1. Introduction
With the marked progress of molecular and cellular
research and technology over the last few decades, notable advances
have been made in the elucidation of the mechanisms underlying
tumor development. The focus of cancer therapy has shifted from
traditional therapy, including surgery, radiotherapy and
chemotherapy (1), to biotherapy,
including cancer immunotherapy, which was declared the annual
scientific breakthrough in 2013 by Science (2).
Cancer immunotherapy has incomparable advantages
over traditional cancer treatment, which may prolong the
progression-free survival and overall survival of patients by
unleashing an immune system attack against tumor cells in multiple
targets and directions (3).
Immunotherapy, including immune checkpoint
inhibitors, adoptive cellular therapy and tumor vaccines, is
considered a milestone in cancer therapeutic strategies and is
leading to a paradigm transformation in cancer management (4). Chimeric antigen receptor (CAR) T-cell
therapy is a novel approach to cell adoptive immunotherapy after
the advent of cell therapies, including the use of
tumor-infiltrating lymphocytes, cytokine-induced killer (CIK)
cells, natural killer cells, dendritic cells (DC)-CIK cells and
T-cell receptor-engineered T-cells.
To date, the US Food and Drug Administration (FDA)
and European Medicines Agency have approved two CAR T-cell
therapies, Novartis' Kymriah (tisagenlecleucel; CTL019) and Kite's
Yescarta (Axicabtagene ciloleucel; KTE-C19) (5), of which Kymriah was aimed to treat
relapsed B-cell acute lymphoblastic leukemia and Yescarta was
indicated for the treatment of adult patients with relapsed or
refractory large B-cell lymphoma (6). Furthermore, hundreds of novel CAR
T-cell therapy clinical trials are underway worldwide with
government support, indicating that CAR T-cell therapy has become
widely accepted by both official agencies and the public due to its
marked efficacy in treating certain malignancies (7,8).
The aim of the present review was to provide an
overview of the principle and clinical application of CAR T-cell
therapies prior to addressing major challenges and proposing
possible solutions and a future outlook.
2. CAR T-cell therapy and the construction
of CAR molecules
CAR T-cells are extracted autologous or allogeneic
T-cells that have been genetically modified to express major
histocompatibility complex-unrestricted and antigen-specific
receptors on the surface, which are able to recognize and attack
antigen-bearing tumor cells after reinfusion (9). In brief, CAR T-cell therapy is a
method that activates the patient's own immune system to eradicate
cancer cells.
The accuracy and efficiency of CAR T-cell therapy
largely depend on the structure of the CARs. CARs consist of an
extracellular antigen recognition domain, hinge region and
transmembrane domain, which is riveted to an intracellular
signaling domain. The extracellular domain is usually a
single-chain variable fragment (scFv) derived from an antibody,
with the ability to specifically target tumor cells with the
tumor-specific antigen. The antibodies are usually derived from
humans or mice or from humanization of murine antibodies. The hinge
is the crystalline fragment (Fc) region of immunoglobulin (Ig),
mediating the antigen-antibody reaction and the elimination of
antibodies. At the same time, it may provide a flexible connection
between binding sites and contribute to the binding affinity. The
transmembrane domain has the role of an anchor to the cell membrane
and connects the ectodomain and endodomain, which affects the
transmission of activating signals (10). The intracellular signaling domain,
consisting of costimulatory domains and T-cell activation moieties,
is involved in signal transduction, which is initiated by the
binding of the extracellular domain to the antigen, in order to
activate the immune system to eliminate the target cell (11).
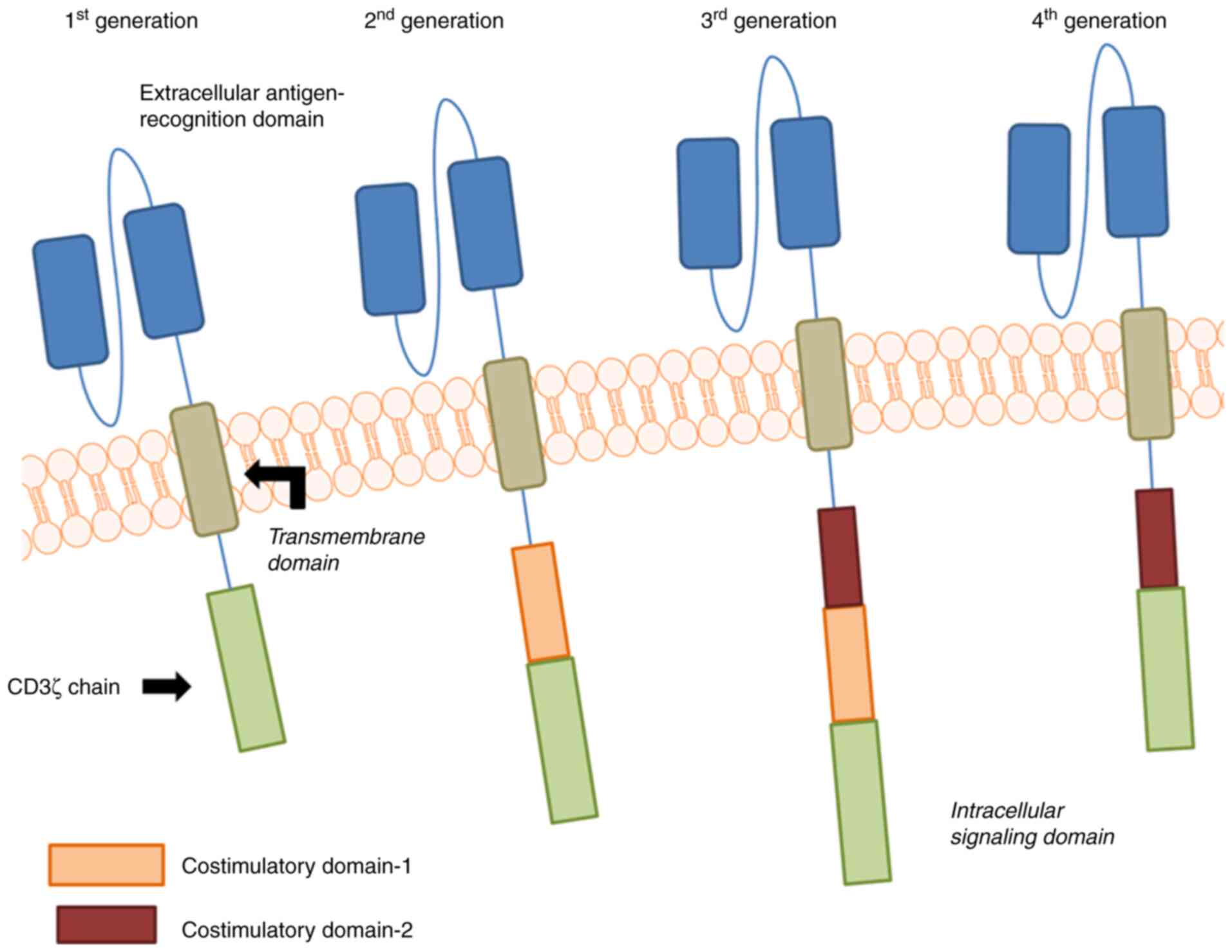
CARs may be classified into four generations: The
first generation is a fusion protein linked to the extracellular
scFv and CD3ζ chain of the T-cell activation signal region, but
cells lack long-term survival and robust proliferation. Therefore,
by incorporating costimulatory endodomain molecules into CD3ζ, such
as 41BB and/or CD28, the persistence and antitumor ability of CAR
T-cells in the body have been significantly improved (12). Of note, according to whether one or
two co-stimulatory domains are added, the CARs are referred to as
second- or third-generation CARs, respectively (Fig. 1) (13).
