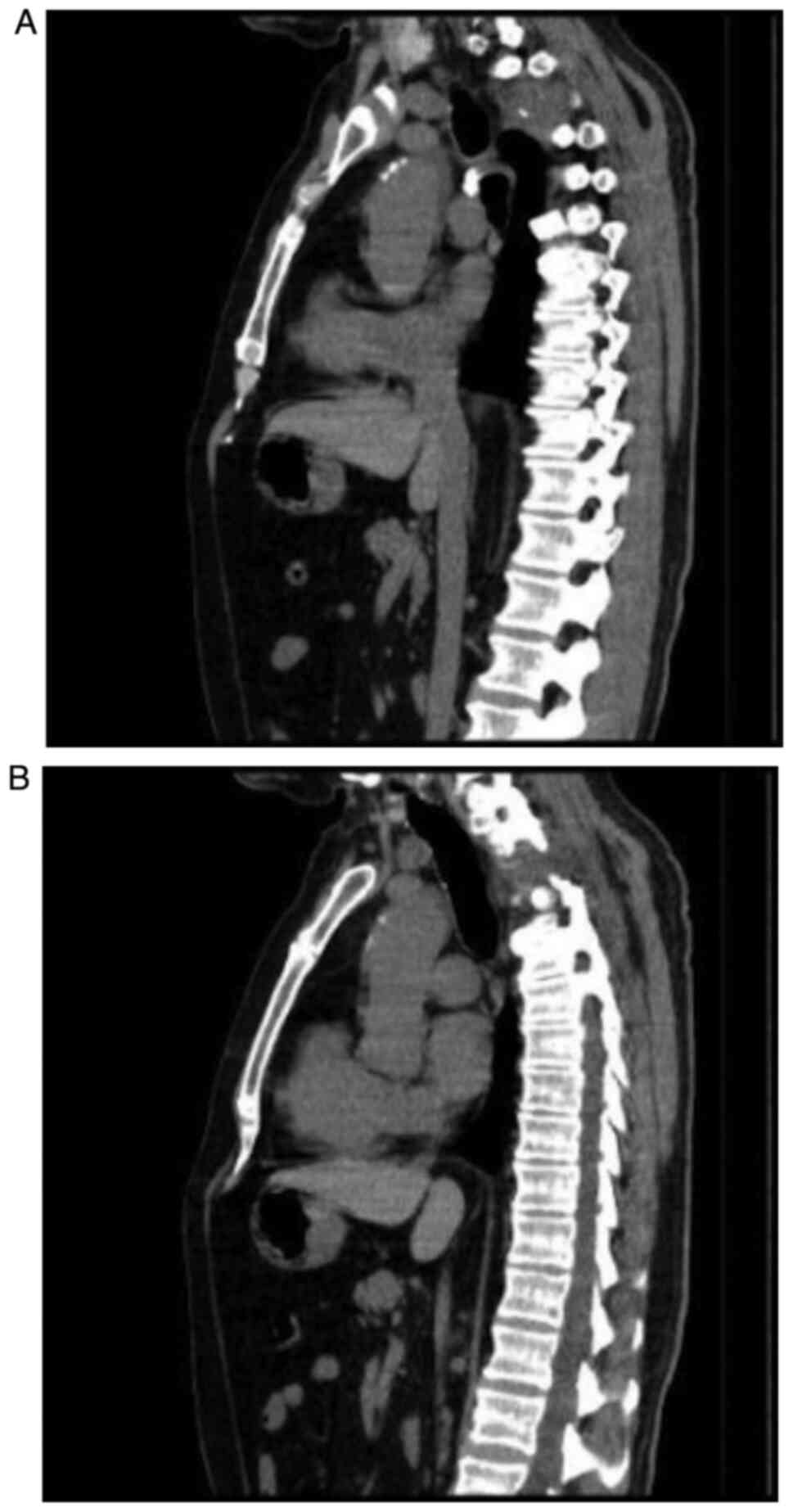
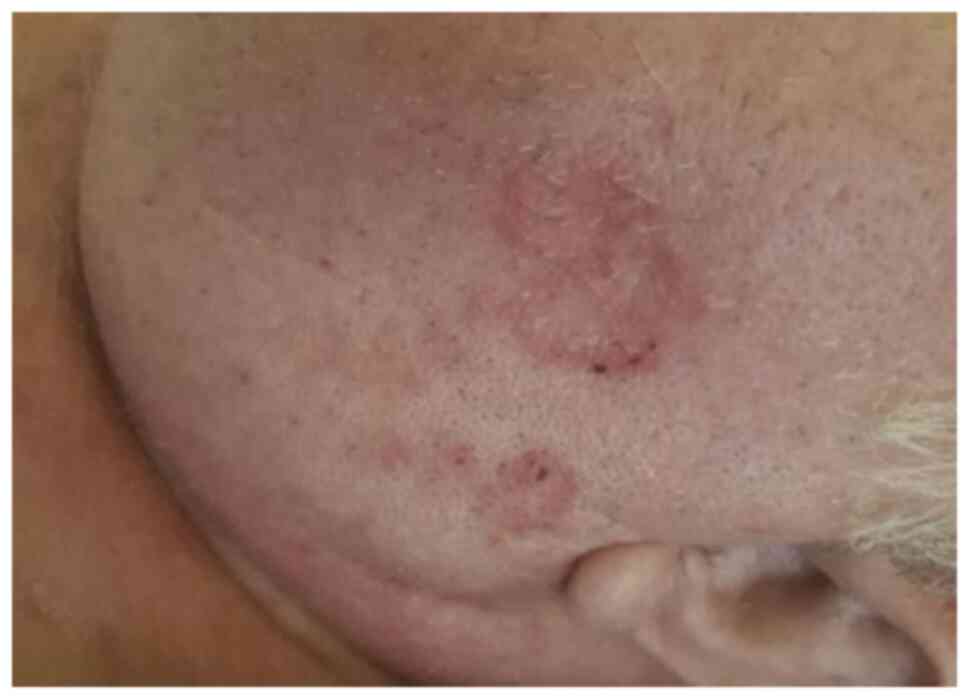
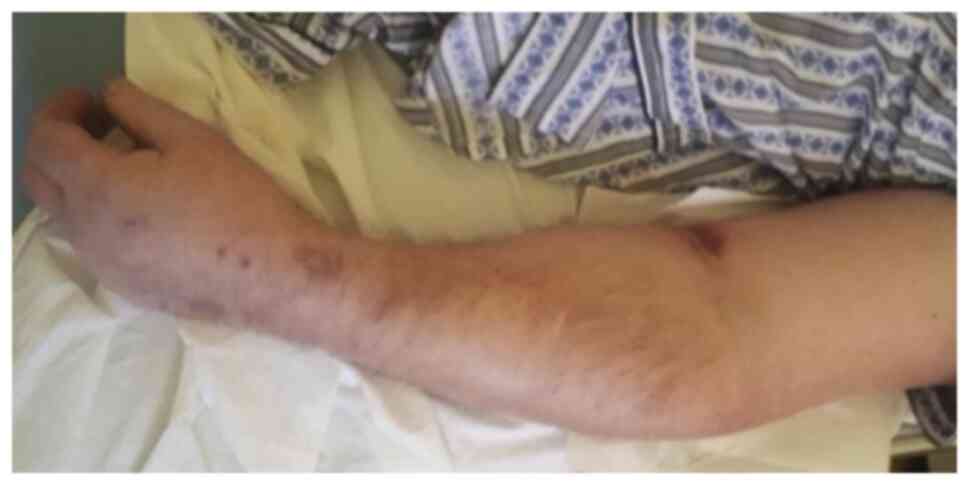
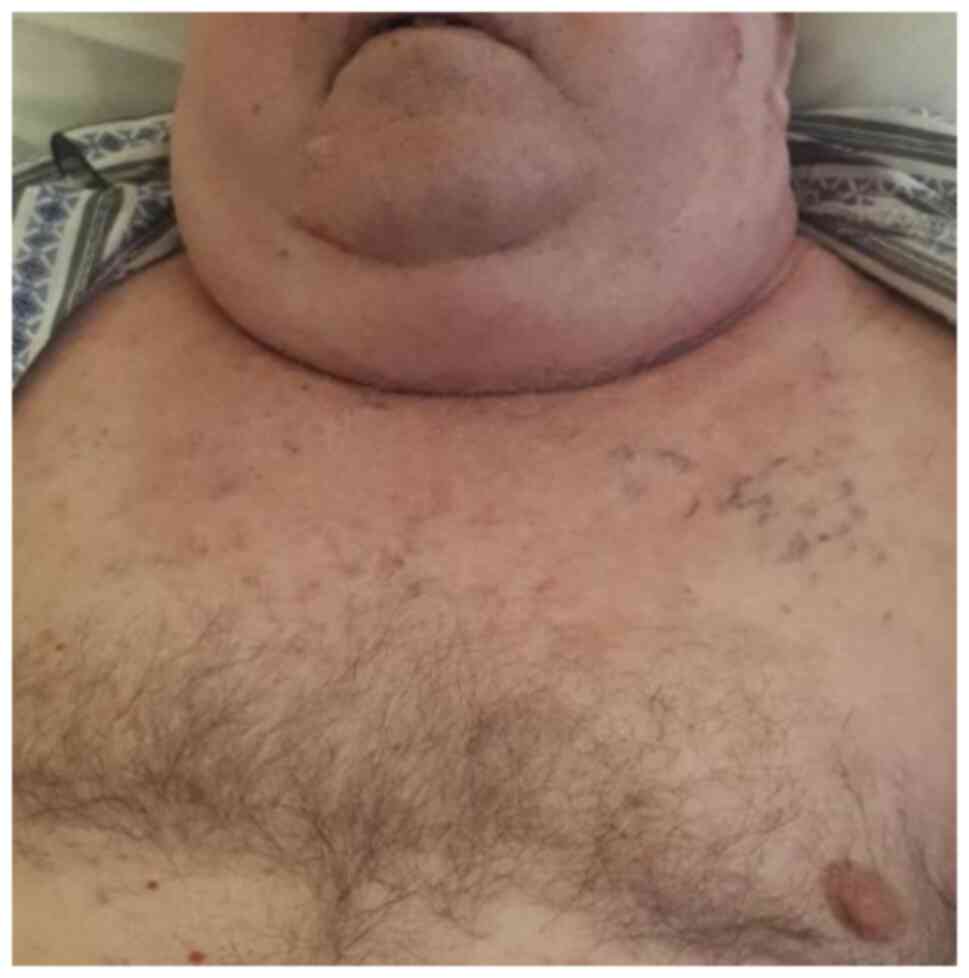
|
1
|
Craescu M, Rebegea L, Ivan I, Dumitru M,
Serban C and Firescu D: Therapeutic challenges in a case of trachea
neuroendocrine tumor. Acta Med Mediterr. 35:1493–1496. 2019.
|
|
2
|
Fekete GL, Cotoi OS and Fekete JE:
Multiple nodular cutaneous metastases as the first clinical sign of
signet ring cell gastric carcinoma: Case report. Acta
Dermatovenerol Croat. 20:34–37. 2012.PubMed/NCBI
|
|
3
|
Riihimäki M, Hemminki A, Fallah M, Thomsen
