Introduction
Obesity and asthma are among the most significant
public health problems worldwide. An expanding body of
epidemiological evidence and longitudinal data suggest that there
is a link between obesity and asthma in both children and adults
(1-5).
Furthermore, obesity is a major risk factor for asthma (6-9).
However, managing patients with asthma who are also impacted by
obesity is challenging, as they are frequently less likely to
respond to conventional asthma therapies (10). Therefore, investigating the
potential mechanisms underlying obesity-associated asthma is of
great significance for the treatment of these patients.
In previous years, there has been an immense
interest in the potential role of adipokines, factors secreted by
adipocytes, in the development or worsening of asthma among obese
individuals (11). Adipokines,
including leptin, adiponectin and resistin, regulate energy
homeostasis via hunger and satiety control (12). An imbalance of adipokines may
promote pro-inflammatory responses and lead to inflammation. An
increased leptin/adiponectin ratio may be an important mediator of
type 2 diabetes and abdominal obesity-associated cardiovascular
diseases (13). In obese patients,
an increased serum concentration of leptin and resistin, and
decreased adiponectin levels have been detected (14). In a six-year follow-up study
performed in 246 obese and 532 non-obese children aged 6-11 years,
Zhang et al (15)
illustrated that the levels of leptin and leptin-to-adiponectin
ratio in obese children were significantly higher than those in
non-obese children. Given the role of the increased
leptin/adiponectin ratio in systemic inflammation, it is plausible
to hypothesize that there is an effect of increased leptin in the
airway inflammation of respiratory diseases, such as asthma.
Using a newly developed analytic method, a
longitudinal study from France demonstrated that leptin is an
important factor mediating the association between high body mass
index (BMI) and persistent asthma over time (16). Through collecting bronchoalveolar
lavage (BAL) by bronchoscopy from lean and obese asthmatics and
controls, Holguin et al (17) demonstrated that increases of the BMI
were positively associated with the concentration of leptin in BAL
fluid (BALF). A pilot study (18)
evaluating the levels of leptin in exhaled breath condensate (EBC)
from overweight asthmatic pediatric patients and normal children
indicated that the leptin levels in EBC were significantly higher
in the obese and asthmatic children as compared with those in
healthy subjects, demonstrating that leptin may represent a
non-invasive marker of airway inflammation in children. However,
whether there is an effect of leptin on airway inflammation in
obese patients with asthma and the way in which leptin affects
airway inflammation remain to be elucidated.
The NOD-, LRR- and pyrin domain-containing protein 3
(NLRP3) inflammasome, a multi-protein complex consisting of NLRP3
as the sensor, apoptosis-associated speck-like protein containing
caspase-recruitment domain (ASC) as the adaptor and caspase 1 as
the effector, is known to be associated with a variety of responses
to a wide range of microbial pathogens, inflammatory diseases,
cancer and metabolic and autoimmune disorders (19). A growing body of evidence has
suggested a role of the NLRP3 inflammasome in the inflammation of
respiratory diseases, such as asthma and chronic obstructive
pulmonary disease (COPD) (20,21).
Kim et al (22) indicated
that the NLRP3 inflammasome was required in the lung inflammation
of obese asthmatic mice. Using an ozone-induced mouse model of
acute allergic airway inflammation, Xu et al (23) demonstrated that the activation of
the NLRP3 inflammasome caused by mitochondrial reactive oxygen
species (mtROS) may have an important role in the pathogenesis of
ozone-induced airway inflammation.
On the other hand, two recent studies have
demonstrated that leptin may be a novel activator and modulator of
the NLRP3 inflammasome in RAW 264.7 cells (24) and breast cancer cells (25). Bronchial epithelial cells (BECs)
also express leptin receptors (26). Thus, it is reasonable to postulate
that leptin may activate mtROS-NLRP3 inflammasomes in BECs,
resulting in airway inflammation. Targeting the mtROS-NLRP3
inflammasome pathway may potentially be a therapeutic direction for
the management of obese asthma.
Mitoquinone (mitoQ), a ubiquinone moiety linked to a
lipophilic triphenylphosphonium cation by a 10-carbon alkyl chain,
is a novel mitochondrial-targeted antioxidant (27). The oral administration of mitoQ is
safe and numerous in vivo studies have demonstrated that
mitoQ is able to protect against oxidative damage in a number of
diseases, including cardiac ischemia-reperfusion injury (28), hypertension (29), sepsis (30) and metabolic syndrome (31). Dashdorj et al (32) indicated that mitoQ has
anti-inflammatory effects, as the activation of the NLRP3
inflammasome and the expression of its downstream cytokines were
significantly decreased in a mouse model of experimental colitis
after treatment of mitoQ. Another study demonstrated that mitoQ may
ameliorate diabetic nephropathy via the inhibition of
mtROS/thioredoxin-interacting protein (TXNIP)/NLRP3/IL-1β axis
activation (33). However, whether
there is a role of mitoQ in the airway inflammation of obese asthma
has remained to be elucidated.
In the present study, leptin and mitoQ were used to
treat the human bronchial epithelial cell line BEAS-2 to
investigate the activation of the mtROS-NLRP3 inflammasome by
leptin and the potential role of mitoQ in leptin-pretreated BEAS-2
cells.
Materials and methods
Chemicals and reagents
BEAS-2B cells (no. ATCC® CRL-9609™) were
obtained from the American Type Culture Collection and were tested
negative for mycoplasma. DMEM-high glucose, FBS and Trypsin-0.25%
EDTA were from Gibco (Thermo Fisher Scientific, Inc.). Recombinant
human leptin was purchased from Novoprotein Scientific Inc.
Lipopolysaccharide (LPS) was purchased from Sigma-Aldrich (Merck
KGaA). mitoQ was purchased from Focus Biomolecules. DMSO and
penicillin-streptomycin (pen-strep) were obtained from Solarbio.
The Cell Counting Kit-8 (CCK-8) was purchased from Dojindo
Molecular Technologies. MitoSOX™ Red mitochondrial superoxide
indicator for live-cell imaging (cat. no. M36008),
MitoTracker® Red CM-H2XRos (cat. no. M7513) and
TRIzol® reagent were purchased from Invitrogen (Thermo
Fisher Scientific, Inc.). DAPI staining solution was obtained from
Beyotime Institute of Biotechnology, Inc. PrimeScript™ RT Master
Mix (cat. no. RR036A) was obtained from Takara Bio, Inc.
LightCycler® 480 SYBR-Green I Master Mix (cat. no.
04707516001) was purchased from Roche Diagnostics. ELISA kit (cat.
no. ml058059; Shanghai Enzyme-linked Biotechnology Co., Ltd.) and
all other chemicals of the highest purity available were purchased
from local companies.