Until the emergence of the fourth generation of CAR
T-cells redirected for universal cytokine-mediated killing, the
exploration of the structure of CARs advanced a step further by
adding an encoded vector on the basis of the previous structure to
induce the expression of corresponding cytokines and enhance other
effectors of the immune system in order to release cytokines in the
immunosuppressive tumor microenvironment to recruit and activate
more immune cells to strengthen the antitumor activity of CAR
T-cells (14). Consequently, due to
the high efficacy and selectivity, CAR T-cell therapy is becoming a
promising novel therapeutic strategy which is inspiring increased
research efforts.
3. Clinical applications of CAR T-cell
therapy
Hematological malignancies
CAR T-cell therapy holds great promise for
hematological malignancies and is widely used in the management of
lymphoma, leukemia and myeloma (15).
In August 2017, the first CAR T-cell therapy,
Kymriah, was historically approved for the treatment of certain
pediatric and young adult patients with B-cell acute lymphoblastic
leukemia (B-ALL), with an overall response rate of 52% in a
clinical trial (16). In October of
the same year, a second CAR T-cell therapy named Yescarta was
approved for the treatment of adult patients with certain types of
relapsed/refractory (R/R) B-cell non-Hodgkin lymphoma. Both these
therapies are anti-CD19 CAR T-cell therapies with the ability to
induce durable responses.
CD19 is a member of the Ig superfamily and is a type
of transmembrane glycoprotein and a biomarker for lymphoma
primarily expressed on malignant B-cells (17). As a popular immunotherapy target,
CD19 is widely used in the treatment of hematological malignancies
for its considerable efficacy. In a clinical trial using anti-CD19
CAR T-cell therapy for ALL, the response rate achieved was up to
81% with a complete remission rate of 78% (18).
Furthermore, CD-19-targeted CAR T-cell therapy may
be applied for treating patients with chronic lymphocytic leukemia
(CLL), but at present, it is mostly used in clinical trials and is
lacking official approval (19). As
a B-cell malignancy, CLL is the most common type of adult leukemia
in western countries and is associated with defects of the immune
system. There are several potentially effective treatment options
available, but CLL cannot be cured with traditional therapies and
the progression of the disease is inevitable. A previous study
demonstrated that in patients with R/R CLL, CD19-directed CAR
T-cells were able to clear tumor cells in the body with an overall
response rate of 57% (20).
In addition to the CD19 antigen, novel targets are
being continuously identified. In a clinical trial of CAR T-cell
therapy targeting CD22, 73% (11/15) of B-ALL patients achieved
complete remission after receiving anti-CD22 CAR T-cell therapy
(21). A retrospective analysis
demonstrated that conventional CD19 may be combined with aberrant
antigens, such as CD123, to form CARs with bi-specific antigens,
which may significantly improve the efficacy when treating R/R
B-ALL (22). Furthermore, CD123 is
considered a promising target for acute myeloid leukemia (AML)
(23). It was previously
demonstrated that CD33-targeted CAR T-cells may clear leukemia
without causing myelosuppression in the treatment of AML (24).
In addition to leukemia, CAR T-cell therapy also
demonstrated great potential in the management of multiple myeloma
(MM). MM is a hematological malignancy with the second-highest
annual incidence among hematological tumors, caused by gene
mutations during B-lymphocyte differentiation into plasma cells. In
an experiment using biepitope-targeting CAR T-cells against B-cell
maturation antigen (BCMA) in 17 cases of R/R MM, the overall
response rate reached 88.2% (25).
As a transmembrane glycoprotein, BCMA belongs to the tumor necrosis
factor receptor superfamily, which has an important role in
long-term plasma cell survival and B-cell differentiation into
plasma cells. In addition, BCMA is important for the survival of
malignant plasma cells and its upregulation may promote MM-cell
growth, while its downregulation inhibits MM-cell proliferation.
Furthermore, BCMA is usually expressed at higher levels in
malignant plasma cells compared to normal plasma cells, which may
be an important factor for the effectiveness of CAR T-cell therapy
when applied to patients with MM (26). Other clinical trials also reported
promising results. A clinical study including patients with MM who
received BCMA-targeting CAR T-cell therapy reported an overall
response rate of 81% (27). In
addition, CD38 and CD138 may be potential targets for MM treatment
strategies (28,29).
Solid tumors
CAR T-cell therapy has achieved great successes in
treating hematological malignancies, but its application in solid
tumors has not been as successful due to the lack of optimal
antigens and the immunosuppressive tumor microenvironment
(TME).
