Introduction
Hepatic ischemia-reperfusion injury (HIRI) generally
occurs during hemorrhagic shock, hepatectomy and liver
transplantation (1). In the United
States, liver transplantation accounted for ~23% transplant
procedures in 2015, where one of the most formidable barriers to
successful liver transplantation is ischemia-reperfusion injury
(2). However, despite the prominent
clinical presence of HIRI, there are currently no effective drugs
available to prevent IR injury.
During hepatic IR, liver injury caused by hypoxia is
further exacerbated by the restoration of blood flow (3). A large number of free radicals and
reactive oxygen species produced as a result of IR, including
superoxide, hydrogen peroxide and hydroxyl radicals, have been
shown to exert serious deleterious effects on hepatocytes (4,5).
During the oxidative stress reaction, the nuclear erythroid
2-related factor 2 (Nrf2)/heme oxygenase (HO)-1 pathway is one of
the most important antioxidant systems (6). It has also been reported that
apoptosis induced by mitochondrial permeability transition (MPT)
leads to caspase (CASP) activation and that MPT also serves an
important role in the pathogenesis of IR injury (7). Therefore, it was hypothesized that
suppressing apoptosis and oxidative stress following acute injury
may be key to treating hepatic IR.
Gastrodin is an organic compound that can be
extracted from the dried root blocks of the orchid plant Gastrodia
elata Blume, which has a long history of clinical application in
Chinese medicine (8,9). Gastrodin (Gas; PubChem CID, 115027;
Fig. S1) is the major active
ingredient in Rhizome Gastrodiae. According to previous studies,
Gas has been found to protect liver cells, neurons and
cardiomyocytes by inhibiting oxidative stress and parenchymal
apoptosis (4,10); however, to the best of our
knowledge, no studies have investigated its role in hepatic IR. As
such, regarding the potential application of Gas, the present study
aimed to investigate the role and mechanism of action of Gas
pretreatment at various times during hepatic IR in mice by
constructing a mouse hepatic IR model.
Materials and methods
Animal studies
A total of 48 6-week-old male (20-23 g) C57/BL6 mice
were purchased from Shanghai Slark Laboratory Animal Co., Ltd. The
animals were housed in a pathogen-free animal room at the Hubei
Animal Experiment Center of Tongji University at 25±3˚C and 55±5%
humidity, with a 12-h light/dark cycle, a standard experimental
diet and free access to drinking water. Forty-eight C57/BL6 mice
were randomly divided into four groups (12 mice/group): i) Gas
(cat. no. 62499-27-8; MedChemExpress) pretreatment + hepatic IR
model (IR + Gas group), whereby 300 mg/kg/day Gas, the dose
reported in the literature (4), was
continuously administered into the stomach for 8 days prior to the
establishment of the hepatic IR model; ii) hepatic IR model (IR +
vehicle group), in which an equal volume of normal saline was
administered to the stomach for 8 consecutive days prior to the
establishment of the hepatic IR model; iii) Gas pretreatment + sham
group (Sham + Gas group), whereby Gas was continuously administered
to the stomach for 8 days, then the abdominal cavity was opened and
the portal vein, hepatic vein and hepatic artery were simply
separated to simulate a sham operation; and iv) sham operation
group (Sham + vehicle group), whereby an equal volume of normal
saline was administered to the stomach for 8 days prior to the sham
operation. Following 2 and 24 h of modeling, six mice were randomly
selected from each group. The mice were euthanized by overdoes of
inhalant anesthetics, followed by exsanguination under anesthesia
to ensure death. When the mice showed no respiration, no heartbeat
and no response to any external stimuli, they were considered dead.
Samples required for testing were collected.
Establishment of hepatic IR model
Prior to the experimental procedure, the mice were
fasted for 12 h and were kept water-free for 4 h. The model of
hepatic IR in male mice was established using the Pringle's method
(11). The mice were anesthetized
by inhalation of isoflurane (oxygen flow rate, 1.0 l/min; induction
dose, 3%; maintenance dose, 2%). Then the mice were placed on the
operating table and the abdominal cavity and liver was exposed
along the white line of the abdomen. Microvascular forceps were
used to clamp the vessels in the left and middle lobe of the liver,
causing 70% liver ischemia. The color of the left lobe liver and
middle lobe liver tissue immediately changed from reddish brown to
light red. Following 60 min of ischemia, the microvascular forceps
were removed to restore the liver blood supply and at the same
time, restore the anatomical position of the abdominal organs. The
abdominal wall was sutured with double layers and the mouse liver
IR model was successfully constructed.
H&E staining
Liver segments from the left lobe of the liver from
each group of mice were cut, fixed with 4% paraformaldehyde for 24
h at room temperature, routinely dehydrated at room temperature
using an increasing ethanol gradient followed by xylene. The
sections were then embedded in paraffin and cut into 5-mm-thick
tissue sections. The sections were subsequently stained with
H&E (hematoxylin was stained for 5 min, 1% HCl ethanol for 1-3
sec and eosin staining for 3 min, all at room temperature) and
visualized using a Nikon light microscope at x400 magnification
(Nikon Corporation). The severity of liver pathological damage was
evaluated using the Suzuki standard method (Table SI) (12). Three experienced pathologists scored
each sample in a blinded manner.
Tissue and serum indicators
Serum was obtained at 2 and 24 h following the
operation. Alanine transaminase (ALT) and aspartate transaminase
(AST) levels in the serum were detected using ALT and AST kits
(cat. nos. C009-2-1 and C010-2-1, respectively; Nanjing Jiancheng
Bioengineering Institute), according to the manufacturer's
protocol. The content of superoxide dismutase (SOD),
malondialdehyde (MDA) and glutathione (GSH) in the liver tissues
was analyzed using SOD, MDA and GSH kits, respectively (cat. nos.
A001-3-2, A003-1-2 and A006-2-1, respectively; Nanjing Jiancheng
Bioengineering Institute).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from the liver tissues of
each group of mice using TRIzol® reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). Total RNA was reverse transcribed
into cDNA using PrimeScript™ RT Master Mix (Perfect Real Time; cat.
no. RR036B; Takara Bio, Inc.). The temperature protocol were as
follows: 37˚C for 15 min, 85˚C for 5 sec. RT-qPCR was subsequently
performed using TB Green® Premix Ex Taq™ (cat. no.
RR042B; Takara Bio, Inc.). The full thermocycling conditions for
qPCR are as follows: Initial denaturation at 95˚C for 30 sec,
followed by 40 cycles of 95˚C for 5 sec and 60˚C for 30 sec. The
primer sequences are presented in Tables SII and SIII. Expression levels were quantified
using the 2-ΔΔCq method (13) and normalized to β-actin.
