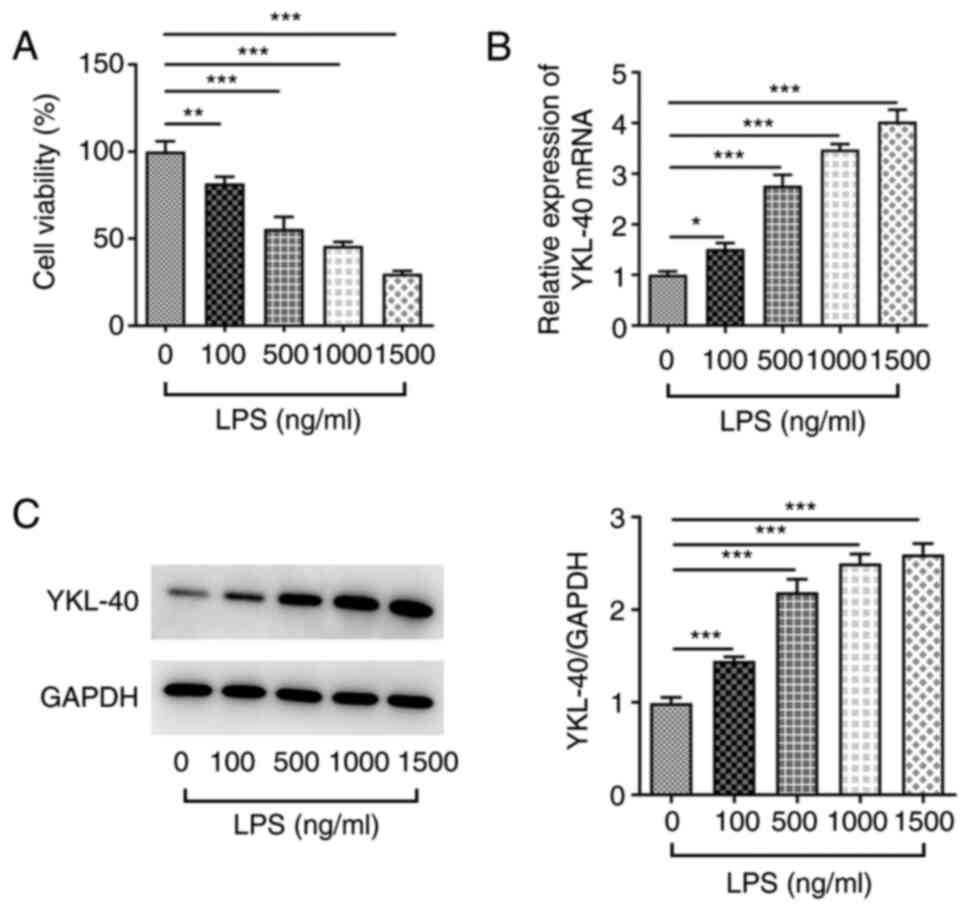
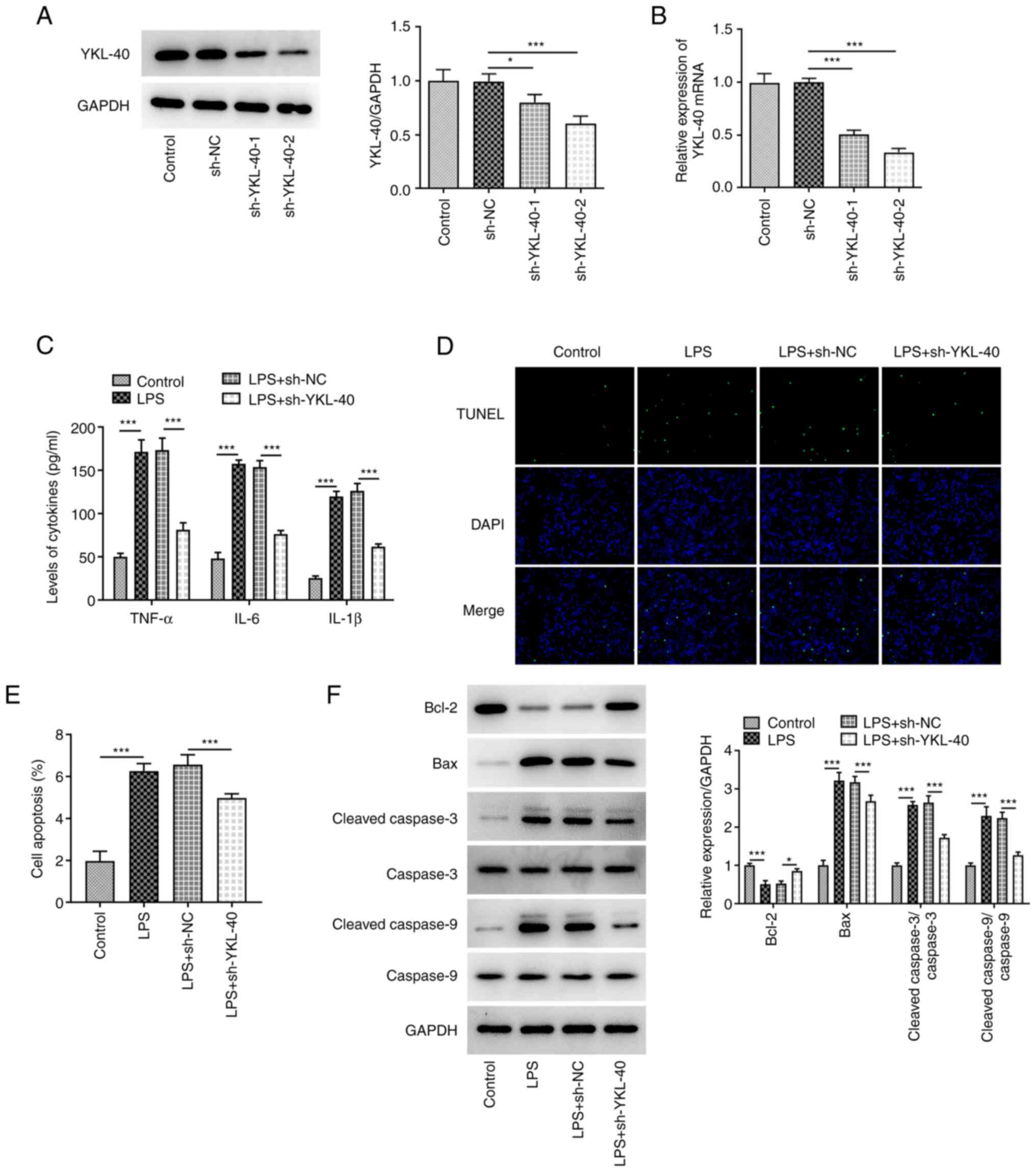
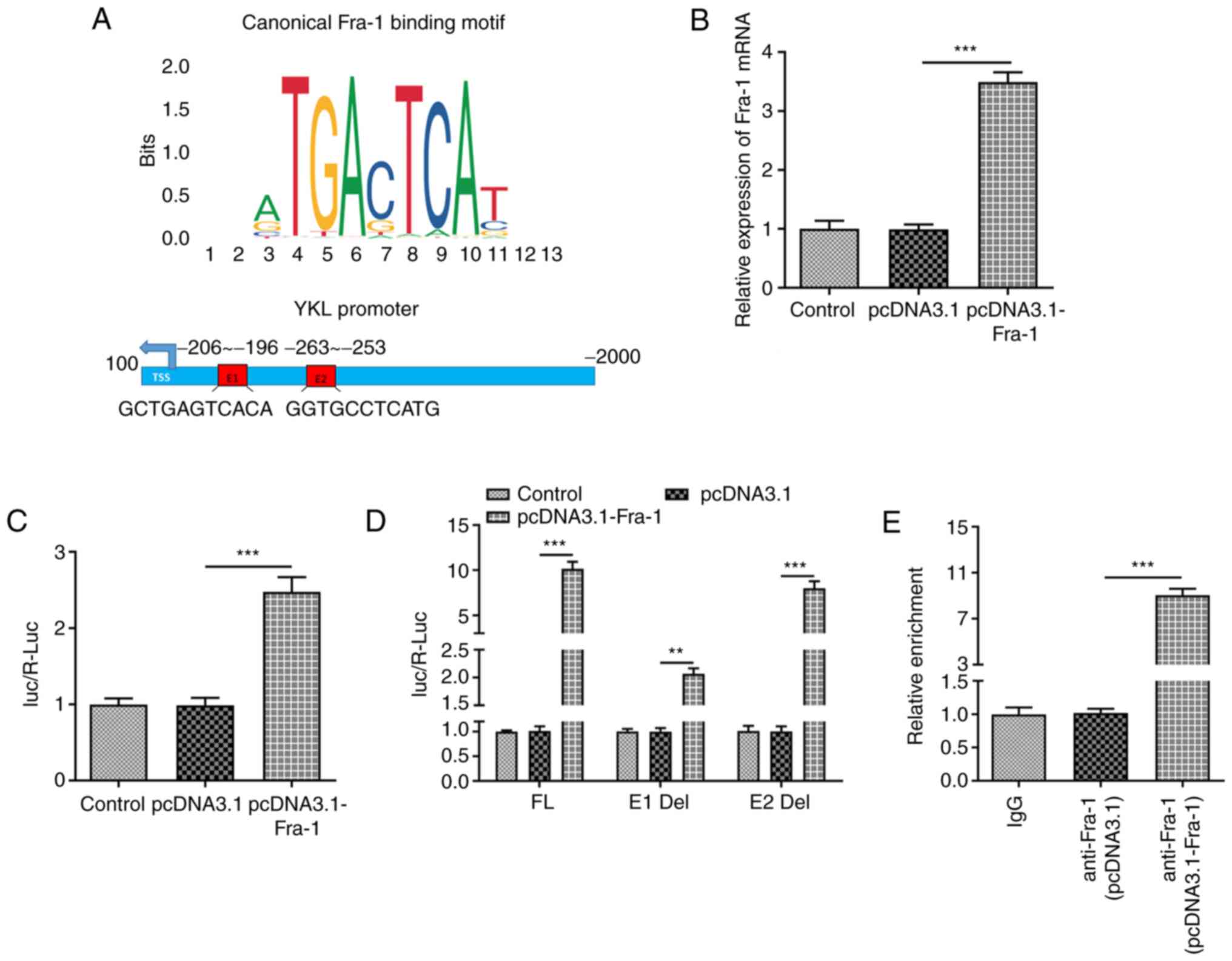
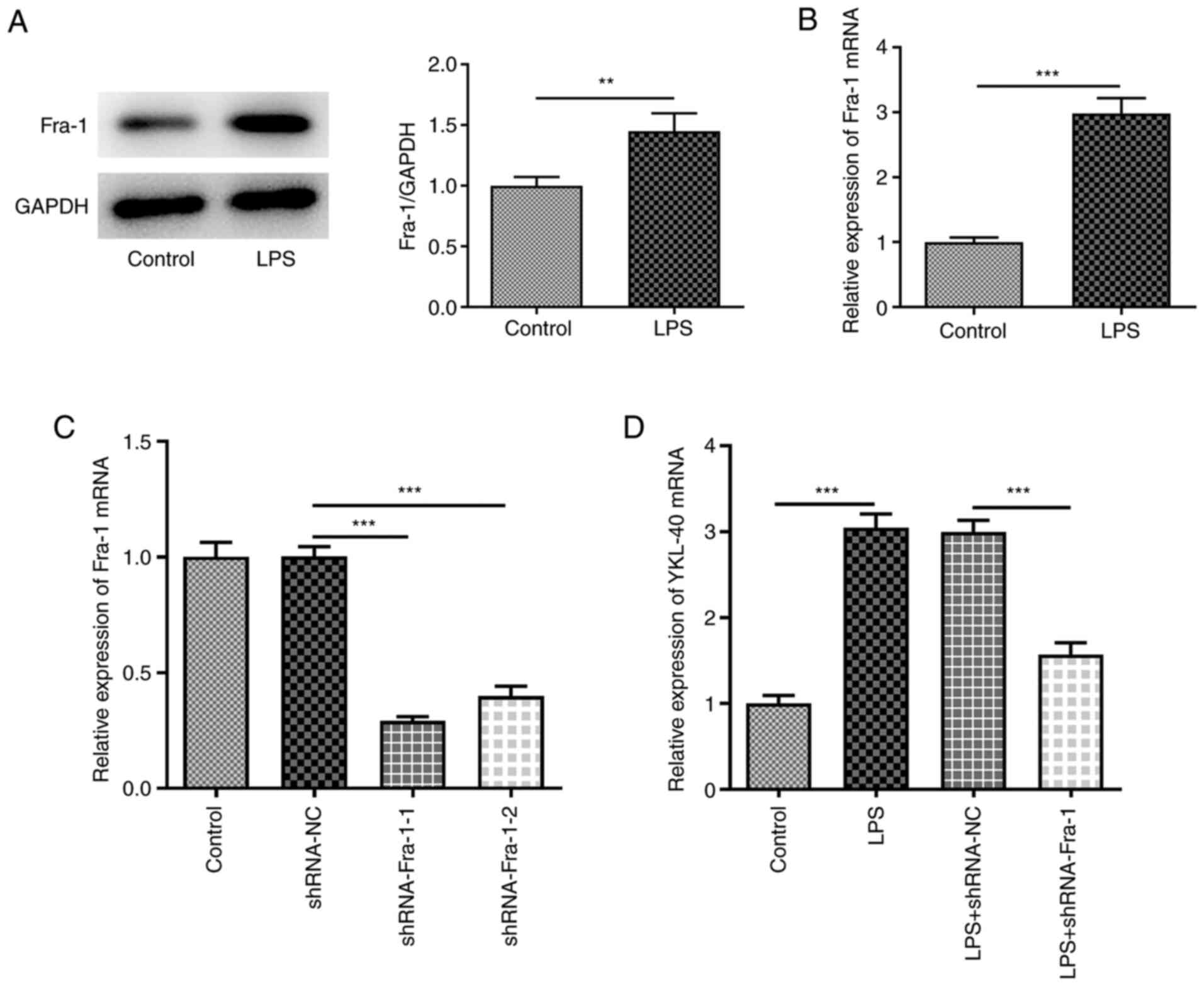
|
1
|
Amigoni A, Pettenazzo A, Stritoni V and
Circelli M: Surfactants in acute respiratory distress syndrome in
infants and children: Past, present and future. Clin Drug Investig.
37:729–736. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Matthay MA and Zemans RL: The acute
respiratory distress syndrome: Pathogenesis and treatment. Annu Rev
Pathol. 6:147–163. 2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ware LB and Matthay MA: The acute