Cell culture
BEAS-2B cells were routinely cultured in
high-glucose DMEM supplemented with 10% FBS and 1% pen-strep in an
incubator at 37˚C in a humidified atmosphere containing 5%
CO2. Cells were harvested using Trypsin-0.25% EDTA
solution and all treatments were performed on cells at their 4th
passages to ensure the stability of cells.
CCK-8 viability assay
In order to determine the optimal concentration and
effect time of leptin, LPS and mitoQ, a CCK-8 assay was used.
BEAS-2B cells were seeded in flat-bottom 96-well plates at
2x104 cells/well in triplicate with 100 µl of medium 24
h prior to the treatments. For the treatments, culture media was
replaced with 100 µl of fresh complete medium that was supplemented
with different concentrations of leptin (0, 1, 20, 40, 60, 80, 100
and 200 ng/ml) or LPS (0, 1, 2.5, 5, 10, 25, 50 and 100 µg/µl) or
mitoQ (0, 100, 200, 500, 1,000, 1,500, 2,000 and 5,000 nM) in an
incubator at 37˚C in a humidified atmosphere containing 5%
CO2. At the end of the drug incubation periods (1, 6,
24, 48 or 72 h), the culture media were discarded and the plates
were washed with PBS three times. Subsequently, 10 µl CCK-8
solution was added, followed by incubation in the incubator for 1 h
at 37˚C. The optical density at 450 nm (OD450) was then
determined on a microplate reader (Bio-Rad Laboratories, Inc.). The
cell viability reflecting the cytotoxicity of drugs was calculated
according to the instructions of the kit, using the following
formula: Cell viability=(A-C)/(B-C), where A is the
OD450 of the experimental group (culture medium with
cells, drugs and CCK8 solution), B the OD450 of the
control group (culture medium with cells and CCK8 solution) and C
the OD450 of the empty group (culture medium with CCK8
solution but without cells).
Cell groups
After determining the optimal concentration and
effect time of leptin, LPS and mitoQ, BEAS-2B cells were seeded in
flat-bottom 6-well plates at 2x105 cells/well. According
to the treatments with the drugs, the cells were divided into 6
groups: Control group (negative control), DMSO group (solvent
control), LPS group (positive control), LPS + mitoQ group, Leptin
group and Leptin + mitoQ group. For LPS + mitoQ group, 5 µg/µl LPS
was added 6 h prior to detection and after that 200 nM mitoQ was
added 1 h before detection. For Leptin + mitoQ group, 100 ng/ml
leptin was added 24 h before collection and after that 200 nM mitoQ
was added 1 h before collection. All the treatments were performed
in an incubator at 37˚C in a humidified atmosphere containing 5%
CO2.
mitoTracker measurement
After the drug treatments, the culture media were
removed and prewarmed in a staining solution containing 200 nM
MitoTracker® probe for 30 min at 37˚C in the dark. After
staining was complete, the staining solution was replaced with
fresh prewarmed PBS and cells were then observed using an inverted
fluorescence microscope (magnification, x100; Ti-U; Nikon
Corporation). The mean fluorescence intensity (MFI) was analyzed
and calculated using ImageJ software 1.48v (National Institutes of
Health).
mtROS measurement
After the treatments, the culture media were
discarded and the plates were washed with PBS three times and
loaded with MitoSOX™ reagent. First, 1 ml of 5 µM MitoSOX™ reagent
was added as a working solution to cover the cells. Subsequently,
the cells were incubated for 10 min at 37˚C in the dark. Next,
cells were washed gently three times with warm PBS and fixed with
4% formaldehyde for 15 min at room temperature. Cells were washed
again and covered with warm PBS. The cells were observed using an
inverse fluorescence microscope (magnification, x100; Nikon
Corporation). The MFI was analyzed and calculated using ImageJ
software.
RNA extraction, complementary (c)DNA
synthesis and reverse transcription-quantitative (RT-q)PCR
analysis
Total RNA of cells was extracted using
TRIzol® reagent. The quality and concentration of total
RNA were determined using a Nanodrop® 2000
spectrophotometer (Thermo Fisher Scientific, Inc.). cDNA was
synthesized from 500 ng RNA using the PrimeScript™ RT Master Mix,
according to the manufacturer's protocol. qPCR was performed in
triplicate using LightCycler® 480 SYBR-Green I Master
Mix on the LightCycler® 480 PCR system (Roche). The
thermocycling protocol consisted of an initial denaturing step at
95˚C for 5 min, followed by 40 cycles of 10 sec at 95˚C and an
extension step at 60˚C for 60 sec. The primer sequences are listed
in Table I. Relative mRNA
expression levels were measured using the 2-∆∆Cq method
(34) and normalized to the level
of GAPDH.
 | Table ISequence information of primers. |
Table I
Sequence information of primers.
| Gene | Primer
sequences |
|---|
| GAPDH | F:
5'-GGAGAAGGCTGGGGCTCAT-3' |
| | R:
5'-TGGGTGGCAGTGATGGCA-3' |
| NLRP3 | F:
5'-CATAGGACCGCTCTGCACTG-3' |
| | R:
5'-CAGGTCTCGTGGTGATGAGC-3' |
| CASP1 | F:
5'-ACAGGCATGACAATGCTGCT-3' |
| | R:
5'-CAGGAACGTGCTGTCAGAGG-3' |
| IL-1β | F:
5'-TGCCACCTTTTGACAGTGATG-3' |
| | R:
5'-TGATGTGCTGCTGCGAGATT-3' |
ELISA
The levels of cytokine IL-1β in the cell culture
supernatants were determined by ELISA according to the
manufacturer's protocol. The OD450 value was measured on
a microplate reader (Bio-Rad Laboratories, Inc.) and the
concentrations of IL-1β in the samples were calculated according to
the OD450.
Statistical analysis
Values are expressed as the mean ± standard error of
the mean of at least 3 samples. Experiments were performed in
triplicate. All values were analyzed by one-way ANOVA among groups
using SPSS (version 19.0; IBM Corp.). Tukey's test was used for
further comparison between two groups. P<0.05 was considered to
indicate a statistically significant difference. The graphs were
drawn using GraphPad Prism 5 (GraphPad Software, Inc.).