Continuous research and ongoing experiments aim at
constructing solid tumor-associated antigens. For instance, CAR
T-cells against human epidermal growth factor receptor 2 (HER2) may
not only recognize and kill HER2+ breast cancer cells
but also induce regression of experimental breast cancer in
vivo (30). In addition, a
previous study demonstrated that phosphatidylinositol 3
(GPC3)-targeted CAR T-cell therapy may also be a potential
treatment for liver cancer (31).
In a recent study, a CAR (hYP7) T-cell model was constructed, which
was able to eliminate GPC3-positive hepatocellular cancer (HCC)
cells, possibly by inducing perforin- and granzyme-mediated
apoptosis or reducing Wnt signaling in tumor cells (32), emphasizing the potential of CAR
T-cell therapy targeting GPC3 in the treatment of patients with
HCC. In addition, GPC3-targeting CAR T-cells expressing IL-12 have
been constructed, which may significantly enhance the function of
CAR T-cells with relatively fewer side effects (33).
A study demonstrated that prostate-specific membrane
antigen and MUC1 may serve as potential targets in non-small cell
lung cancer (34). Furthermore,
mesothelin, anthrax toxin receptor 1 (ANTXR1; also known as tumor
endothelial marker 8) and mucin 3A are possible targets for CAR
T-cell therapy in gastric adenocarcinomas (35). Anti-CD22/anti-CD19 CAR T-cell
therapy has been successfully used to treat R/R B-cell lymphoma
involving the gastrointestinal tract. Out of 14 patients, 10
achieved an objective response and 7 patients achieved a complete
response (36). Previous studies
have indicated that programmed cell death protein 1 knockout may
enhance the lytic activity of epidermal growth factor receptor
variant III (EGFRvIII)-CAR T-cells against programmed death-ligand
1 EGFRvIII glioblastoma multiforme (GBM) cells, which may provide a
novel therapeutic strategy for GBM (37). In addition, CAR T-cell therapy is
also applied to treat solid tumor types, including pancreatic
cancer, renal cell carcinoma, ovarian cancer, colorectal cancer and
melanoma (38).
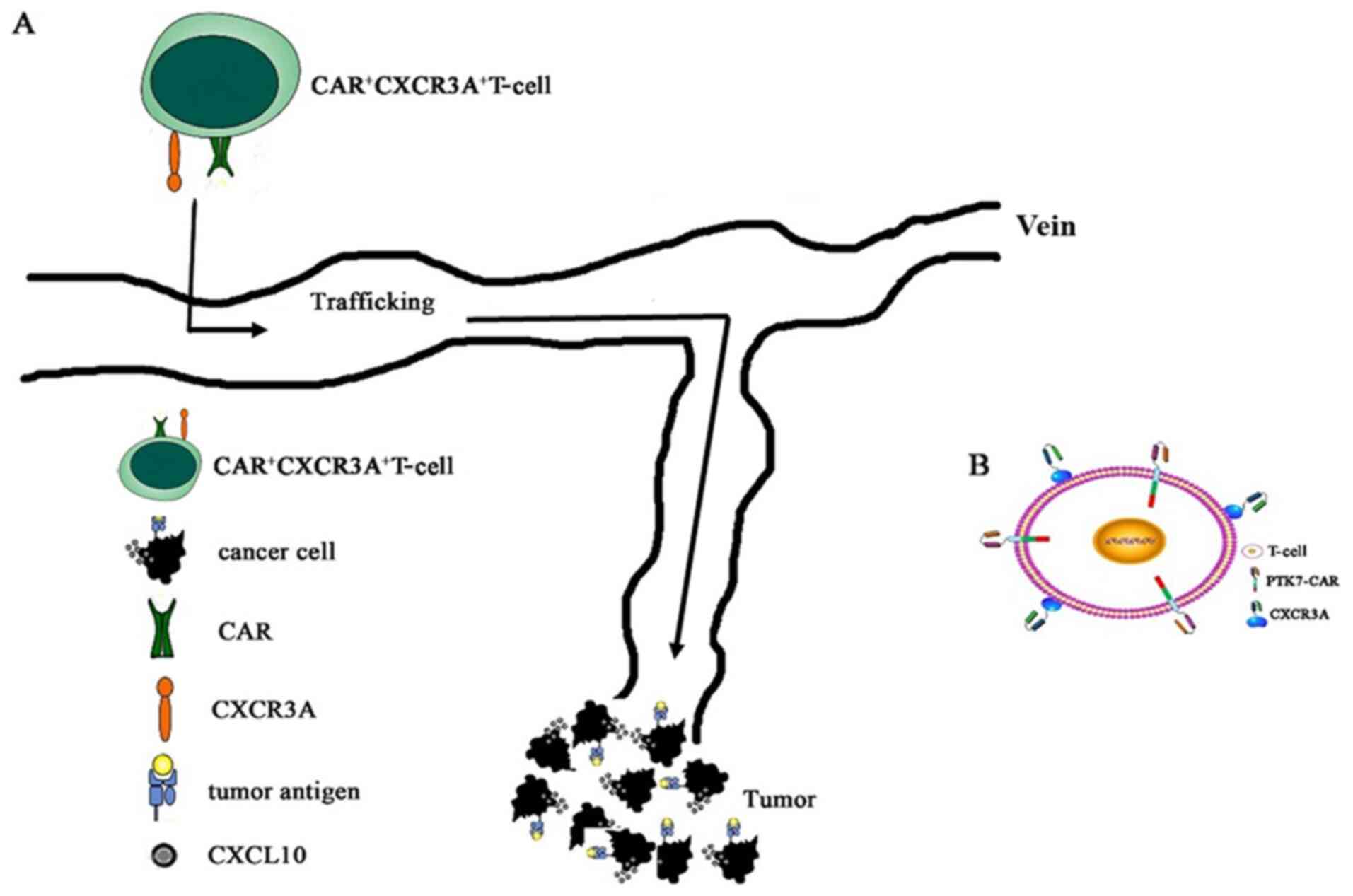
Regarding the use of CAR T-cells for the management
of solid tumors, our group has also performed various preliminary
investigations. A number of tumor antigens have been screened to be
used for CAR T-cell therapy, among which, our previous study
constructed a humanized protein tyrosine kinase 7-CAR through
genetic engineering to perform genetic modification of lymphocytes
(Fig. 2) (39).
