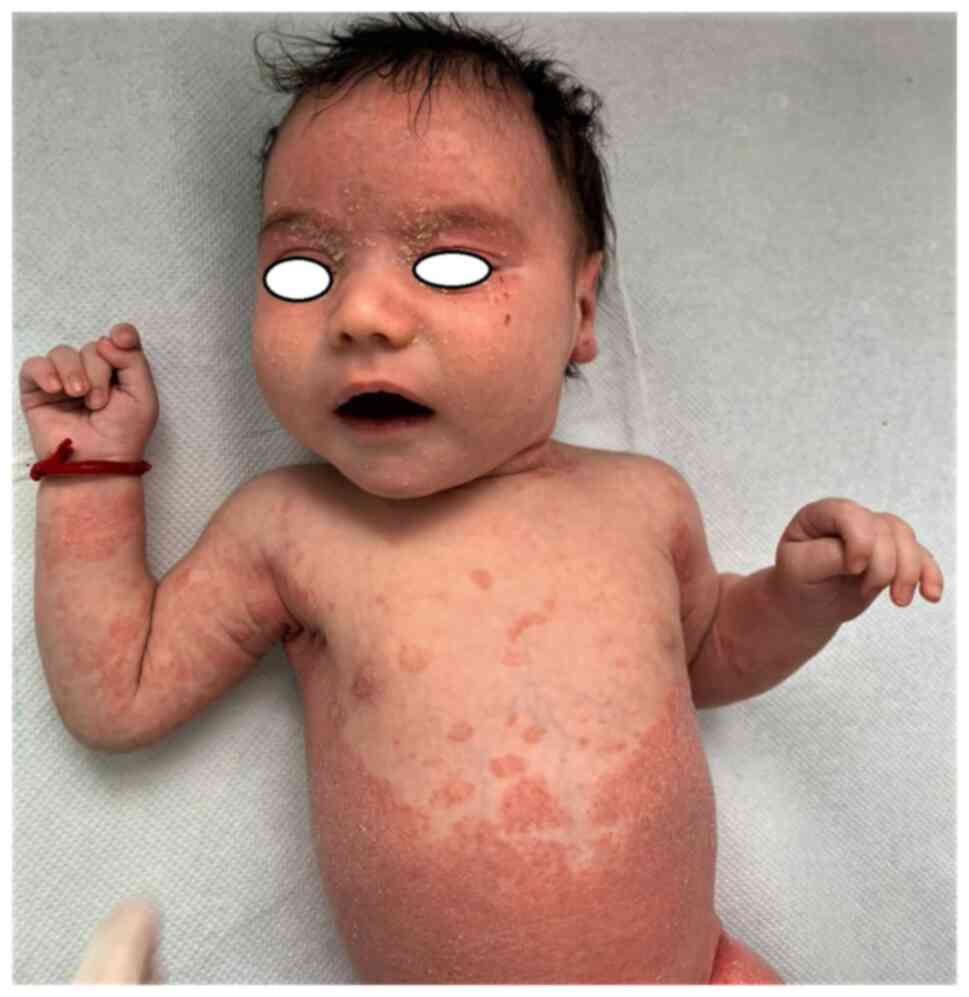
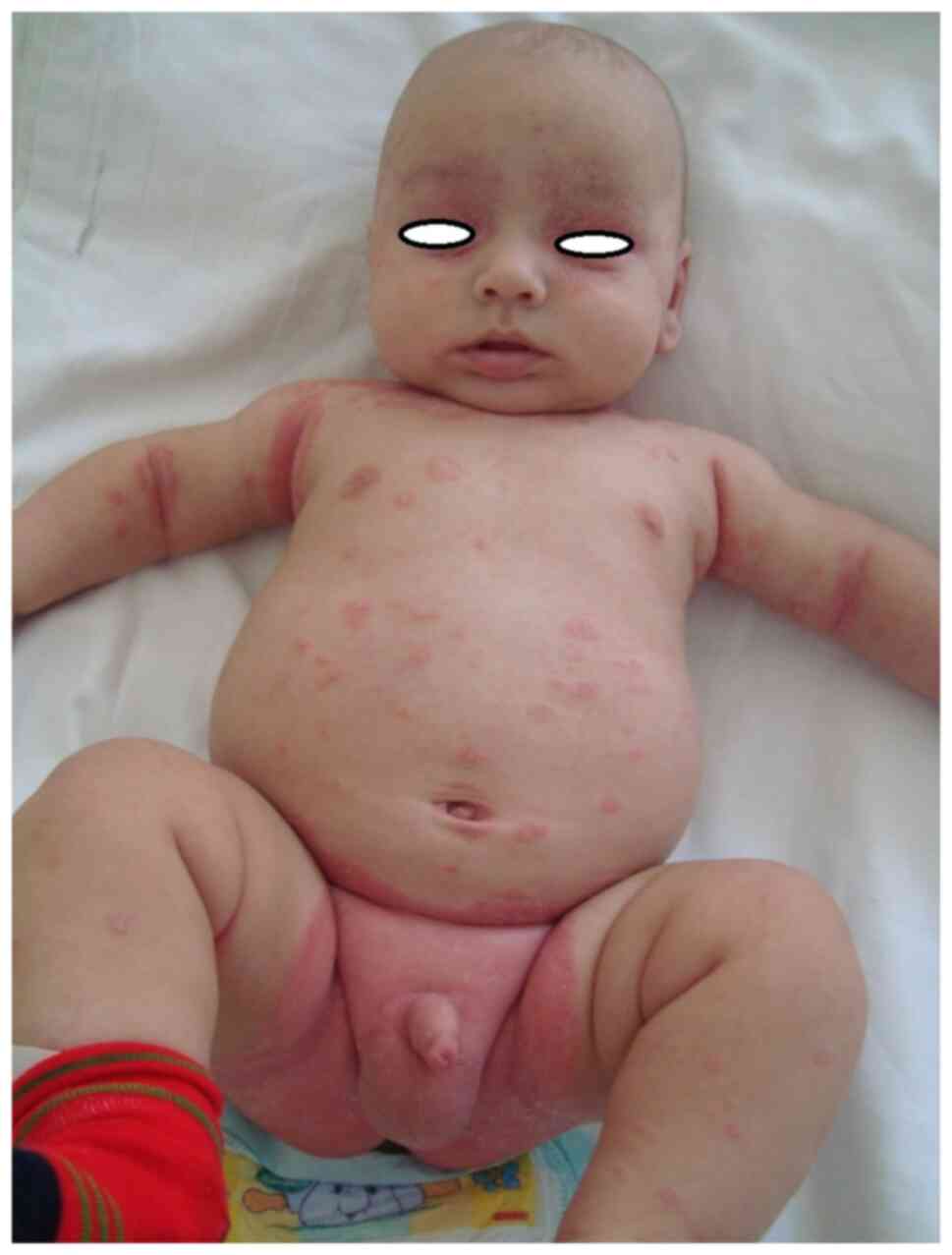
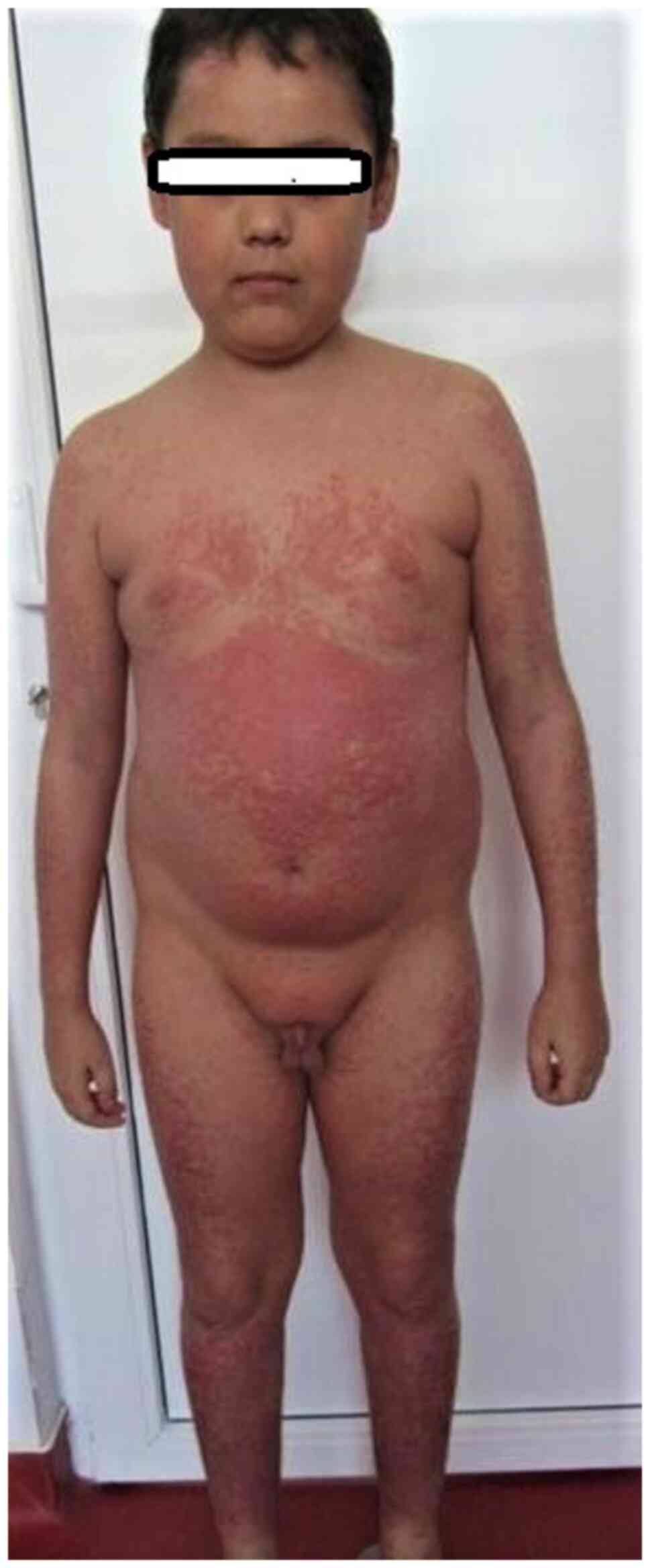