Results
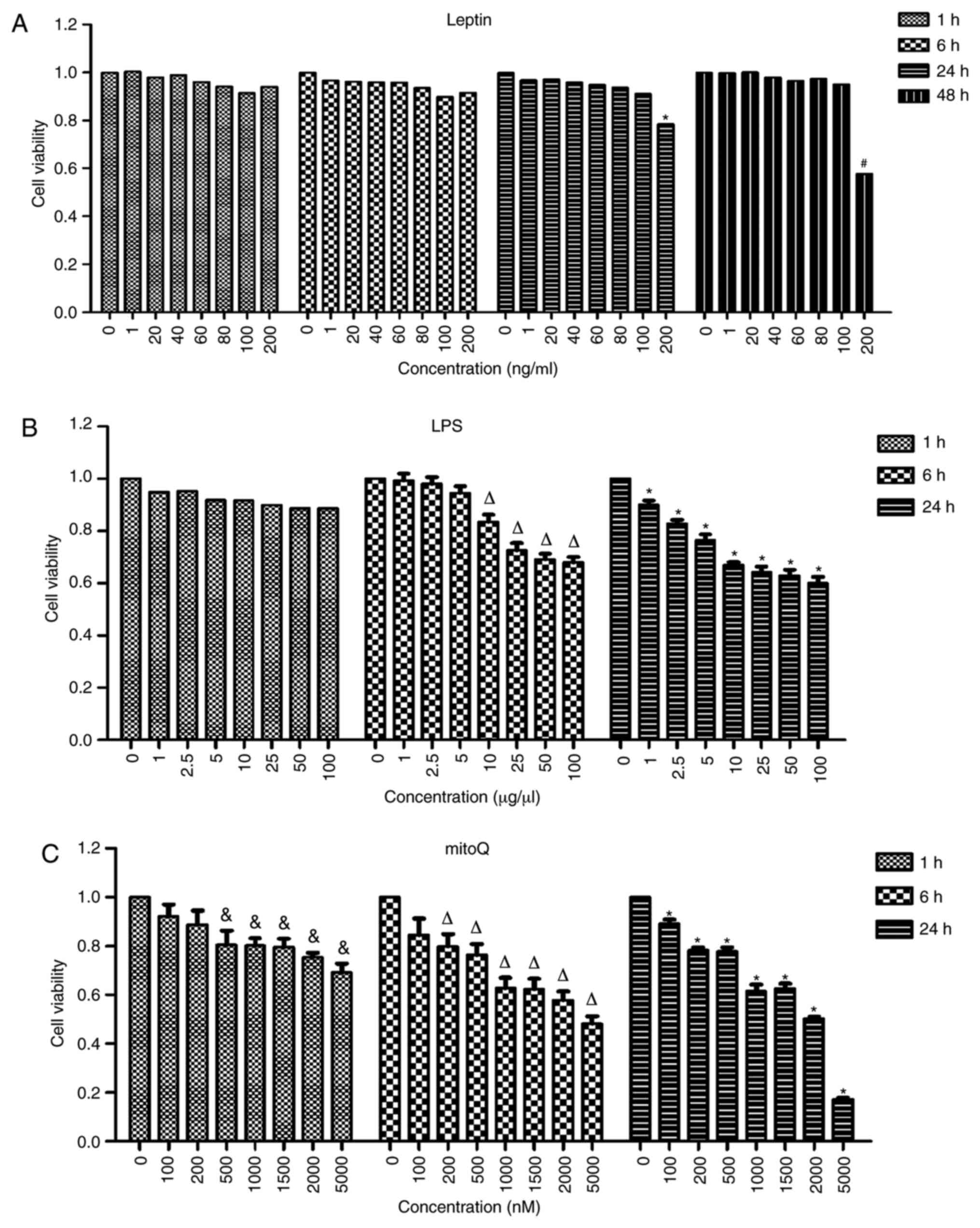
Determination of the optimal
concentrations and incubation times for LPS, leptin and mitoQ
In order to determine the optimal concentrations and
incubation times, the cells were treated with different
concentrations of drugs and incubated for different time periods.
The highest concentration incubated with the lowest incubation time
that did not result in a significantly effect on cell viability was
considered to be the optimal concentration. No significant
differences were observed among the different concentrations of
leptin after 1 and 6 h of treatment. After 24 h of treatment with
leptin, the viability of the cells treated at the concentration of
200 ng/ml became significantly lower than that of untreated cells
(P<0.05). However, there was no significant difference between
cells treated with leptin at a concentration of 100 ng/ml and
untreated cells after 24 h of treatment. Thus, 100 ng/ml was used
as the final concentration of leptin and the incubation time
selected was 24 h (Fig. 1A).
Similarly, after 6 h of incubation with LPS, the
viability of cells treated at the concentration of 10 µg/µl was
significantly decreased compared with that of untreated cells
(P<0.01). However, the viability of cells treated at the
concentration of 5 µg/µl was not significantly different compared
with that of untreated cells. Furthermore, the viability of cells
treated with mitoQ at the concentration of 500 nM was significantly
decreased after 1 h of treatment as compared with that of untreated
cells (P<0.05), while that of cells treated with 200 nM mitoQ
was not significantly affected. Thus, in the subsequent
experiments, cells were treated with 5 µg/µl LPS for 6 h (Fig. 1B) and 200 nM mitoQ for 1 h (Fig. 1C). Therefore, for LPS + mitoQ group,
5 µg/µl LPS was added 6 h prior to detection and 200 nM mitoQ was
added 1 h before detection. For Leptin + mitoQ group, 100 ng/ml
leptin was added 24 h before collection and 200 nM mitoQ was added
1 h before collection.
