Introduction
Gastric cancer is the fifth most common cancer
worldwide (1). Surgical treatment
is the first choice for gastric cancer; however, the 5-year
survival rate is only 20-30% due to low early diagnosis rates and
high postoperative recurrence and metastasis rates (2).
Lidocaine is a local anesthetic which can
effectively inhibit the biological activity of a variety of cancer
types. In vivo and in vitro studies have indicated
the antitumor effects of lidocaine in gastric cancer (3-5).
Mechanistically, lidocaine has been found to decrease Src
activation (6-8).
A recent study suggested that lidocaine can inhibit the
proliferative and invasive capabilities of c-Met positive MKN45
cells (3). c-Met is a member of the
protein tyrosine kinase receptor superfamily, which is encoded by
the mesenchymal-epithelial transition (MET) proto-oncogene and
mainly produced by epithelial cells (9). c-Met expression in gastric cancer
tissues is higher compared with that in healthy tissues and is
associated with invasion, metastasis and poor prognosis of gastric
cancer, but is not associated with sex, age, size, location or
differentiation degree of the tumor (10-12).
MET amplification has been found to be common feature in gastric
cancer and its inhibition contributes to apoptosis of gastric
cancer cells (13-16).
c-Src, a non-receptor tyrosine kinase, is closely associated with
the proliferation and survival of cancer cells (17,18).
Hepatocyte growth factor (HGF) functions as a natural endogenous
ligand of the MET receptor, which forms a signaling pathway with
c-Met, which is closely related to the occurrence, development,
metastasis and prognosis of gastric cancer (9,19,20).
In most types of gastric cancer, the inhibition of this signaling
pathway exerts an antiproliferative effect and induces apoptosis in
gastric cancer cells (13).
c-Met/c-Src signaling has been reported to play a vital role on the
growth of MET-activated gastric cancer cells (13). Although the potential anti-tumor
effects of lidocaine have been reported, the role and mechanism of
action of lidocaine remain unclear. The present study aimed to
investigate the efficacy of lidocaine against the malignant
behavior and proliferation of gastric cancer cells and its
mechanism of action.
Materials and methods
Cell culture
Human gastric carcinoma cells MKN45 (ATCC), a
c-MET-positive cell line, were cultured in RPMI-1640 medium (Thermo
Fisher Scientific, Inc.) containing 10% FBS at 37˚C after
resuscitation until adherent. Once the cell density reached 80%,
cells were digested with 0.25% trypsin and passaged at a 1:3 ratio
of cells: Medium. MKN45 cells were continuously cultured using the
same conditions for subsequent experiments.
Cell Counting Kit-8 (CCK-8) assay
MKN45 cells were cultured to the logarithmic stage
and seeded into 96-well plates (2x103 cells/well). After
overnight culture, the culture solution was discarded. Media
containing lidocaine (Selleck Chemicals) at final concentrations of
1, 5 and 10 mM was added to the cells for incubation for 48 h at
37˚C. Each lidocaine concentration treatment was performed as five
separate assays. In analyzing the influence of lidocaine on the
sensitivities of cells for cisplatin (cat. no. A10221; Adooq
Bioscience) or 5-FU (cat. no. CSN19496, CSNpharm, Inc.), cells were
cultured in RPMI-1640 medium containing cisplatin (0.25 or 0. 5
µg/ml) or in combination with lidocaine (10 mM) or HGF (40 ng/ml,
cat. no. AP3513; Adooq Bioscience). Following culture for 48 h, 10
µl CCK-8 solution was added for incubation for 2 h at 37˚C (Abcam).
The absorbance at a wavelength of 450 nm was detected using a
microplate reader.
Western blotting
Total protein was extracted from MKN45 cells using
RIPA lysis buffer (Beijing Solarbio Science & Technology Co.,
Ltd.). Protein concentration was determined using a BCA kit. A
total of 30 µg protein was obtained, mixed with loading buffer and
loaded into 10% polyacrylamide gels. Polyacrylamide gel
electrophoresis was performed to separate the proteins. The
proteins were then transferred to PVDF membranes and 5% skimmed
milk was used to block the membranes at room temperature for 1 h.
Subsequently, the membranes were incubated with primary antibodies
[c-Met, cat. no. ab216574, 1:1,000; phosphorylated (p)-c-Met, cat.
no. ab5662, 1:1,000; c-Src, cat. no. ab16885, 1:1,000; p-c-Src,
cat. no. ab40660, 1:2,000; N-cadherin, cat. no. ab76011, 1:5,000;
vimentin, cat. no. ab92547, 1:2,000; GAPDH, ab8245, 1:10,000; all
from Abcam] at 4˚C overnight and secondary antibodies [horseradish
peroxidase (HRP)-conjugated rabbit anti-mouse, cat. no. ab47827,
1:10,000; HRP-conjugated goat anti-Rabbit IgG, ab97040, 1:10,000;
Abcam] at room temperature for 1 h. Protein bands were visualized
using a gel imaging system (Bio-Rad Laboratories, Inc.) following
addition of ECL developing reagent (Beyotime Institute of
Biotechnology). The gray value of protein bands was analyzed using
Image J software 1.46r (National Institutes of Health). The
grayscale ratio of target proteins to GADPH was then
calculated.
Wound healing assay
MKN45 cells were seeded into 6-well plates
(2x106 cells/well) and cultured for 24 h. When cell
confluence reached 100%, linear scratches were made using a 200-µl
sterile pipette tip and photographed as the controls. After being
washed with PBS, the cells were treated with serum-free medium
containing lidocaine (10 mM) or HGF (40 ng/ml) at 37˚C for 48 h.