respiratory distress syndrome. N Engl J Med. 342:1334–1349.
2000.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Jung YJ, Park YY, Huh JW and Hong SB: The
effect of human adipose-derived stem cells on
lipopolysaccharide-induced acute respiratory distress syndrome in
mice. Ann Transl Med. 7(674)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Li D, Sun T, Chi L, Zhao D and Li W:
Acupoint catgut embedding improves the lipopolysaccharide-induced
acute respiratory distress syndrome in rats. Biomed Res Int.
2020(2394734)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Pooladanda V, Thatikonda S, Bale S,
Pattnaik B, Sigalapalli DK, Bathini NB, Singh SB and Godugu C:
Nimbolide protects against endotoxin-induced acute respiratory
distress syndrome by inhibiting TNF-α mediated NF-κB and HDAC-3
nuclear translocation. Cell Death Dis. 10(81)2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lee CG, Hartl D, Lee GR, Koller B,
Matsuura H, Da Silva CA, Sohn MH, Cohn L, Homer RJ, Kozhich AA, et
al: Role of breast regression protein 39 (BRP-39)/chitinase
3-like-1 in Th2 and IL-13-induced tissue responses and apoptosis. J
Exp Med. 206:1149–1166. 2009.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Johansen JS: Studies on serum YKL-40 as a
biomarker in diseases with inflammation, tissue remodelling,
fibroses and cancer. Dan Med Bull. 53:172–209. 2006.PubMed/NCBI
|
|
9
|
Kastrup J: Can YKL-40 be a new
inflammatory biomarker in cardiovascular disease? Immunobiology.
217:483–491. 2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Jafari-Nakhjavani MR, Ghorbanihaghjo A,
Bagherzadeh-Nobari B, Malek-Mahdavi A and Rashtchizadeh N: Serum
YKL-40 levels and disease characteristics in patients with
rheumatoid arthritis. Caspian J Intern Med. 10:92–97.
