|
1
|
Meschia JF, Klaas JP, Brown RD Jr and
Brott TG: Evaluation and management of atherosclerotic carotid
stenosis. Mayo Clin Proc. 92:1144–1157. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sarikas A, Carrier L, Schenke C, Doll D,
Flavigny J, Lindenberg KS, Eschenhagen T and Zolk O: Impairment of
the ubiquitin-proteasome system by truncated cardiac myosin binding
protein C mutants. Cardiovasc Res. 66:33–44. 2005.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Cheng SF, Brown MM, Simister RJ and
Richards T: Contemporary prevalence of carotid stenosis in patients
presenting with ischaemic stroke. Br J Surg. 106:872–878.
2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
De Reuck JL: Pathophysiology of carotid
artery disease and related clinical syndromes. Acta Chir Belg.
104:30–34. 2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sherman DG: The carotid artery and stroke.
Am Fam Phys. 40 (5 Suppl):41S–44S, 7S-9S. 1989.PubMed/NCBI
|
|
6
|
Fanelli F, Boatta E, Cannavale A, Corona
M, Lucatelli P, Wlderk A, Cirelli C and Salvatori FM: Carotid
artery stenting: Analysis of a 12-year single-center experience. J
Endovasc Ther. 19:749–756. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
AbuRahma AF, Abu-Halimah S, Hass SM,
Nanjundappa A, Stone PA, Mousa A, Lough E and Dean LS: Carotid
artery stenting outcomes are equivalent to carotid endarterectomy
outcomes for patients with post-carotid endarterectomy stenosis. J
Vasc Surg. 52:1180–1187. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Dangas GD, Claessen BE, Caixeta A, Sanidas
EA, Mintz GS and Mehran R: In-stent restenosis in the drug-eluting
stent era. J Am Coll Cardiol. 56:1897–1907. 2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zhang H, Ren KF, Chang H, Wang JL and Ji
J: Surface-mediated transfection of a pDNA vector encoding short
hairpin RNA to downregulate TGF-β1 expression for the prevention of
in-stent restenosis. Biomaterials. 116:95–105. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bagyura Z, Kiss L, Berta B, Szilágyi Á,
Hirschberg K, Széplaki G, Lux Á, Szelid Z, Soós P and Merkely B:
High rate of in-stent restenosis after coronary intervention in
carriers of the mutant mannose-binding lectin allele. BMC
Cardiovasc Disord. 17(4)2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Santulli G: microRNAs distinctively
regulate vascular smooth muscle and endothelial cells: Functional
implications in angiogenesis, atherosclerosis, and in-stent
restenosis. Adv Exp Med Biol. 887:53–77. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yuan Y, Liu X, Hao S, He Q and Shen Z:
Plasma levels of miR-143 and miR-145 are associated with coronary
in-stent restenosis within 1 year of follow-up after drug-eluting
stent implantation. Ann Transl Med. 8(756)2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
He M, Gong Y, Shi J, Pan Z, Zou H, Sun D,
Tu X, Tan X, Li J, Li W, et al: Plasma microRNAs as potential
noninvasive biomarkers for in-stent restenosis. PLoS One.