4. Current challenges and treatment
principles of CAR T-cell therapy
Cytokine release syndrome (CRS)
CRS is a systemic inflammatory state that occurs as
a result of robust immune activation. As one of the most frequent
serious adverse events associated with CAR T-cell therapy, CRS is
reported in patients who receive CAR T-cell therapy with an
incidence of 57-93%, depending on the type of immunotherapy and the
disease burden (40).
CRS is mainly triggered by a large number of
cytokines and inflammatory factors, and interleukin (IL)-6 has a
key role in mediating CRS. However, the levels of other cytokines
also increase over the course of CRS, including IL-2, interferon-γ,
tumor necrosis factor, GM-CSF, IL-5, IL-8 and IL-10(41).
As a systemic inflammatory response, CRS involves
multiple systems and has various aspects, such as circulatory,
breathing, urinary and digestive system disorders. The patient may
develop fever, skin rash, cardiac dysfunction, respiratory failure,
renal failure, nausea, vomiting, hepatic dysfunction, disseminated
intravascular coagulopathy and neurotoxicity after undergoing CAR
T-cell therapy (42,43).
Although most cases of CRS are self-limited, it may
prove life-threatening without timely and effective treatment
(44). For instance, respiratory
symptoms are common in mild cases. CRS may manifest as cough and
tachypnea, but it may progress to acute respiratory distress
syndrome with hypoxemia and dyspnea. When respiratory dysfunction
occurs, mechanical ventilation is usually required. Similarly, if
cardiovascular (CV) side effects occur, patients must be promptly
transferred to the intensive care unit where fluids and pressors
may be administered to effectively manage tachycardia and
hypotension (45). A study on CRS
and cardiotoxicity caused by CAR T-cell therapy reported that heart
injury and CV events are common in adult patients following CAR
T-cell therapy, but a shorter time from CRS onset to the
utilization of tocilizumab was associated with a lower rate of CV
events (46). Therefore, it is
crucial to ensure timely detection and treatment for patients
receiving CAR T-cell to prevent further damage to the body from
CRS.
Of note, patients with mild and moderate symptoms
may require supportive measures, such as antibiotics and
antipyretics, while tocilizumab is usually required in severe
cases. As the first-line medicine in the clinical treatment of CRS,
tocilizumab may ameliorate CRS by antagonising IL-6(47). Furthermore, tocilizumab has already
demonstrated clinical efficacy in treating CRS, without
compromising the antitumor effects, and clinical improvement is
usually observed within hours. As a consequence, tocilizumab was
approved by the FDA in August 2017 and is currently considered as
the mainstay of treatment for severe CRS (48).
Of note, both tocilizumab and steroids may be used
to treat CRS. However, the use of tocilizumab is favored over
corticosteroids, as the latter may adversely affect the treatment
efficacy (49).
Regarding the treatment of R/R chronic leukemia,
studies have confirmed that the combination of ibrutinib and
anti-CD19 CAR T-cell immunotherapy was able to effectively improve
the antitumor efficacy of CAR T-cells and reduce the incidence of
CRS (50). The use of dual-target
chimeric antigen receptor T-cells may also be considered. A case
report on a patient with R/R primary central nervous system (PCNS)
diffuse large B-cell lymphoma treated with anti-CD19- and
anti-CD70-specific CAR T-cell therapy indicated that CAR T-cells
were able to effectively target PCNS lymphomas and maintain
disease-free survival without inducing CRS (51). Another study reported that, with the
use of real-time monitoring of cytokines, CAR T-cell therapy
against CD19 may be safely applied in patients with R/R leukemia
(52).
Immune effector-related neurotoxicity
syndrome (ICANS)
ICANS is the second most common complication of CAR
T-cell therapy with an incidence of 12-55%.
ICANS may manifest with a diverse range of symptoms,
spanning from mild confusion with tremor, dysgraphia, mild
expressive aphasia and apraxia, to seizures, somnolence, global
aphasia and, in a minority of cases, death (53,54).
Fatal cerebral edema has also been reported (55). Another previous study indicated that
patients with a higher tumor burden were more likely to develop
neurotoxic symptoms when receiving high-dose CAR T-cell therapy. Of
the 25 adult patients who developed ICANS following CAR T-cell
therapy, 12 (48%) developed grade 1-2 neurotoxicity, whereas 13
(52%) presented with grade 3-4 neurotoxicity (56). In a phase I study including 53 adult
patients with B-ALL treated with CD19 CAR T-cell therapy, grade 1
and 2 neurotoxicity was observed in 11 patients, and grade 3 and 4
neurotoxicity was observed in 22 patients (57). Although the pathophysiology of the
neurotoxicity has yet to be fully elucidated, there is evidence of
breakdown of the blood-brain barrier in patients with severe
neurotoxicity and the protein concentration in the cerebrospinal
fluid (CSF) is correlated with the grade of neurotoxicity,
indicating that there is increased permeability of the blood-CSF
barrier during ICANS. It was previously reported that the
neurotoxicity of CAR T-cell therapy may be associated with the
proliferation of CAR T-cells in the central nervous system. A phase
I study enrolled 21 pediatric and young adult patients (aged 1-30
years) with R/R ALL or non-Hodgkin lymphoma (58) and reported that those who developed
neurotoxicity had higher concentrations of CAR T-cells in their
CSF. In this trial, the numbers of CAR T-cells in the CSF of
patients exhibiting neurotoxicity were higher compared with those
in patients who did not receive this therapy. Similar to CRS, ICANS
is also self-limited and the therapeutic interventions are mainly
supportive, aimed at attenuating the immune system response by
using steroids (59).
Tumor lysis syndrome (TLS)
TLS refers to the metabolic disorder caused by the
excessive release of uric acid, potassium and phosphorus following
the death of a large number of cancer cells within a short time
period.
In general, the symptoms are non-specific, including
nausea, fatigue, numbness, hallucinations, cramps and palpitations.
TLS is a common tumor emergency and, if left untreated, may lead to
kidney failure, heart failure and death in severe cases (60). When patients with a high tumor
burden receive CAR T-cell therapy, plenty of tumor cells are killed
by CAR T-cells within a short period, releasing their contents into
the blood and leading to the development of TLS. Therefore, when
administering CAR T-cell therapy, the results of blood tests and
the characteristics of the cancer should be taken into
consideration to evaluate the risk of developing TLS. Furthermore,
adequate preventive measures are required to reduce the risk of TLS
(61,62).