Images were then captured using a light microscope (magnification,
x200; Olympus Corporation). The migration rate was calculated using
the formula: Migration rate=(T0 h area-T48 h area)/T0 h area
x100%.
Transwell assay
The invasive capabilities of MKN45 cells were
detected using Transwell assays. Matrigel (50 mg/l) was added to
the upper chamber of the Transwell at 4˚C to dry for 5 h (0.4 µM
pore size; BD Biosciences). Once the cells were digested and
collected, the cell suspension (5x104 cells/200 µl) was
prepared using RPMI-1640 medium containing no FBS and seeded into
the Transwell upper chamber. Medium containing 10% FBS was added
into the lower chamber. After 48 h at 37˚C, the chamber was removed
and cells in the upper chamber were wiped off using a cotton swab.
The cells were fixed with 4% paraformaldehyde for 15 min at room
temperature and then stained with 0.1% crystal violet for 30 min at
room temperature (Sigma Aldrich; Merck KGaA) and observed under a
light microscope (magnification x200).
TUNEL assay
Following treatment of MKN45 cells, cells
(1x106 cells) were collected added to polylysine slides
and fixed using 4% paraformaldehyde for 25 min at room temperature.
Subsequently, 0.2 % Triton X-100 was added to the sections and
incubated for 5 min at room temperature. Apoptotic cells were
stained with TUNEL reagent (50 µl) for 1 h at room temperature and
DAPI for 10 min (0.4 µg/ml, blue) in the dark, according to the
instructions of the TUNEL Fluorescence Assay kit (Roche
Diagnostics). Apoptosis was observed under a fluorescence
microscope (Roche Diagnostics). In total, five non-overlapping
fields were randomly selected. The apoptosis levels of the cells
were calculated using the following method: (The number of positive
cells in each field/the total number of all cells in the field)
x100%.
Statistical analysis
Data are presented as the mean ± SD. Experimental
data among the various groups were compared using one-way ANOVAs
followed by post hoc Tukey's tests using GraphPad 7.0 (GraphPad
Software, Inc.). Each experiment was repeated at least three
times.
Results
Effects of lidocaine on the
c-Met/c-Src axis in MKN45 cells
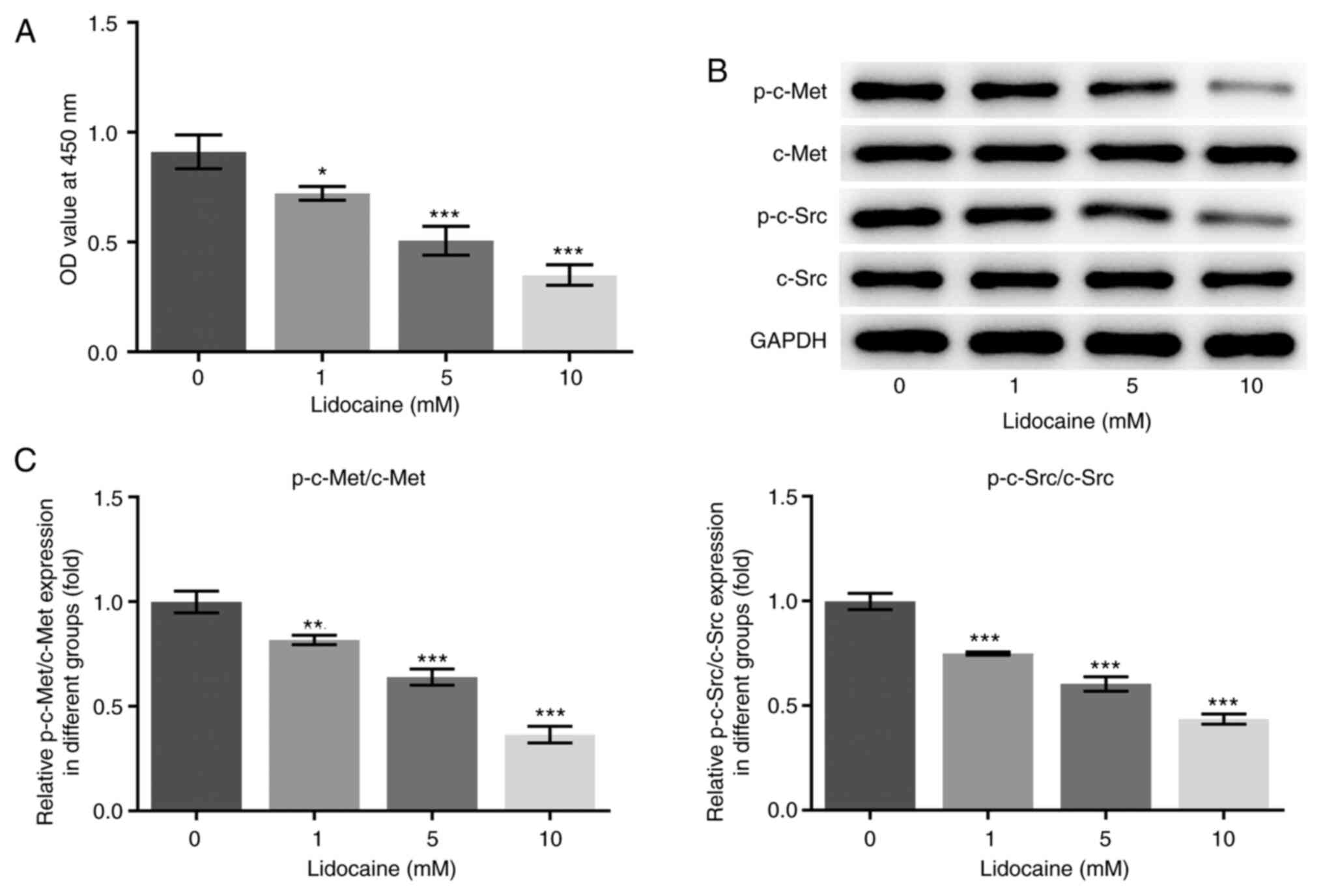
To evaluate the effects of lidocaine on cell growth,
MKN45 cells were exposed to various concentrations of lidocaine for
48 h and the cell viability was measured. Various concentrations of
lidocaine were found to inhibit MKN45 cell proliferation in a
dose-dependent manner (Fig. 1A),
indicating that MKN45 cells are sensitive to lidocaine. To further
investigate the mechanism of action of lidocaine, western blot
assays were performed to analyze the protein expression and
phosphorylation levels of c-Met and c-Src. Although there were no
marked changes in the expression levels of c-Met and c-Src in MKN45
cells upon treatment with various concentrations of lidocaine, a
significant decrease was observed in the phosphorylation levels of
c-Met and c-Src (Fig. 1B and
C).