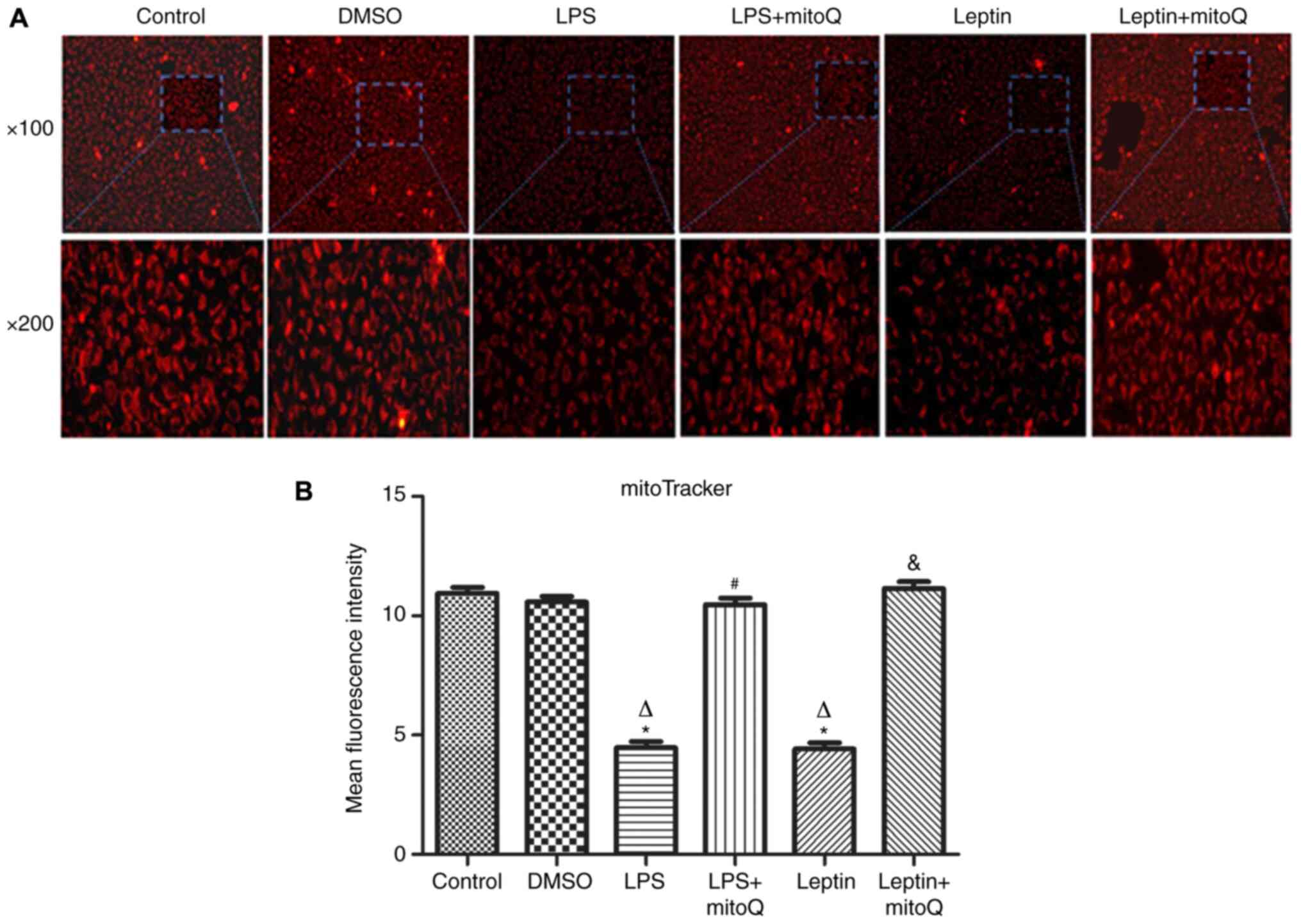
Effects of mitoQ on the mitochondrial
membrane integrity in the LPS and leptin-pretreated BEAS-2
cells
The mitoTracker probe, a kind of mitochondrial
membrane tracer, was used to detect the membrane integrity of
mitochondria. As presented in Fig.
2A and B, the mitochondrial
membrane of the control group and DMSO group was intact and the
MFIs were 10.945±0.254 and 10.587±0.233, respectively. However,
after treatment with LPS and leptin, the mitochondrial membrane was
disrupted and the MFIs of the groups (4.474±0.264 and 4.416±0.267,
respectively) were significantly decreased compared with those of
the control and DMSO groups (P<0.01), indicating that the
mitochondria were impaired. However, the MFI of the LPS + mitoQ
group (10.47±0.271) or the Leptin + mitoQ group (11.14±0.294) was
significantly increased compared with that in the LPS group or
Leptin group (P<0.01), suggesting that the impaired
mitochondrial membrane may be recovered following treatment with
mitoQ.
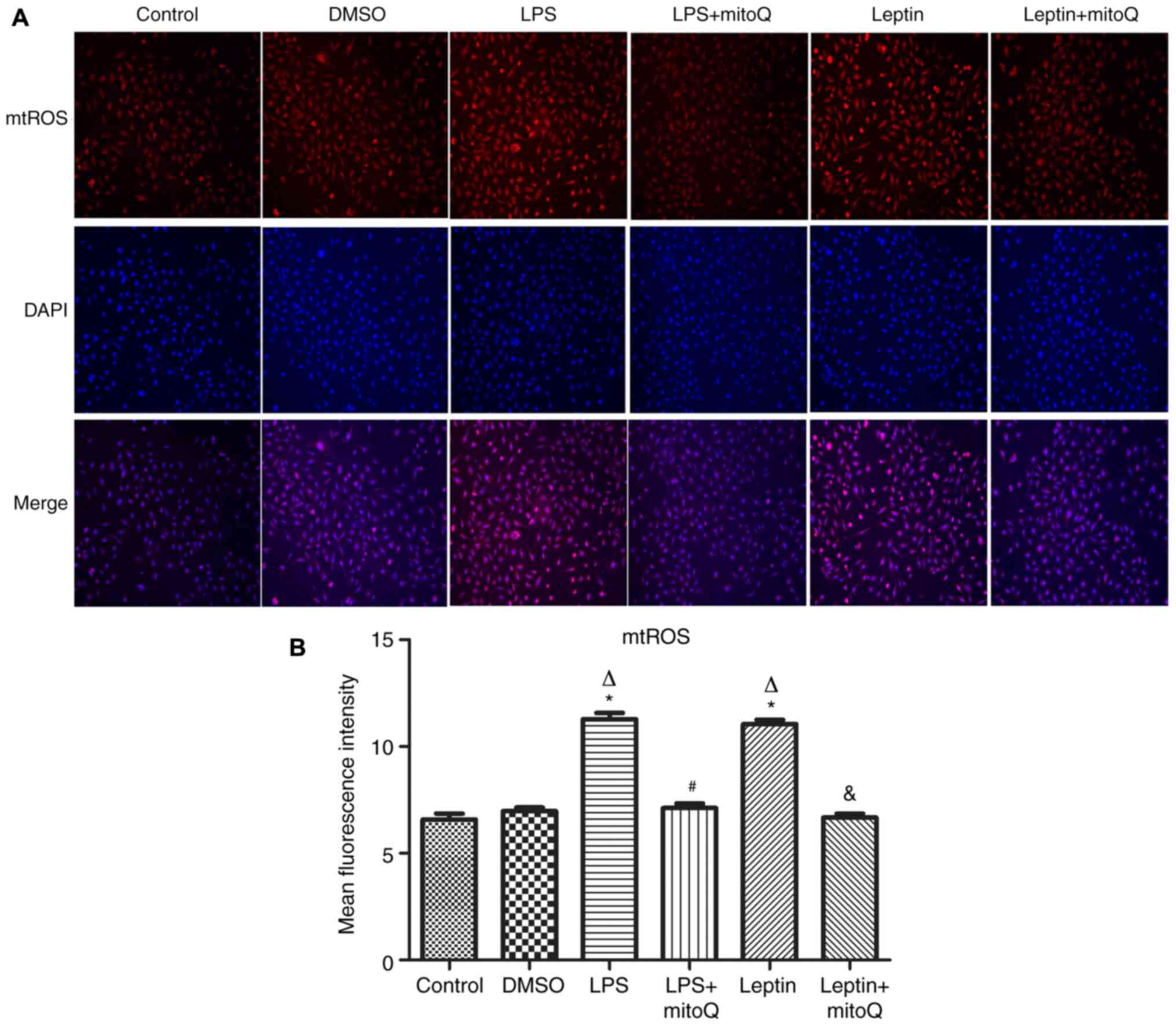
Effects of mitoQ on the production of
mtROS in the LPS and leptin-pretreated BEAS-2 cells
Next, to observe the influence of mitoQ on the
production of mtROS, the mitoSOX probe was used to detect the
production of mtROS after LPS and leptin treatment. In the LPS
group as the positive control, the production of mtROS was
significantly augmented (MFI, 11.282±0.286) when compared with that
in the control group and DMSO group (6.585±0.271 and 6.965±0.178,
respectively; P<0.01). Similarly, after treatment with leptin,
the production of mtROS was also significantly increased compared
with that in the control and DMSO groups (P<0.01), revealing the
potential effect of leptin on the overproduction of mtROS in BEAS-2
cells. After treatment with mitoQ, the overproduction of mtROS
caused by LPS or leptin was significantly decreased to normal
levels (P<0.01), confirming the scavenger role of mitoQ on mtROS
(Fig. 3A and B).