Homing disorders
Another cause for the lack of response in numerous
patients receiving CAR T-cell therapy is the poor homing of T-cells
into tumors.
This may be caused by the lack of optimal
tumor-associated or tumor-specific antigens. An optimal target
should have wide tumor coverage and be highly specific for the
tumor in order to ensure safety and efficacy. Unlike hematological
cancer cells carrying homogeneous target antigens, the antigens on
solid tumor cells are heterogeneous and may differ between the
primary and metastatic stages of the same tumor; therefore, finding
a suitable antigen may prove difficult (63).
The second reason for homing disorders is the
immunosuppressive TME. The lack of sufficient blood supply, low pH
and certain cytokines prevent the continued antitumor activity of
the immune system (64). In
addition, fibroblasts and myeloid cells may form a physical barrier
around the tumor to prevent CAR T-cells from entering the TME.
Furthermore, a variety of chemokines secreted by solid tumors may
lead to immune escape and make the penetration of CAR T-cells less
efficient. As a consequence, it is difficult for CAR T-cells to
adequately home to and infiltrate the tumor.
In order to solve these problems, methods for
improving the treatment efficacy of CAR T-cells in solid tumors
have been under investigation. In a study by Katz et al
(65), CAR T-cell therapy was
applied to patients with liver metastases through percutaneous
hepatic artery infusions, which limited extrahepatic toxicity while
optimizing treatment efficacy. Other methods, such as engineering
CAR T-cells to express cytokines, such as IL-7 and CCL19, may
enhance the efficacy of this therapeutic application by recruiting
T-cells and DCs to tumor tissues (66). In addition, as a chemokine receptor,
C-X-C motif chemokine receptor 3 has a key role in activating
T-cells and is involved in the development and persistence of
tissue-resident memory CD8+ T-cells (67).
Poor proliferation and short-term
persistence
As well as the antigen loss, poor persistence of the
infused CAR T-cells is one of the major causes of treatment
failure, resulting from programmed cell death or exhaustion of
T-cells. Of note, the exhaustion of T-cells is complex and may be
affected by a variety of factors, including the starting state of
T-cells, patient age and the TME (68). It has been reported that CAR T-cells
may be functionally suppressed under adverse conditions, including
poor blood supply, low pH and hypoxia (69). Furthermore, tumors secrete cytokines
to recruit various types of immunosuppressive cells that may
secrete anti-inflammatory cytokines and/or express ligands for
immune checkpoint receptors, which potentially causes the
exhaustion of CAR T-cells (70). As
a consequence, these negative regulators lead to the poor expansion
or short-term persistence of CAR T-cells.
On-target, off-tumor toxicity
On-target off-tumor toxicity is an adverse effect
associated with CAR T-cell-induced injury of normal tissues. Since
tumor-associated antigens may also be weakly expressed in normal
tissues (71), potential damage of
normal tissues may occur during immunotherapy (72). A severe event was reported during
the treatment of a patient with colon cancer metastasis to the
lungs and liver (73). In that
case, the patient developed respiratory distress within 15 min
after the CAR T-cell infusion and succumbed to the condition 5 days
later. The final conclusion was that the patient's death was caused
by the localization of a large number of CAR T-cells in the lungs
and severe injury of the pulmonary epithelial cells.
Current research indicates that the introduction of
suicide gene systems may be used to address the possible on-target
off-tumor toxicity (74).
Furthermore, by engineering CAR T-cells to express two or more
receptors, such CARs may recognize the tumor cells more
specifically, while normal tissues are protected as they only
express one antigen (75).
5. Conclusions and outlook
Compared with traditional therapy, immunotherapy
exhibits higher specificity and stronger antitumor efficacy.
Furthermore, this field is rapidly advancing due to continuous
scientific breakthroughs.
Although CAR T-cell therapy is rapidly developing
and appears to hold great promise, several issues remain to be
resolved, such as increasing the ability of CAR T-cells to
infiltrate solid tumor tissues, enhancing their specificity when
applied to various solid tumors and increasing their ability to
cope with the immunosuppressive TME.
With the continuous development of tumor
immunotherapy, the related anticancer drugs are expected to be more
widely used in the future. Therefore, the incidence of
immune-related adverse reactions is expected to increase over the
next few years and effectively reducing various adverse reactions
should be the focus of future research. In addition, traditional
CARs have a fixed design, which limits their clinical application
due to the high manufacturing cost, lengthy production process and
an inherent risk of production failure; therefore, the design of a
universal CAR represents a new goal as well as a significant
challenge (76).
Acknowledgements
Not applicable.
Funding
This work was supported by the National College Student
Innovation and Entrepreneurship Training Program (grant no.
202010366031), the Science and Technology Project of the Health
Planning Committee of Sichuan (grant nos. 19PJ057 and 20PJ177),
Sichuan Science and Technology Program (grant nos. 2018SZ0377 and
2018JY0219) and Nanchong Science and Technology Program (grant nos.
19SXHZ0451 and 18SXHZ0366).
Availability of data and materials
Not applicable.
Authors' contributions
KL and FL designed the review. YL and GS performed
the literature search, selection of articles and wrote the paper.
SL checked every edition and made revisions. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Panagopoulou TI and Rafiq QA: CAR-T
immunotherapies: Biotechnological strategies to improve safety,
efficacy and clinical outcome through CAR engineering. Biotechnol
Adv. 37(107411)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Couzin-Frankel J: Breakthrough of the year
2013. Cancer immunotherapy. Science. 342:1432–1433. 2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Tan S, Li D and Zhu X: Cancer
immunotherapy: Pros, cons and beyond. Biomed Pharmacother.
124(109821)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Peng M, Mo Y, Wang Y, Wu P, Zhang Y, Xiong
F, Guo C, Wu X, Li Y, Li X, et al: Neoantigen vaccine: An emerging
tumor immunotherapy. Mol Cancer. 18(128)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Yáñez L, Sánchez-Escamilla M and Perales
MA: CAR T cell toxicity: Current management and future directions.
