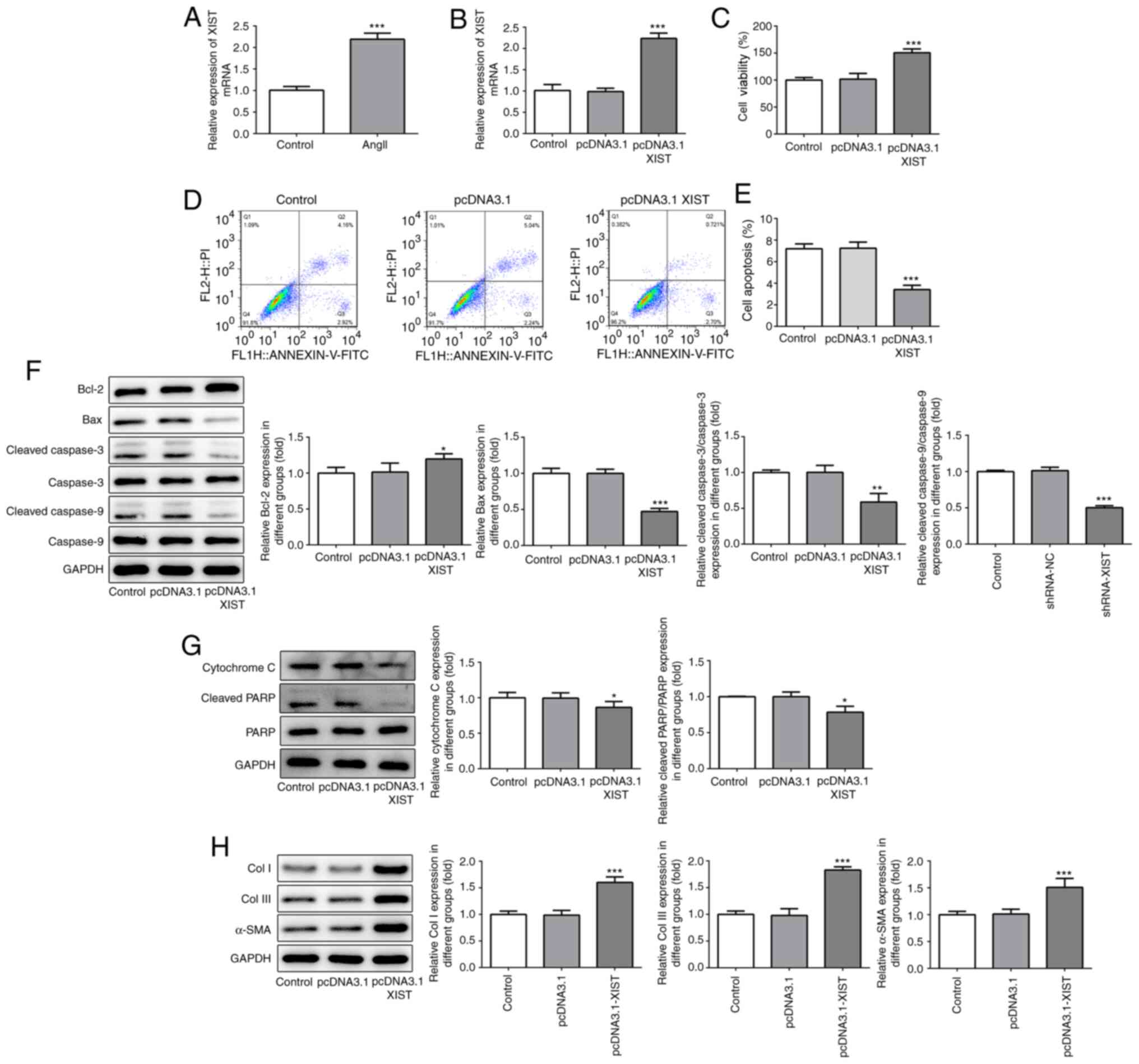
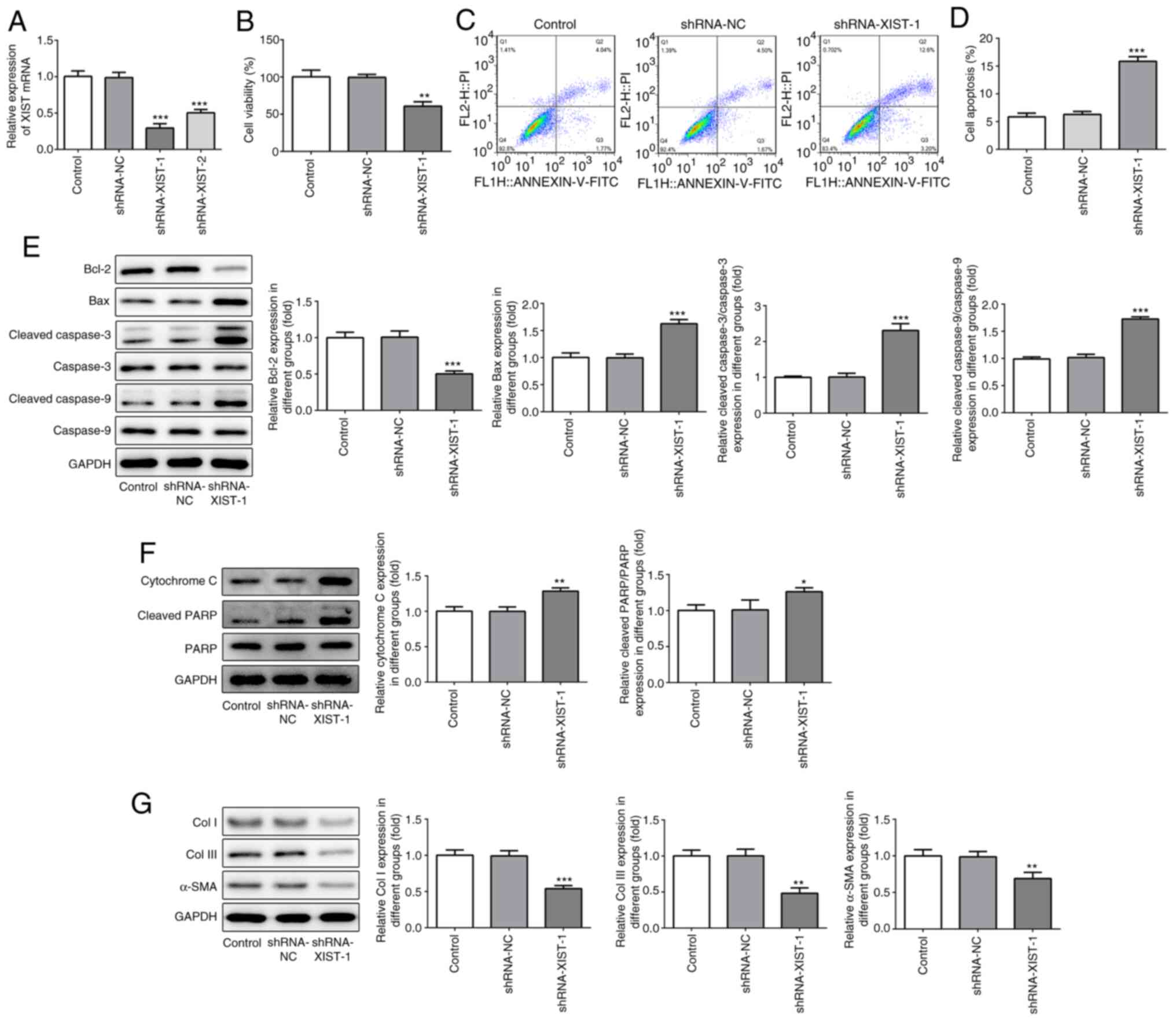
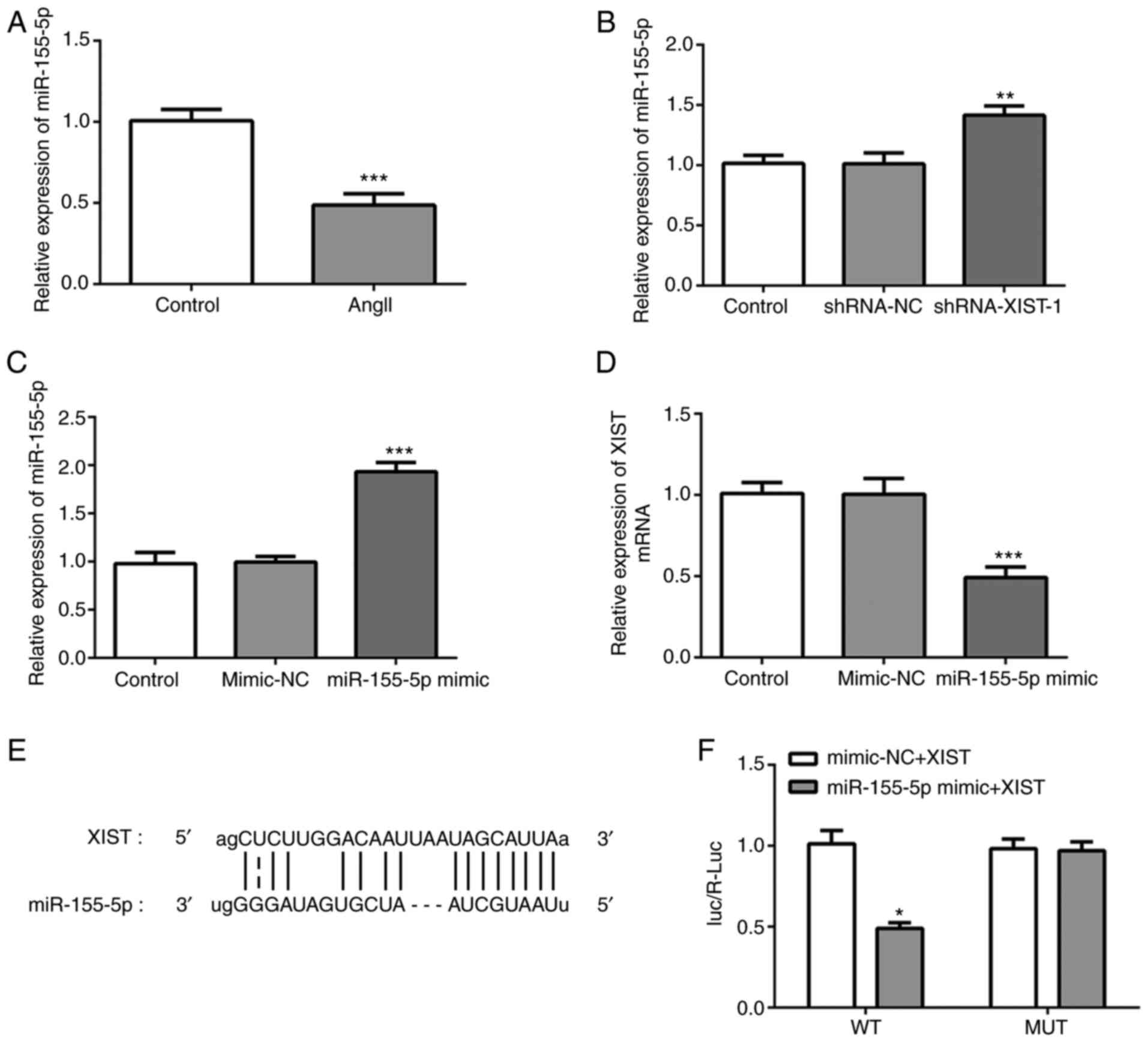
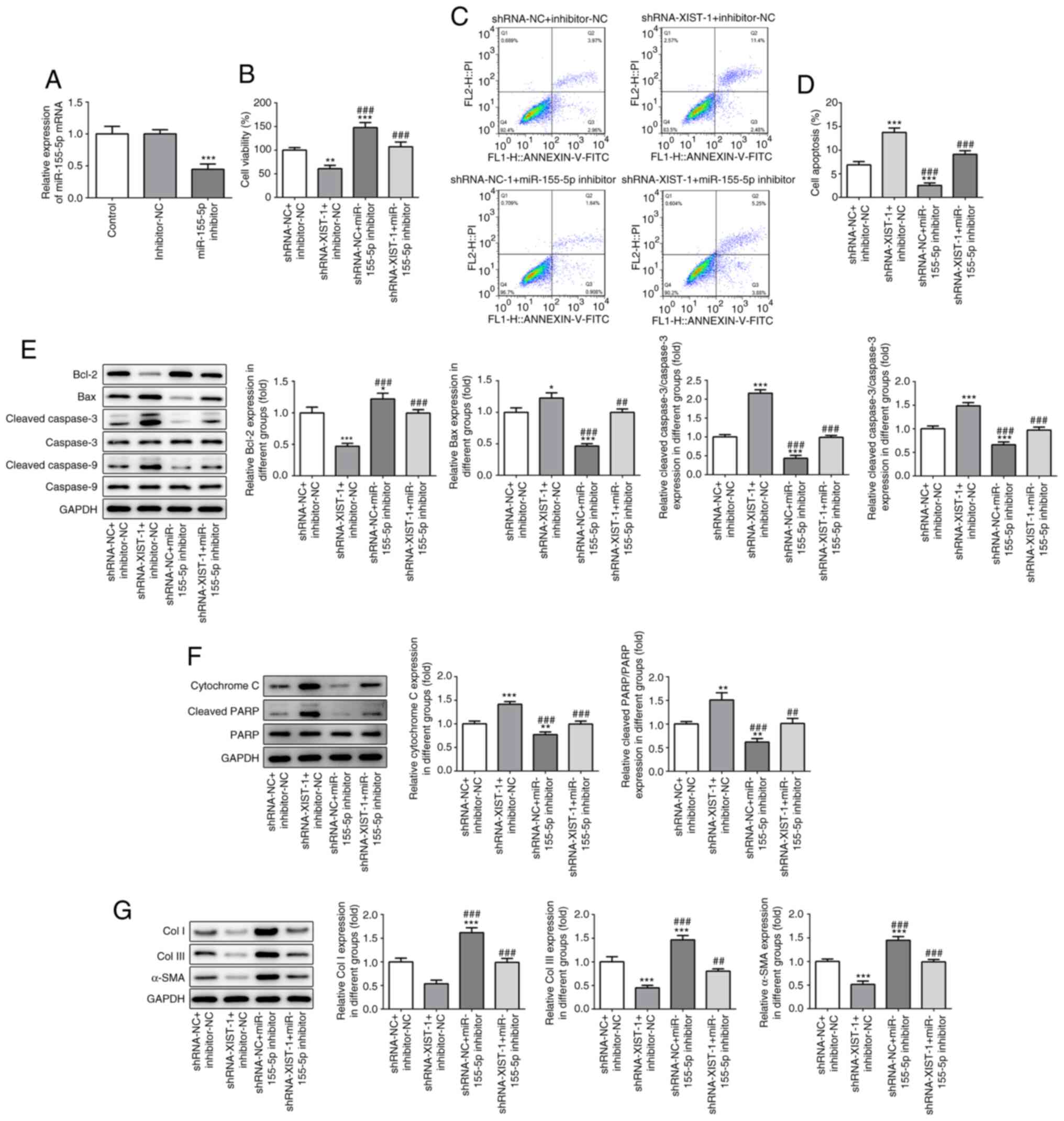
|
1
|
Davidson SM, Ferdinandy P, Andreadou I,
Bøtker HE, Heusch G, Ibáñez B, Ovize M, Schulz R, Yellon DM,
Hausenloy DJ, et al: Multitarget strategies to reduce myocardial
ischemia/reperfusion injury: JACC review topic of the week. J Am
Coll Cardiol. 73:89–99. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kurose H and Mangmool S: Myofibroblasts
and inflammatory cells as players of cardiac fibrosis. Arch Pharm
Res. 39:1100–1113. 2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Cho N, Razipour SE and McCain ML: Featured