Western blotting
Western blotting was performed RIPA lysis buffer
(Beyotime Institute of Biotechnology) from liver specimens. Equal
amounts of protein (30 µg; quantified using the bicinchoninic acid
protein assay) were separated using 10% SDS-PAGE and then
wet-transferred onto a PVDF membrane (cat. no. IPVH00010; EMD
Millipore). The membranes were blocked with 5% non-fat milk for 1 h
at room temperature and subsequently incubated with the following
primary antibodies at 4˚C overnight: Anti-Nrf2 (cat. no.
16396-1-AP; 1:1,000; ProteinTech Group, Inc.), anti-HO-1 (cat. no.
10701-1-AP; 1:1,000; ProteinTech Group, Inc.), anti-Bax (cat. no.
ab32503; 1:1,000; Abcam), anti-Bcl-2 (cat. no. ab32124; 1:1,000;
Abcam), anti-CASP-3 (cat. no. ab32351; 1:1,000; Abcam) and
anti-β-actin (cat. no. MA1-140; 1:5,000; Thermo Fisher Scientific,
Inc.). Following the primary antibody incubation, the membranes
were incubated with the horseradish peroxidase-conjugated goat
anti-rabbit IgG secondary antibody (cat. no. A0208; 1:10,000;
Beyotime Institute of Biotechnology) and goat anti-mouse IgG (cat.
no. A0216; 1:10,000; Beyotime Institute of Biotechnology) at room
temperature for 1 h. The protein bands were visualized by BeyoECL
Plus (Beyotime Institute of Biotechnology). QuantityOne v4.6.6
software (Bio-Rad Laboratories, Inc.) was used for analysis and the
ratio of the gray value of the target protein band to the gray
value of the internal reference β-actin protein band was used to
represent the relative expression levels of the target protein.
Statistical analysis
GraphPad Prism software (GraphPad Software, Inc.)
and SPSS 19.0 software (IBM Corp.) were used for statistical
analysis. One-way ANOVA, followed by Bonferroni's correction or
Tamhane's T2 post-hoc test, was used to determine the statistical
differences between groups. A Kruskal Wallis test, followed by
post-hoc Dunn's test, was used to evaluate non-parametric data. All
data are presented as the mean ± SEM, or median (25-75th
percentile). P<0.05 was considered to indicate a statistically
significant difference.
Results
Gas pretreatment alleviates the degree
of liver IR injury in mice
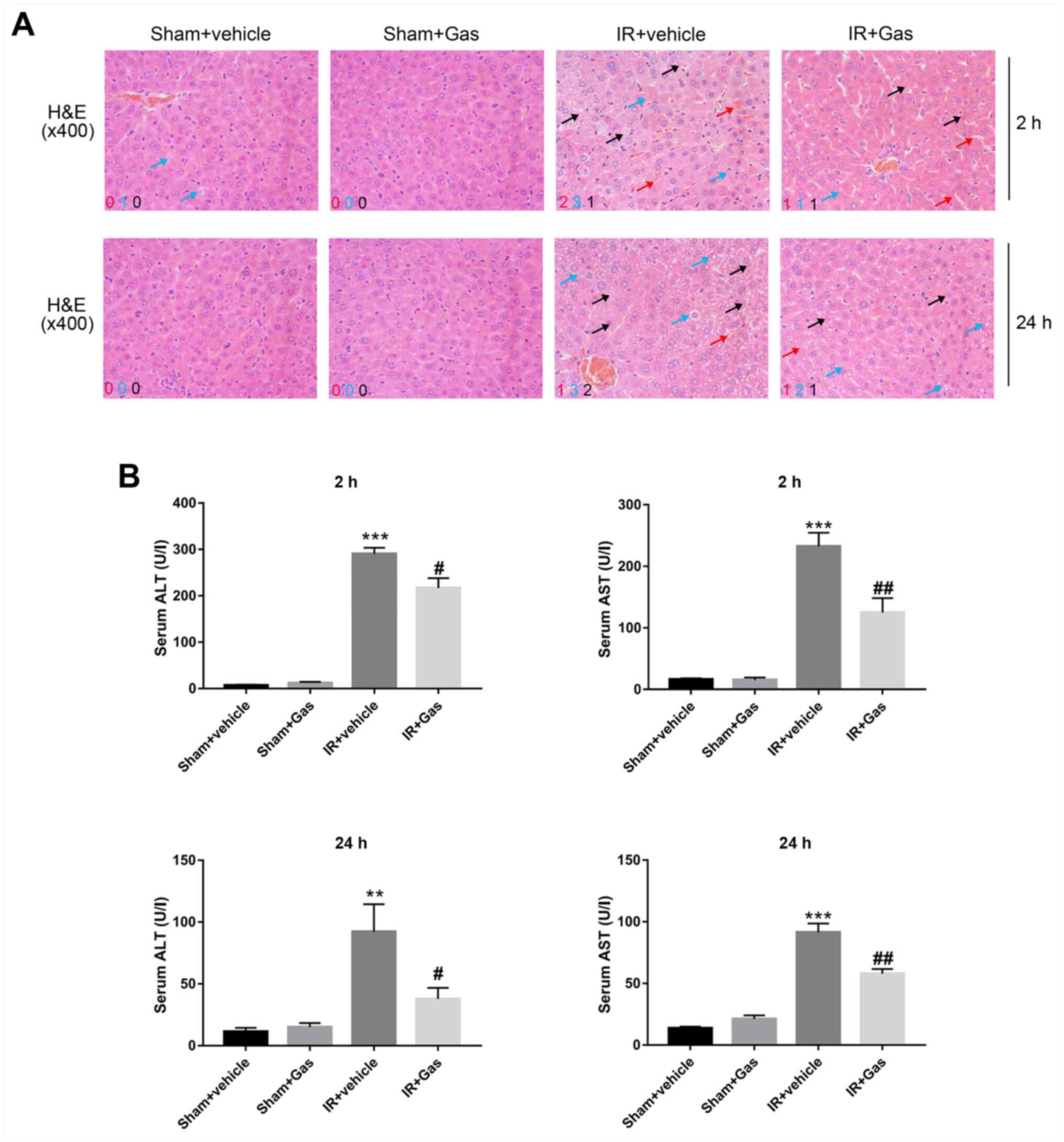
H&E staining and a Suzuki's score were used to
evaluate the liver histopathological changes of mice in each group.