HemaSphere. 3(e186)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ma CC, Wang ZL, Xu T, He ZY and Wei YQ:
The approved gene therapy drugs worldwide: from 1998 to 2019.
Biotechnol Adv. 40(107502)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Mullard A: FDA approves first CAR T
therapy. Nat Rev Drug Discov. 16(669)2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
No authors listed. FDA approves second CAR
T-cell therapy. Cancer Discov. 8:5–6. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Huang X and Yang Y: Driving an improved
CAR for cancer immunotherapy. J Clin Invest. 126:2795–2798.
2016.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Huang R, Li X, He Y, Zhu W, Gao L, Liu Y,
Gao L, Wen Q, Zhong JF, Zhang C, et al: Recent advances in CAR-T
cell engineering. J Hematol Oncol. 13(86)2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Jackson HJ, Rafiq S and Brentjens RJ:
Driving CAR T-cells forward. Nat Rev Clin Oncol. 13:370–383.
2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Depil S, Duchateau P, Grupp SA, Mufti G
and Poirot L: ‘Off-the-shelf’ allogeneic CAR T cells: Development
and challenges. Nat Rev Drug Discov. 19:185–199. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Schmidts A and Maus MV: Making CAR T cells
a solid option for solid tumors. Front Immunol.
9(2593)2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Chmielewski M and Abken H: TRUCKs: The
fourth generation of CARs. Expert Opin Biol Ther. 15:1145–1154.
2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Li D, Li X, Zhou WL, Huang Y, Liang X,
Jiang L, Yang X, Sun J, Li Z, Han WD, et al: Genetically engineered
T cells for cancer immunotherapy. Signal Transduct Target Ther.
4(35)2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Schuster SJ, Bishop MR, Tam CS, Waller EK,
Borchmann P, McGuirk JP, Jäger U, Jaglowski S, Andreadis C, Westin
JR, et al: JULIET Investigators: Tisagenlecleucel in adult relapsed
or refractory diffuse large B-cell lymphoma. N Engl J Med.
380:45–56. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yamada S, Kaneko MK, Sayama Y, Asano T,
Sano M, Yanaka M, Nakamura T, Okamoto S, Handa S, Komatsu Y, et al:
Development of novel mouse monoclonal antibodies against human
CD19. Monoclon Antib Immunodiagn Immunother. 39:45–50.
2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Schubert ML, Hückelhoven A, Hoffmann JM,
Schmitt A, Wuchter P, Sellner L, Hofmann S, Ho AD, Dreger P and
Schmitt M: Chimeric antigen receptor T cell therapy targeting
CD19-positive leukemia and lymphoma in the context of stem cell
transplantation. Hum Gene Ther. 27:758–771. 2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Iovino L and Shadman M: Novel therapies in
chronic lymphocytic leukemia: A rapidly changing landscape. Curr
Treat Options Oncol. 21(24)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Porter DL, Hwang WT, Frey NV, Lacey SF,
Shaw PA, Loren AW, Bagg A, Marcucci KT, Shen A, Gonzalez V, et al:
Chimeric antigen receptor T cells persist and induce sustained
remissions in relapsed refractory chronic lymphocytic leukemia. Sci
Transl Med. 7(303ra139)2015.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Fry TJ, Shah NN, Orentas RJ,
Stetler-Stevenson M, Yuan CM, Ramakrishna S, Wolters P, Martin S,
Delbrook C, Yates B, et al: CD22-targeted CAR T cells induce
remission in B-ALL that is naive or resistant to CD19-targeted CAR
immunotherapy. Nat Med. 24:20–28. 2018.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Liu J, Tan X, Ma YY, Liu Y, Gao L, Gao L,
Kong P, Peng XG, Zhang X and Zhang C: Study on the prognostic value
of aberrant antigen in patients with acute B lymphocytic leukemia.
Clin Lymphoma Myeloma Leuk. 19:e349–e358. 2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Qin H, Edwards JP, Zaritskaya L, Gupta A,
Mu CJ, Fry TJ, Hilbert DM and LaFleur DW: Chimeric antigen
receptors incorporating D domains targeting CD123 direct potent
mono- and bi-specific antitumor activity of T cells. Mol Ther.
27:1262–1274. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Borot F, Wang H, Ma Y, Jafarov T, Raza A,
Ali AM and Mukherjee S: Gene-edited stem cells enable CD33-directed
immune therapy for myeloid malignancies. Proc Natl Acad Sci USA.
116:11978–11987. 2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Xu J, Chen LJ, Yang SS, Sun Y, Wu W, Liu
YF, Xu J, Zhuang Y, Zhang W, Weng XQ, et al: Exploratory trial of a
biepitopic CAR T-targeting B cell maturation antigen in
relapsed/refractory multiple myeloma. Proc Natl Acad Sci USA.
116:9543–9551. 2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
D'Agostino M and Raje N: Anti-BCMA CAR
T-cell therapy in multiple myeloma: Can we do better? Leukemia.
34:21–34. 2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Brudno JN, Maric I, Hartman SD, Rose JJ,
Wang M, Lam N, Stetler-Stevenson M, Salem D, Yuan C, Pavletic S, et
al: T cells genetically modified to express an anti-B-cell
maturation antigen chimeric antigen receptor cause remissions of
poor-prognosis relapsed multiple myeloma. J Clin Oncol.
36:2267–2280. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Morandi F, Horenstein AL, Costa F,
Giuliani N, Pistoia V and Malavasi F: CD38: A target for
immunotherapeutic approaches in multiple myeloma. Front Immunol.
9(2722)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Vasuthasawat A, Yoo EM, Trinh KR,
Lichtenstein A, Timmerman JM and Morrison SL: Targeted
immunotherapy using anti-CD138-interferon α fusion proteins and
bortezomib results in synergistic protection against multiple
myeloma. MAbs. 8:1386–1397. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Sun M, Shi H, Liu C, Liu J, Liu X and Sun
Y: Construction and evaluation of a novel humanized HER2-specific
chimeric receptor. Breast Cancer Res. 16(R61)2014.PubMed/NCBI View
Article : Google Scholar
|
|
31
|
Gao H, Li K, Tu H, Pan X, Jiang H, Shi B,
Kong J, Wang H, Yang S, Gu J, et al: Development of T cells
redirected to glypican-3 for the treatment of hepatocellular
carcinoma. Clin Cancer Res. 20:6418–6428. 2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Li D, Li N, Zhang YF, Fu H, Feng M,
Schneider D, Su L, Wu X, Zhou J, Mackay S, et al: Persistent
polyfunctional chimeric antigen receptor T cells that target
glypican 3 eliminate orthotopic hepatocellular carcinomas in mice.