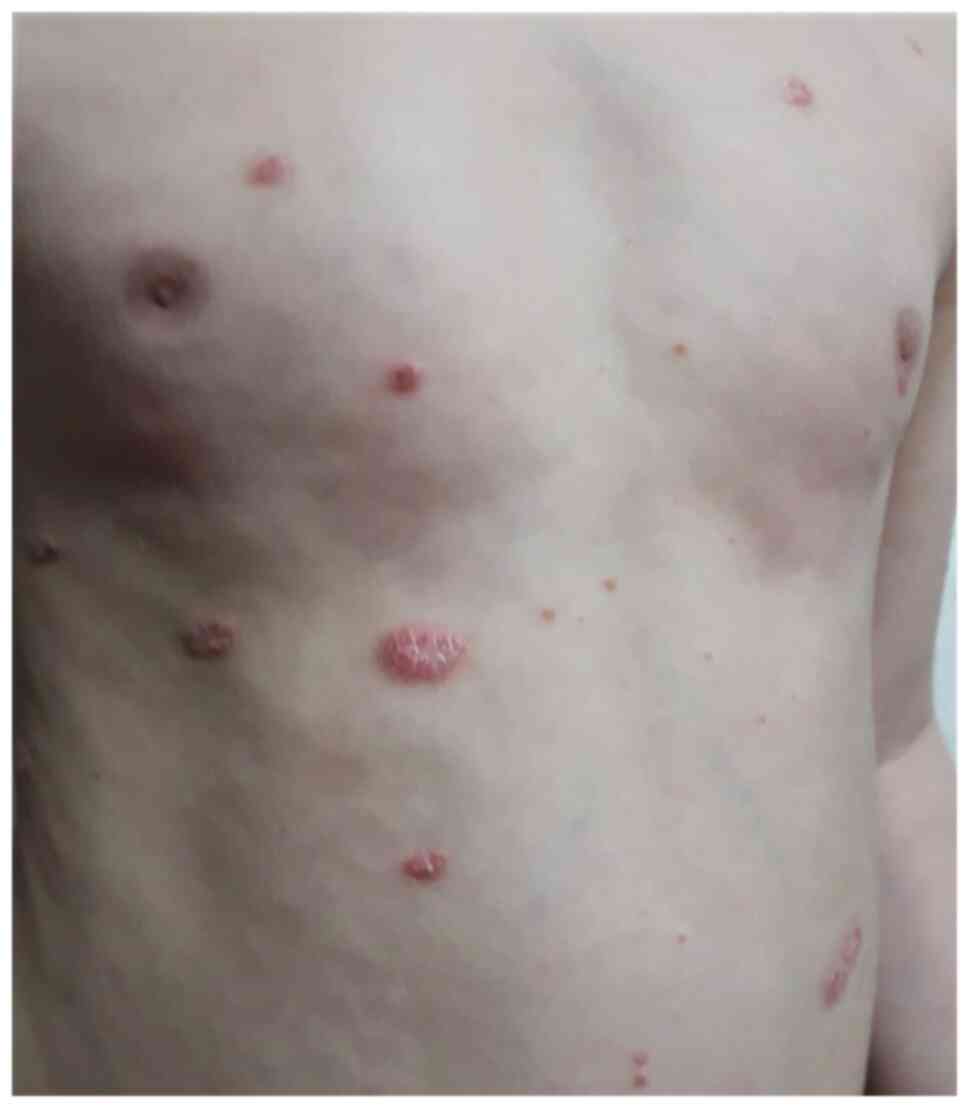
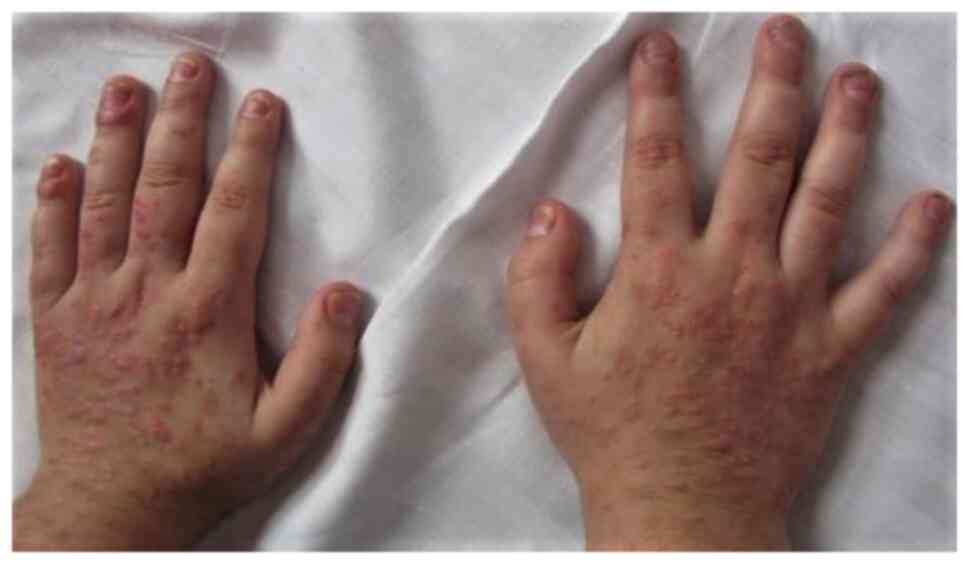
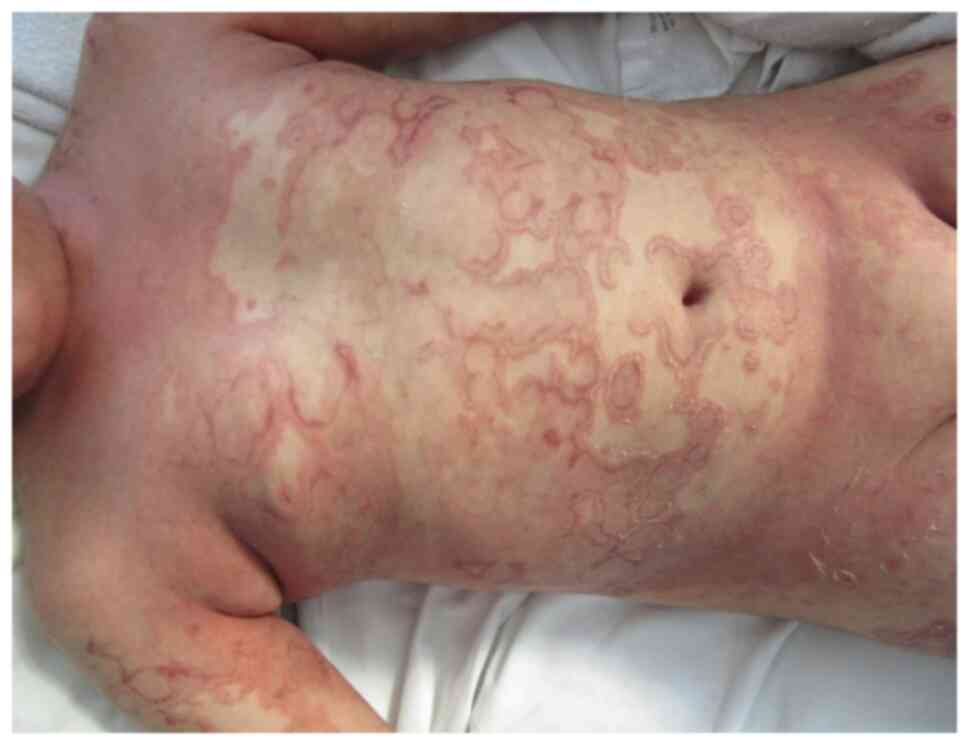
