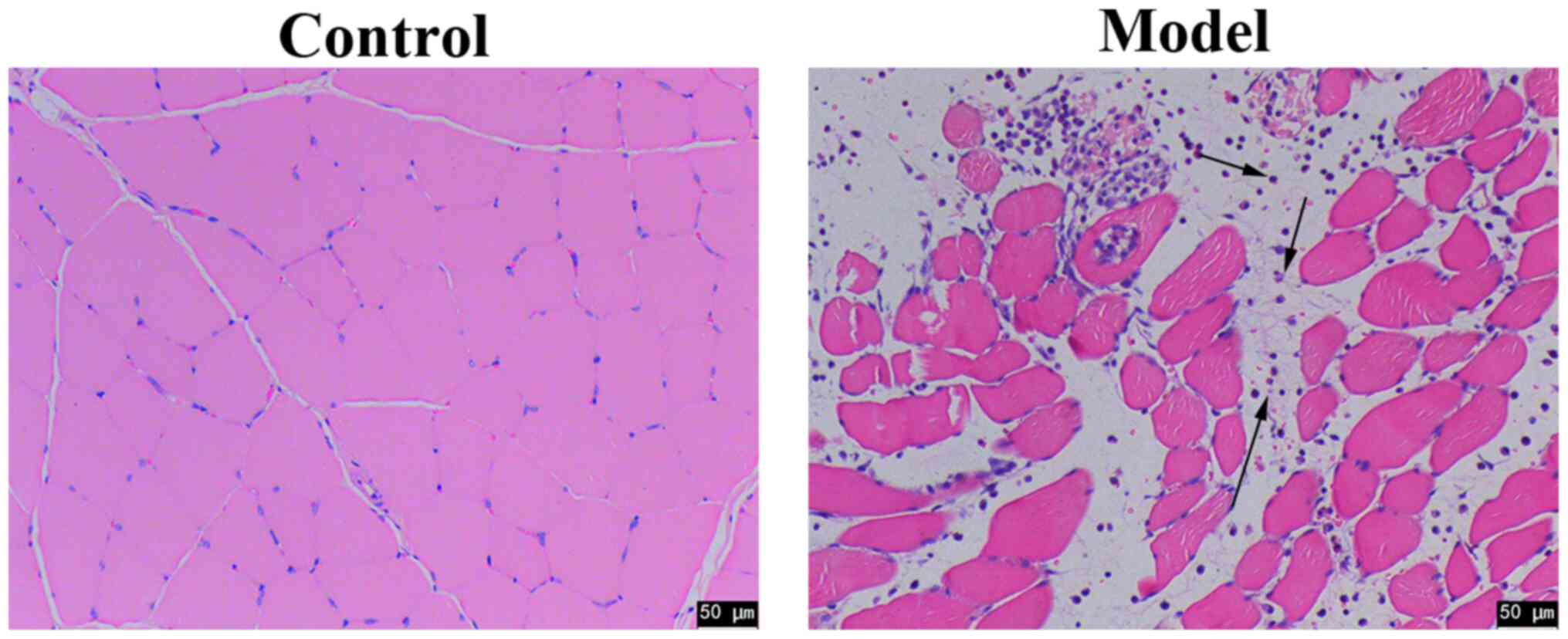
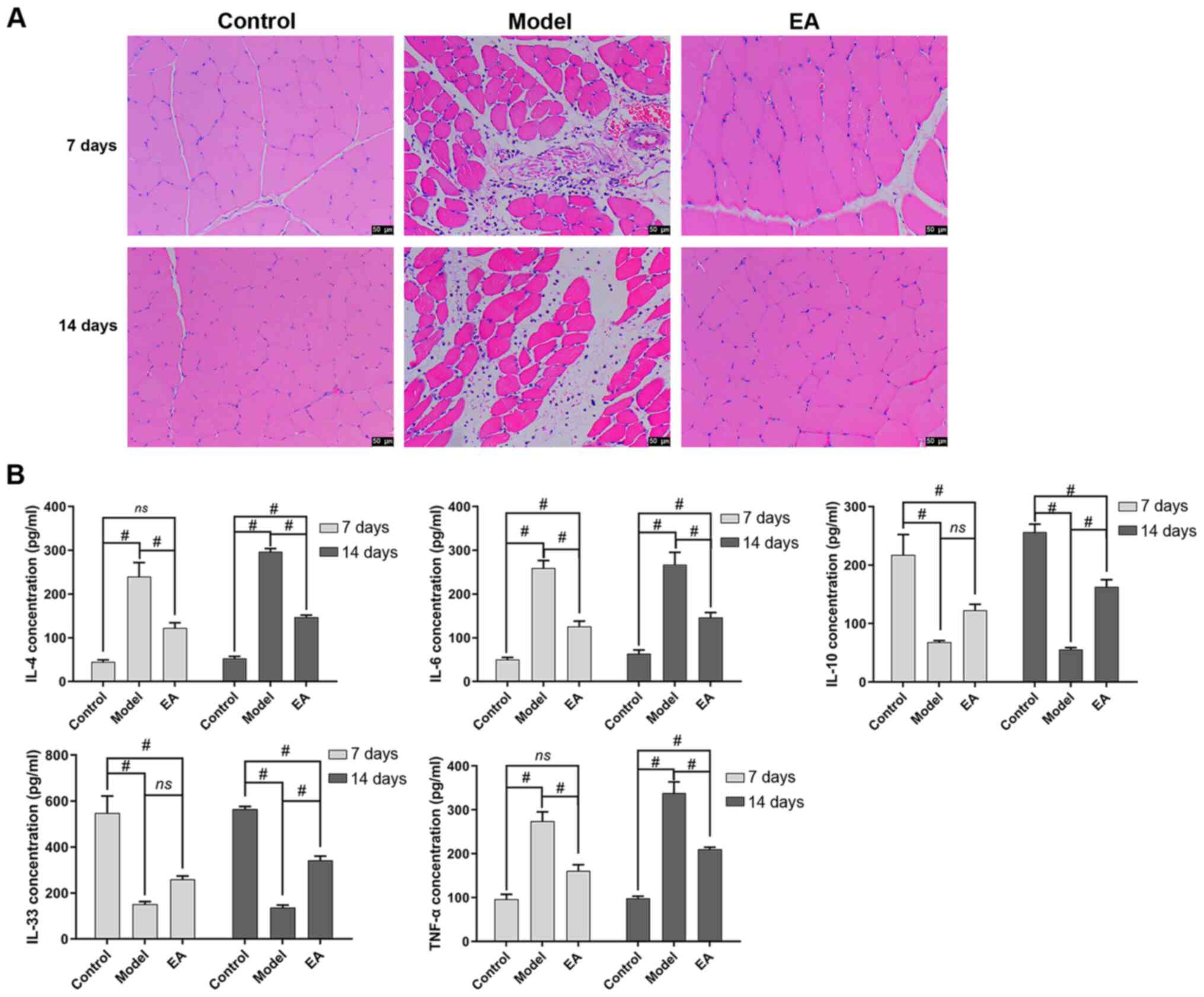
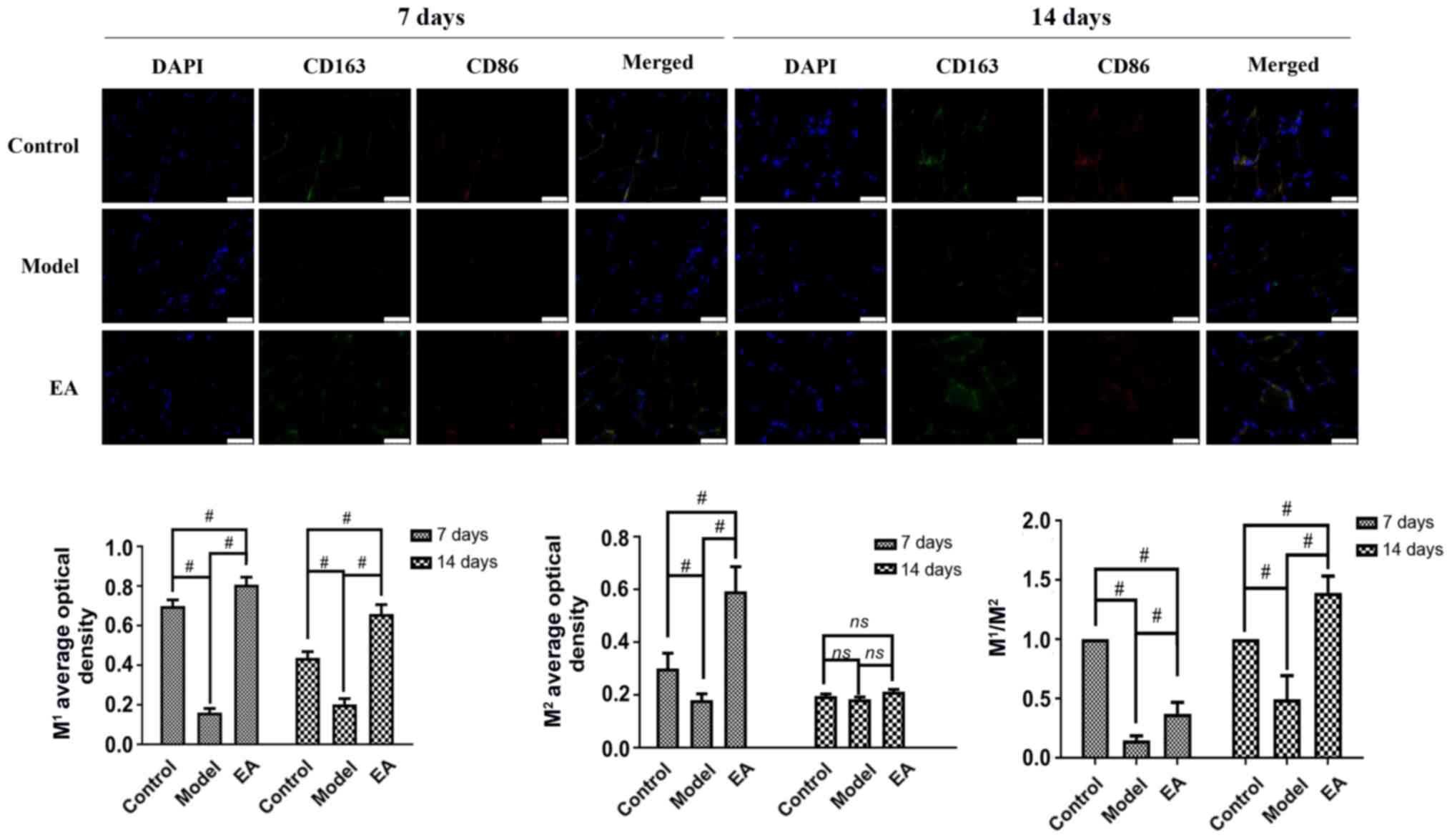
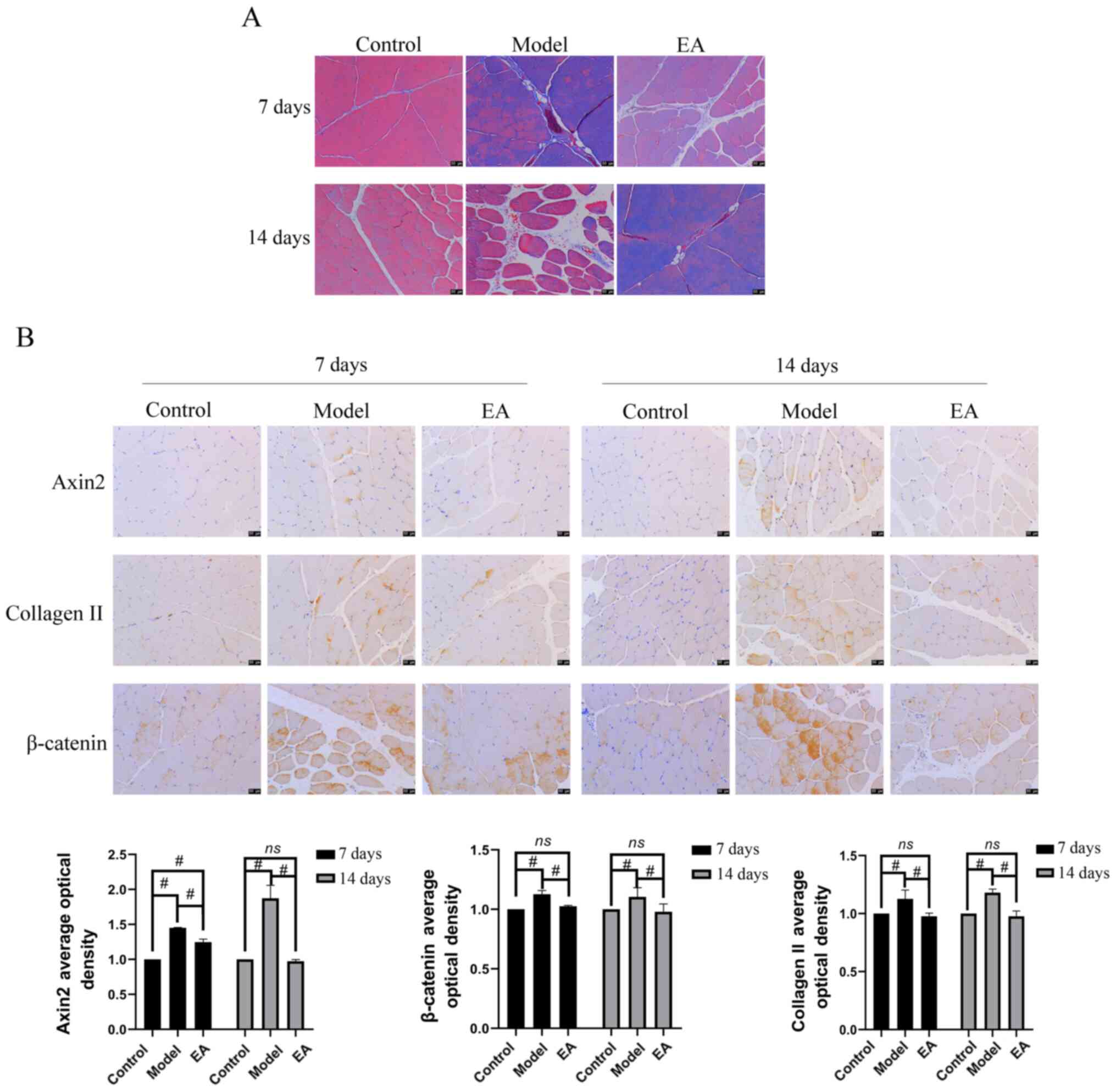
|
1
|
Liao CH, Lin LP, Yu TY, Hsu CC, Pang JS
and Tsai WC: . Ibuprofen inhibited migration of skeletal muscle
cells in association with downregulation of p130cas and CrkII
expressions. Skelet Muscle. 9(23)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Huard J, Li Y and Fu FH: Muscle injuries
and repair: Current trends in research. J Bone Joint Surg Am.
84:822–832. 2002.PubMed/NCBI
|
|
3
|
Baoge L, Van Den Steen E, Rimbaut S,
Philips N, Witvrouw E, Almqvist KF, Vanderstraeten G and Vanden