Gastroenterology. 158:2250–2265.e20. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Liu Y, Di S, Shi B, Zhang H, Wang Y, Wu X,
Luo H, Wang H, Li Z and Jiang H: Armored inducible expression of
IL-12 enhances antitumor activity of glypican-3-targeted chimeric
antigen receptor-engineered T cells in hepatocellular carcinoma. J
Immunol. 203:198–207. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wei X, Lai Y, Li J, Qin L, Xu Y, Zhao R,
Li B, Lin S, Wang S, Wu Q, et al: PSCA and MUC1 in non-small-cell
lung cancer as targets of chimeric antigen receptor T cells.
OncoImmunology. 6(e1284722)2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Sotoudeh M, Shirvani SI, Merat S,
Ahmadbeigi N and Naderi M: MSLN (Mesothelin), ANTXR1 (TEM8), and
MUC3A are the potent antigenic targets for CAR T cell therapy of
gastric adenocarcinoma. J Cell Biochem. 120:5010–5017.
2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zeng C, Cheng J, Li T, Huang J, Li C,
Jiang L, Wang J, Chen L, Mao X, Zhu L, et al: Efficacy and toxicity
for CD22/CD19 chimeric antigen receptor T-cell therapy in patients
with relapsed/refractory aggressive B-cell lymphoma involving the
gastrointestinal tract. Cytotherapy. 22:166–171. 2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Zhu H, You Y, Shen Z and Shi L:
EGFRvIII-CAR-T cells with PD-1 knockout have improved anti-glioma
activity. Pathol Oncol Res. 26:2135–2141. 2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Zhang BL, Li D, Gong YL, Huang Y, Qin DY,
Jiang L, Liang X, Yang X, Gou HF, Wang YS, et al: Preclinical
evaluation of chimeric antigen receptor-modified T cells specific
to epithelial cell adhesion molecule for treating colorectal
cancer. Hum Gene Ther. 30:402–412. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Liu K, Song G, Zhang X, Li Q, Zhao Y, Zhou
Y, Xiong R, Hu X, Tang Z and Feng G: PTK7 is a novel oncogenic
target for esophageal squamous cell carcinoma. World J Surg Oncol.
15(105)2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Gutgarts V, Jain T, Zheng J, Maloy MA,
Ruiz JD, Pennisi M, Jaimes EA, Perales MA and Sathick J: Acute
kidney injury after CAR-T cell therapy: Low incidence and rapid
recovery. Biol Blood Marrow Transplant. 26:1071–1076.
2020.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Aldoss I, Khaled SK, Budde E and Stein AS:
Cytokine release syndrome with the novel treatments of acute
lymphoblastic leukemia: Pathophysiology, prevention, and treatment.
Curr Oncol Rep. 21(4)2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Riegler LL, Jones GP and Lee DW: Current
approaches in the grading and management of cytokine release
syndrome after chimeric antigen receptor T-cell therapy. Ther Clin
Risk Manag. 15:323–335. 2019.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Shimabukuro-Vornhagen A, Gödel P, Subklewe
M, Stemmler HJ, Schlößer HA, Schlaak M, Kochanek M, Böll B and von
Bergwelt-Baildon MS: Cytokine release syndrome. J Immunother
Cancer. 6(56)2018.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Chou CK and Turtle CJ: Assessment and
management of cytokine release syndrome and neurotoxicity following
CD19 CAR-T cell therapy. Expert Opin Biol Ther. 20:653–664.
2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Thakar MS, Kearl TJ and Malarkannan S:
Controlling cytokine release syndrome to harness the full potential
of CAR-based cellular therapy. Front Oncol. 9(1529)2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Alvi RM, Frigault MJ, Fradley MG, Jain MD,
Mahmood SS, Awadalla M, Lee DH, Zlotoff DA, Zhang L, Drobni ZD, et
al: Cardiovascular events among adults treated with chimeric
antigen receptor T-cells (CAR-T). J Am Coll Cardiol. 74:3099–3108.
2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Liu D and Zhao J: Cytokine release
syndrome: Grading, modeling, and new therapy. J Hematol Oncol.
11(121)2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Gödel P, Shimabukuro-Vornhagen A and von
Bergwelt-Baildon M: Understanding cytokine release syndrome.
Intensive Care Med. 44:371–373. 2018.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Acharya UH, Dhawale T, Yun S, Jacobson CA,
Chavez JC, Ramos JD, Appelbaum J and Maloney DG: Management of
cytokine release syndrome and neurotoxicity in chimeric antigen
receptor (CAR) T cell therapy. Expert Rev Hematol. 12:195–205.
2019.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Gauthier J, Hirayama AV, Purushe J, Hay
KA, Lymp J, Li DH, Yeung CCS, Sheih A, Pender BS, Hawkins RM, et
al: Feasibility and efficacy of CD19-targeted CAR T cells with
concurrent ibrutinib for CLL after ibrutinib failure. Blood.
135:1650–1660. 2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Tu S, Zhou X, Guo Z, Huang R, Yue C, He Y,
Li M, Chen Y, Liu Y, Chang LJ, et al: CD19 and CD70 dual-target
chimeric antigen receptor T-cell therapy for the treatment of
relapsed and refractory primary central nervous system diffuse
large B-cell lymphoma. Front Oncol. 9(1350)2019.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Shen D, Song H, Xu X, Xu W, Wang D, Liang
J, Fang M, Liao C, Chen X, Li S, et al: Chimeric antigen receptor T
cell therapy can be administered safely under the real-time
monitoring of Th1/Th2 cytokine pattern using the cytometric bead
array technology for relapsed and refractory acute lymphoblastic
leukemia in children. Pediatr Hematol Oncol. 37:288–299.