HGF suppresses the efficacy of
lidocaine in MKN45 cells via the c-Met/c-Src axis
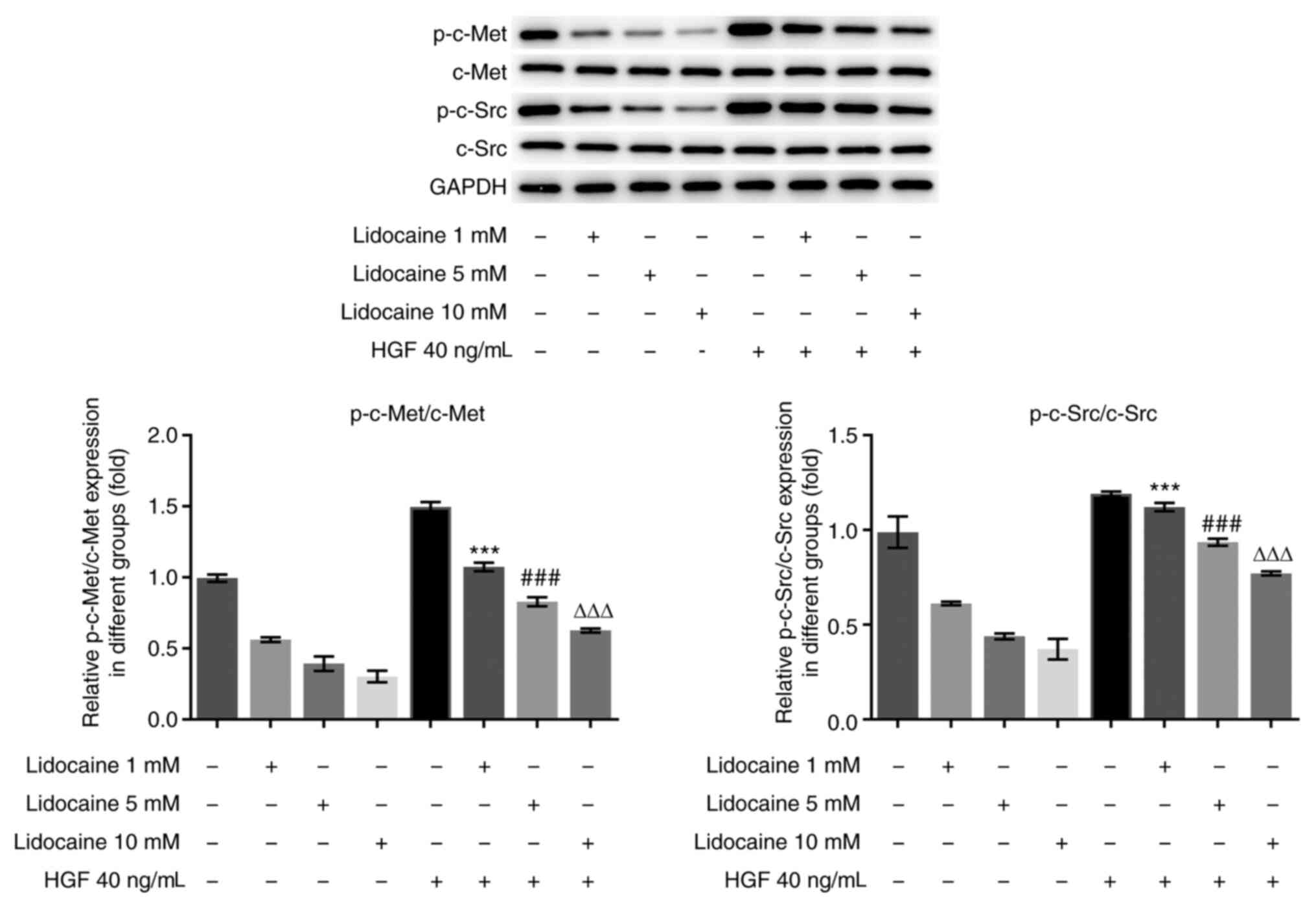
The aforementioned findings indicated that there was
an inhibitory effect of lidocaine on c-Met and c-Src. It has been
reported that c-Met/c-Src activation is closely related to the
overall survival rate of patients with gastric cancer and is
considered as a potential therapeutic target for gastric cancer
(20-24).