Bossche LC: Treatment of skeletal muscle injury: A review. ISRN
Orthop. 2012(689012)2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Garg K, Corona BT and Walters TJ:
Therapeutic strategies for preventing skeletal muscle fibrosis
after injury. Front Pharmacol. 6(87)2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
McDermott MM, Dayanidhi S, Kosmac K, Saini
S, Slysz J, Leeuwenburgh C, Hartnell L, Sufit R and Ferucci L: .
Walking Exercise Therapy Effects on Lower Extremity Skeletal Muscle
in Peripheral Artery Disease. Circ Res. 128:1851–1867.
2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Mahdy MAA: Skeletal muscle fibrosis: An
overview. Cell Tissue Res. 375:575–588. 2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tsai WC, Yu TY, Chang GJ, Lin LP, Lin MS
and Pang JS: Platelet-Rich Plasma Releasate Promotes Regeneration
and Decreases Inflammation and Apoptosis of Injured Skeletal
Muscle. Am J Sports Med Jul. 46:1980–1986. 2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Border WA and Noble NA: Transforming
growth factor beta in tissue fibrosis. N Engl J Med. 331:1286–1292.
1994.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Delaney K, Kasprzycka P, Ciemerych MA and
Zimowska M: The role of TGF-β1 during skeletal muscle regeneration.
Cell Biol Int. 41:706–715. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Fang H and Judd RL: Adiponectin Regulation
and Function. Compr Physiol. 8:1031–1063. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Delos D, Leineweber MJ, Chaudhury S,
Alzoobaee S, Gao Y and Rodeo SA: The effect of platelet-rich plasma
on muscle contusion healing in a rat model. Am J Sports Med.
42:2067–2074. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Li H, Hicks JJ, Wang L, Oyster N,
Philippon MJ, Hurwitz S, Hogan MV and Huard J: Customized
platelet-rich plasma with transforming growth factor β1
neutralization antibody to reduce fibrosis in skeletal muscle.