Article: TGF-β1 1 dominates extracellular matrix rigidity for
inducing differentiation of human cardiac fibroblasts to
myofibroblasts. Exp Biol Med (Maywood). 243:601–612.
2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Yuan X, Pan J, Wen L, Gong B, Li J, Gao H,
Tan W, Liang S, Zhang H and Wang X: MiR-144-3p enhances cardiac
fibrosis after myocardial infarction by targeting PTEN. Front Cell
Dev Biol. 7(249)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Puvvula PK: LncRNAs regulatory networks in
cellular senescence. Int J Mol Sci. 20(2615)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Yarani R, Mirza AH, Kaur S and Pociot F:
The emerging role of lncRNAs in inflammatory bowel disease. Exp Mol
Med. 50:1–14. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Xiao L, Gu Y, Sun Y, Chen J, Wang X, Zhang
Y, Gao L and Li L: The long noncoding RNA XIST regulates cardiac
hypertrophy by targeting miR-101. J Cell Physiol. 234:13680–13692.
2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
McKiernan PJ, Molloy K, Cryan SA,
McElvaney NG and Greene CM: Long noncoding RNA are aberrantly
expressed in vivo in the cystic fibrosis bronchial epithelium. Int
J Biochem Cell Biol. 52:184–191. 2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Wang Y, Liang Y, Luo J, Nie J, Yin H, Chen
Q, Dong J, Zhu J, Xia J and Shuai W: XIST/miR-139 axis regulates
bleomycin (BLM)-induced extracellular matrix (ECM) and pulmonary
fibrosis through β-catenin. Oncotarget. 8:65359–65369.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yang J, Shen Y, Yang X, Long Y, Chen S,
Lin X, Dong R and Yuan J: Silencing of long noncoding RNA XIST
protects against renal interstitial fibrosis in diabetic
nephropathy via microRNA-93-5p-mediated inhibition of CDKN1A. Am J
Physiol Renal Physiol. 317:F1350–F1358. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Liu Y, Gao L, Guo S, Zhao X, Li R, Yan X,
Li Y, Wang S, Niu X, Yao L, et al: Kaempferol alleviates
angiotensin II-induced cardiac dysfunction and interstitial
fibrosis in mice. Cell Physiol Biochem. 43:2253–2263.