As such, it was hypothesized that the c-Met/c-Src pathway may
mediate the suppressive effects of lidocaine on MKN45 cell
proliferation. To validate this hypothesis, MKN45 cells were
pre-treated with HGF (40 ng/ml), followed by treatment with various
concentrations of lidocaine (1, 5 and 10 mM) for 48 h (Fig. 2). HGF is a known activator of c-MET
(25). A marked increase in the
phosphorylation levels of c-Met and c-Src was observed in MKN45
cells treated with the various concentrations of lidocaine in the
presence of HGF, when compared with lidocaine treatment alone. In
addition, no marked changes in the expression of c-Met and c-Src
were observed in MKN45 cells following lidocaine or HGF
treatment.
Lidocaine suppresses the malignant
behavior of MKN45 cells
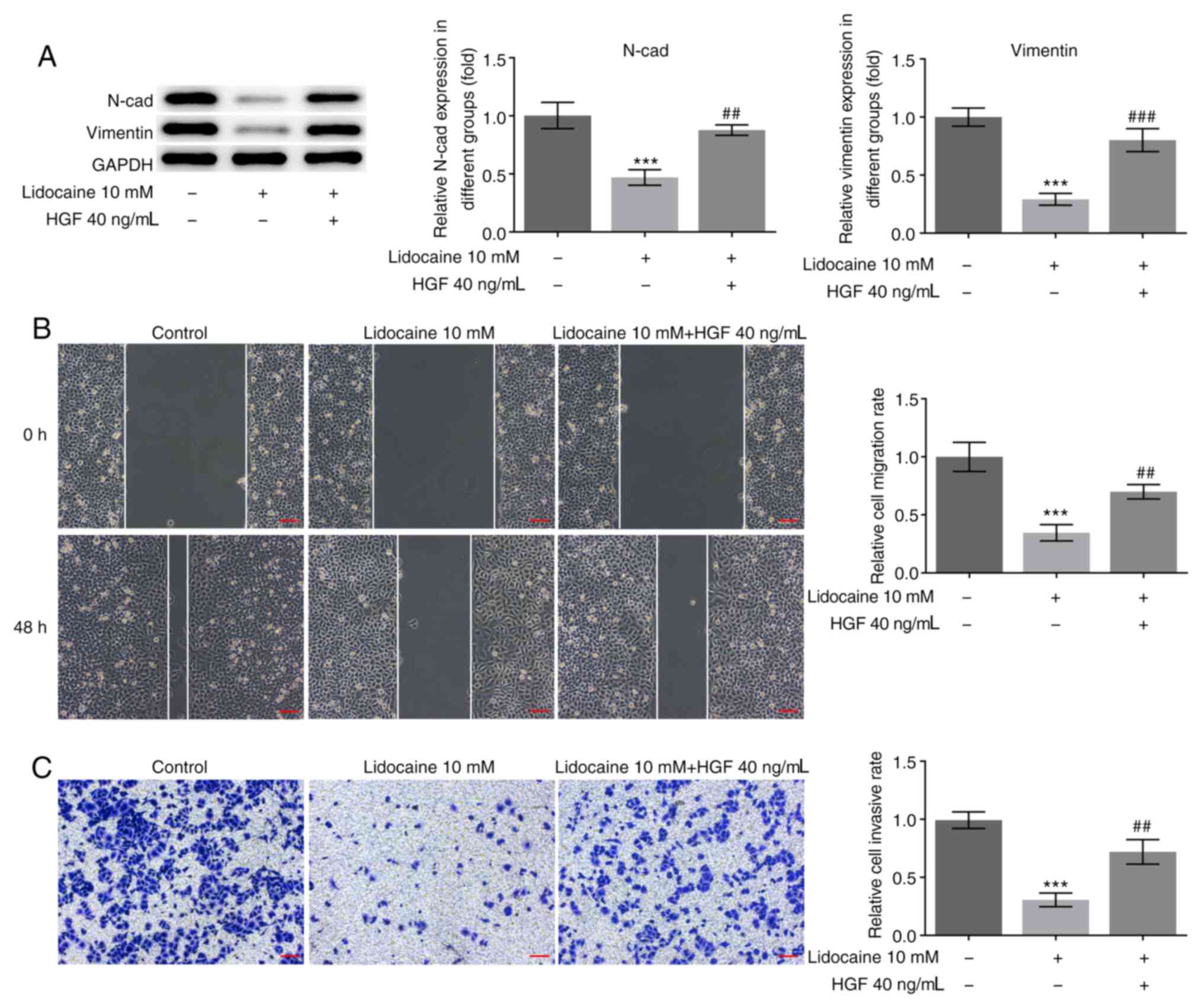
Subsequently, the present study further evaluated
the influence of lidocaine on the malignant behavior of MKN45 cells
through analyzing the abilities of cell migration and invasion, as
well as the expression of EMT-related markers. Western blot assays
were performed to analyze the expression of N-cadherin and
vimentin, while migration and invasion were investigated using
wound healing and Transwell assays. A significant reduction in
N-cadherin and vimentin protein expression levels was found in
MKN45 cells exposed to lidocaine, while HGF reversed these effects
(Fig. 3A). Metastasis is one of the
factors that impede successful treatment of patients with gastric
cancer. Therefore, in vitro migration and invasion assays
were subsequently performed in MKN45 cells following lidocaine or
HGF treatment. MKN45 cells exposed to 10 mM lidocaine exhibited
significantly higher levels of both migration and invasion, while
HGF treatment reversed these effects (Fig. 3B and C).