Biomaterials. 87:147–156. 2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wehling-Henricks M, Jordan MC, Gotoh T,
Grody WW, Roos KP and Tidball JG: Arginine metabolism by
macrophages promotes cardiac and muscle fibrosis in mdx muscular
dystrophy. PLoS One. 5(e10763)2010.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Nozaki M, Ota S, Terada S, Li Y, Uehara K,
Gharaibeh B, Fu FH and Huard J: Timing of the administration of
suramin treatment after muscle injury. Muscle Nerve. 46:70–79.
2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ifrim Chen F, Antochi AD and Barbilian AG:
Acupuncture and the retrospect of its modern research. Rom J
Morphol Embryol. 60:411–418. 2019.PubMed/NCBI
|
|
16
|
Martins L, Gallo CC, Honda TSB, Alves PT,
Stilhano RS, Rosa DS, Koh TJ and Han SW: Skeletal muscle healing by
M1-like macrophages produced by transient expression of exogenous
GM-CSF. Stem Cell Res Ther. 11(473)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Li JP, Xu T and Xu SS: Expression and
significance of TNF-α and MMP-1 in acupuncture to inhibit skeletal
muscle fibrosis. Annual Meeting of Sports Physiology Committee of
Chinese Physiological Society and Academic Seminar on ‘Sports and
Health’, 2013.
|
|
18
|
Chen JDZ, Ni M and Yin J:
Electroacupuncture treatments for gut motility disorders.
Neurogastroenterol Motil. 30(e13393)2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Han H and Li M: Clinical observation of
acupuncture combined with exercise therapy for acute lumbar sprain.
Lishizhen Med Mater Med Res. 23:244–245. 2012.
|
|
20
|
Zhang J, Xiao Z, Qu C, Cui W, Wang X and
Du J: CD8 T cells are involved in skeletal muscle regeneration
through facilitating MCP-1 secretion and Gr1(high) macrophage
infiltration. J Immunol. 193:5149–5160. 2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Bayer ML, Magnusson SP and Kjaer M: Tendon
Research Group Bispebjerg. Early versus Delayed Rehabilitation
after Acute Muscle Injury. N Engl J Med. 377:1300–1301.
2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Tidball JG: Mechanisms of muscle injury,
repair, and regeneration. Compr Physiol. 1:2029–2062.
2011.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Smith RS and Chang FC: Traumatic rupture
of the aorta: Still a lethal injury. Am J Surg. 152:660–663.
1986.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Deng XY, Wu ZB and Yang Z: Fibrosis in
skeletal muscle:celluar and molecular mechanism. Chin J RHP.
20:142–147. 2014.
|
|
25
|
DeNardo DG and Ruffell B: Macrophages as
regulators of tumour immunity and immunotherapy. Nat Rev Immunol
Jun. 19:369–382. 2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Xia Y, Rao L, Yao H, Wang Z, Ning P and
Chen X: Engineering Macrophages for Cancer Immunotherapy and Drug
Delivery. Adv Mater. 32(e2002054)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zhang J, Xiao Z, Qu C, Cui W, Wang X and
Du J: CD8 T cells are involved in skeletal muscle regeneration
through facilitating MCP-1 secretion and Gr1(high) macrophage
infiltration. J Immunol. 193:5149–5160. 2014.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Zhou SN, Xu YY and Hu HT: Research
progress of acupuncture therapy on immune cells. Zhejiang JTradit
Chin Med. 55:74–75. 2020.
|
|
29
|
Zhang SY, Hu X and Tang H: Effect of
acupuncture on white adipose tissue macrophage polarization induced
by high fat diet in obese mice. Chin Acupunc. 37:1205–1211.
2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ding J, Chang YY, Li WJ, Guo B, Lu M and
Collage XM: The effects of DAPT intervention on the expression of
α-SMA and MMP-1/TIMP-1 in rats cardiac fibroblasts. J Clin Cardiol.
