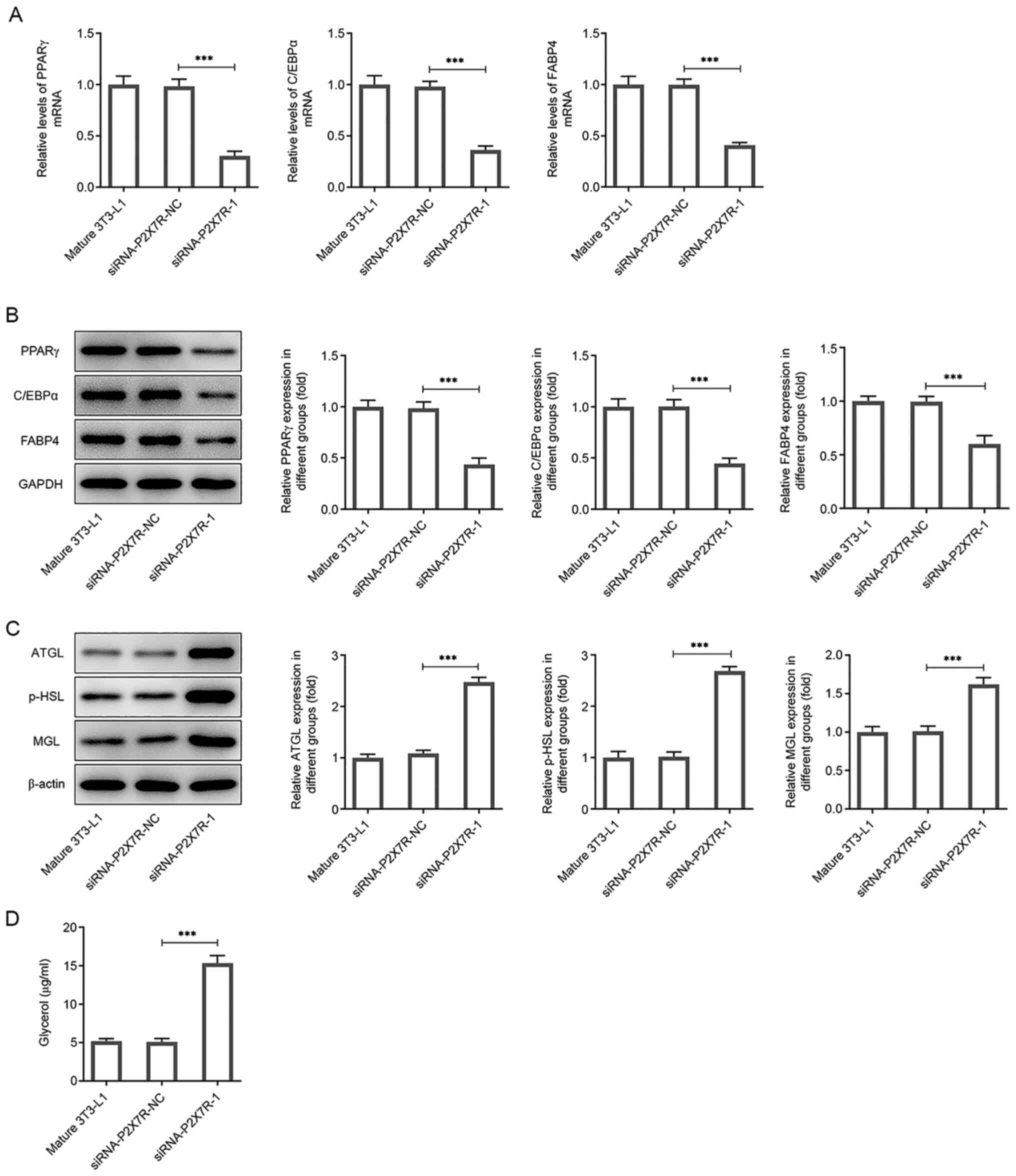
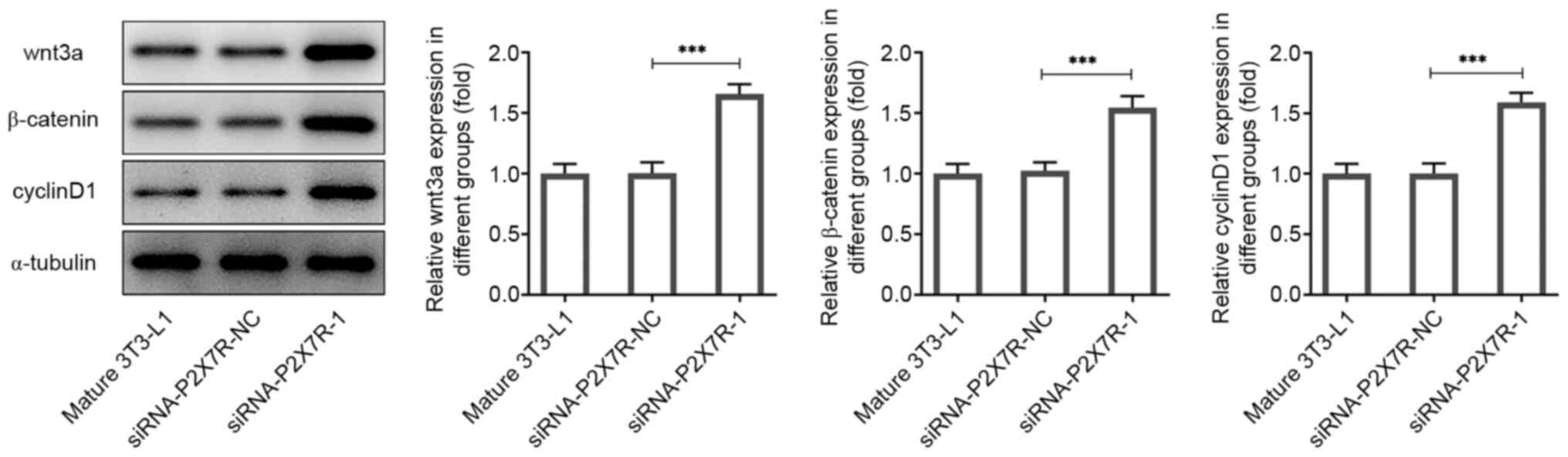
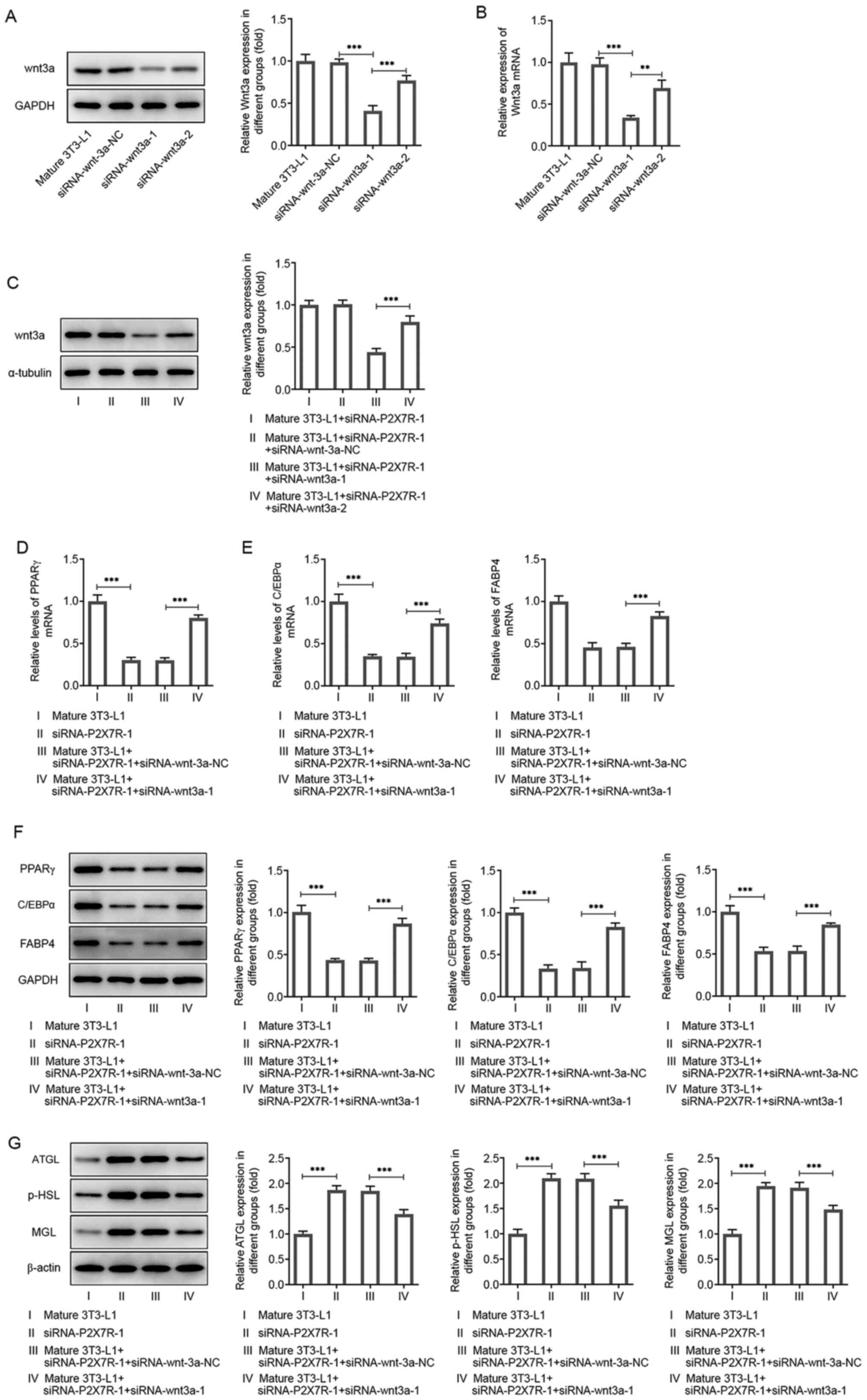
|
1
|
Bou M, Todorcevic M, Rodriguez J, Capilla
E, Gutierrez J and Navarro I: Interplay of adiponectin, TNFα and
insulin on gene expression, glucose uptake and PPARү, AKT and TOR
pathways in rainbow trout cultured adipocytes. Gen Comp Endocrinol.
205:218–225. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Saxton SN, Clark BJ, Withers SB, Eringa EC
and Heagerty AM: Mechanistic links between obesity, diabetes, and
blood pressure: Role of perivascular adipose tissue. Physiol Rev.
99:1701–1763. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kahn CR, Wang G and Lee KY: Altered
adipose tissue and adipocyte function in the pathogenesis of
metabolic syndrome. J Clin Invest. 129:3990–4000. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bleich SN, Vercammen KA, Zatz LY, Frelier
JM, Ebbeling CB and Peeters A: Interventions to prevent global
childhood overweight and obesity: A systematic review. Lancet
Diabetes Endocrinol. 6:332–346. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Bhupathiraju SN and Hu FB: Epidemiology of
obesity and diabetes and their cardiovascular complications. Circ
Res. 118:1723–1735. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Gallagher EJ and LeRoith D: Obesity and
diabetes: The increased risk of cancer and cancer-related
mortality. Physiol Rev. 95:727–748. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tang QQ and Lane MD: Adipogenesis: From
stem cell to adipocyte. Annu Rev Biochem. 81:715–736.
2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Haczeyni F, Bell-Anderson KS and Farrell
GC: Causes and mechanisms of adipocyte enlargement and adipose
expansion. Obes Rev. 19:406–420. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sawamoto A, Nakanishi M, Okuyama S,
Furukawa Y and Nakajima M: Heptamethoxyflavone inhibits
adipogenesis via enhancing PKA signaling. Eur J Pharmacol.