H&E pathological sections revealed that the Sham + Gas and Sham
+ vehicle groups had complete hepatic lobules without the presence
of abnormal inflammatory cell infiltration, fragments or massive
necrosis (Fig. 1A). No significant
differences were observed in the Suzuki's score (Table I) and in serum ALT and AST levels
(Fig. 1B).
 | Table ISuzuki's score of each group at
different time-points. |
Table I
Suzuki's score of each group at
different time-points.
| | Suzuki's score |
|---|
| Group | 2 h | 24 h |
|---|
| Sham + vehicle
group | 0.00
(0.00-1.00) | 0.50
(0.00-1.00) |
| Sham + Gas
group | 0.00
(0.00-1.00) | 1.00
(0.00-1.00) |
| IR + vehicle
group | 6.50
(5.50-8.00)a | 7.00
(6.75-8.00)a |
| IR + Gas group | 4.00
(4.00-6.00)b | 5.50
(5.00-7.00)b |
The pathological changes in the IR groups appeared
to be different. Compared with the IR + vehicle group, the liver
pathological changes, such as liver congestion, vacuole-like
changes and necrosis, were markedly alleviated in the IR + Gas
group at 2 and 24 h (Fig. 1A).
Following Gas pretreatment, the pathological results of the IR +
Gas group revealed that the structure of the liver lobules was
intact, the red blood cell depositions in the central vein and
hepatic sinuses were reduced, and the proportion of cytoplasmic
vacuoles and necrotic cells were reduced. The IR + Gas group
displayed a lower Suzuki's score and decreased serum levels of ALT
and AST at 2 h post-modeling, with the trends being the same at 24
h (Fig. 1; Table I).
Pathological and serological results confirmed that
Gas pretreatment itself did not cause damage to the liver tissue
and function. Gas pretreatment appeared to have reduced the
occurrence of congestion, vacuole-like changes and necrosis
following hepatic IR. Therefore, subsequent experiments focused on
the effect of Gas pretreatment on mice hepatic IR.
Gas pretreatment enhances the ability
of mice to resist oxidative stress
Oxygen free radicals can attack polyunsaturated
fatty acids in biofilms, decompose lipid hydroperoxides and damage
liver tissue (14-16).
Following hepatic IR modeling, due to the effect of Gas, the liver
SOD activity in the IR + Gas group was significantly increased at
both the 2 and 24 h time-points, while the MDA content was
significantly decreased compared with that in the IR + vehicle
group (Fig. 2). In addition,
compared with those in the IR + vehicle group, liver GSH content in
the IR + Gas group showed an upward trend at the time-point of 2 h
without significant difference, but increased significantly at the
time-point of 24 h.
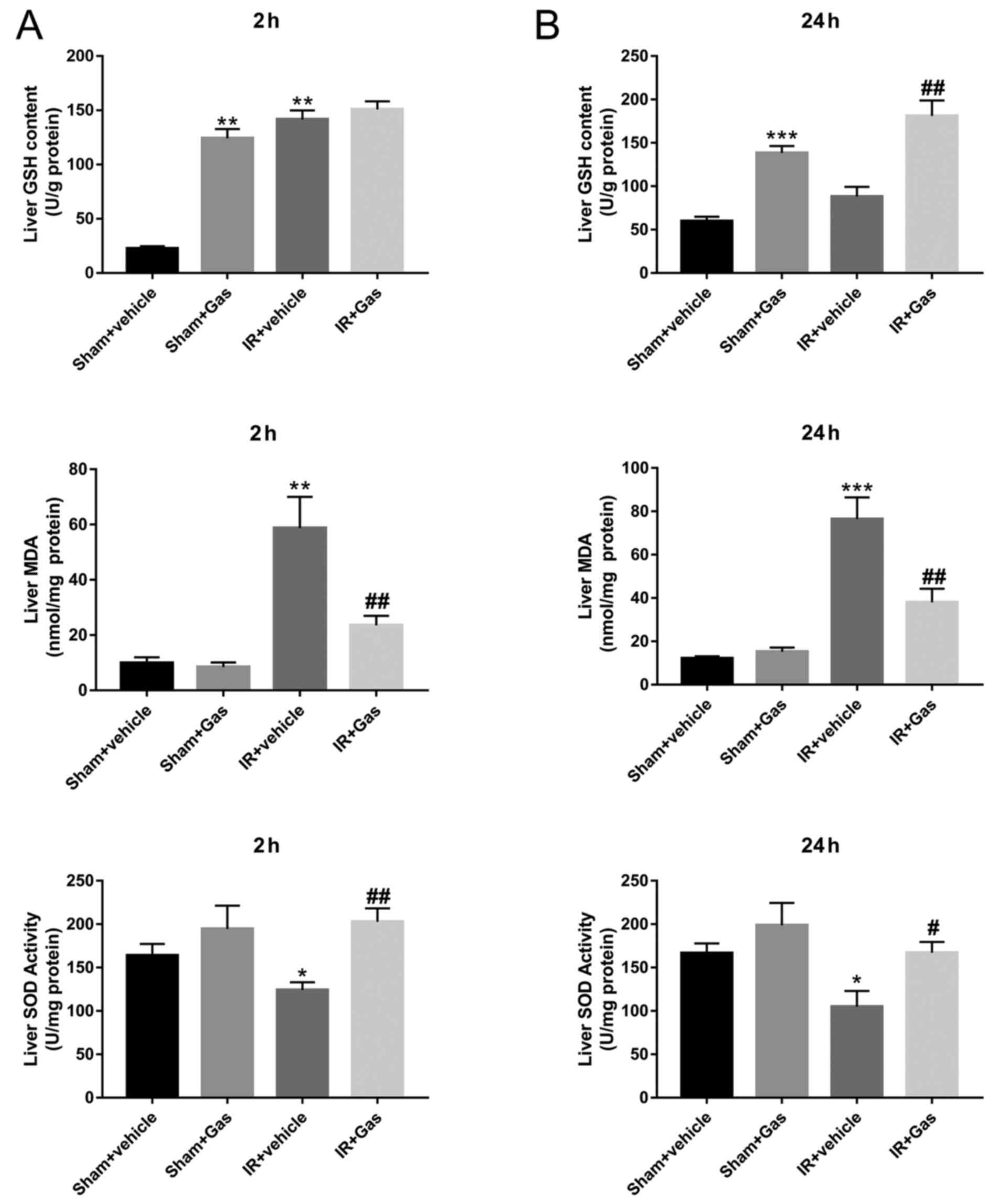 | Figure 2Gas pretreatment enhances the ability
of mice to resist oxidative stress. (A) Liver tissue biochemical
indexes of SOD, GSH and MDA at 2 h following hepatic
ischemia-reperfusion modeling. (B) Liver tissue biochemical indexes
of SOD, GSH and MDA at 24 h following hepatic IR modeling (n=6
mice). *P<0.05, **P<0.01,
***P<0.001 vs. the Sham + vehicle group;
#P<0.05, ##P<0.01 vs. the IR + vehicle
group. Gas, gastrodin; GSH, glutathione; IR, ischemia-reperfusion;
MDA, malondialdehyde; SOD, superoxide dismutase. |
To investigate whether Gas pretreatment enhanced the
antioxidant capacity of normal mice, the Sham + vehicle group was
compared with the Sham + Gas group. The results confirmed that the
liver GSH content in the Sham + Gas group was significantly higher
compared with that in the Sham + vehicle group (Fig. 2). Although there were no differences
observed in SOD activity and MDA content between the Sham groups;
however, they were significantly changed following HIRI. The
expression levels of the SOD gene were also analyzed; the results
revealed that compared with the IR + vehicle group, SOD expression
levels were significantly increased following Gas pretreatment
(Figs. 3B and 4B).
