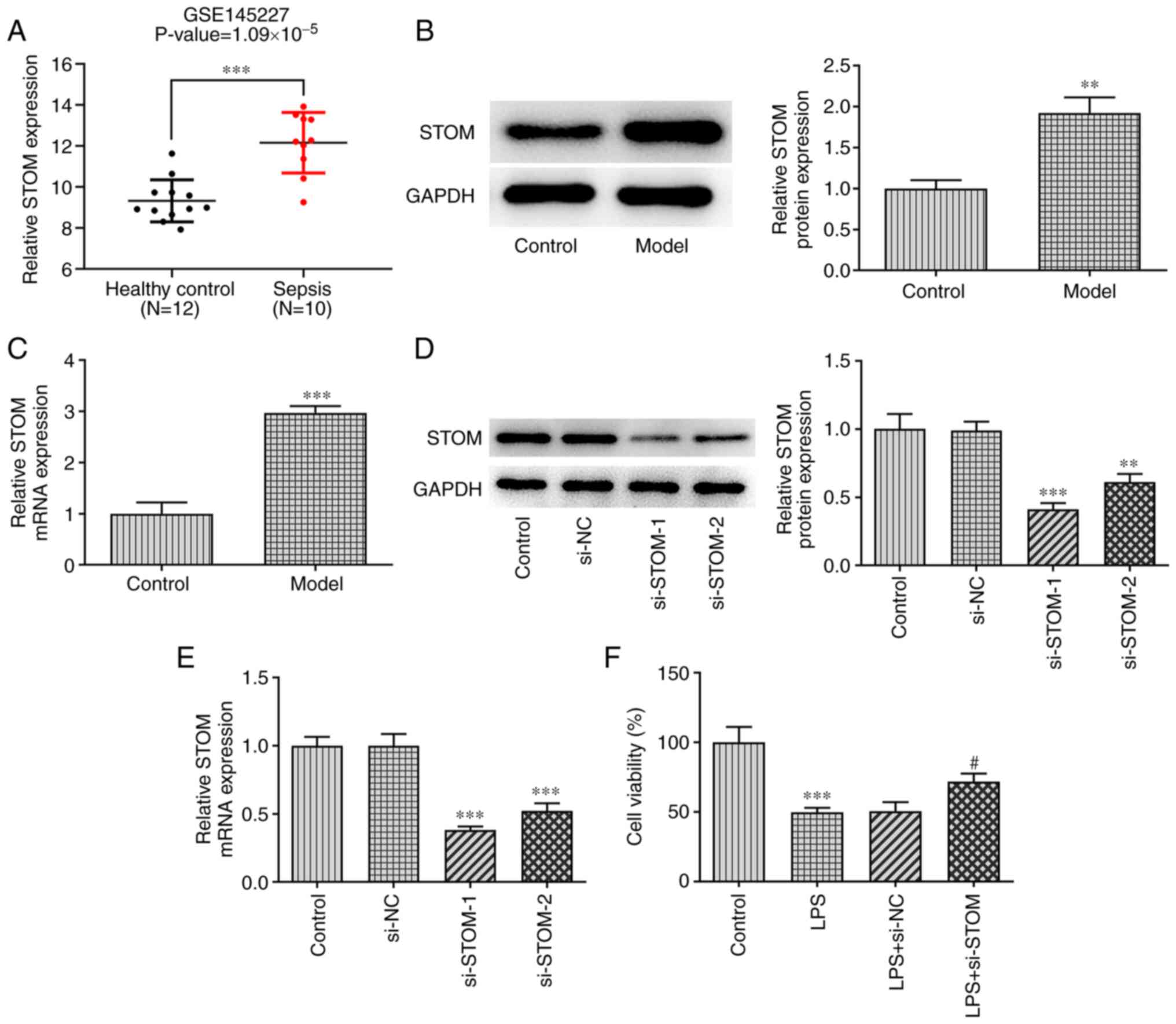
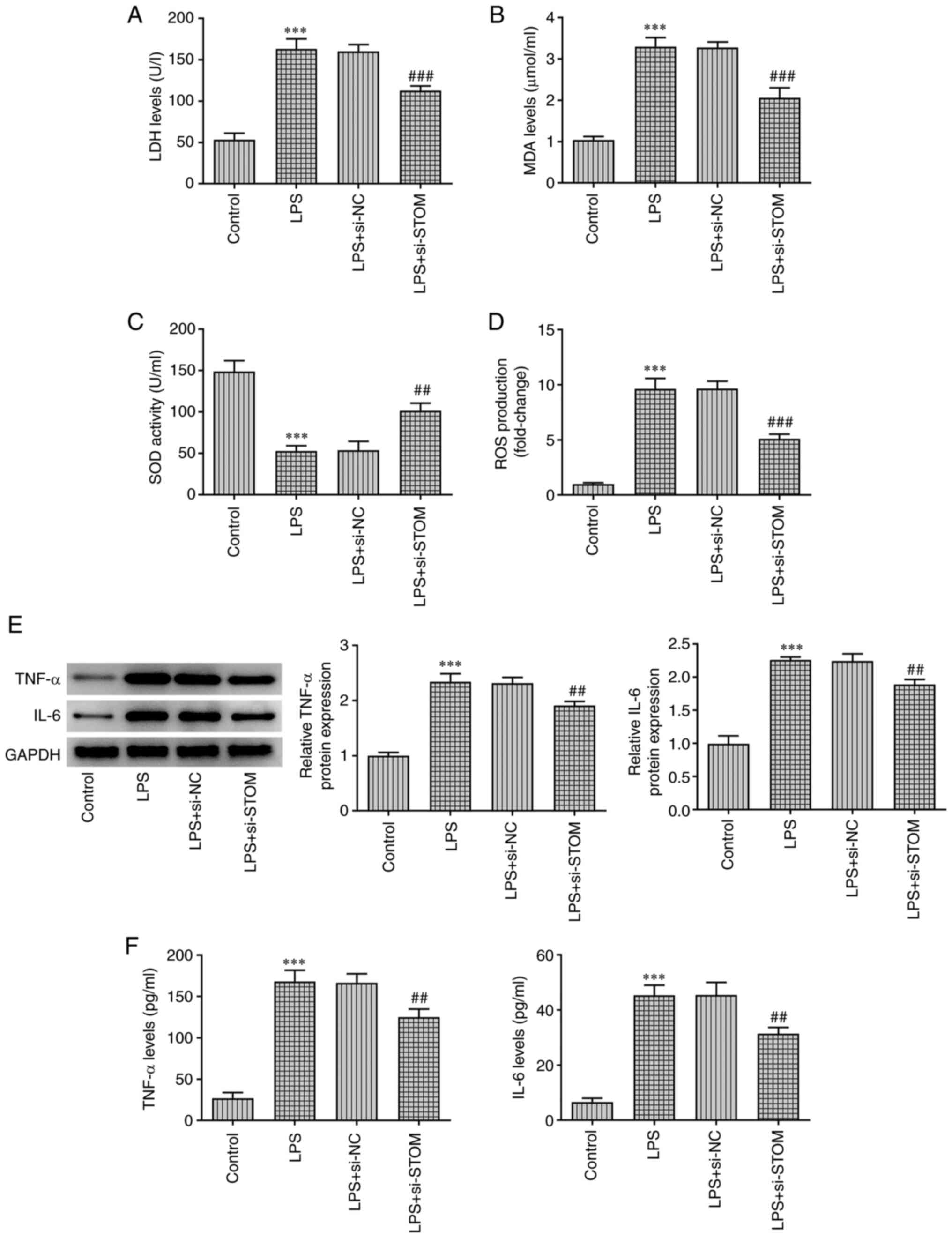
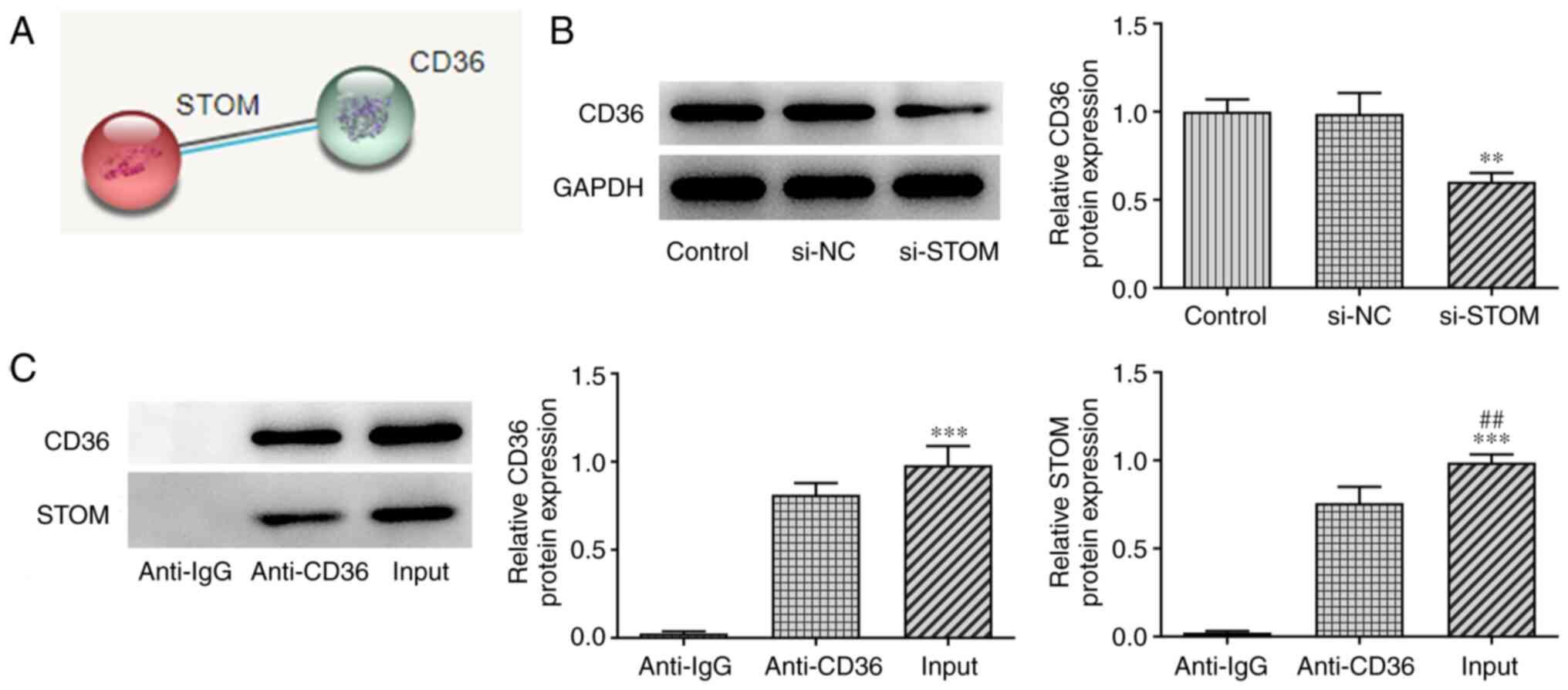
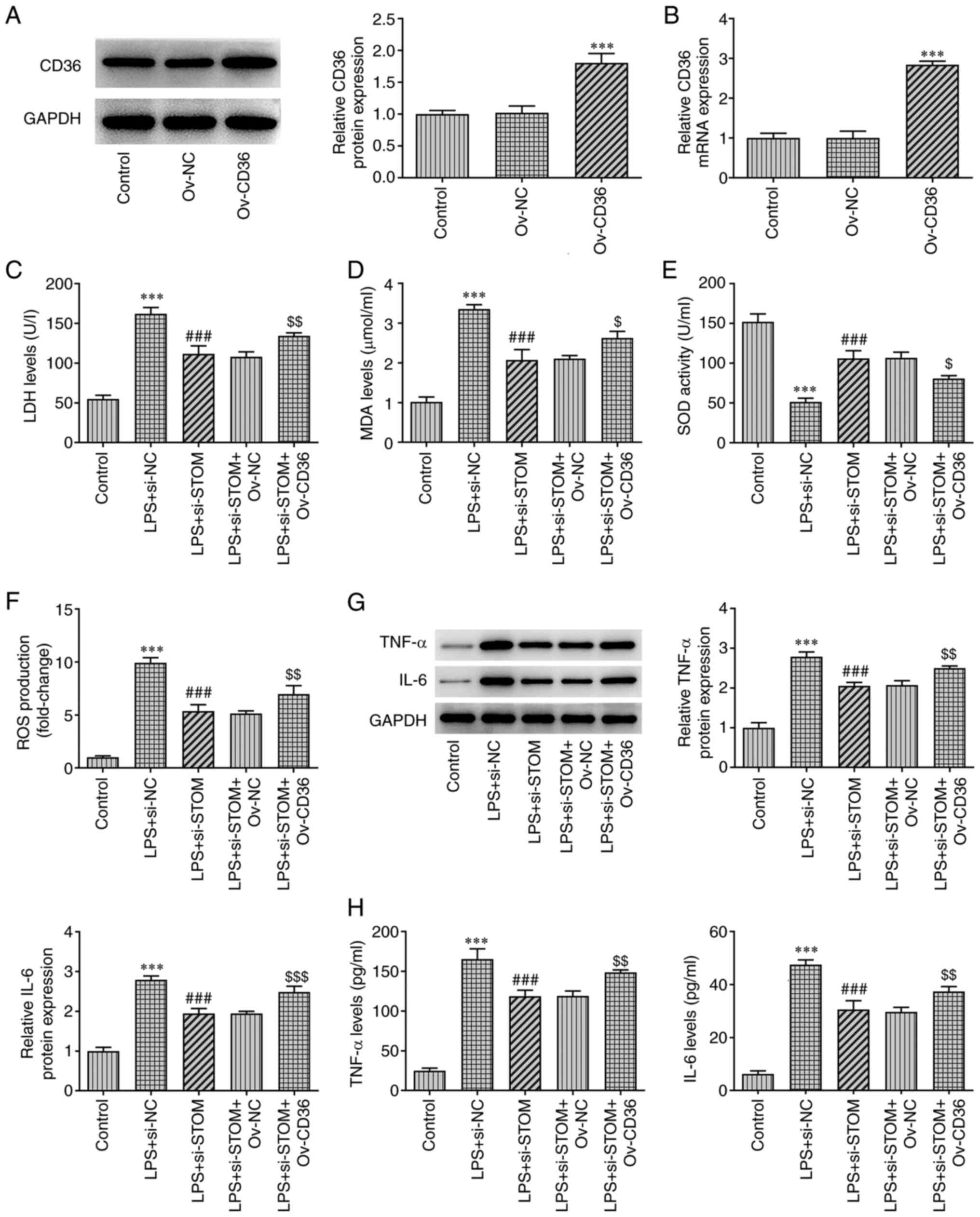
|
1
|
Singer M, Deutschman CS, Seymour CW,
Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche
JD, Coopersmith CM, et al: The third international consensus
definitions for sepsis and septic shock (Sepsis-3). JAMA.
315:801–810. 2016.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Salomao R, Ferreira BL, Salomão MC, Santos
SS, Azevedo LCP and Brunialti MKC: Sepsis: Evolving concepts and
challenges. Braz J Med Biol Res. 52(e8595)2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Fleischmann C, Scherag A, Adhikari NK,
Hartog CS, Tsaganos T, Schlattmann P, Angus DC and Reinhart K:
International Forum of Acute Care Trialists. Assessment of global
incidence and mortality of hospital-treated sepsis. Current