2020.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Cornillon J, Hadhoum N, Roth-Guepin G,
Quessar A, Platon L, Ouachée-Chardin M, Nicolas-Virelizier E,
Naudin J, Moreau AS, Masouridi-Levrat S, et al: Management of CAR-T
cell-related encephalopathy syndrome in adult and pediatric
patients: Recommendations of the French Society of Bone Marrow
transplantation and cellular Therapy (SFGM-TC). Bull Cancer.
107:S12–S17. 2020.PubMed/NCBI View Article : Google Scholar : (In French).
|
|
54
|
Kennedy LB and Salama AKS: A review of
cancer immunotherapy toxicity. CA Cancer J Clin. 70:86–104.
2020.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Turtle CJ, Hay KA, Hanafi LA, Li D,
Cherian S, Chen X, Wood B, Lozanski A, Byrd JC, Heimfeld S, et al:
Durable molecular remissions in chronic lymphocytic leukemia
treated with CD19-specific chimeric antigen receptor-modified T
cells after failure of Ibrutinib. J Clin Oncol. 35:3010–3020.
2017.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Karschnia P, Jordan JT, Forst DA,
Arrillaga-Romany IC, Batchelor TT, Baehring JM, Clement NF,
Gonzalez Castro LN, Herlopian A, Maus MV, et al: Clinical
presentation, management, and biomarkers of neurotoxicity after
adoptive immunotherapy with CAR T cells. Blood. 133:2212–2221.
2019.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Santomasso BD, Park JH, Salloum D, Riviere
I, Flynn J, Mead E, Halton E, Wang X, Senechal B, Purdon T, et al:
Clinical and biological correlates of neurotoxicity associated with
CAR T-cell therapy in patients with B-cell acute lymphoblastic
leukemia. Cancer Discov. 8:958–971. 2018.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Lee DW, Kochenderfer JN, Stetler-Stevenson
M, Cui YK, Delbrook C, Feldman SA, Fry TJ, Orentas R, Sabatino M,
Shah NN, et al: T cells expressing CD19 chimeric antigen receptors
for acute lymphoblastic leukaemia in children and young adults: A
phase 1 dose-escalation trial. Lancet. 385:517–528. 2015.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Oved JH, Barrett DM and Teachey DT:
Cellular therapy: Immune-related complications. Immunol Rev.
290:114–126. 2019.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Gupta A and Moore JA: Tumor lysis
syndrome. JAMA Oncol. 4(895)2018.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Feldmann A, Arndt C, Koristka S, Berndt N,
Bergmann R and Bachmann MP: Conventional CARs versus modular CARs.
Cancer Immunol Immunother. 68:1713–1719. 2019.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Neelapu SS: Managing the toxicities of CAR
T-cell therapy. Hematol Oncol. 37 (Suppl 1):48–52. 2019.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Xia AL, Wang XC, Lu YJ, Lu XJ and Sun B:
Chimeric-antigen receptor T (CAR-T) cell therapy for solid tumors:
Challenges and opportunities. Oncotarget. 8:90521–90531.
2017.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Greenbaum U, Yalniz FF, Srour SA, Rezvani
K, Singh H, Olson A, Blumenschein G Jr, Hong DS, Shpall EJ and
Kebriaei P: Chimeric antigen receptor therapy: How are we driving
in solid tumors? Biol Blood Marrow Transplant. 26:1759–1769.
2020.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Katz SC, Burga RA, McCormack E, Wang LJ,
Mooring W, Point GR, Khare PD, Thorn M, Ma Q, Stainken BF, et al:
Phase I hepatic immunotherapy for metastases study of
intra-arterial chimeric antigen receptor-modified T-cell therapy
for CEA+ liver metastases. Clin Cancer Res.
21:3149–3159. 2015.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Adachi K, Kano Y, Nagai T, Okuyama N,
Sakoda Y and Tamada K: IL-7 and CCL19 expression in CAR-T cells
improves immune cell infiltration and CAR-T cell survival in the
tumor. Nat Biotechnol. 36:346–351. 2018.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Alanio C, Barreira da Silva R, Michonneau
D, Bousso P, Ingersoll MA and Albert ML: CXCR3/CXCL10 Axis Shapes
Tissue Distribution of Memory Phenotype CD8+ T Cells in
Nonimmunized Mice. J Immunol. 200:139–146. 2018.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Liu L, Bi E, Ma X, Xiong W, Qian J, Ye L,
Su P, Wang Q, Xiao L, Yang M, et al: Enhanced CAR-T activity
against established tumors by polarizing human T cells to secrete
interleukin-9. Nat Commun. 11(5902)2020.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Knochelmann HM, Smith AS, Dwyer CJ, Wyatt
MM, Mehrotra S and Paulos CM: CAR T Cells in solid tumors:
Blueprints for building effective therapies. Front Immunol.
9(1740)2018.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Morgan MA and Schambach A: Engineering
CAR-T cells for improved function against solid tumors. Front
Immunol. 9(2493)2018.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Yu S, Yi M, Qin S and Wu K: Next
generation chimeric antigen receptor T cells: Safety strategies to
overcome toxicity. Mol Cancer. 18(125)2019.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Akbari P, Huijbers EJM, Themeli M,
Griffioen AW and van Beijnum JR: The tumor vasculature an
attractive CAR T cell target in solid tumors. Angiogenesis.
22:473–475. 2019.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Morgan RA, Yang JC, Kitano M, Dudley ME,
Laurencot CM and Rosenberg SA: Case report of a serious adverse
event following the administration of T cells transduced with a
chimeric antigen receptor recognizing ERBB2. Mol Ther. 18:843–851.
2010.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Gargett T and Brown MP: The inducible
caspase-9 suicide gene system as a ‘safety switch’ to limit
on-target, off-tumor toxicities of chimeric antigen receptor T
cells. Front Pharmacol. 5(235)2014.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Kloss CC, Condomines M, Cartellieri M,
Bachmann M and Sadelain M: Combinatorial antigen recognition with
balanced signaling promotes selective tumor eradication by
engineered T cells. Nat Biotechnol. 31:71–75. 2013.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Liu D, Zhao J and Song Y: Engineering
switchable and programmable universal CARs for CAR T therapy. J
Hematol Oncol. 12(69)2019.PubMed/NCBI View Article : Google Scholar
|