865(172758)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Im DU, Kim SC, Chau GC and Um SH:
Carbamazepine enhances adipogenesis by inhibiting Wnt/β-catenin
expression. Cells. 8(1460)2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Prestwich TC and Macdougald OA:
Wnt/beta-catenin signaling in adipogenesis and metabolism. Curr
Opin Cell Biol. 19:612–617. 2007.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chen X, Ayala I, Shannon C, Fourcaudot M,
Acharya NK, Jenkinson CP, Heikkinen S and Norton L: The diabetes
gene and Wnt pathway effector TCF7L2 regulates adipocyte
development and function. Diabetes. 67:554–568. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Guo Y, Mishra A, Weng T, Chintagari NR,
Wang Y, Zhao C, Huang C and Liu L: Wnt3a mitigates acute lung
injury by reducing P2X7 receptor-mediated alveolar epithelial type
I cell death. Cell Death Dis. 5(e1286)2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sindhavajiva PR, Sastravaha P,
Arksornnukit M and Pavasant P: Purinergic 2X7 receptor activation
regulates WNT signaling in human mandibular-derived osteoblasts.
Arch Oral Biol. 81:167–174. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Gnoni A, Siculella L, Paglialonga G,
Damiano F and Giudetti AM: 3,5-diiodo-L-thyronine increases de novo
lipogenesis in liver from hypothyroid rats by SREBP-1 and
ChREBP-mediated transcriptional mechanisms. IUBMB Life. 71:863–872.
2019.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Vadrot N, Duband-Goulet I, Cabet E,
Attanda W, Barateau A, Vicart P, Gerbal F, Briand N, Vigouroux C,
Oldenburg AR, et al: The p. R482W substitution in A-type lamins
deregulates SREBP1 activity in Dunnigan-type familial partial
lipodystrophy. Hum Mol Genet. 24:2096–2109. 2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Hou XX, Dong HR, Sun LJ, Yang M, Cheng H
and Chen YP: Purinergic 2X7 receptor is involved in the podocyte
damage of obesity-related glomerulopathy via activating
nucleotide-binding and oligomerization domain-like receptor protein
3 inflammasome. Chin Med J (Engl). 131:2713–2725. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
North RA: Molecular physiology of P2X
receptors. Physiol Rev. 82:1013–1067. 2002.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Su QQ, Tian YY, Liu ZN, Ci LL and Lv XW:
Purinergic P2X7 receptor blockade mitigates alcohol-induced
steatohepatitis and intestinal injury by regulating MEK1/2-ERK1/2
signaling and egr-1 activity. Int Immunopharmacol. 66:52–61.
2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Beaucage KL, Xiao A, Pollmann SI, Grol MW,
Beach RJ, Holdsworth DW, Sims SM, Darling MR and Dixon SJ: Loss of
P2X7 nucleotide receptor function leads to abnormal fat
distribution in mice. Purinergic Signal. 10:291–304.
2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Gao H, Li D, Yang P, Zhao L, Wei L, Chen Y
and Ruan XZ: Suppression of CD36 attenuates adipogenesis with a
reduction of P2X7 expression in 3T3-L1 cells. Biochem Biophys Res
Commun. 491:204–208. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Han Y, Lee SH, Lee IS and Lee KY:
Regulatory effects of 4-methoxychalcone on adipocyte
differentiation through PPARγ activation and reverse effect on
TNF-α in 3T3-L1 cells. Food Chem Toxicol. 106:17–24.
2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Lee CW, Seo JY, Lee J, Choi JW, Cho S, Bae
JY, Sohng JK, Kim SO, Kim J and Park YI: 3-O-Glucosylation of
quercetin enhances inhibitory effects on the adipocyte
differentiation and lipogenesis. Biomed Pharmacother. 95:589–598.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ma J, Liao H, Lin Y, Huang K, Zhu J and
Wang Y: Molecular characterization, expression analysis of Chemerin
gene and its potential role in intramuscular adipocyte
differentiation of goat. Anim Biotechnol. 31:382–390.