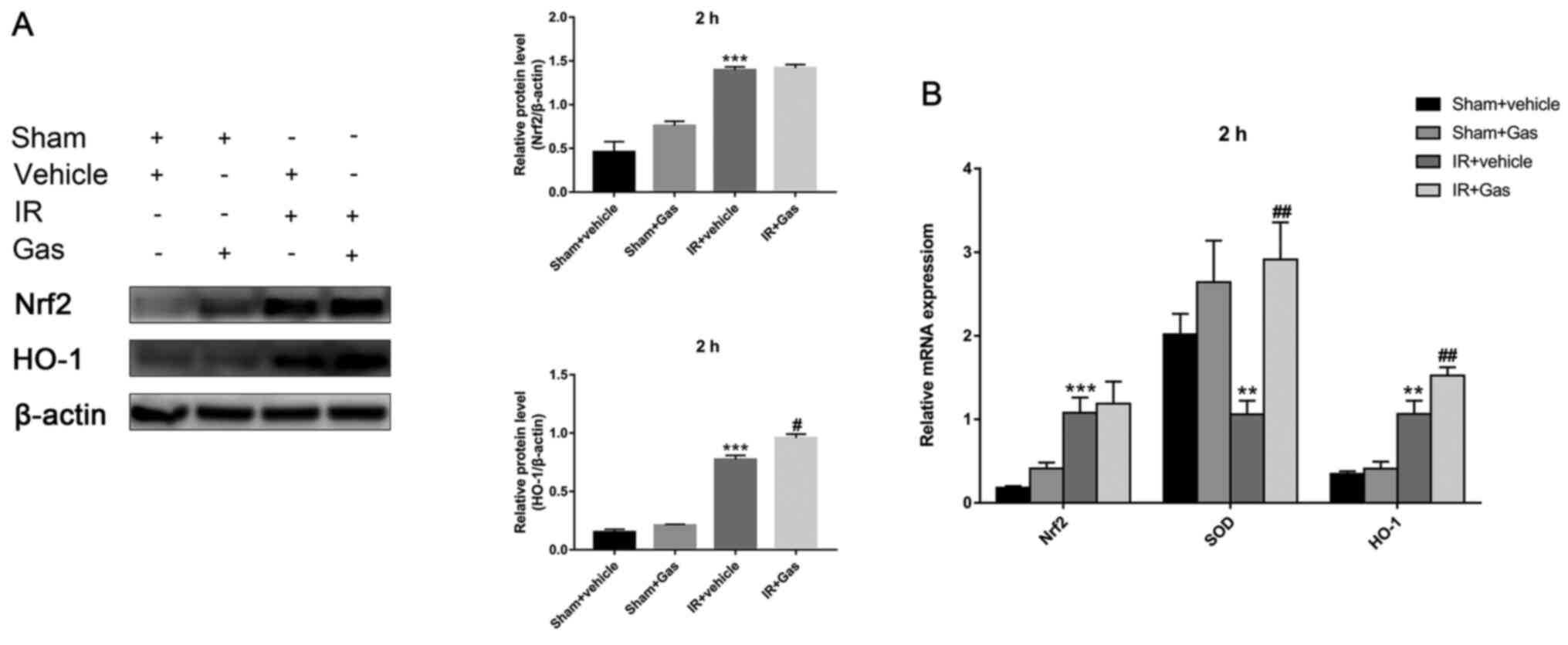
Gas pretreatment upregulates Nrf2 and
HO-1 expression levels in the liver
Subsequently, the present study investigated whether
Gas pretreatment affected the expression levels of liver Nrf2 and
HO-1.