32:735–738. 2016.
|
|
31
|
Wang SQ, Chang Y, Ma XW and Wang F:
Effects of endurence exercise of different intensity on cardiac
collagen of rats and regulation of MMP-1/TIMP-1. Chi Sport Sci
Technol. 51:60–66. 2015.
|
|
32
|
Wang QY, Wang WJ, Xi J, Cai K, Wang P,
Liang JD, Han J and He GZ: Effect of polysaccharides from eucommiae
cortex on expressions of genes of I, III collagen, MMP-1, TIM MP-1
and TGF-β1 from hepatic fibrosis in rat models. Chin J Exp Trad Med
Formu. 24:153–158. 2018.
|
|
33
|
Spinale FG: Myocardial matrix remodeling
and the matrix metalloproteinases: Influence on cardiac form and
function. Physiol Rev. 87:1285–1342. 2007.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Xiao W, Liu Y and Chen P: Macrophage
depletion impairs skeletal muscle regeneration: The roles of
pro-fibrotic factors, inflammation, and oxidative stress.
Inflammation. 39:2016–2028. 2016.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Gihring A, Gärtner F, Liu C, Hoenicka M,
Wabitsch M, Knippschild U and Xu P: Influence of Obesity on the
Organization of the Extracellular Matrix and Satellite Cell
Functions After Combined Muscle and Thorax Trauma in C57BL/6J Mice.
Front Physiol. 11(849)2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Dong G, Wang M, Gu G, Li S, Sun X, Li Z,
Cai H and Zhu Z: MACC1 and HGF are associated with survival in
patients with gastric cancer. Oncol Lett. 15:3207–3213.
2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Nalbandian M, Radak Z and Takeda M:
Lactate Metabolism and Satellite Cell Fate. Front Physiol.
11(610983)2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Chen X and Li Y: Role of matrix
metalloproteinases in skeletal muscle: Migration, differentiation,
regeneration and fibrosis. Cell Adhes Migr. 3:337–341.
2009.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Zheng LF, Chen LF, Chen PJ and Xiao WH:
Fat deposition in skeletal muscle and its regulatory mechanism.
Acta Physiologica Sinicaa. 69:344–350. 2017.PubMed/NCBI(in Chinese).
|
|
40
|
Miyazaki D, Nakamura A, Fukushima K,
Yoshida K, Takeda S and Ikeda S: Matrix metalloproteinase-2
ablation in dystrophin-deficient mdx muscles reduces angiogenesis
resulting in impaired growth of regenerated muscle fibers. Hum Mol
Genet. 20:1787–1799. 2011.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zheng LF: Study on the role and mechanism
of macrophages in the repair of skeletal muscle contusion. Shanghai
university of sport, 2018 (in Chinese).
|
|
42
|
Carlson ME, Conboy MJ, Hsu M, Barchas L,
Jeong J, Agrawal A, Mikels AJ, Agrawal S, Schaffer DV and Conboy
IM: Relative roles of TGF-beta1 and Wnt in the systemic regulation
and aging of satellite cell responses. Aging Cell. 8:676–689.
2009.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Fjeldborg K, Pedersen SB, Møller HJ,
Christiansen T, Bennetzen M and Richelsen B: Human adipose tissue
macrophages are enhanced but changed to an anti-inflammatory
profile in obesity. J Immunol Res. 2014(309548)2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Rana T, Jiang C, Liu G, Miyata T, Antony
V, Thannickal VJ and Liu RM: PAI-1 Regulation of TGF-β1-induced
Alveolar Type II Cell Senescence, SASP Secretion, and SASP-mediated
Activation of Alveolar Macrophages. Am J Respir Cell Mol Biol.
62:319–330. 2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Lorts A, Schwanekamp JA, Baudino TA,
McNally EM and Molkentin JD: Deletion of periostin reduces muscular
dystrophy and fibrosis in mice by modulating the transforming
growth factor-β pathway. Proc Natl Acad Sci USA. 109:10978–10983.
2012.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Bedair HS, Karthikeyan T, Quintero A, Li Y
and Huard J: Angiotensin II receptor blockade administered after
injury improves muscle regeneration and decreases fibrosis in
normal skeletal muscle. Am J Sports Med. 36:1548–1554.
2008.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Roffe S, Hagai Y, Pines M and Halevy O:
Halofuginone inhibits Smad3 phosphorylation via the PI3K/Akt and
MAPK/ERK pathways in muscle cells: Effect on myotube fusion. Exp
Cell Res. 316:1061–1069. 2010.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Huebner KD, Jassal DS, Halevy O, Pines M
and Anderson JE: Functional resolution of fibrosis in mdx mouse
dystrophic heart and skeletal muscle by halofuginone. Am J Physiol
Heart Circ Physiol. 294:H1550–H1561. 2008.PubMed/NCBI View Article : Google Scholar
|