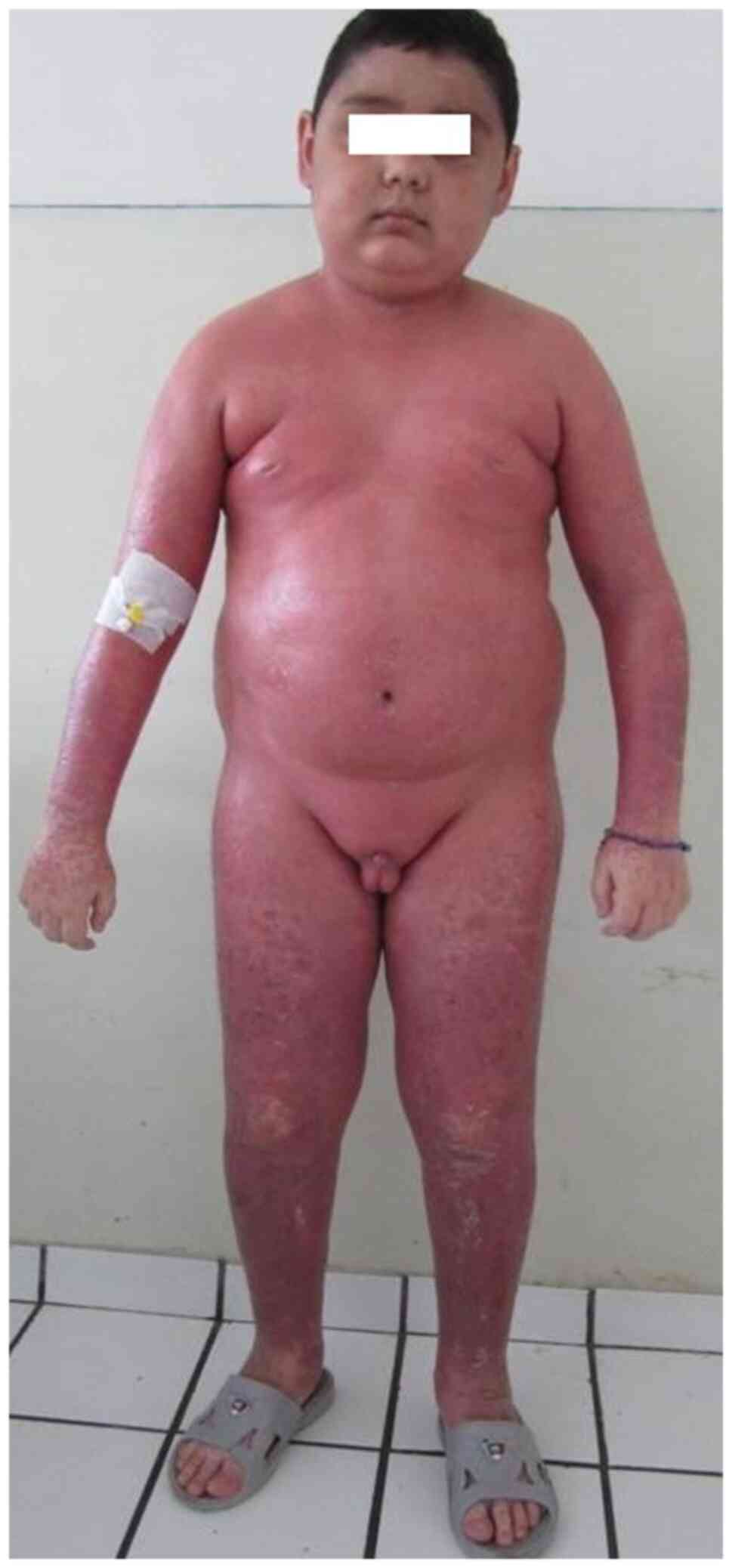
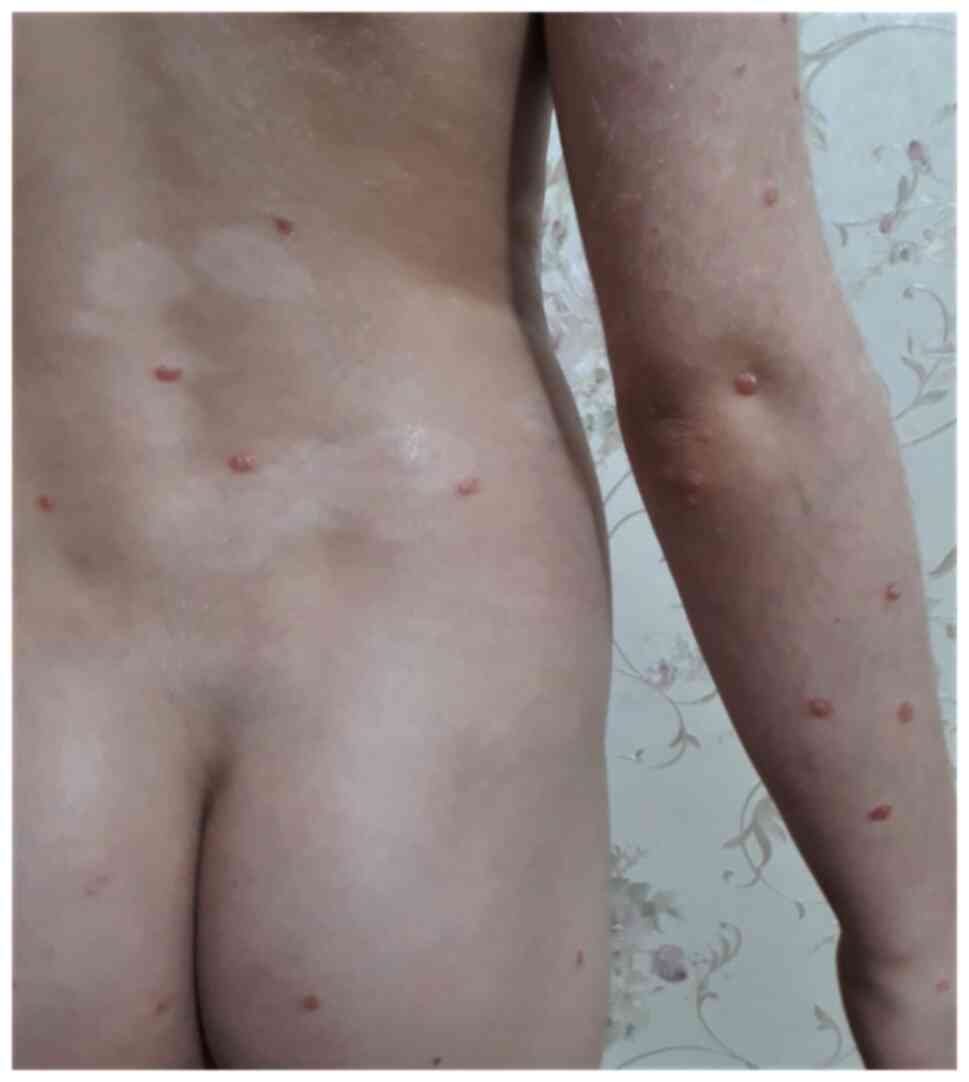
|
1
|
Patterson JW: The psoriasiform reaction
pattern. In: Weedon's Skin Pathology. 5th edition. Elsevier,
Philadelphia, PA, pp99-120.e11, 2021.
|
|
2
|
Tollefson MM: Diagnosis and management of
Psoriasis in children. Pediatr Clin North Am. 61:261–277.