At 2 h following hepatic IR modeling, compared with
the IR + vehicle group, the HO-1 expression levels in the liver of
the IR + Gas group were significantly upregulated; however, there
were no significant differences observed between Nrf2 mRNA and
protein expression levels (Fig. 3A
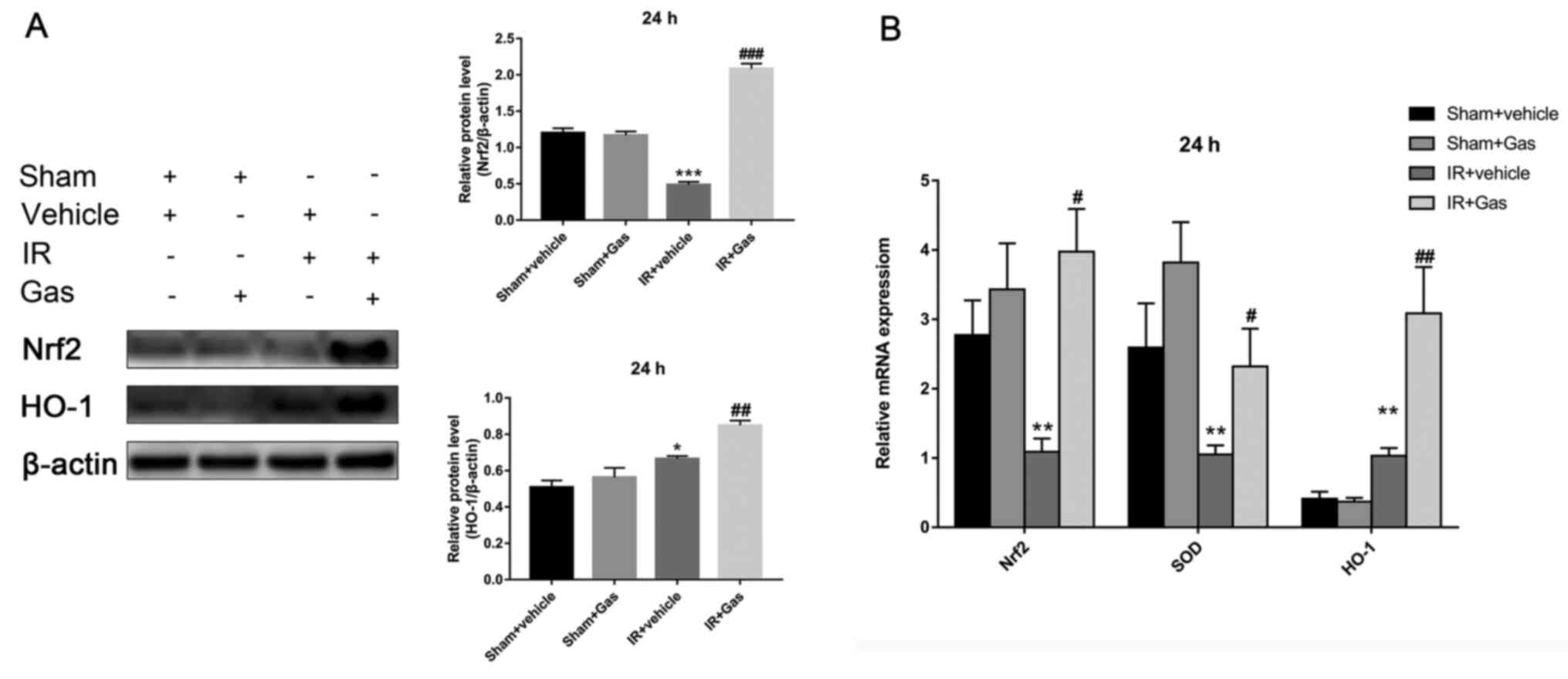
and B). However, at 24 h after
hepatic IR modeling, the liver Nrf2 and HO-1 expression levels in
the IR + Gas group demonstrated similar changes, with significantly
upregulated expression levels (Fig.
4A and B).
The expression levels of Nrf2 appeared to fluctuate
over time, which was not only observed between the IR groups, but
also between the sham groups (Figs.
3A and 4A).
Gas pretreatment improves the
antiapoptotic ability of the liver through regulating the
expression levels of Bax, Bcl-2 and CASP-3
The present study also investigated the expression
levels of Bax, Bcl-2 and CASP-3 in the liver of each group of mice
(Figs. 5 and 6). The results revealed that Gas
significantly downregulated the expression levels of Bax and
cleaved CASP-3, while upregulating Bcl-2 expression levels at 2 and
24 h post hepatic IR modeling (Figs.
5B and 6B). This trend was
further confirmed at the protein expression level, as detected by
western blotting (Figs. 5A and
6A). Meanwhile, no significant
differences were observed between the Sham + vehicle and the Sham +
Gas groups (Figs. 5 and 6). These results suggested that Gas
pretreatment may not cause damage to normal mice liver tissue.
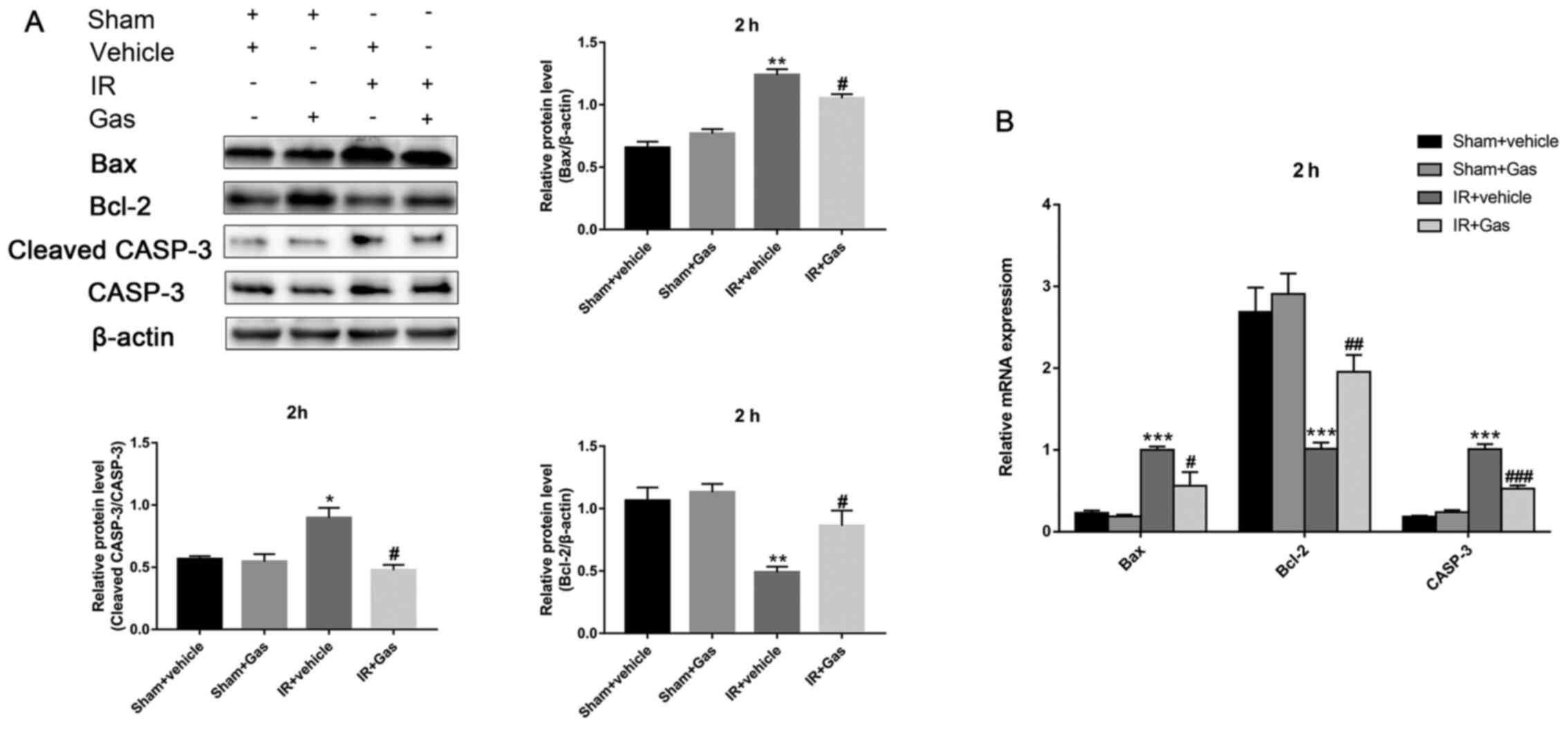 | Figure 5Gas pretreatment improves the
antiapoptotic ability of the liver through regulating the
expression levels of Bax, Bcl-2 and CASP-3. (A) Protein expression
levels of Bax, Bcl-2 and CASP-3 at 2 h following hepatic IR
modeling were analyzed using western blotting (n=3). (B) RT-qPCR
was used to analyze the expression levels of Bax, Bcl-2 and CASP-3
at 2 h post hepatic ischemic-reperfusion modeling (n=6 mice).