H, Sundquist K, Sundquist J and Hemminki K: Metastatic sites and
survival in lung cancer. Lung Cancer. 86:78–84. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
National Comprehensive Cancer Network
(NCCN): NCCN Clinical Practice Guidelines in Oncology (NCCN
guidelines): Non-small cell lung cancer. Version 5.2019 - June 7,
2019. NCCN, Plymouth Meeting, PA, 2019. https://www.nccn.org/guidelines/category_1#si.
Accessed July 30, 2019.
|
|
5
|
Nomori H, Watanabe K, Ohtusuka T, Naruke
T, Suemasu K and Uno K: The size of metastatic foci and lymph nodes
yielding false-negative and false-positive lymph node staging with
positron emission tomography in patients with lung cancer. J Thorac
Cardiovasc Surg. 127:1087–1092. 2004.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Niculet E, Chioncel V, Elisei AM, Miulescu
M, Buzia OD, Nwabudike LC, Craescu M, Draganescu M, Bujoreanu F,
Marinescu E, et al: Multifactorial expression of IL-6 with update
on COVID-19 and the therapeutic strategies of its blockade
(Review). Exp Ther Med. 21(263)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Dela Cruz CS, Tanoue LT and Matthay RA:
Lung cancer: Epidemiology, etiology and prevention. Clin Chest Med.