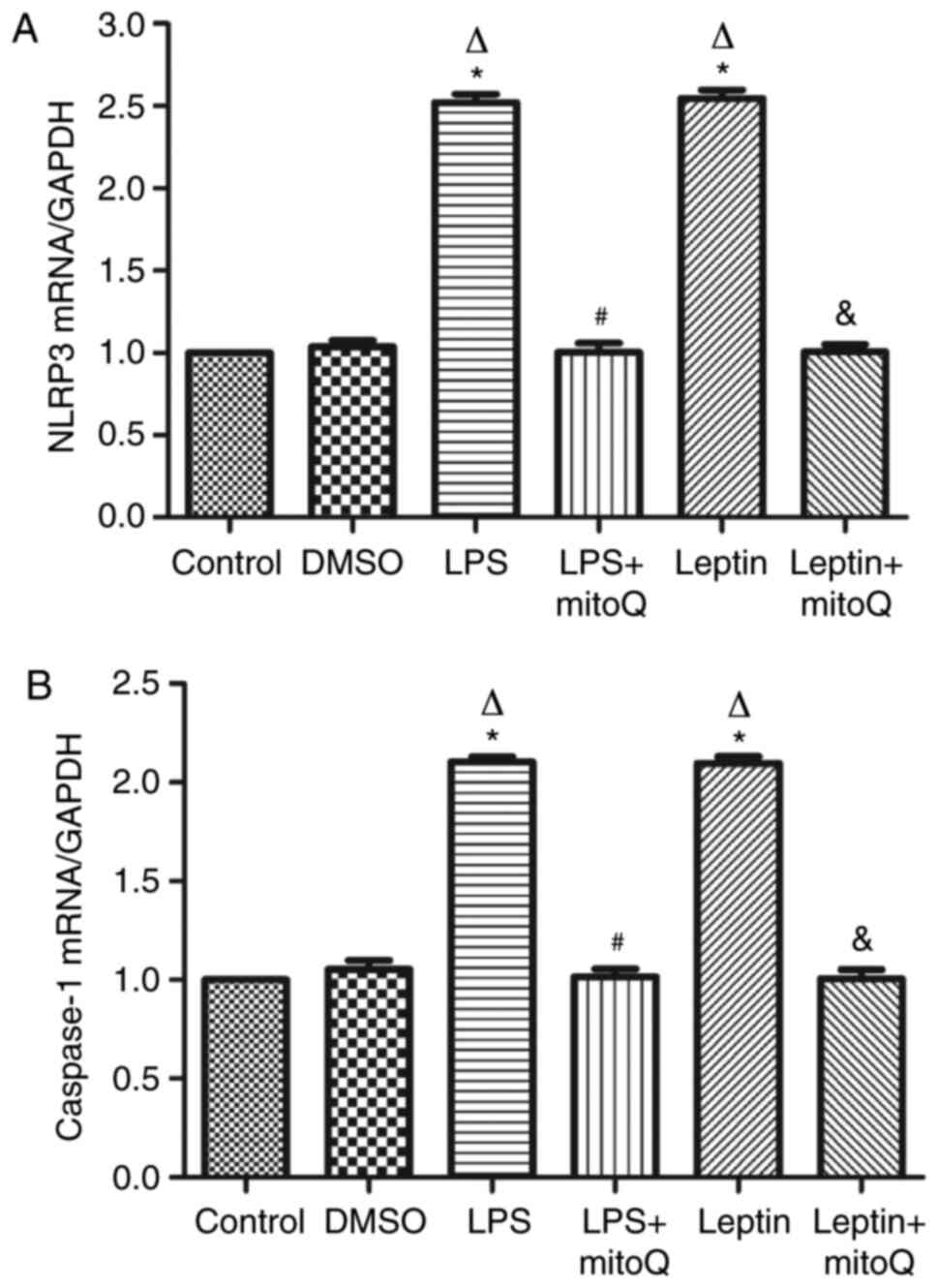
Effects of mitoQ on the expression
levels of NLRP3 and caspase-1 mRNA in the LPS and leptin-pretreated
BEAS-2 cells
The NLRP3 inflammasome may be activated by the
overproduction of mtROS. In accordance with the aforementioned
results, the expression levels of NLRP3 and caspase-1 mRNA in the
LPS- or leptin-pretreated BEAS-2 cells were ~2.5-fold higher than
those in the control and DMSO groups (P<0.01). Following
subsequent treatment with mitoQ, their expression levels of NLRP3
and caspase-1 were significantly decreased (P<0.01), suggesting
that mitoQ may reverse BEAS-2 cells from leptin-induced injury,
partly through inhibiting the mtROS-NLRP3 inflammasome signaling
pathway (Fig. 4A and B).