*P<0.05, **P<0.01,
***P<0.001 vs. the Sham + vehicle group;
#P<0.05, ##P<0.01,
###P<0.001 vs. the IR + vehicle group. CASP-3,
caspase 3; Gas, gastrodin; IR, ischemia-reperfusion; RT-qPCR,
reverse transcription-quantitative PCR. |
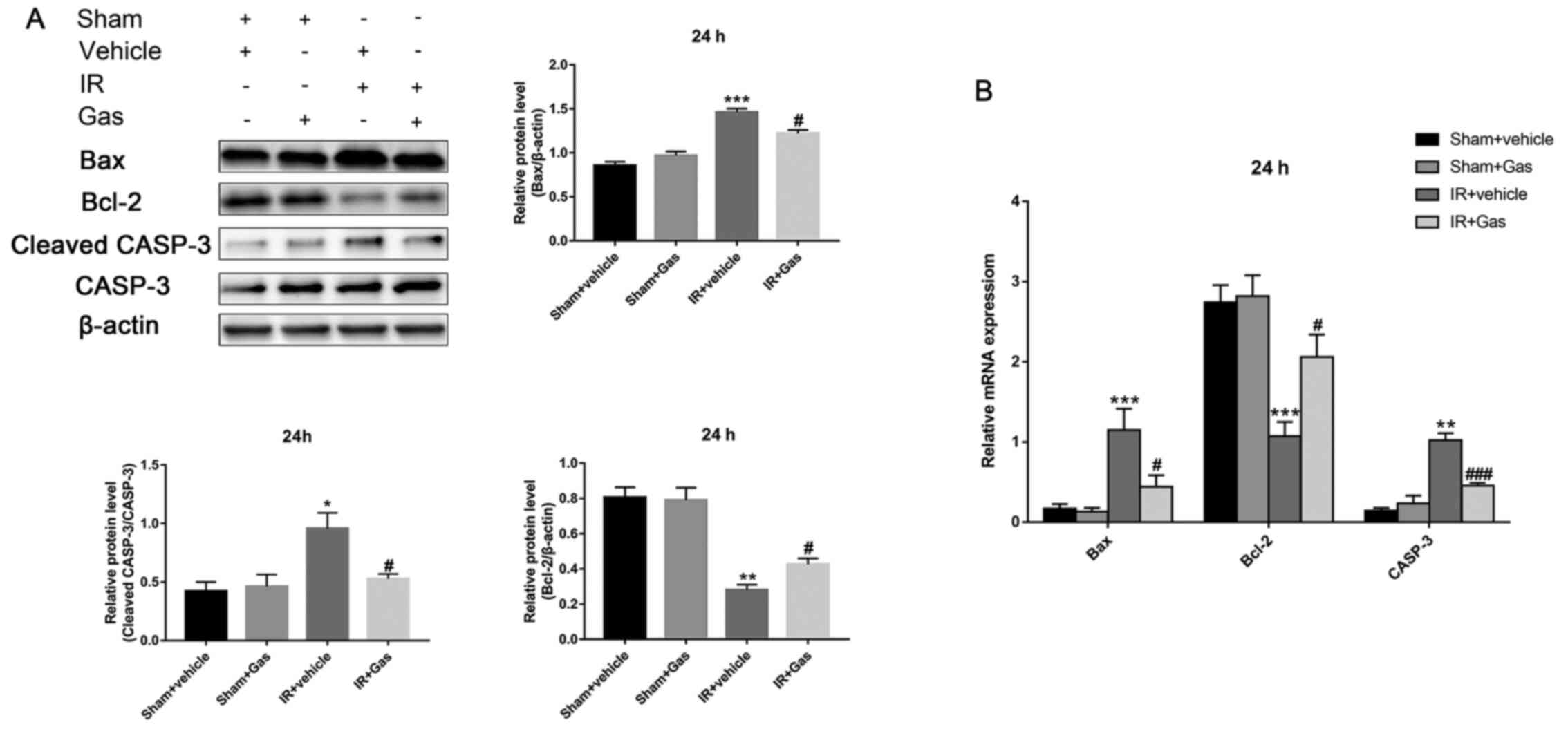 | Figure 6Gas pretreatment improves the
antiapoptotic ability of the liver through regulating the
expression levels of Bax, Bcl-2 and CASP-3. (A) Western blotting
analysis of Bax, Bcl-2 and CASP-3 protein expression levels at 24 h
post hepatic IR modeling (n=3). (B) Reverse
transcription-quantitative PCR analysis of Bax, Bcl-2 and CASP-3
mRNA expression levels at 24 h post hepatic IR modeling (n=6 mice).
*P<0.05, **P<0.01,
***P<0.001 vs. the Sham + vehicle group;
#P<0.05, ###P<0.001 vs. the IR +
vehicle group. CASP-3, caspase 3; Gas, gastrodin; IR,
ischemia-reperfusion; RT-qPCR, reverse transcription-quantitative
PCR. |
Discussion
Previous studies have reported that Gas improves the
ability of cells to resist hypoxia and apoptosis, and exerts
protective effects on the mouse heart and following cerebral IR
(4,17-19).
As such, the present study aimed to determine the role of Gas in
mice hepatic IR.
To determine the concentration and dosage of Gas
pretreatment, previous literature reports were used (4). A concentration of 300 mg/kg/day was
administered by gavage 8 days before modeling and the changes in
the related indicators were analyzed at 2 and 24 h. The results of
the present study revealed IR caused significant pathological
changes within the two time-points by the end of the model,
indicating that the hepatic IR model was established successfully
and that the liver tissue was severely damaged. Thus, the present
study aimed to investigate the role of Gas preconditioning in
HIRI.