Lidocaine inhibits the apoptosis of
MKN45 cells
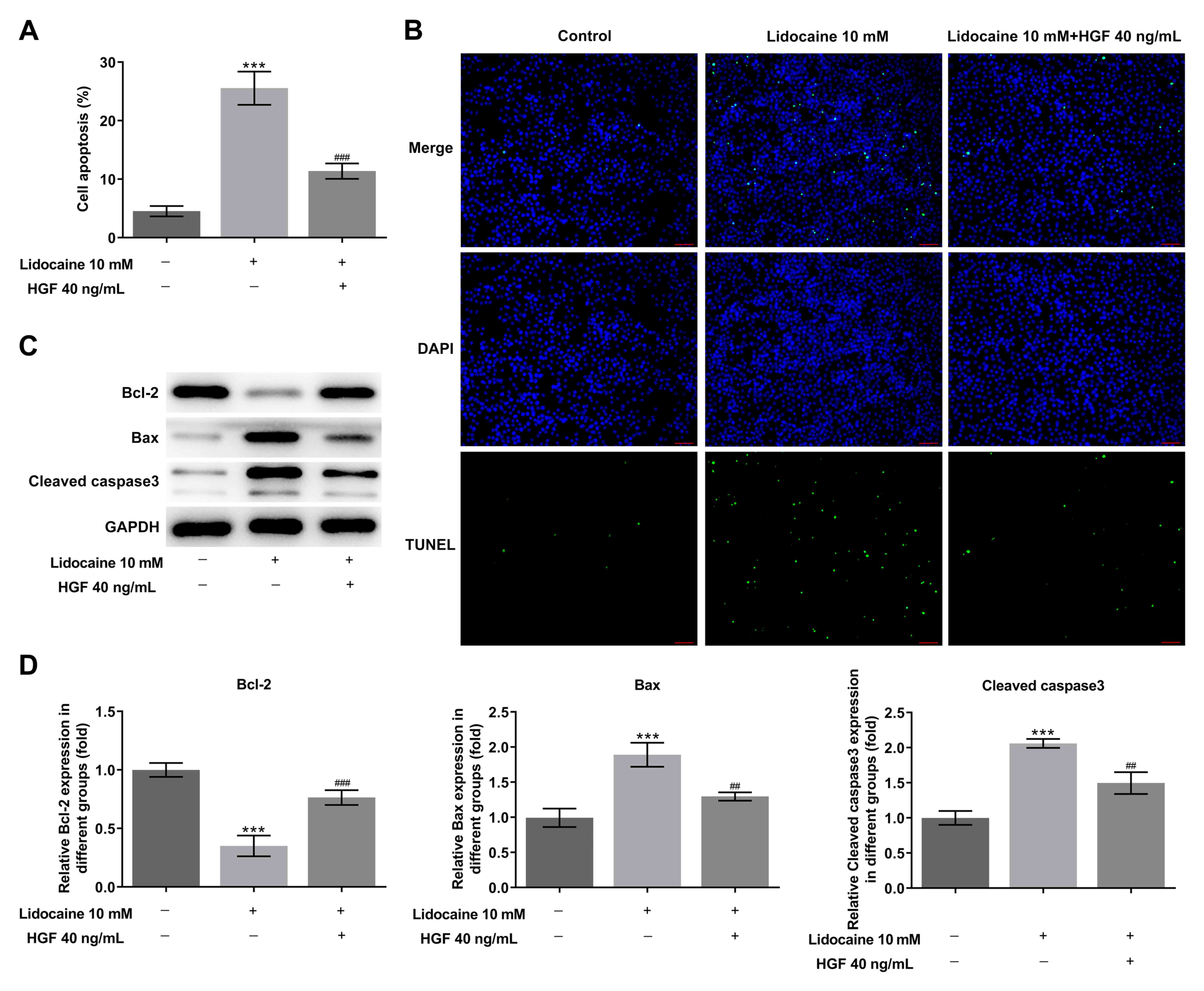
Fluorescein-dUTP was used to analyze cell apoptosis
using a TUNEL assay. Following treatment of MKN45 cells with
lidocaine (10 mM), a marked increase in the number of cells showing
green fluorescence was observed, indicating an increase in the
extent of cell apoptosis compared with the control group. HGF (40
ng/ml) treatment significantly reduced the promoting effects of
lidocaine on cell apoptosis (Fig.
4A and B). The expression of
apoptosis-related proteins (Bcl-2, Bax and cleaved caspase 3) in
MKN45 cells was detected using western blot assays. Significant
changes in all of the aforementioned protein expression levels in
cells exposed to lidocaine were found compared with the control
group (Fig. 4C and D). The expression levels of the
pro-apoptotic protein Bcl-2 were significantly decreased, whilst
Bax and cleaved caspase3 expression was significantly increased.
The results indicated that lidocaine induced cell apoptosis by
potentially activating the intrinsic c-Met/c-Src pathway.