estimates and limitations. Am J Respir Crit Care Med. 193:259–272.
2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Thompson BT, Chambers RC and Liu KD: Acute
respiratory distress syndrome. N Engl J Med. 377:562–572.
2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sawa T: The molecular mechanism of acute
lung injury caused by pseudomonas aeruginosa: From bacterial
pathogenesis to host response. J Intensive Care.
2(10)2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bellani G, Laffey JG, Pham T, Fan E,
Brochard L, Esteban A, Gattinoni L, van Haren F, Larsson A, McAuley
DF, et al: Epidemiology, patterns of care, and mortality for
patients with acute respiratory distress syndrome in intensive care
units in 50 countries. JAMA. 315:788–800. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Stewart GW: Stomatin. Int J Biochem Cell
Biol. 9:271–274. 1997.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Genetet S, Desrames A, Chouali Y, Ripoche
P, Lopez C and Mouro-Chanteloup I: Stomatin modulates the activity
of the anion exchanger 1 (AE1, SLC4A1). Sci Rep.
7(46170)2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Nagarajan A, Dogra SK, Sun L, Gandotra N,
Ho T, Cai G, Cline G, Kumar P, Cowles RA and Wajapeyee N:
Paraoxonase 2 facilitates pancreatic cancer growth and metastasis
by stimulating GLUT1-mediated glucose transport. Mol Cell.
67:685–701.e686. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chen JC, Cai HY, Wang Y, Ma YY, Song LN,
Yin LJ, Cao DM, Diao F, Li YD and Lu J: Up-regulation of stomatin
expression by hypoxia and glucocorticoid stabilizes
membrane-associated actin in alveolar epithelial cells. J Cell Mol
Med. 17:863–872. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wang D, Qi H, Li A, Deng F, Xu Y, Hu Z and
Liu Q: Coexisting overexpression of STOML1 and STOML2 proteins may
be associated with pathology of oral squamous cell carcinoma. Oral
Surg Oral Med Oral Pathol Oral Radiol. 129:591–599.e593.
2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Long G and Yang C: A sixgene support
vector machine classifier contributes to the diagnosis of pediatric
septic shock. Mol Med Rep. 21:1561–1571. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang M, Li C and Shi W: Stomatin-like
protein-2 confers neuroprotection effect in oxygen-glucose
deprivation/reoxygenation-injured neurons by regulating AMPK/Nrf2
signalling. J Drug Target. 28:600–608. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
McGarry T, Biniecka M, Veale DJ and Fearon
U: Hypoxia, oxidative stress and inflammation. Free Radic Biol Med.