Similar to previous studies, the levels of three
biochemical indicators, SOD, GSH and MDA, were determined to
evaluate the ability of antioxidative stress following Gas
pretreatment (20). The
experimental results confirmed that Gas pretreatment significantly
enhanced the antioxidative stress capacity following hepatic IR in
mice. Therefore, these findings suggested that Gas pretreatment may
help the mouse liver to accelerate the elimination of superoxide
anion free radicals, stabilize enzymes containing thiol groups and
prevent hemoglobin and other cofactors from oxidative damage during
HIRI. In addition, Gas increased the content of GSH in the liver of
normal mice, suggesting an increased resistance to attacks from
oxygen free radicals and reactive oxygen species. This effect was
consistent with previously published studies (21,22).
A previous study has reported that Nrf2 regulates
HO-1 expression levels by binding to antioxidative stress elements
on the HO-1 promoter (23). Nrf2
protected organisms from oxidative stress by regulating the
expression levels of antioxidant enzymes (24). In addition, ischemic hypoxia
stimulation has been shown to induce the production of HO-1, which
exerts protective antioxidant functions (6,25,26).
Consistent with these findings, the present experimental results
revealed that Gas pretreatment upregulated the mRNA and protein
expression levels of Nrf2 and HO-1 at 24 h post hepatic IR
modeling, with the two demonstrating a similar trend. However,
compared with the Sham + vehicle group at 2 and 24 h post IR
modeling, Nrf2 mRNA and protein expression levels in the IR +
vehicle group were upregulated at first, but then decreased,
whereas HO-1 mRNA and protein expression levels demonstrated an
upward trend. Therefore, there did not appear to be a similar trend
between liver Nrf2 and HO-1 expression levels in the IR + vehicle
group at the 24 h time-point after establishment of the model.
However, pretreatment with Gas produced a similar change between
Nrf2 and HO-1 expression levels at 24 h post modeling, rather than
at 2 h. This finding suggested that there may be another regulatory
mechanism acting between Nrf2 and HO-1 under gastrodin action in
the subacute time window (2-24 h) following HIRI. According to a
previous study, Gas promotes the nuclear transfer of Nrf2(20). This phenomenon may explain the
experimental results; however, the associated mechanism of action
requires further research.
Necrosis has been considered as the main outcome of
dying tissue cells from acute injury. However, this conclusion has
not been confirmed after inhibition at key regulatory sites during
necrosis in acute liver injury models (27). Concurrently, the process of
apoptosis, another form of cell death, has attracted increasing
attention. HIRI was significantly improved by inhibiting
proapoptotic molecules, since apoptosis is one of the predominant
forms of liver cell death (28).
Previous studies have revealed that by inhibiting c-Jun NH2
terminal kinase 2, the release of mitochondrial proapoptotic
molecules was decreased (29,30),
thereby reducing the apoptosis of hepatocytes and markedly
improving adverse IR events (3). In
addition, Bax and Bcl-2 regulate cell survival or resistance by
promoting signal transduction (4).
CASP-3 exists as a precursor and functions following its cleavage
to cleaved CASP-3(31).
Furthermore, the expression levels of CASP-3 mRNA were identified
to be upregulated in association with the occurrence of apoptosis
(32). Therefore, the present study
investigated the relationship between Gas and the expression levels
of the apoptosis-related proteins, Bax, Bcl-2 and CASP-3. The
experimental results revealed that Gas pretreatment may reduce the
incidence of hepatocyte apoptosis.
In conclusion, the findings of the present study
revealed that Gas pretreatment increased the content of GSH in the
liver, enhanced the activity of SOD, upregulated the expression
levels of antioxidant-related genes Nrf2 and HO-1, and regulated
the expression levels of apoptosis-related proteins, Bax, Bcl -2
and CASP-3, thereby significantly alleviating HIRI. These results
may provide insights into a novel therapeutic strategy for the
clinical treatment and prevention of liver failure following liver
surgery.
Supplementary Material
Chemical structural formula of
gastrodin.
Suzuki's criteria of hepatic ischemic
reperfusion.
Reverse transcription-quantitative PCR
primer sequences.
Primer information.
Acknowledgements
The authors would like to thank pathologists (Dr
Xuyou Zhu, Dr Tingting Zhang and Dr Yu Zeng) of the Pathology
Department of Shanghai Tongji Hospital, Shanghai, China, for
scoring the experimental samples.
Funding
The present study was supported by grants from the National
Natural Science Foundation of China (grant no. 81873567) and the
Shanghai Municipal Commission of Health and Family Planning (grant
no. WSJ1607).
Availability of data and materials
The data used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
WY and RL conceived and designed the study. BS, JJ
and XZ collected the tissue samples and established the mouse
model. DY, ZC, YZ, MZ and YQ performed the experiments and acquired
the data. BS and WY confirm the authenticity of all the raw data.
WY, RL, XZ, BS and JJ interpreted the data and prepared and revised
the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Animal Ethics
Committee of Tongji University (approval no. TJHBLAC-2019-116;
Shanghai, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dery KJ, Nakamura K, Kadono K, Hirao H,
Kageyama S, Ito T, Kojima H, Kaldas FM, Busuttil RW and
Kupiec-Weglinski JW: Human Antigen R (HuR): A new regulator of heme
oxygenase-1 cytoprotection in mouse and human liver transplant
injury. Hepatology. 72:1056–1072. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Covington SM, Bauler LD and Toledo-Pereyra
LH: Akt: A therapeutic target in hepatic ischemia-reperfusion
injury. J Invest Surg. 30:47–55. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Peralta C, Jimenez-Castro MB and
Gracia-Sancho J: Hepatic ischemia and reperfusion injury: Effects
on the liver sinusoidal milieu. J Hepatol. 59:1094–1106.
2013.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Han X, Shi H, Liu K, Zhong L, Wang F and
You Q: Protective effect of gastrodin on myocardial
ischemia-reperfusion injury and the expression of Bax and Bcl-2.
Exp Ther Med. 17:4389–4394. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ju C, Colgan SP and Eltzschig HK:
Hypoxia-inducible factors as molecular targets for liver diseases.
J Mol Med (Berl). 94:613–627. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Qiu M, Xiao F, Wang T, Piao S, Zhao W,
Shao S, Yan M and Zhao D: Protective effect of Hedansanqi Tiaozhi
Tang against non-alcoholic fatty liver disease in vitro and in vivo
through activating Nrf2/HO-1 antioxidant signaling pathway.