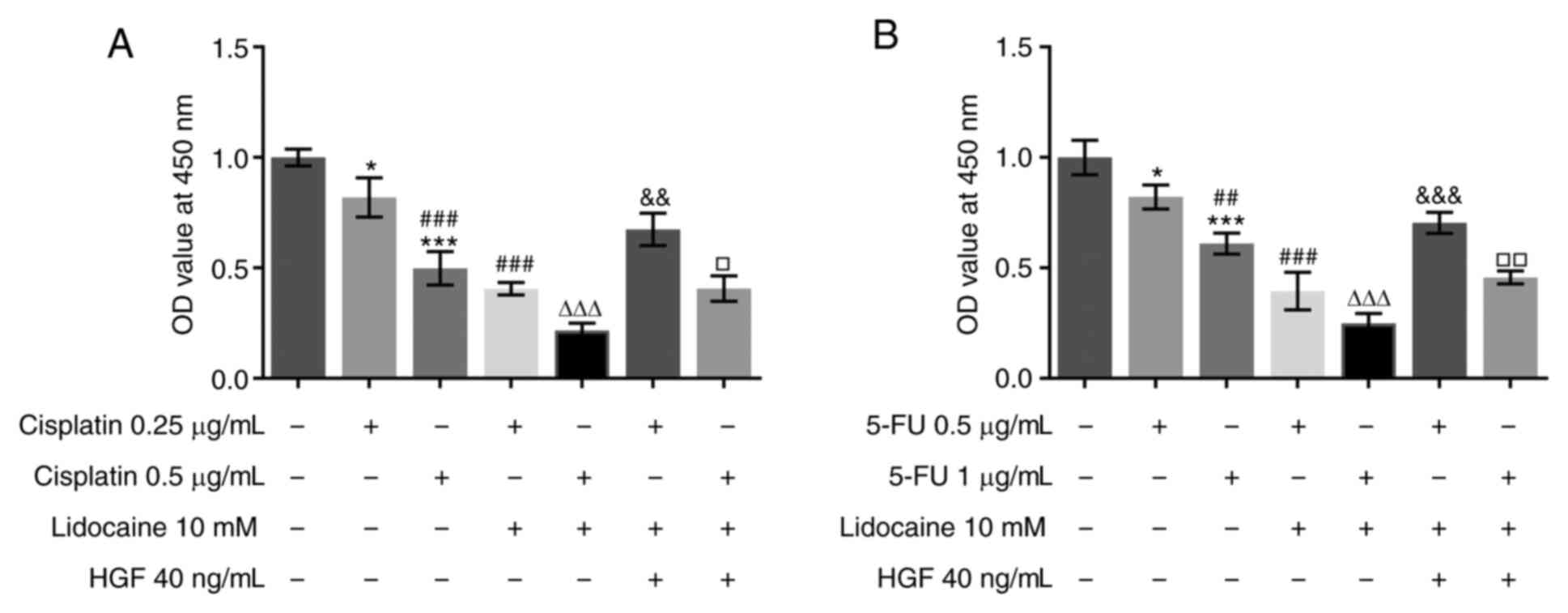
Lidocaine enhances the sensitivity of
cells to chemotherapy
The resistance of tumor cells to chemotherapy drugs
limits the efficacy of drugs and is a major obstacle to effective
cancer chemotherapy (26). The
sensitivity of MKN45 cells exposed to lidocaine in combination with
cisplatin to chemotherapy was assessed using a CCK-8 assay.
Cisplatin inhibited cell viability in a dose-dependent manner, the
effects of which were significantly enhanced when combined with
lidocaine treatment (Fig. 5A). The
synergistic inhibitory effects of lidocaine were also observed in
MKN45 cells treated with 5-FU (Fig.
5B). Moreover, the promoting effects of HGF on cell viability
were observed in cells treated with lidocaine, cisplatin when
compared with co-treatment of cisplatin and lidocaine. Similarly,
the effects of the combination treatment of 5-FU and lidocaine on
cell viability were reversed by HGF.
Discussion
Lidocaine is a local anesthetic, with strong and
lasting effects and good surface penetration (27). Intravenous lidocaine injections may
effectively relieve pain and reduce fentanyl consumption during the
early postoperative period (28).
Recent studies have demonstrated a possible regulatory mechanism of
action for lidocaine through suppressing the proliferation of
gastric cancer cells, which is associated with ERK1/2
phosphorylation, the MAPK pathway and altering the expression
profiles of microRNAs (3-5).
EMT activation is involved in the cell invasion and metastasis of a
variety of cancer types (29,30).
In the present study, lidocaine downregulated the expression levels
of the EMT markers, N-cadherin and vimentin, the effects of which
were inhibited by HGF treatment. The HGF/c-Met pathway has
previously been found to induce EMT in gastric cancer cells
(31). Based on these experimental
data, lidocaine may suppress the EMT process by inhibiting the
activation of the c-Met/c-Src signaling pathway.
The proliferative and anti-apoptotic functions of
HGF have been previously confirmed in gastric cancer cells,
consistent with the results of the present study (32). Abnormal HGF levels and apoptosis
dysregulation are closely associated with the pathogenesis of
gastric cancer (33). Detection of
growth factors and apoptosis-related proteins has revealed an
increased HGF expression and dysregulated Bax/Bcl-2 in patients
with gastric cancer (34). The
present study found that lidocaine decreased Bcl-2 levels and
increased the levels of Bax and cleaved caspase3 through the
c-Met/c-Src pathway, and also revealed that lidocaine may promote
mitochondrial mediated apoptosis pathway to induce cell apoptosis.
Similar roles for lidocaine in apoptosis have also been reported a
number of malignancies, including lymphoma, colorectal cancer and
cervical cancer (35-37).
The present study demonstrated that promoting apoptosis through
lidocaine treatment resulted in the inhibition of cell
proliferation, indicating the potential anti-tumor effects of
lidocaine on gastric cancer cells.
HGF was shown to suppress the effects of lidocaine
in MKN45 cells. HGF is cytotoxic and serves as a ligand for c-Met
(38). HGF/c-Met signaling plays a
vital role in normal biological functions as well as cancer
pathology, where it has been implicated in tumor metastasis
(23,39). Inhibition of this pathway may
sensitize tumor cells to chemotherapy (24). It has previously been shown that
c-MET-mediates the malignant behaviors in NT2D1 non-seminoma cells,
as well as being implicated in the recruitment of c-Src, which has
been associated with the aggressiveness of some types of cancer
(40). In the present study,
lidocaine alone markedly suppressed cell proliferation, while its
combination with cisplatin resulted in the synergistic suppression
of cell proliferation. Furthermore, HGF treatment reversed these
effects, indicating that the inhibition of c-Met/c-Src with
lidocaine enhanced the anti-proliferative effects of cisplatin.