2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kerkhof PCM and van de Nestlé FO:
Psoriasis. In: Dermatology. 4th edtion. Philadelphia, PA,
pp138-160, 2018.
|
|
4
|
Menter A, Cordoro KM, Davis DMR,
Kroshinsky D, Paller AS, Armstrong AW, Connor C, Elewski BE,
Gelfand JM, Gordon KB, et al: Joint American Academy of
Dermatology-National Psoriasis Foundation guidelines of care for
the management and treatment of psoriasis in pediatric patients. J
Am Acad Dermatol. 82:161–201. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Tkach VY, Voloshynovych MS, Girnyk GY and
Kozak NV: Clinical features and the course of psoriasis in
children. Przegl Dermatol. 107:476–480. 2020.
|
|
6
|
Tollefson MM, Crowson CS, McEvoy MT and
Maradit Kremers H: Incidence of psoriasis in children: A
population-based study. J Am Acad Dermatol. 62:979–987.
2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Murzina E: Pediatric Psoriasis: Clinical
Features and Course. OAJBS. 1(OAJBS.ID.000147)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Gelfand JM, Weinstein R, Porter SB,
Neimann AL, Berlin JA and Margolis DJ: Prevalence and treatment of
psoriasis in the United Kingdom: A population-based study. Arch
Dermatol. 141:1537–1541. 2005.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Pinson R, Sotoodian B and Fiorillo L:
Psoriasis in children. Psoriasis (Auckl). 6:121–129.
2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Parisi R, Symmons DP, Griffiths CE and
Ashcroft DM: Identification and Management of Psoriasis and
Associated ComorbidiTy (IMPACT) project team. Global epidemiology
of psoriasis: A systematic review of incidence and prevalence. J
Invest Dermatol. 133:377–385. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Paller AS, Singh R, Cloutier M,
Gauthier-Loiselle M, Emond B, Guérin A and Ganguli A: Prevalence of
psoriasis in children and adolescents in the United States: A
claims-based analysis. J Drugs Dermatol. 17:187–194.
2018.PubMed/NCBI
|
|
12
|
Seyhan M, Coşkun BK, Sağlam H, Ozcan H and
Karincaoğlu Y: Psoriasis in childhood and adolescence: Evaluation
of demographic and clinical features. Pediatr Int. 48:525–530.
2006.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kwon HH, Na SJ, Jo SJ and Youn JI:
Epidemiology and clinical features of pediatric psoriasis in
tertiary referral psoriasis clinic. J Dermatol. 39:260–264.
2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ran D, Cai M and Zhang X: Genetics of
psoriasis: A basis for precision medicine. Precis Clin Med.
2:120–130. 2019.
|
|
15
|
Paller AS, Siegfried EC, Pariser DM, Rice
KC, Trivedi M, Iles J, Collier DH, Kricorian G and Langley RG:
Long-term safety and efficacy of etanercept in children and
adolescents with plaque psoriasis. J Am Acad Dermatol.
74:280–287.e1-e3. 2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kliegman RM, Geme JWS, Blum NJ, Shah SS,
Tasker RC and Wilson KM: Diseases of the epidermis. In: Nelson
Textbook of Pediatrics. Kliegman RM and Geme JWS (eds). Elsevier,
Philadelphia, PA, pp3501-3510.e1, 2020.
|
|
17
|
Rendon A and Schäkel K: Psoriasis
pathogenesis and treatment. Int J Mol Sci. 20(1475)2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Batani A, Brănișteanu DE, Ilie MA, Boda D,
Ianosi S, Ianosi G and Caruntu C: Assessment of dermal papillary
and microvascular parameters in psoriasis vulgaris using in vivo
reflectance confocal microscopy. Exp Ther Med. 15:1241–1246.
2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Căruntu C, Boda D, Căruntu A, Rotaru M,
Baderca F and Zurac S: In vivo imaging techniques for psoriatic
lesions. Rom J Morphol Embryol. 55 (Suppl 3):S1191–S1196.
2014.PubMed/NCBI
|
|
20
|
Das RP, Jain AK and Ramesh V: Current
concepts in the pathogenesis of psoriasis. Indian J Dermatol.
54:7–12. 2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Qing M, Liu P, Zhu W, Chen M, Chen M and
Kuang Y: Analysis for 208 children with psoriasis vulgaris. Zhong
Nan Da Xue Xue Bao Yi Xue Ban. 45:804–811. 2020.PubMed/NCBI View Article : Google Scholar : (In Chinese,
English).
|
|
22
|
Nguyen TT: Papulosquamous eruptions. In:
Conn's Current Therapy 2021. Kellerman RD and Rakel D (eds).