Phytomedicine. 67(153140)2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Theruvath TP, Czerny C, Ramshesh VK, Zhong
Z, Chavin KD and Lemasters JJ: C-Jun N-terminal kinase 2 promotes
graft injury via the mitochondrial permeability transition after
mouse liver transplantation. Am J Transplant. 8:1819–1828.
2008.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Matias M, Silvestre S, Falcao A and Alves
G: Gastrodia elata and epilepsy: Rationale and therapeutic
potential. Phytomedicine. 23:1511–1526. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Li JJ, Liu SJ, Liu XY and Ling EA: Herbal
compounds with special reference to gastrodin as potential
therapeutic agents for microglia mediated neuroinflammation. Curr
Med Chem. 25:5958–5974. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hu Y, Li C and Shen W: Gastrodin
alleviates memory deficits and reduces neuropathology in a mouse
model of Alzheimer's disease. Neuropathology. 34:370–377.
2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Matsumata T, Kanematsu T, Shirabe K,
Yamagata M, Utsunomiya T, Furuta T and Sugimachi K: Modified
technique of pringle's maneuver in resection of the liver. Surg
Gynecol Obstet. 172:245–246. 1991.PubMed/NCBI
|
|
12
|
Li Y, Li T and Qi H: Protective effect of
minocycline on hepatic ischemia-reperfusion injury in rats. Zhong
Nan Da Xue Xue Bao Yi Xue Ban. 39:1137–1144. 2014.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Smedsrød B, De Bleser PJ, Braet F,
Lovisetti P, Vanderkerken K, Wisse E and Geerts A: Cell biology of
liver endothelial and Kupffer cells. Gut. 35:1509–1516.
1994.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kaplowitz N: Drug-induced liver injury.
Clin Infect Dis. 38 (Suppl 2):S44–S48. 2004.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Senoner T, Schindler S, Stattner S, Ofner
D, Troppmair J and Primavesi F: Associations of oxidative stress
and postoperative outcome in liver surgery with an outlook to
future potential therapeutic options. Oxid Med Cell Longev.
2019(3950818)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yang P, Han Y, Gui L, Sun J, Chen YL, Song
R, Guo JZ, Xie YN, Lu D and Sun L: Gastrodin attenuation of the
inflammatory response in H9c2 cardiomyocytes involves inhibition of
NF-κB and MAPKs activation via the phosphatidylinositol 3-kinase
signaling. Biochem Pharmacol. 85:1124–1133. 2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Chen Pz, Jiang Hh, Wen B, Ren SC, Chen Y,
Ji WG, Hu B, Zhang J, Xu F and Zhu ZR: Gastrodin suppresses the
amyloid β-induced increase of spontaneous discharge in the
entorhinal cortex of rats. Neural Plast.
2014(320937)2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhan HD, Zhou HY, Sui YP, Du XL, Wang WH,
Dai L, Sui F, Huo HR and Jiang TL: The rhizome of Gastrodia elata
Blume-An ethnopharmacological review. J Ethnopharmacol.
189:361–385. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Qu LL, Yu B, Li Z, Jiang WX, Jiang JD and
Kong WJ: Gastrodin ameliorates oxidative stress and proinflammatory
response in nonalcoholic fatty liver disease through the AMPK/Nrf2
pathway. Phytother Res. 30:402–411. 2016.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Dai JN, Zong Y, Zhong LM, Li YM, Zhang W,
Bian LG, Ai QL, Liu YD, Sun J and Lu D: Gastrodin inhibits
expression of inducible NO synthase, cyclooxygenase-2 and
proinflammatory cytokines in cultured LPS-stimulated microglia via
MAPK pathways. PLoS One. 6(e21891)2011.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Peng Z, Wang S, Chen G, Cai M, Liu R, Deng
J, Liu J, Zhang T, Tan Q and Hai C: Gastrodin alleviates cerebral
ischemic damage in mice by improving anti-oxidant and
anti-inflammation activities and inhibiting apoptosis pathway.
Neurochem Res. 40:661–673. 2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Masuda Y, Vaziri ND, Takasu C, Li S,
Robles L, Pham C, Le A, Vo K, Farzaneh SH, Stamos MJ and Ichii H:
Salutary effect of pre-treatment with an Nrf2 inducer on ischemia
reperfusion injury in the rat liver. Gastroenterol Hepatol (Que).
1:1–7. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
24
|
Chen Z, Zhong H, Wei J, Lin S, Zong Z,
Gong F, Huang X, Sun J, Li P, Lin H, et al: Inhibition of Nrf2/HO-1
signaling leads to increased activation of the NLRP3 inflammasome
in osteoarthritis. Arthritis Res Ther. 21(300)2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tanaka Y, Maher JM, Chen C and Klaassen
CD: Hepatic ischemia-reperfusion induces renal heme oxygenase-1 via
NF-E2-related factor 2 in rats and mice. Mol Pharmacol. 71:817–825.
2007.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Bauer M and Bauer I: Heme oxygenase-1:
Redox regulation and role in the hepatic response to oxidative
stress. Antioxid Redox Signal. 4:749–758. 2002.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Schwabe RF and Luedde T: Apoptosis and
necroptosis in the liver: A matter of life and death. Nat Rev
Gastroenterol Hepatol. 15:738–752. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Lei K and Davis RJ: JNK phosphorylation of
Bim-related members of the Bcl2 family induces Bax-dependent
apoptosis. Proc Natl Acad Sci USA. 100:2432–2437. 2003.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Tournier C, Hess P, Yang DD, Xu J, Turner
TK, Nimnual A, Bar-Sagi D, Jones SN, Flavell RA and Davis RJ:
Requirement of JNK for stress-induced activation of the cytochrome
c-mediated death pathway. Science. 288:870–874. 2000.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Liu Q, Rehman H, Krishnasamy Y,
Schnellmann RG, Lemasters JJ and Zhong Z: Improvement of liver
injury and survival by JNK2 and iNOS deficiency in liver
transplants from cardiac death mice. J Hepatol. 63:68–74.
2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Shalini S, Dorstyn L, Dawar S and Kumar S:
Old, new and emerging functions of caspases. Cell Death Differ.
22:526–539. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Lossi L, Castagna C and Merighi A:
Caspase-3 mediated cell death in the normal development of the
mammalian cerebellum. Int J Mol Sci. 19(3999)2018.PubMed/NCBI View Article : Google Scholar
|