Taken together, the findings of the present study
provided evidence supporting a potential role of lidocaine against
the malignant behavior of gastric cancer cells and provided a novel
insight into the mechanisms of action of lidocaine in gastric
cancer.
Acknowledgements
No applicable.
Funding
The present study was supported by the Pharmaceutical Research
Fund of Guangdong Province Hospital (approval no. 2020XC25;
Zhongshan, China).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author and the first
author on reasonable request.
Authors' contributions
WZ, ZTX, MYT, YWW and CYZ conceived and designed the
study, performed the experiment, collected, analyzed and
interpreted the data, and revised the manuscript. WZ and CYZ wrote
the manuscript. All authors read and approved the final manuscript.
WZ and CYZ confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
No applicable.
Patient consent for publication
No applicable.
Competing interests
All authors declare that they have no competing
interests.
References
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386.
2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Casamayor M, Morlock R, Maeda H and Ajani
J: . Targeted literature review of the global burden of gastric
cancer. Ecancermedicalscience. 12(883)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Sui H, Lou A, Li Z and Yang J: . Lidocaine
inhibits growth, migration and invasion of gastric carcinoma cells
by up-regulation of miR-145. BMC Cancer. 19(233)2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Yang W, Cai J, Zhang H, Wang G and Jiang
W: . Effects of lidocaine and ropivacaine on gastric cancer cells
through down-regulation of ERK1/2 phosphorylation in vitro.
Anticancer Res. 38:6729–6735. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ye L, Zhang Y, Chen YJ and Liu Q:
Anti-tumor effects of lidocaine on human gastric cancer cells in
vitro. Bratisl Lek Listy. 120:212–217. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Piegeler T, Votta-Velis EG, Bakhshi FR,
Mao M, Carnegie G, Bonini MG, Schwartz DE, Borgeat A, Beck-Schimmer
B and Minshall RD: Endothelial barrier protection by local
anesthetics: Ropivacaine and lidocaine block tumor necrosis
factor-α-induced endothelial cell src activation. Anesthesiology.
120:1414–1428. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Piegeler T, Votta-Velis EG, Liu G, Place
AT, Schwartz DE, Beck-Schimmer B, Minshall RD and Borgeat A:
Antimetastatic potential of amide-linked local anesthetics:
Inhibition of lung adenocarcinoma cell migration and inflammatory
src signaling independent of sodium channel blockade.
Anesthesiology. 117:548–559. 2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Wall TP, Crowley PD, Sherwin A, Foley AG
and Buggy DJ: Effects of lidocaine and src inhibition on metastasis
in a murine model of breast cancer surgery. Cancers (Basel).
11(1414)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Matsumoto K, Umitsu M, De Silva DM, Roy A
and Bottaro DP: Hepatocyte growth factor/MET in cancer progression
and biomarker discovery. Cancer Sci. 108:296–307. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Han SU, Lee HY, Lee JH, Kim WH, Nam H, Kim
H, Cho YK, Kim MW and Lee KU: Modulation of E-cadherin by
hepatocyte growth factor induces aggressiveness of gastric
carcinoma. Ann Surg. 242:676–683. 2005.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Lee KH, Choi EY, Hyun MS, Jang BI, Kim TN,
Kim SW, Song SK, Kim JH and Kim JR: Hepatocyte growth factor/c-met
signaling in regulating urokinase plasminogen activator in human
stomach cancer: A potential therapeutic target for human stomach
cancer. Korean J Intern Med. 21:20–27. 2006.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Park WS, Oh RR, Kim YS, Park JY, Shin MS,
Lee HK, Lee SH, Yoo NJ and Lee JY: Absence of mutations in the
kinase domain of the met gene and frequent expression of met and
HGF/SF protein in primary gastric carcinomas. APMIS. 108:195–200.
2000.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Okamoto W, Okamoto I, Yoshida T, Okamoto
K, Takezawa K, Hatashita E, Yamada Y, Kuwata K, Arao T, Yanagihara
K, et al: Identification of c-src as a potential therapeutic target
for gastric cancer and of MET activation as a cause of resistance
to c-src inhibition. Mol Cancer Ther. 9:1188–1197. 2010.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kuniyasu H, Yasui W, Kitadai Y, Yokozaki
H, Ito H and Tahara E: Frequent amplification of the c-met gene in
scirrhous type stomach cancer. Biochem Biophys Res Commun.
189:227–232. 1992.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Nessling M, Solinas-Toldo S, Wilgenbus KK,
Borchard F and Lichter P: Mapping of chromosomal imbalances in
gastric adenocarcinoma revealed amplified protooncogenes MYCN, MET,
WNT2, and ERBB2. Genes Chromosomes Cancer. 23:307–316.
1998.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sakakura C, Mori T, Sakabe T, Ariyama Y,
Shinomiya T, Date K, Hagiwara A, Yamaguchi T, Takahashi T, Nakamura
Y, et al: Gains, losses, and amplifications of genomic materials in
primary gastric cancers analyzed by comparative genomic
hybridization. Genes Chromosomes Cancer. 24:299–305.
1999.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Irby RB and Yeatman TJ: Role of src
expression and activation in human cancer. Oncogene. 19:5636–5642.
2000.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kim LC, Song L and Haura EB: Src kinases
as therapeutic targets for cancer. Nat Rev Clin Oncol. 6:587–595.
2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Anestis A, Zoi I and Karamouzis MV:
Current advances of targeting HGF/c-Met pathway in gastric cancer.
Ann Transl Med. 6(247)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Moosavi F, Giovannetti E, Saso L and
Firuzi O: HGF/MET pathway aberrations as diagnostic, prognostic,
and predictive biomarkers in human cancers. Crit Rev Clin Lab Sci.