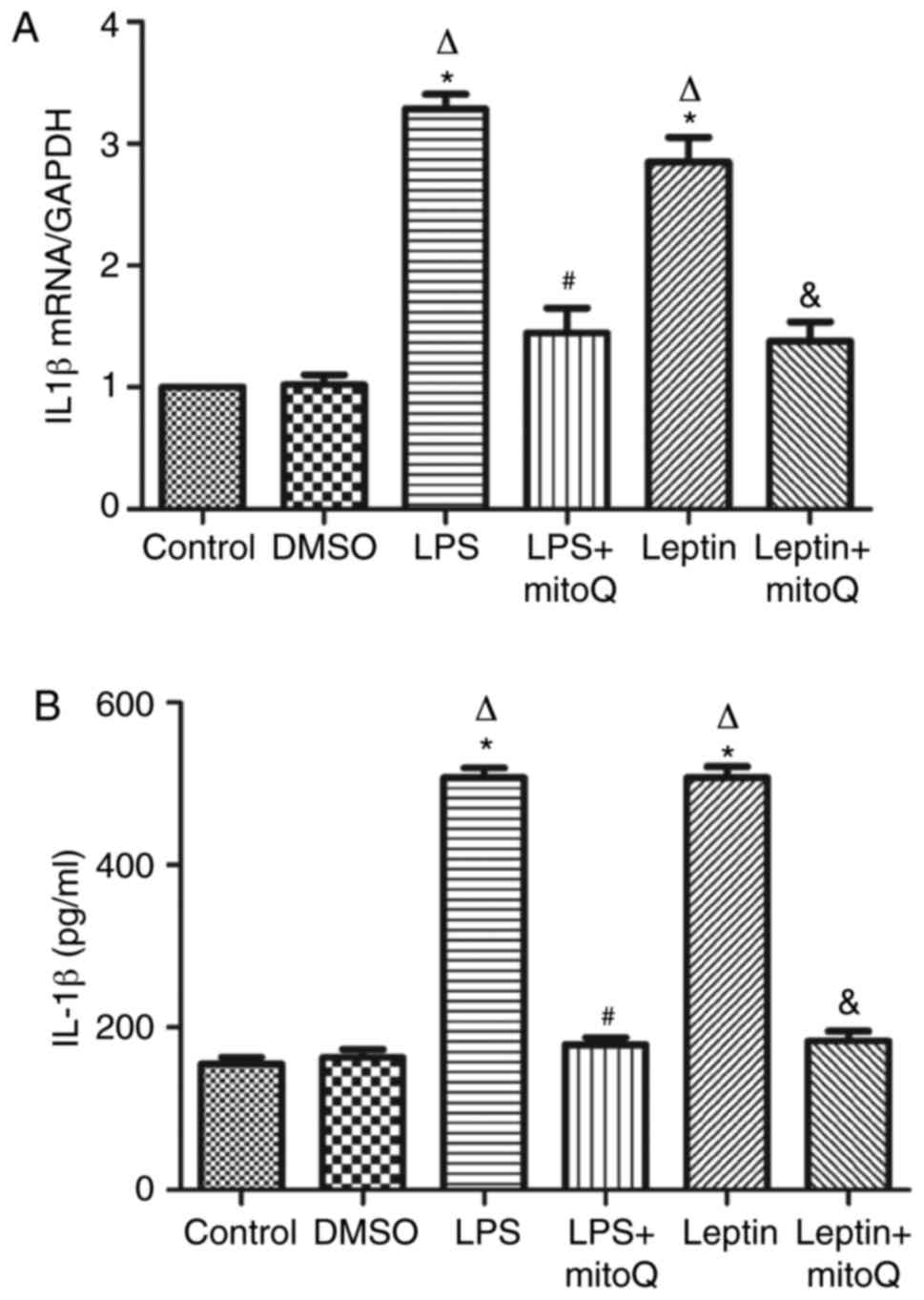
Effects of mitoQ on the expression of
IL-1β in the LPS and leptin-pretreated BEAS-2 cells
As presented in Fig.
5A, the mRNA expression levels of IL-1β in the LPS and
leptin-pretreated BEAS-2 cells were ~3 times higher than those in
the control and DMSO groups (P<0.01). The changes in the protein
levels of IL-1β in the supernatant of the LPS and leptin groups
were similar to those in mRNA expression (Fig. 5B). By contrast, the mRNA expression
and protein levels in the supernatant of the LPS + mitoQ and Leptin
+ mitoQ groups were significantly decreased (P<0.01).
Discussion
Airway inflammation is the key characteristic of
asthma. As a distinct asthma phenotype, the airway inflammation of
obese asthma is different from that of conventional asthma, making
it difficult to control the condition (11). Leptin, a 16-kDa protein derived from
the obesity gene, is secreted predominantly by adipocytes (35). The serum leptin concentrations are
4-6 times greater in severely obese patients compared with those in
lean patients (36). Studies have
demonstrated that leptin concentrations, either in sputum or in
BALF, are highly associated with serum leptin concentrations
(17,37). Bruno et al (38) revealed that in the airway epithelial
cells, the expression levels of leptin and its receptor LEPR were
higher in normal subjects than in smokers or in patients with
mild-to-severe COPD. In a previous study, it was also revealed that
the expression level of leptin in the BALF was significantly
increased in obese mice with asthma (39). Thus, it is conceivable that
obesity-associated increases in airway leptin may have an effect on
the airway inflammation of asthma.
The NLRP3 inflammasome is a multi-protein complex
involved in the innate immune system. Formation of the NLRP3
inflammasome activates caspase 1, which in turn cleaves pro-IL-1β
and pro-IL-18, leading to the release of IL-1β and IL-18. The NLRP3
inflammasome may be activated by a diverse series of endogenous and
exogenous agonists and the generation of mtROS is thought to be an
essential and proximal upstream event of NLRP3 activation (40). A growing body of evidence has
suggested a role of the NLRP3 inflammasome-linked cytokine IL-1β in
severe, steroid-resistant asthma (41). Using an ovalbumin- and LPS-induced
mouse model of neutrophil dominant allergic airway disease, Kim
et al (42) revealed that
mtROS have a crucial role in the pathogenesis of allergic airway
inflammation through activating the NLRP3 inflammasome in airway
epithelial cells. Consistent with previous results, the present
study revealed that in BEAS-2 cells treated with LPS, the
mitochondrial membrane was disrupted, followed by an upregulation
of mtROS. Furthermore, similar to LPS, treating cells with leptin
also led to significantly increased mtROS levels, suggesting that
leptin may serve as an activator of the NLRP3 inflammasome.
Next, the transcriptional expression levels of NLRP3
and caspase-1 were detected by RT-qPCR in the present study. The
results demonstrated that the expression levels of NLRP3 and
caspase-1 mRNA were significantly upregulated in both leptin- and
LPS-pretreated groups. Furthermore, to validate the potential role
of leptin in activating the NLRP3 inflammasome, leptin receptor,
LEPR was knocked down in the cells by transfection with LEPR RNA
interference lentivirus. The mRNA expression of NLRP3 and caspase-1
were not increased following leptin treatment compared with those
in the control group (data not shown). Similarly, upregulation of
IL-1β in the supernatant of leptin-pretreated cells was also
detected, further indicating that leptin may promote airway
inflammation through activating the NLRP3 inflammasome pathway.
However, a limitation of the present study was that the protein
levels of NLRP3 inflammasome were not detected. In a further study,
the protein levels of NLRP3 inflammasome should be measured.
MitoQ is an orally active mitochondria-targeted
antioxidant that has the ability to scavenge mtROS. Two clinical
trials have been performed to investigate the benefits of mitoQ.
One is a human phase II trial in Parkinson's disease, the PROTECT
trial (www.clinicaltrials.gov; no.