9(e112043)2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
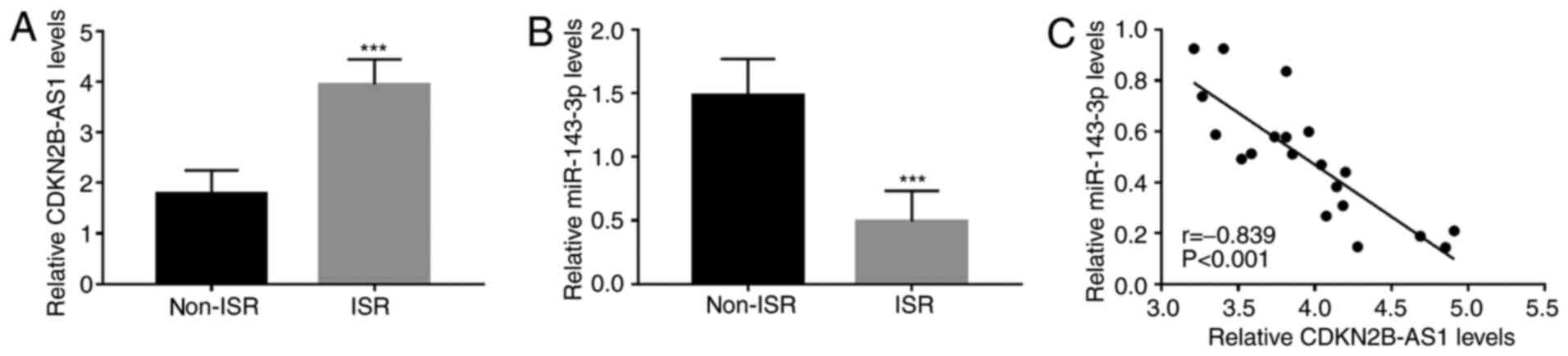
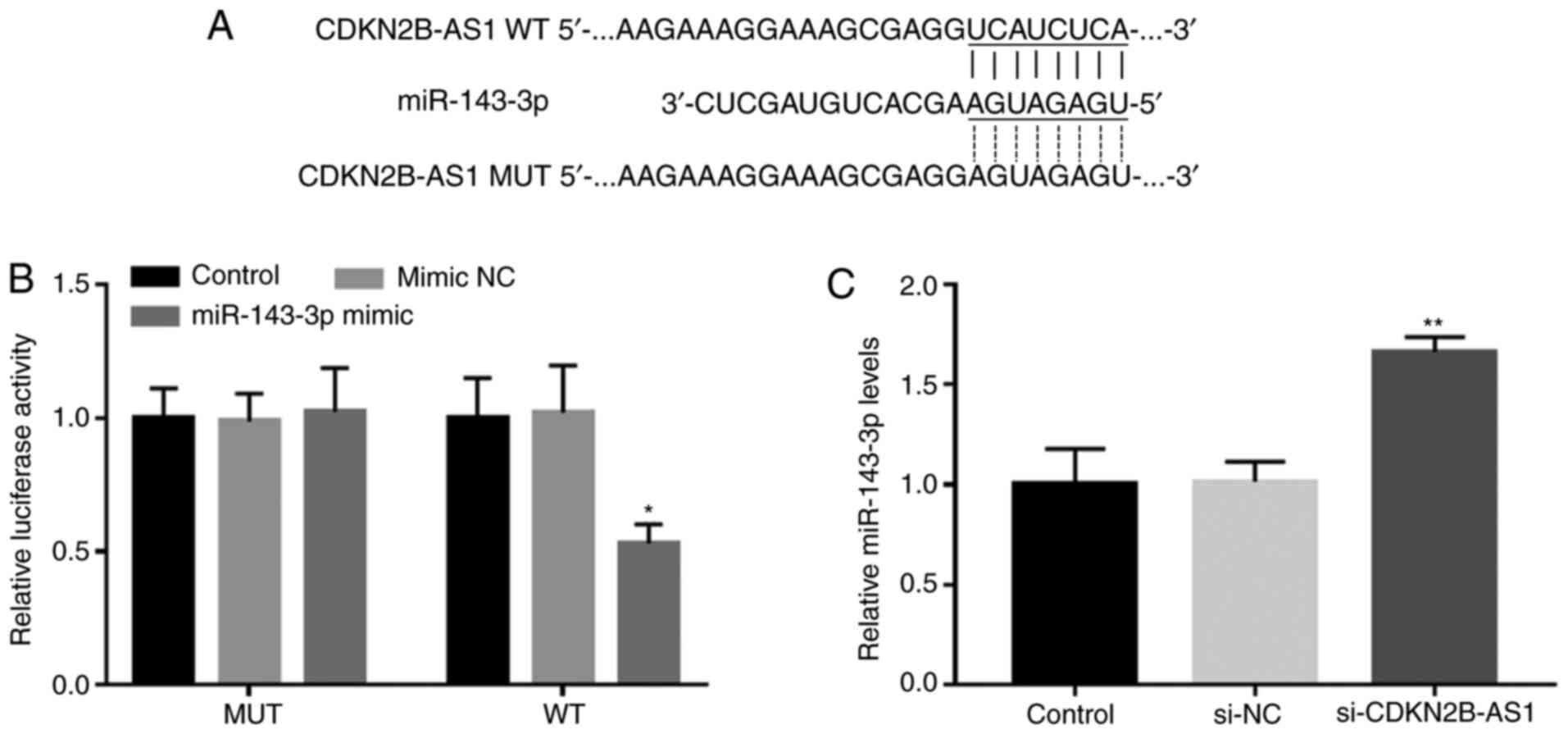
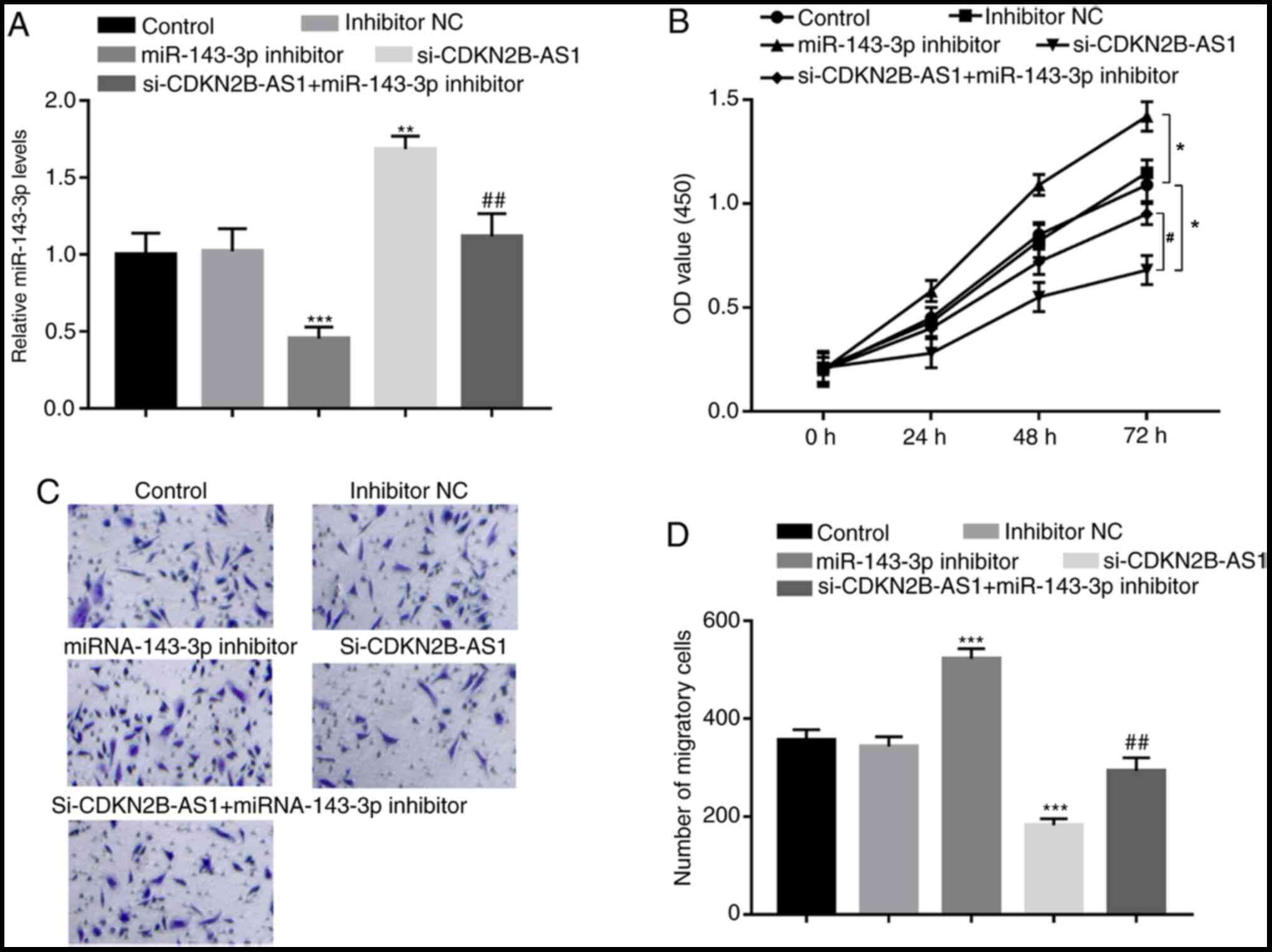
Xu C, Zhai J and Fu Y: LncRNA CDKN2B-AS1
promotes the progression of ovarian cancer by miR-143-3p/SMAD3 axis
and predicts a poor prognosis. Neoplasma. 67:782–793.