125:15–24. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wang J and Li Y: CD36 tango in cancer:
Signaling pathways and functions. Theranostics. 9:4893–4908.
2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Banesh S and Trivedi V: Therapeutic
potentials of scavenger receptor cd36 mediated innate immune
responses against infectious and non-infectious diseases. Curr Drug
Discov Technol. 17:299–317. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Bocharov AV, Wu T, Baranova IN, Birukova
AA, Sviridov D, Vishnyakova TG, Remaley AT, Eggerman TL, Patterson
AP and Birukov KG: Synthetic amphipathic helical peptides targeting
CD36 attenuate lipopolysaccharide-induced inflammation and acute
lung injury. J Immunol. 197:611–619. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Li J and Liu S: LncRNA GAS5 suppresses
inflammatory responses and apoptosis of alveolar epithelial cells
by targeting miR-429/DUSP1. Exp Mol Pathol.
113(104357)2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Bai Z, Li Y, Li Y, Pan J, Wang J and Fang
F: Long noncoding RNA and messenger RNA abnormalities in pediatric
sepsis: A preliminary study. BMC Med Genomics.
13(36)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Shao L, Meng D, Yang F, Song H and Tang D:
Irisin-mediated protective effect on LPS-induced acute lung injury
via suppressing inflammation and apoptosis of alveolar epithelial
cells. Biochem Biophys Res Commun. 487:194–200. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Hu Q, Wang Q, Han C and Yang Y: Sufentanil
attenuates inflammation and oxidative stress in sepsis-induced
acute lung injury by downregulating KNG1 expression. Mol Med Rep.
22:4298–4306. 2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zeng M, Huang C, Zheng H, Chen Q, He W and
Deng Y: Effects of ghrelin on iNOS-derived NO promoted LPS-induced
pulmonary alveolar epithelial A549 cells apoptosis. Cell Physiol
Biochem. 49:1840–1855. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Basu A, Harper S, Pesciotta EN, Speicher
KD, Chakrabarti A and Speicher DW: Proteome analysis of the
triton-insoluble erythrocyte membrane skeleton. J Proteomics.
128:298–305. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Li Y, Li Y, Bai Z, Pan J, Wang J and Fang
F: Identification of potential transcriptomic markers in developing
pediatric sepsis: A weighted gene co-expression network analysis
and a case-control validation study. J Transl Med.
15(254)2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Cao W, Zhang B, Ding F, Zhang W, Sun B and
Liu Z: Expression of SLP-2 was associated with invasion of
esophageal squamous cell carcinoma. PLoS One.
8(e63890)2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Guo XY, Guo HF and Guo HM: Clinical
significance of SLP-2 in epithelial ovarian cancer and its
regulatory effect on the notch signaling pathway. Eur Rev Med
Pharmacol Sci. 24:1666–1671. 2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Yang CT, Li JM, Li LF, Ko YS and Chen JT:
Stomatin-like protein 2 regulates survivin expression in non-small
cell lung cancer cells through β-catenin signaling pathway. Cell
Death Dis. 9(425)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zhou C, Li Y, Wang G, Niu W, Zhang J, Wang
G, Zhao Q and Fan L: Enhanced SLP-2 promotes invasion and
metastasis by regulating Wnt/β-catenin signal pathway in colorectal
cancer and predicts poor prognosis. Pathol Res Pract. 215:57–67.
2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kucuk I, Tanoglu A, Öncü K, Yılmaz I, Kara
M, Beyazıt Y, Akyol T, Kaplan M, Özarı HO and Yazgan Y:
Immunohistochemical activity of prohibitin-2 and stomatin-like
protein-2 in patients with ulcerative colitis. Turk J
Gastroenterol. 27:233–238. 2016.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Pu X, Dong C, Zhu W, Li W and Jiang H:
Silencing stomatin-like protein 2 attenuates tumor progression and
inflammatory response through repressing CD14 in liver cancer. Onco
Targets Ther. 12:7361–7373. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Park YM: CD36, a scavenger receptor
implicated in atherosclerosis. Exp Mol Med. 46(e99)2014.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Pepino MY, Kuda O, Samovski D and Abumrad
NA: Structure-function of CD36 and importance of fatty acid signal
transduction in fat metabolism. Annu Rev Nutr. 34:281–303.
2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Li X, Zhang X, Pang L, Yao L, ShangGuan Z
and Pan Y: Agaricus bisporus-derived β-glucan enter macrophages and
adipocytes by CD36 receptor. Nat Prod Res. 34:3253–3256.
2020.PubMed/NCBI View Article : Google Scholar
|