NCT00329056) (43). Although no
difference has yet been observed compared with the placebo, this
trial suggested that mitoQ may be safely administered to patients
for 1 year. The other is the CLEAR trial on patients with chronic
hepatitis C virus (clinicaltrials.gov; no. NCT00433108) (44). This study identified a decrease in
markers of liver damage, indicating a clinical benefit provided by
mitoQ in humans. Another study assessed the lungs of ozone-exposed
mice and human airway smooth muscle cells (ASMC) isolated from
bronchial biopsy specimens from patients with COPD and indicated
that mitoQ reduced airway inflammation and inhibited TGF-β-induced
ASMC proliferation (45).
Consistent with the results of previous studies, the
present study suggested that mitoQ decreased the production of
mtROS induced by leptin in BEAS-2 cells, which further inhibited
the expression of NLRP3 and caspase-1 mRNA. The increase of IL-1β
was also restored after treatment with mitoQ. The in vitro
results of the present study provide a molecular basis for mitoQ
having a potential role in managing obese asthma through targeting
the mtROS-NLRP3 inflammasome pathway. However, the study did not
use any group treated with mitoQ alone to observe the effect of
mitoQ on the cells, which may be another limitation in the present
study.
In conclusion, the results of the present study
suggested that leptin may induce or worsen airway inflammation
partly through activating the mtROS-NLRP3 inflammasome pathway,
which may be a potential mechanism underlying obesity-associated
asthma. The mitochondrial-targeted antioxidant mitoQ may have
potential therapeutic effects in obese asthma. However, the present
study was only performed on cells. Future in vivo studies in
obese mice with asthma are required in order to further investigate
the role of mitoQ in airway inflammation.
Acknowledgements
The authors thank Professor Changchong Li
(Discipline of Pediatric Respiratory Medicine, the Second
Affiliated Hospital of Wenzhou Medical University, Wenzhou, China)
for contributing to the design of the study and revising the
manuscript.
Funding
The present study was supported by Zhejiang Provincial Natural
Science Foundation of China (grant no. LQ19H010002).
Availability of data and materials
All of the data used and/or analyzed in the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LC and GY conceived and designed the current study.
LC and HL performed the experiments. LZ analyzed the data and
prepared the figures. All authors read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Figueroa-Muñoz JI, Chinn S and Rona RJ:
Association between obesity and asthma in 4-11 year old children in
the UK. Thorax. 56:133–137. 2001.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Beuther DA and Sutherland ER: Overweight,
obesity, and incident asthma: A meta-analysis of prospective
epidemiologic studies. Am J Respir Crit Care Med. 175:661–666.
2007.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Quinto KB, Zuraw BL, Poon KY, Chen W,
Schatz M and Christiansen SC: The association of obesity and asthma
severity and control in children. J Allergy Clin Immunol.
128:964–969. 2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Papoutsakis C, Priftis KN, Drakouli M,
Prifti S, Konstantaki E, Chondronikola M, Antonogeorgos G and
Matziou V: Childhood overweight/obesity and asthma: Is there a
link? A systematic review of recent epidemiologic evidence. J Acad
Nutr Diet. 113:77–105. 2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Jiang D, Wang L, Bai C and Chen O:
Association between abdominal obesity and asthma: A meta-analysis.
Allergy Asthma Clin Immunol. 15(16)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Barros R, Moreira P, Padrão P, Teixeira
VH, Carvalho P, Delgado L and Moreira A: Obesity increases the
prevalence and the incidence of asthma and worsens asthma severity.
Clin Nutr. 36:1068–1074. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rastogi D: Quantifying the contribution of
obesity to incident childhood asthma: It's about time. Pediatrics.
142(e20182979)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Lang JE, Bunnell HT, Hossain MJ, Wysocki
T, Lima JJ, Finkel TH, Bacharier L, Dempsey A, Sarzynski L, Test M
and Forrest CB: Being overweight or obese and the development of
asthma. Pediatrics. 142(e20182119)2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Rzehak P, Wijga AH, Keil T, Eller E,
Bindslev-Jensen C, Smit HA, Weyler J, Dom S, Sunyer J, Mendez M, et
al: Body mass index trajectory classes and incident asthma in
childhood: Results from 8 European Birth Cohorts-a Global Allergy
and Asthma European Network initiative. J Allergy Clin Immunol.
131:1528–1536. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mohan A, Grace J, Wang BR and Lugogo N:
The effects of obesity in asthma. Curr Allergy Asthma Rep.
19(49)2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Muc M, Mota-Pinto A and Padez C:
Association between obesity and asthma-epidemiology,
pathophysiology and clinical profile. Nutr Res Rev. 29:194–201.
2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Trayhurn P, Bing C and Wood IS: Adipose
tissue and adipokines-energy regulation from the human perspective.
J Nutr. 136 (Suppl 7):S1935–S1939. 2006.PubMed/NCBI View Article : Google Scholar
|
|
13
|
López-Jaramillo P, Gómez-Arbeláez D,
López-López J, López-López C, Martínez-Ortega J, Gómez-Rodríguez A
and Triana-Cubillos S: The role of leptin/adiponectin ratio in
metabolic syndrome and diabetes. Horm Mol Biol Clin Investig.
18:37–45. 2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Balistreri CR, Caruso C and Candore G: The
role of adipose tissue and adipokines in obesity-related
inflammatory diseases. Mediators Inflamm.
2010(802078)2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zhang M, Cheng H, Zhao X, Hou D, Yan Y,
Cianflone K, Li M and Mi J: Leptin and leptin-to-adiponectin ratio
predict adiposity gain in nonobese children over a six-year period.
Child Obes. 13:213–221. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Li Z, Leynaert B, Dumas O, Diaz Gil O,
Garcia-Aymerich J, Fito Colomer M, Le Moual N, Pison C, Romieu I,
Siroux V, et al: Role of leptin in the association between body
adiposity and persistent asthma: A longitudinal study. Obesity
(Silver Spring). 27:894–898. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Holguin F, Rojas M, Brown LA and
Fitzpatrick AM: Airway and plasma leptin and adiponectin in lean
and obese asthmatics and controls. J Asthma. 48:217–223.
2011.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bodini A, Tenero L, Sandri M, Maffeis C,
Piazza M, Zanoni L, Peroni D, Boner A and Piacentini G: Serum and
exhaled breath condensate leptin levels in asthmatic and obesity
children: A pilot study. J Breath Res. 11(46005)2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Davis BK, Wen H and Ting JP: The
inflammasome NLRs in immunity, inflammation, and associated
diseases. Annu Rev Immunol. 29:707–735. 2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Birrell MA and Eltom S: The role of the
NLRP3 inflammasome in the pathogenesis of airway disease. Pharmacol
Ther. 130:364–370. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Theofani E, Semitekolou M, Morianos I,
Samitas K and Xanthou G: Targeting NLRP3 inflammasome activation in
severe asthma. J Clin Med. 8(1615)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kim HY, Lee HJ, Chang YJ, Pichavant M,
Shore SA, Fitzgerald KA, Iwakura Y, Israel E, Bolger K, Faul J, et
al: Interleukin-17-producing innate lymphoid cells and the NLRP3
inflammasome facilitate obesity-associated airway hyperreactivity.