32:605–644. 2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhou H, Wu A, Fu W, Lv Z and Zhang Z:
Significance of semaphorin-3A and MMP-14 protein expression in
non-small cell lung cancer. Oncol Lett. 7:1395–1400.
2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Gheorghe I, Tatu AL, Lupu I, Thamer O,
Cotar AI, Pircalabioru GG, Popa M, Cristea VC, Lazar V and
Chifiriuc MC: Molecular characterization of virulence and
resistance features in Staphylococcus aureus clinical strains
isolated from cutaneous lesions in patients with drug adverse
reactions. Rom Biotech Lett. 22:12321–12327. 2017.
|
|
10
|
Hwang SJE and Fernández-Peñas P: Adverse
reactions to biologics: Melanoma (Ipilimumab, Nivolumab,
Pembrolizumab). Curr Probl Dermatol. 53:82–92. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Lee M and Seetharamu N: An atypical
presentation of lichen planus-like reaction from pembrolizumab.
Case Rep Dermatol Med. 2019(4065437)2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nwabudike LC and Tatu AL: Reply to
Gambichler T et al: Altered epigenetic pathways and cell
cycle dysregulation in healthy appearing skin of patients with
koebnerized squamous cell carcinomas following skin surgery. J Eur
Acad Dermatol Venereol. 33:e3–e4. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Flaten HK and Monte AA: The
pharmacogenomic and metabolomic predictors of ACE inhibitor and
angiotensin II receptor blocker effectiveness and safety.
Cardiovasc Drugs Ther. 31:471–482. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Johnson JA: Advancing management of
hypertension through pharmacogenomics. Ann Med. 44 (Suppl
1):S17–S22. 2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Tatu AL, Elisei AM, Chioncel V, Miulescu M
and Nwabudike LC: Immunologic adverse reactions of β-blockers and
the skin (Review). Exp Ther Med. 18:955–959. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Nwabudike LC, Elisei AM, Buzia OD,
Miulescu M and Tatu AL: Statins. A review on structural
perspectives, adverse reactions and relations with non-melanoma
skin cancer. Rev Chim (Bucharest). 69:2557–2562. 2018.
|
|
17
|
Tatu AL, Ciobotaru OR, Miulescu M, Buzia
OD, Elisei AM, Mardare N, Diaconu C, Robu S and Nwabudike LC:
Hydrochlorothiazide: Chemical structure, therapeutic, phototoxic
and carcinogenetic effects in dermatology. Rev Chim (Bucharest).
69:2110–2114. 2018.
|
|
18
|
Dobre M, Georgescu C, Stefanescu V,
Cuciureanu M, Nechita A and Arbune M: Homeostatic changes during
anticonvulsant medication in children. Farmacia. 63:402–406.
2015.
|
|
19
|
Jáuregui-Garrido B and Jáuregui-Lobera I:
Interactions between antihypertensive drugs and food. Nutr Hosp.
27:1866–1875. 2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Fidler-Benaoudia MM, Torre LA, Bray F,
Ferlay J and Jemal A: Lung cancer incidence in young women vs.
young men: A systematic analysis in 40 countries. Int J Cancer.
147:811–819. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Özmen S and Ceylan O: Trends in lung
cancer incidence within the last 10 years: An Eastern Anatolian
single center experience. J Surg Med. 4:112–115. 2020.
|
|
22
|
Blandino G and Di Agostino S: New
therapeutic strategies to treat human cancers expressing mutant p53
proteins. J Exp Clin Cancer Res. 37(30)2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
NSCLC Meta-analyses Collaborative Group.