Elsevier, Philadelphia, PA, pp1056-1061, 2021.
|
|
23
|
Kim SK, Kang HY, Kim YC and Lee ES:
Clinical comparison of psoriasis in Korean adults and children:
Correlation with serum anti-streptolysin O titers. Arch Dermatol
Res. 302:295–299. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Say M, Boralévi F, Lenormand C, Bursztejn
AC, Estève E, Phan A, Bourrat E, Lacour JP, Richard MA, Acher A, et
al: Clinical and therapeutic aspects of linear psoriasis: A study
of 30 cases. Am J Clin Dermatol. 19:609–615. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Thomas L, Azad J and Takwale A: Management
of nail psoriasis. Clin Exp Dermatol. 46:3–8. 2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Uber M, Carvalho VO, Abagge KT, Robl Imoto
R and Werner B: Clinical features and nail clippings in 52 children
with psoriasis. Pediatr Dermatol. 35:202–207. 2018.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Narla S and Silverberg JI: 15838
Autoimmune comorbidities of psoriasis in US adults and children. J
Am Acad Dermatol. 83(AB48)2020.
|
|
28
|
Brandon TG, Manos CK, Xiao R, Ogdie A and
Weiss PF: Pediatric psoriatic arthritis: A population-based cohort
study of risk factors for onset and subsequent risk of inflammatory
comorbidities. J Psoriasis Psoriatic Arthritis. 3:131–136.
2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Thomas J and Parimalam K: Treating
pediatric plaque psoriasis: Challenges and solutions. Pediatric
Health Med Ther. 7:25–38. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Caruntu C, Boda D, Dumitrascu G,
Constantin C and Neagu M: Proteomics focusing on immune markers in
psoriatic arthritis. Biomark Med. 9:513–528. 2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Brihan I, Hălmăjan A, Boda D, Ianoși SL,
Fekete GL and Zdrîncă M: Role of osteodensitometry in osteoporosis
and osteopenia in psoriatic arthritis. Exp Ther Med.
20(188)2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Taranu T, Constantin M, Ungureanu D, Esanu
IM and Toader MP: IL6 is correlated with metabolic syndrome
parameters in oral lichen planus. In: 2015 E-Health and
Bioengineering Conference (EHB). pp1-4, 2015. http://toc.proceedings.com/29132webtoc.pdf.
|
|
33
|
Brihan I, Ianoși SL, Boda D, Hălmăjan A,
Zdrîncă M and Fekete LG: Implications of self-esteem in the quality
of life in patients with psoriasis. Exp Ther Med.
20(202)2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Gånemo A, Wahlgren CF and Svensson Å:
Quality of life and clinical features in Swedish children with
psoriasis. Pediatr Dermatol. 28:375–379. 2011.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Branisteanu DC, Stoleriu G, Branisteanu
DE, Boda D, Branisteanu CI, Maranduca MA, Moraru A, Stanca HT,
Zemba M and Balta F: Ocular cicatricial pemphigoid (Review). Exp
Ther Med. 20:3379–3382. 2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Karabulut AA, Yalvac IS, Vahaboglu H,
Nurozler AB and Duman S: Conjunctival impression cytology and
tear-film changes in patients with psoriasis. Cornea. 18:544–548.
1999.PubMed/NCBI
|
|
37
|
Kilic B, Dogan U, Parlak AH, Goksugur N,
Polat M, Serin D and Ozmen S: Ocular findings in patients with
psoriasis. Int J Dermatol. 52:554–559. 2013.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Cruz NFSD, Brandão LS, Cruz SFSD, Cruz
SASD, Pires CAA and Carneiro FRO: Ocular manifestations of
psoriasis. Arq Bras Oftalmol. 81:219–225. 2018.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Erbagci I, Erbagci Z, Gungor K and Bekir
N: Ocular anterior segment pathologies and tear film changes in
patients with psoriasis vulgaris. Acta Med Okayama. 57:299–303.
2003.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Salek SS, Pradeep A, Guly C, Ramanan AV
and Rosenbaum JT: Uveitis and juvenile psoriatic arthritis or
psoriasis. Am J Ophthalmol. 185:68–74. 2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Mahé E: Childhood psoriasis. Eur J
Dermatol. 26:537–548. 2016.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Coondoo A, Phiske M, Verma S and Lahiri K:
Side-effects of topical steroids: A long overdue revisit. Indian
Dermatol Online J. 5:416–425. 2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Ogawa E, Sato Y, Minagawa A and Okuyama R:
Pathogenesis of psoriasis and development of treatment. J Dermatol.
45:264–272. 2018.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Negrei C, Ginghina O, Caruntu C,
Burcea-Dragomiroiu GTA, Jinescu G and Boda D: Investigation
relevance of methotrexate polyglutamates in biological systems by
high performance liquid chromatography. REV CHIM (Bucharest).
66:766–768. 2015.
|
|
45
|
Negrei C, Caruntu C, Ginghina O,
Burcea-Dragomiroiu GTA, Toderescu CD and Boda D: Qualitative and
quantitative determination of methotrexate polyglutamates in
erythrocytes by high performance liquid chromatography. REV CHIM
(Bucharest). 66:607–610. 2015.
|
|
46
|
Boda D, Negrei C, Nicolescu F and Balalau
C: Assessment of some oxidative stress parameters in methotrexate
treated psoriasis patients. Farmacia. 62:704–710. 2014.
|
|
47
|
Nguyen TU and Koo J: Etanercept in the
treatment of plaque psoriasis. Clin Cosmet Investig Dermatol.