Nat Med. 20:54–61. 2013.PubMed/NCBI View
Article : Google Scholar
|
|
23
|
Xu M, Wang L, Wang M, Wang H, Zhang H,
Chen Y, Wang X, Gong J, Zhang JJ, Adcock IM, et al: Mitochondrial
ROS and NLRP3 inflammasome in acute ozone-induced murine model of
airway inflammation and bronchial hyperresponsiveness. Free Radic
Res. 53:780–790. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Fu S, Liu L, Han L and Yu Y: Leptin
promotes IL18 secretion by activating the NLRP3 inflammasome in RAW
264.7 cells. Mol Med Rep. 16:9770–9776. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Raut PK, Kim SH, Choi DY, Jeong GS and
Park PH: Growth of breast cancer cells by leptin is mediated via
activation of the inflammasome: Critical roles of estrogen receptor
signaling and reactive oxygen species production. Biochem
Pharmacol. 161:73–88. 2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Löllmann B, Grüninger S, Stricker-Krongrad
A and Chiesi M: Detection and quantification of the leptin receptor
splice variants Ob-Ra, b, and, e in different mouse tissues.
Biochem Biophys Res Commun. 238:648–652. 1997.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Murphy MP: Understanding and preventing
mitochondrial oxidative damage. Biochem Soc Trans. 44:1219–1226.
2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Adlam VJ, Harrison JC, Porteous CM, James
AM, Smith RA, Murphy MP and Sammut IA: Targeting an antioxidant to
mitochondria decreases cardiac ischemia-reperfusion injury. FASEB
J. 19:1088–1095. 2005.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Graham D, Huynh NN, Hamilton CA, Beattie
E, Smith RA, Cochemé HM, Murphy MP and Dominiczak AF:
Mitochondria-targeted antioxidant MitoQ10 improves endothelial
function and attenuates cardiac hypertrophy. Hypertension.
54:322–328. 2009.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lowes DA, Thottakam BM, Webster NR, Murphy
MP and Galley HF: The mitochondria-targeted antioxidant MitoQ
protects against organ damage in a lipopolysaccharide-peptidoglycan
model of sepsis. Free Radic Biol Med. 45:1559–1565. 2008.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Mercer JR, Yu E, Figg N, Cheng KK, Prime
TA, Griffin JL, Masoodi M, Vidal-Puig A, Murphy MP and Bennett MR:
The mitochondria-targeted antioxidant MitoQ decreases features of
the metabolic syndrome in ATM+/-/ApoE-/-
mice. Free Radic Biol Med. 52:841–849. 2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Dashdorj A, Jyothi KR, Lim S, Jo A, Nguyen
MN, Ha J, Yoon KS, Kim HJ, Park JH, Murphy MP and Kim SS:
Mitochondria-targeted antioxidant MitoQ ameliorates experimental
mouse colitis by suppressing NLRP3 inflammasome-mediated
inflammatory cytokines. BMC Med. 11(178)2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Han Y, Xu X, Tang C, Gao P, Chen X, Xiong
X, Yang M, Yang S, Zhu X, Yuan S, et al: Reactive oxygen species
promote tubular injury in diabetic nephropathy: The role of the
mitochondrial ros-txnip-nlrp3 biological axis. Redox Biol.
16:32–46. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zhang Y, Proenca R, Maffei M, Barone M,
Leopold L and Friedman JM: Positional cloning of the mouse obese
gene and its human homologue. Nature. 372:425–432. 1994.PubMed/NCBI View
Article : Google Scholar
|
|
36
|
Maffei M, Halaas J, Ravussin E, Pratley
RE, Lee GH, Zhang Y, Fei H, Kim S, Lallone R, Ranganathan S, et al:
Leptin levels in human and rodent: Measurement of plasma leptin and
ob RNA in obese and weight-reduced subjects. Nat Med. 1:1155–1161.
1995.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Sood A, Seagrave JC, Herbert G, Harkins M,
Qualls C and Schuyler M: Asthma is associated with lower
adiponectin concentrations in sputum than controls. Am Thorac Soc
Int Confer. 185(A6502)2012.
|
|
38
|
Bruno A, Chanez P, Chiappara G, Siena L,
Giammanco S, Gjomarkaj M, Bonsignore G, Bousquet J and Vignola AM:
Does leptin play a cytokine-like role within the airways of COPD
patients? Eur Respir J. 26:398–405. 2005.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Chong L, Liu L, Zhu L, Li H, Shao Y, Zhang
H and Yu G: Expression levels of predominant adipokines and
activations of STAT3, STAT6 in an experimental mice model of obese
asthma. Iran J Allergy Asthma Immunol. 18:62–71. 2019.PubMed/NCBI
|
|
40
|
Xiao Y, Xu W and Su W: NLRP3 inflammasome:
A likely target for the treatment of allergic diseases. Clin Exp
Allergy. 48:1080–1091. 2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Kim RY, Pinkerton JW, Essilfie AT,
Robertson AAB, Baines KJ, Brown AC, Mayall JR, Ali MK, Starkey MR,
Hansbro NG, et al: Role for NLRP3 inflammasome-mediated,
IL-1β-dependent responses in severe, steroid-resistant asthma. Am J
Respir Crit Care Med. 196:283–297. 2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Kim SR, Kim DI, Kim SH, Lee H, Lee KS, Cho
SH and Lee YC: NLRP3 inflammasome activation by mitochondrial ROS
in bronchial epithelial cells is required for allergic
inflammation. Cell Death Dis. 5(e1498)2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Snow BJ, Rolfe FL, Lockhart MM, Frampton
CM, O'Sullivan JD, Fung V, Smith RA, Murphy MP and Taylor KM:
Protect Study Group. A double-blind, placebo-controlled study to
assess the mitochondria-targeted antioxidant MitoQ as a
disease-modifying therapy in Parkinson's disease. Move Disord.
25:1670–1674. 2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Gane EJ, Weilert F, Orr DW, Keogh GF,
Gibson M, Lockhart MM, Frampton CM, Taylor KM, Smith RA and Murphy
MP: The mitochondria-targeted anti-oxidant mitoquinone decreases
liver damage in a phase II study of hepatitis C patients. Liver
Int. 30:1019–1026. 2010.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Wiegman CH, Michaeloudes C, Haji G, Narang
P, Clarke CJ, Russell KE, Bao W, Pavlidis S, Barnes PJ, Kanerva J,
et al: Oxidative stress-induced mitochondrial dysfunction drives
inflammation and airway smooth muscle remodeling in patients with
chronic obstructive pulmonary disease. J Allergy Clin Immunol.
136:769–780. 2015.PubMed/NCBI View Article : Google Scholar
|