Arriagada R, Auperin A, Burdett S, Higgins JP, Johnson DH, Le
Chevalier T, Le Pechoux C, Parmar MK, Pignon JP, et al: Adjuvant
chemotherapy, with or without postoperative radiotherapy, in
operable non-small-cell lung cancer: Two meta-analyses of
individual patient data. Lancet. 375:1267–1277. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Sui H, Ma N, Wang Y, Li H, Liu X, Su Y and
Yang J: Anti-PD-1/PD-L1 therapy for non-small-cell lung cancer:
Toward personalized medicine and combination strategies. J Immunol
Res. 2018(6984948)2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Biton J, Mansuet-Lupo A, Pécuchet N,
Alifano M, Ouakrim H, Arrondeau J, Boudou-Rouquette P, Goldwasser
F, Leroy K, Goc J, et al: TP53, STK11, and EGFR mutations predict
tumor immune profile and the response to anti-PD-1 in lung
adenocarcinoma. Clin Cancer Res. 24:5710–5723. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wu CH and Hwang MJ: Risk stratification
for lung adenocarcinoma on EGFR and TP53 mutation status,
chemotherapy, and PD-L1 immunotherapy. Cancer Med. 8:5850–5861.
2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Sanlorenzo M, Vujic I, Daud A, Algazi A,
Gubens M, Luna SA, Lin K, Quaglino P, Rappersberger K and
Ortiz-Urda S: Pembrolizumab cutaneous adverse events and their
association with disease progression. JAMA Dermatol. 151:1206–1212.
2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Simonsen AB, Kaae J, Ellebaek E, Svane IM
and Zachariae C: Cutaneous adverse reactions to anti-PD-1
treatment-a systematic review. J Am Acad Dermatol. 83:1415–1424.
2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Garon EB, Rizvi NA, Hui R, Leighl N,
Balmanoukian AS, Eder JP, Patnaik A, Aggarwal C, Gubens M, Horn L,
et al: Pembrolizumab for the treatment of non-small-cell lung
cancer. N Engl J Med. 372:2018–2028. 2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Fekete GL, Boda D, Căruntu C and Fekete L:
Paraneoplastic pityriasis rubra pilaris in association with
prostate carcinoma: A case report and literature review. Exp Ther
Med. 18:5052–5055. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Coleman E, Panse G, Haldas J, Gettinger SN
and Leventhal JS: Pityriasis rubra pilaris-like erythroderma in the
setting of pembrolizumab therapy responsive to acitretin. JAAD Case
Rep. 4:669–671. 2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Creţu A, Dimitriu A, Brănişteanu D and
Brinişteanu DE: Erythema multiforme-etiopathogenic, clinical and
therapeutic aspects. Rev Med Chir Soc Med Nat Iasi. 119:55–61.
2015.PubMed/NCBI
|
|
33
|
Joshi N (ed): Pembrolizumab/rivaroxaban
interaction. In: Reactions Weekly. Vol 1704. Springer Nature
Switzerland AG, Cham, p304, 2018.
|
|
34
|
Bobeică C, Tatu AL, Crăescu M and
Solovăstru L: Dynamics of digital ulcers in systemic sclerosis. Exp
Ther Med. 20:61–67. 2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Bobeica C, Niculet E, Craescu M, Onisor C,
Bujoreanu F, Draganescu ML, Halip IA and Gheuca-Solovastru L:
Epidemiological profile of systemic sclerosis in the southeast
region of Romania. Exp Ther Med. 21(77)2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Niculet E, Bobeica C and Tatu AL:
Glucocorticoid-induced skin atrophy: The old and the new. Clin
Cosmet Investig Dermatol. 13:1041–1050. 2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Haanen J, Ernstoff M, Wang Y, Menzies A,
Puzanov I, Grivas P, Larkin J, Peters S, Thompson J and Obeid M:
Rechallenge patients with immune checkpoint inhibitors following
severe immune-related adverse events: Review of the literature and
suggested prophylactic strategy. J Immunother Cancer.
8(e000604)2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Winer A, Bodor JN and Borghaei H:
Identifying and managing the adverse effects of immune checkpoint
blockade. J Thorac Dis. 10 (Suppl 3):S480–S489. 2018.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Bhatlapenumarthi V, Patwari A and Harb AJ:
Immune-related adverse events and immune checkpoint inhibitor
tolerance on rechallenge in patients with irAEs: A single-center
experience. J Cancer Res Clin Oncol. 147:2789–2800. 2021.PubMed/NCBI View Article : Google Scholar
|