2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Li H, Han S, Sun Q, Yao Y, Li S, Yuan C,
Zhang B, Jing B, Wu J, Song Y and Wang H: Long non-coding RNA
CDKN2B-AS1 reduces inflammatory response and promotes cholesterol
efflux in atherosclerosis by inhibiting ADAM10 expression. Aging
(Albany NY). 11:1695–1715. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ou M, Li X, Zhao S, Cui S and Tu J: Long
non-coding RNA CDKN2B-AS1 contributes to atherosclerotic plaque
formation by forming RNA-DNA triplex in the CDKN2B promoter.
EBioMedicine. 55(102694)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Huang Y, Xiang B, Liu Y, Wang Y and Kan H:
LncRNA CDKN2B-AS1 promotes tumor growth and metastasis of human
hepatocellular carcinoma by targeting let-7c-5p/NAP1L1 axis. Cancer
Lett. 437:56–66. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zhuang H, Cao G, Kou C and Li D:
Overexpressed lncRNA CDKN2B-AS1 is an independent prognostic factor
for liver cancer and promotes its proliferation. J BUON.
24:1441–1448. 2019.PubMed/NCBI
|
|
19
|
Gasecki AP, Hachinski VC, Mendel T and
Barnett HT: Endarterectomy for symptomatic carotid stenosis. Review
of the European and North American symptomatic carotid surgery
trials. Nebr Med J. 77:121–123. 1992.PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Chen KP, Wang JJ, Wang LJ, Lu J, Qi P, Hu
S, Yang XM, Wang HF and Wang DM: Study of correlation between
carotid artery tortuosity and atherosclerotic carotid artery
stenosis. Zhonghua Wai Ke Za Zhi. 55:608–612. 2017.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
22
|
Puz P, Lasek-Bal A, Urbanek T and
Kazibutowska Z: Assessment of cerebral embolism and vascular
reserve parameters in patients with carotid artery stenosis. Neurol
Neurochir. 50:356–362. 2016.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Wang T, Mei B and Zhang J: Atherosclerotic
carotid stenosis and cognitive function. Clin Neurol Neurosurg.
146:64–70. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Jiang F, Zhang X, Lu YM, Li YG, Zhou X and
Wang YS: Elevated level of miR-17 along with decreased levels of
TIMP-1 and IL-6 in plasma associated with the risk of in-stent
restenosis. Biosci Trends. 13:423–429. 2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
O'Sullivan JF, Neylon A, Fahy EF, Yang P,
McGorrian C and Blake GJ: MiR-93-5p is a novel predictor of
coronary in-stent restenosis. Heart Asia.
11(e011134)2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wang F, Su X, Liu C, Wu M and Li B:
Prognostic value of plasma long noncoding RNA ANRIL for in-stent
restenosis. Med Sci Monit. 23:4733–4739. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zhu L, Zhang Q, Li S, Jiang S, Cui J and
Dang G: Interference of the long noncoding RNA CDKN2B-AS1
upregulates miR-181a-5p/TGFβI axis to restrain the metastasis and
promote apoptosis and senescence of cervical cancer cells. Cancer
Med. 8:1721–1730. 2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Frismantiene A, Philippova M, Erne P and
Resink TJ: Smooth muscle cell-driven vascular diseases and
molecular mechanisms of VSMC plasticity. Cell Signal. 52:48–64.
2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
He Y, Zou P, Lu Y, Jia D, Li X, Yang H,
Tang L, Zhu Z, Tu T, Tai S, et al: Osteoprotegerin promotes intimal
hyperplasia and contributes to in-stent restenosis: Role of an
αVβ3/FAK dependent YAP pathway. J Mol Cell Cardiol. 139:1–13.
2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Shi L, Tian C, Sun L, Cao F and Meng Z:
The lncRNA TUG1/miR-145-5p/FGF10 regulates proliferation and
migration in VSMCs of hypertension. Biochem Biophys Res Commun.
501:688–695. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Tang R, Mei X, Wang YC, Cui XB, Zhang G,
Li W and Chen SY: LncRNA GAS5 regulates vascular smooth muscle cell
cycle arrest and apoptosis via p53 pathway. Biochim Biophys Acta
Mol Basis Dis. 1865:2516–2525. 2019.PubMed/NCBI View Article : Google Scholar
|