2:77–84. 2009.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Branisteanu DE, Voicu CM, Cretu A,
Dimitriu A, Luca MC and Salavastru CM: Adverse reactions of
biological therapy for psoriasis. Rev Med Chir Soc Med Nat Iasi.
119:38–44. 2015.PubMed/NCBI
|
|
49
|
Papp K, Thaçi D, Marcoux D, Weibel L,
Philipp S, Ghislain PD, Landells I, Hoeger P, Kotkin C, Unnebrink
K, et al: Efficacy and safety of adalimumab every other week versus
methotrexate once weekly in children and adolescents with severe
chronic plaque psoriasis: A randomised, double-blind, phase 3
trial. Lancet. 390:40–49. 2017.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Khobzey K, Liskova I, Szegedi A, Pavlovsky
L, Lunder T, Kingo K, Miljković J, Péč J, Bohinc M and Hojnik M:
Effectiveness of adalimumab in the treatment of scalp and nail
affection in patients with moderate to severe plaque psoriasis in
routine clinical practice. Acta Dermatovenerol Alp Pannonica
Adriat. 26:11–14. 2017.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Mocanu M, Toader MP, Rezus E and Taranu T:
Aspects concerning patient adherence to anti-TNFα therapy in
psoriasis: A decade of clinical experience. Exp Ther Med.
18:4987–4992. 2019.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Savage LJ, Wittmann M, McGonagle D and
Helliwell PS: Ustekinumab in the treatment of psoriasis and
psoriatic arthritis. Rheumatol Ther. 2:1–16. 2015.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Cline A, Bartos GJ, Strowd LC and Feldman
SR: Biologic treatment options for pediatric psoriasis and atopic
dermatitis. Children (Basel). 6(103)2019.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Broge T, Nguyen N, Sacks A and Davis M:
Infliximab-associated psoriasis in children with Crohn's disease
may require withdrawal of anti-tumor necrosis factor therapy.
Inflamm Bowel Dis. 19:E75–E77. 2013.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Menter MA and Cush JM: Successful
treatment of pediatric psoriasis with infliximab. Pediatr Dermatol.
21:87–88. 2004.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Skrabl-Baumgartner A, Weger W, Salmhofer W
and Jahnel J: Childhood generalized pustular psoriasis: Longtime
remission with combined infliximab and methotrexate treatment.
Pediatr Dermatol. 32:e13–e14. 2015.PubMed/NCBI View Article : Google Scholar
|
|
57
|
UCB Celltech: (2018). The Use of
Certolizumab Pegol for Treatment of Active Crohn's Disease in
Children and Adolescents (NURTURE). ClinicalTrials.gov Identifier: NCT00899678.
https://clinicaltrials.gov/ct2/show/NCT00899678.
|
|
58
|
UCB BIOSCIENCES GmbH: (2020). Pediatric
Arthritis Study of Certolizumab Pegol (PASCAL). ClinicalTrials.gov Identifier: NCT01550003.
https://clinicaltrials.gov/ct2/show/NCT01550003.
|
|
59
|
Novartis Pharmaceuticals: (2020).
Pediatric Study in Children and Adolescents With Severe Plaque
Psoriasis. ClinicalTrials.gov Identifier:
NCT02471144. https://clinicaltrials.gov/ct2/show/NCT02471144.
|
|
60
|
Novartis Pharmaceuticals: (2020). Study to
Assess the Long-term Safety, Tolerability, Efficacy of Secukinumab
in Pediatric Patients of Age 6 to <18 Years, With Moderate to
Severe Plaque Psoriasis. ClinicalTrials.gov Identifier: NCT03668613.
https://clinicaltrials.gov/ct2/show/NCT03668613.
|
|
61
|
Eli Lilly and Company: (2020). Study of
Ixekizumab (LY2439821) in Children 6 to Less Than 18 Years With
Moderate-to-Severe Plaque Psoriasis (Ixora-peds). ClinicalTrials.gov Identifier: NCT03073200.
https://clinicaltrials.gov/ct2/show/NCT03073200.
|
|
62
|
Janssen Research & Development, LLC:
(2020). A Study to Evaluate the Efficacy, Safety, and
Pharmacokinetics of Subcutaneously Administered Guselkumab for the
Treatment of Chronic Plaque Psoriasis in Pediatric Participants
(PROTOSTAR). ClinicalTrials.gov Identifier:
NCT03451851. https://clinicaltrials.gov/ct2/show/NCT03451851.
|
|
63
|
Bausch Health Americas, Inc. (2021). An
Open-label, Single-dose Study to Evaluate Safety, Tolerability, and
Pharmacokinetics of Brodalumab in Pediatric Subjects. ClinicalTrials.gov Identifier: NCT03240809.
https://clinicaltrials.gov/ct2/show/NCT03240809.
|