2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ostergaard C, Johansen JS, Benfield T,
Price PA and Lundgren JD: YKL-40 is elevated in cerebrospinal fluid
from patients with purulent meningitis. Clin Diagn Lab Immunol.
9:598–604. 2002.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nordenbaek C, Johansen JS, Junker P,
Borregaard N, Sorensen O and Price PA: YKL-40, a matrix protein of
specific granules in neutrophils, is elevated in serum of patients
with community-acquired pneumonia requiring hospitalization. J
Infect Dis. 180:1722–1726. 1999.PubMed/NCBI View
Article : Google Scholar
|
|
13
|
Tong X, Wang D, Liu S, Ma Y, Li Z, Tian P
and Fan H: The YKL-40 protein is a potential biomarker for COPD: A
meta-analysis and systematic review. Int J Chron Obstruct Pulmon
Dis. 13:409–418. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sohn MH, Kang MJ, Matsuura H, Bhandari V,
Chen NY, Lee CG and Elias JA: The chitinase-like proteins breast
regression protein-39 and YKL-40 regulate hyperoxia-induced acute
lung injury. Am J Respir Crit Care Med. 182:918–928.
2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wagner EF and Eferl R: Fos/AP-1 proteins
in bone and the immune system. Immunol Rev. 208:126–140.
2005.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Burch PM, Yuan Z, Loonen A and Heintz NH:
An extracellular signal-regulated kinase 1- and 2-dependent program
of chromatin trafficking of c-Fos and Fra-1 is required for cyclin
D1 expression during cell cycle reentry. Mol Cell Biol.
24:4696–4709. 2004.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Xi X, Yao Y, Liu N and Li P: MiR-297
alleviates LPS-induced A549 cell and mice lung injury via targeting
cyclin dependent kinase 8. Int Immunopharmacol.
80(106197)2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Bein T, Briegel J and Annane D: Steroids
are part of rescue therapy in ARDS patients with refractory
hypoxemia: Yes. Intensive Care Med. 42:918–920. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhao D, Ding R, Mao Y, Wang L, Zhang Z and
Ma X: Heparin rescues sepsis-associated acute lung injury and
lethality through the suppression of inflammatory responses.
Inflammation. 35:1825–1832. 2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hu H, Shi D, Hu C, Yuan X, Zhang J and Sun
H: Dexmedetomidine mitigates CLP-stimulated acute lung injury via
restraining the RAGE pathway. Am J Transl Res. 9:5245–5258.
2017.PubMed/NCBI
|
|
22
|
Zhou H, Wang X and Zhang B: Depression of
lncRNA NEAT1 antagonizes LPS-evoked acute injury and inflammatory
response in alveolar epithelial cells via HMGB1-RAGE signaling.
Mediators Inflamm. 2020(8019467)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Li P, Yao Y, Ma Y and Chen Y: MiR-150
attenuates LPS-induced acute lung injury via targeting AKT3. Int
Immunopharmacol. 75(105794)2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Li P, Yao Y, Ma Y and Chen Y: MiR-30a-5p
ameliorates LPS-induced inflammatory injury in human A549 cells and
mice via targeting RUNX2. Innate Immun. 27:41–49. 2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yamaguchi T, Takada Y, Maruyama K, Shimoda
K, Arai Y, Nango N, Kosaki N, Takaishi H, Toyama Y and Matsuo K:
Fra-1/AP-1 impairs inflammatory responses and chondrogenesis in
fracture healing. J Bone Miner Res. 24:2056–2065. 2009.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Rajasekaran S, Vaz M and Reddy SP:
Fra-1/AP-1 transcription factor negatively regulates pulmonary
fibrosis in vivo. PLoS One. 7(e41611)2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Rajasekaran S, Tamatam CR, Potteti HR,
Raman V, Lee JW, Matthay MA, Mehta D, Reddy NM and Reddy SP:
Visualization of Fra-1/AP-1 activation during LPS-induced
inflammatory lung injury using fluorescence optical imaging. Am J
Physiol Lung Cell Mol Physiol. 309:L414–L424. 2015.PubMed/NCBI View Article : Google Scholar
|