56:533–566. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Summy JM and Gallick GE: Src family
kinases in tumor progression and metastasis. Cancer Metastasis Rev.
22:337–358. 2003.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yan H, Sun Y, Wu Q, Wu Z, Hu M, Sun Y, Liu
Y, Ma Z, Liu S, Xiao W, et al: PELP1 suppression inhibits gastric
cancer through downregulation of c-Src-PI3K-ERK pathway. Front
Oncol. 9(1423)2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Kim HJ, Kang SK, Kwon WS, Kim TS, Jeong I,
Jeung HC, Kragh M, Horak ID, Chung HC and Rha SY: Forty-nine
gastric cancer cell lines with integrative genomic profiling for
development of c-MET inhibitor. Int J Cancer. 143:151–159.
2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Huang KH, Sung IC, Fang WL, Chi CW, Yeh
TS, Lee HC, Yin PH, Li AF, Wu CW, Shyr YM and Yang MH: Correlation
between HGF/c-Met and notch1 signaling pathways in human gastric
cancer cells. Oncol Rep. 40:294–302. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Oh HA, Lee G, Kang HJ, Kim YG, Bae SH, Lee
JL, Lee KH, Hyun MS and Kim DS: Overexpression of c-met protein in
gastric cancer and role of uPAR as a therapeutic target. Cancer Res
Treat. 35:9–15. 2003.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chen C, Tang X, Liu Y, Zhu J and Liu J:
Induction/reversal of drug resistance in gastric cancer by
non-coding RNAs (Review). Int J Oncol. 54:1511–1524.
2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kiriyama S, Oda I, Nishimoto F, Mashimo Y,
Ikehara H and Gotoda T: Pilot study to assess the safety of local
lidocaine injections during endoscopic submucosal dissection for
early gastric cancer. Gastric Cancer. 12:142–147. 2009.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kim TH, Kang H, Choi YS, Park JM, Chi KC,
Shin HY and Hong JH: Pre- and intraoperative lidocaine injection
for preemptive analgesics in laparoscopic gastrectomy: A
prospective, randomized, double-blind, placebo-controlled study. J
Laparoendosc Adv Surg Tech A. 23:663–668. 2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Yao L, Zhang D, Zhao X, Sun B, Liu Y, Gu
Q, Zhang Y, Zhao X, Che N, Zheng Y, et al: Dickkopf-1-Promoted
vasculogenic mimicry in non-small cell lung cancer is associated
with EMT and development of a cancer stem-like cell phenotype. J
Cell Mol Med. 20:1673–1685. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Acloque H, Adams MS, Fishwick K,
Bronner-Fraser M and Nieto MA: Epithelial-mesenchymal transitions:
The importance of changing cell state in development and disease. J
Clin Invest. 119:1438–1449. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
31
|
Toiyama Y, Yasuda H, Saigusa S, Matushita
K, Fujikawa H, Tanaka K, Mohri Y, Inoue Y, Goel A and Kusunoki M:
Co-Expression of hepatocyte growth factor and c-met predicts
peritoneal dissemination established by autocrine hepatocyte growth
factor/c-met signaling in gastric cancer. Int J Cancer.
130:2912–2921. 2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Lee JC, Koh SA, Lee KH and Kim JR: BAG3
contributes to HGF-mediated cell proliferation, migration, and
invasion via the egr1 pathway in gastric cancer. Tumori. 105:63–75.
2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Koh SA and Lee KH: HGF-Mediated S100A11
overexpression enhances proliferation and invasion of gastric
cancer. Am J Transl Res. 10:3385–3394. 2018.PubMed/NCBI
|
|
34
|
Konturek PC, Konturek SJ, Sulekova Z,
Meixner H, Bielanski W, Starzynska T, Karczewska E, Marlicz K,
Stachura J and Hahn EG: Expression of hepatocyte growth factor,
transforming growth factor alpha, apoptosis related proteins bax
and bcl-2, and gastrin in human gastric cancer. Aliment Pharmacol
Ther. 15:989–999. 2001.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kamiya Y, Ohta K and Kaneko Y:
Lidocaine-induced apoptosis and necrosis in U937 cells depending on
its dosage. Biomed Res. 26:231–239. 2005.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Qu X, Yang L, Shi Q, Wang X, Wang D and Wu
G: Lidocaine inhibits proliferation and induces apoptosis in
colorectal cancer cells by upregulating mir-520a-3p and targeting
EGFR. Pathol Res Pract. 214:1974–1979. 2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Zhu J and Han S: Lidocaine inhibits
cervical cancer cell proliferation and induces cell apoptosis by
modulating the lncRNA-MEG3/miR-421/BTG1 pathway. Am J Transl Res.
11:5404–5416. 2019.PubMed/NCBI
|
|
38
|
Arnold L, Enders J and Thomas SM:
Activated HGF-c-met axis in head and neck cancer. Cancers (Basel).
9(169)2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Rucki AA, Xiao Q, Muth S, Chen J, Che X,
Kleponis J, Sharma R, Anders RA, Jaffee EM and Zheng L: Dual
inhibition of hedgehog and c-met pathways for pancreatic cancer
treatment. Mol Cancer Ther. 16:2399–2409. 2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Leonetti E, Gesualdi L, Scheri KC,
Dinicola S, Fattore L, Masiello MG, Cucina A, Mancini R, Bizzarri
M, Ricci G and Catizone A: C-Src recruitment is involved in
c-MET-mediated malignant behaviour of NT2D1 non-seminoma cells. Int
J Mol Sci. 20(320)2019.PubMed/NCBI View Article : Google Scholar
|