2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Rosen ED and MacDougald OA: Adipocyte
differentiation from the inside out. Nat Rev Mol Cell Biol.
7:885–896. 2006.PubMed/NCBI View
Article : Google Scholar
|
|
27
|
Moseti D, Regassa A and Kim WK: Molecular
regulation of adipogenesis and potential anti-adipogenic bioactive
molecules. Int J Mol Sci. 17(124)2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Zhang L, Zhang L, Wang X and Si H:
Anti-adipogenic effects and mechanisms of ginsenoside Rg3 in
pre-adipocytes and obese mice. Front Pharmacol.
8(113)2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Jin H, Lee K, Chei S, Oh HJ, Lee KP and
Lee BY: Ecklonia stolonifera extract suppresses lipid accumulation
by promoting lipolysis and adipose browning in high-fat
diet-induced obese male mice. Cells. 9(871)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Di Virgilio F, Dal Ben D, Sarti AC,
Giuliani AL and Falzoni S: The P2X7 receptor in infection and
inflammation. Immunity. 47:15–31. 2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Coccurello R and Volonte C: P2X7 Receptor
in the management of energy homeostasis: Implications for obesity,
dyslipidemia, and insulin resistance. Front Endocrinol (Lausanne).
11(199)2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Calderon-Dominguez M, Mir JF, Fucho R,
Weber M, Serra D and Herrero L: Fatty acid metabolism and the basis
of brown adipose tissue function. Adipocyte. 5:98–118.
2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Moon MH, Jeong JK, Lee YJ, Seol JW, Ahn
DC, Kim IS and Park SY: 18β-Glycyrrhetinic acid inhibits adipogenic
differentiation and stimulates lipolysis. Biochem Biophys Res
Commun. 420:805–810. 2012.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Seo YJ, Jin H, Lee K, Song JH, Chei S, Oh
HJ, Oh JH and Lee BY: Cardamonin suppresses lipogenesis by
activating protein kinase A-mediated browning of 3T3-L1 cells.
Phytomedicine. 65(153064)2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Lee JE, Schmidt H, Lai B and Ge K:
Transcriptional and epigenomic regulation of adipogenesis. Mol Cell
Biol. 39:e00601–e00618. 2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Walther TC and Farese RV Jr: Lipid
droplets and cellular lipid metabolism. Annu Rev Biochem.
81:687–714. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Suh HJ, Cho SY, Kim EY and Choi HS:
Blockade of lipid accumulation by silibinin in adipocytes and
zebrafish. Chem Biol Interact. 227:53–62. 2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Clevers H: Wnt/beta-catenin signaling in
development and disease. Cell. 127:469–480. 2006.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Xi FX, Wei CS, Xu YT, Ma L, He YL, Shi XE,
Yang GS and Yu TY: MicroRNA-214-3p targeting Ctnnb1 promotes 3T3-L1
preadipocyte differentiation by interfering with the Wnt/β-catenin
signaling pathway. Int J Mol Sci. 20(1816)2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Kang S, Bennett CN, Gerin I, Rapp LA,
Hankenson KD and Macdougald OA: Wnt signaling stimulates
osteoblastogenesis of mesenchymal precursors by suppressing
CCAAT/enhancer-binding protein alpha and peroxisome
proliferator-activated receptor gamma. J Biol Chem.
282:14515–14524. 2007.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Liu J and Farmer SR: Regulating the
balance between peroxisome proliferator-activated receptor gamma
and beta-catenin signaling during adipogenesis. A glycogen synthase
kinase 3beta phosphorylation-defective mutant of beta-catenin
inhibits expression of a subset of adipogenic genes. J Biol Chem.
279:45020–45027. 2004.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Liu J, Wang H, Zuo Y and Farmer SR:
Functional interaction between peroxisome proliferator-activated
receptor gamma and beta-catenin. Mol Cell Biol. 26:5827–5837.
2006.PubMed/NCBI View Article : Google Scholar
|