2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wang ZF, Wang NP, Harmouche S, Philip T,
Pang XF, Bai F and Zhao ZQ: Postconditioning attenuates coronary
perivascular and interstitial fibrosis through modulating
angiotensin II receptors and Angiotensin-converting enzyme 2 after
myocardial infarction. J Surg Res. 211:178–190. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Lin C, Wei D, Xin D, Pan J and Huang M:
Ellagic acid inhibits proliferation and migration of cardiac
fibroblasts by Down-regulating expression of HDAC1. J Toxicol Sci.
44:425–433. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Tang CM, Zhang M, Huang L, Hu ZQ, Zhu JN,
Xiao Z, Zhang Z, Lin QX, Zheng XL, Yang M, et al: CircRNA_000203
enhances the expression of fibrosis-associated genes by
derepressing targets of miR-26b-5p, Col1a2 and CTGF, in cardiac
fibroblasts. Sci Rep. 7(40342)2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zhao N, Yu H, Sun M, Zhang Y, Xu M and Gao
W: MiRNA-711-SP1-collagen-I pathway is involved in the
anti-fibrotic effect of pioglitazone in myocardial infarction. Sci
China Life Sci. 56:431–439. 2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Maruyama T, Nishihara K, Umikawa M,
Arasaki A, Nakasone T, Nimura F, Matayoshi A, Takei K, Nakachi S,
Kariya KI, et al: MicroRNA-196a-5p is a potential prognostic marker
of delayed lymph node metastasis in early-stage tongue squamous
cell carcinoma. Oncol Lett. 15:2349–2363. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Castro-Dominguez Y, Dharmarajan K and
McNamara RL: Predicting death after acute myocardial infarction.
Trends Cardiovasc Med. 28:102–109. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yin Y, Zhang Q, Zhao Q, Ding G, Wei C,
Chang L, Li H, Bei H, Wang H, Liang J, et al: Tongxinluo attenuates
myocardiac fibrosis after acute myocardial infarction in rats via
inhibition of endothelial-to-mesenchymal transition. Biomed Res
Int. 2019(6595437)2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
van Putten S, Shafieyan Y and Hinz B:
Mechanical control of cardiac myofibroblasts. J Mol Cell Cardiol.
93:133–142. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Xie ZY, Wang FF, Xiao ZH, Liu SF, Lai YL
and Tang SL: Long noncoding RNA XIST enhances ethanol-induced
hepatic stellate cells autophagy and activation via miR-29b/HMGB1
axis. IUBMB Life. 71:1962–1972. 2019.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Chen L, Zheng SY, Yang CQ, Ma BM and Jiang
D: MiR-155-5p inhibits the proliferation and migration of VSMCs and
HUVECs in atherosclerosis by targeting AKT1. Eur Rev Med Pharmacol
Sci. 23:2223–2233. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhang Z, Liang K, Zou G, Chen X, Shi S,
Wang G, Zhang K, Li K and Zhai S: Inhibition of miR-155 attenuates
abdominal aortic aneurysm in mice by regulating macrophage-mediated
inflammation. Biosci Rep. 38(BSR20171432)2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chen NX, Kiattisunthorn K, O'Neill KD,
Chen X, Moorthi RN, Gattone VH II, Allen MR and Moe SM: Decreased
microRNA is involved in the vascular remodeling abnormalities in
chronic kidney disease (CKD). PLoS One. 8(e64558)2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhang G, Shi H, Wang L, Zhou M, Wang Z,
Liu X, Cheng L, Li W and Li X: MicroRNA and transcription factor
mediated regulatory network analysis reveals critical regulators
and regulatory modules in myocardial infarction. PLoS One.
10(e0135339)2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhao F, Wu Y, Yang W, Wu D, Wang C and
Zhang F: Inhibition of vascular calcification by microRNA-155-5p is
accompanied by the inactivation of TGF-β1/Smad2/3 signaling
pathway. Acta Histochem. 122(151551)2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Halper J and Kjaer M: Basic components of
connective tissues and extracellular matrix: Elastin, fibrillin,
fibulins, fibrinogen, fibronectin, laminin, tenascins and
thrombospondins. Adv Exp Med Biol. 802:31–47. 2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Jabłońska-Trypuć A, Matejczyk M and
Rosochacki S: Matrix metalloproteinases (MMPs), the main
extracellular matrix (ECM) enzymes in collagen degradation, as a
target for anticancer drugs. J Enzyme Inhib Med Chem. 31 (Suppl
1):S177–S183. 2016.PubMed/NCBI View Article : Google Scholar
|