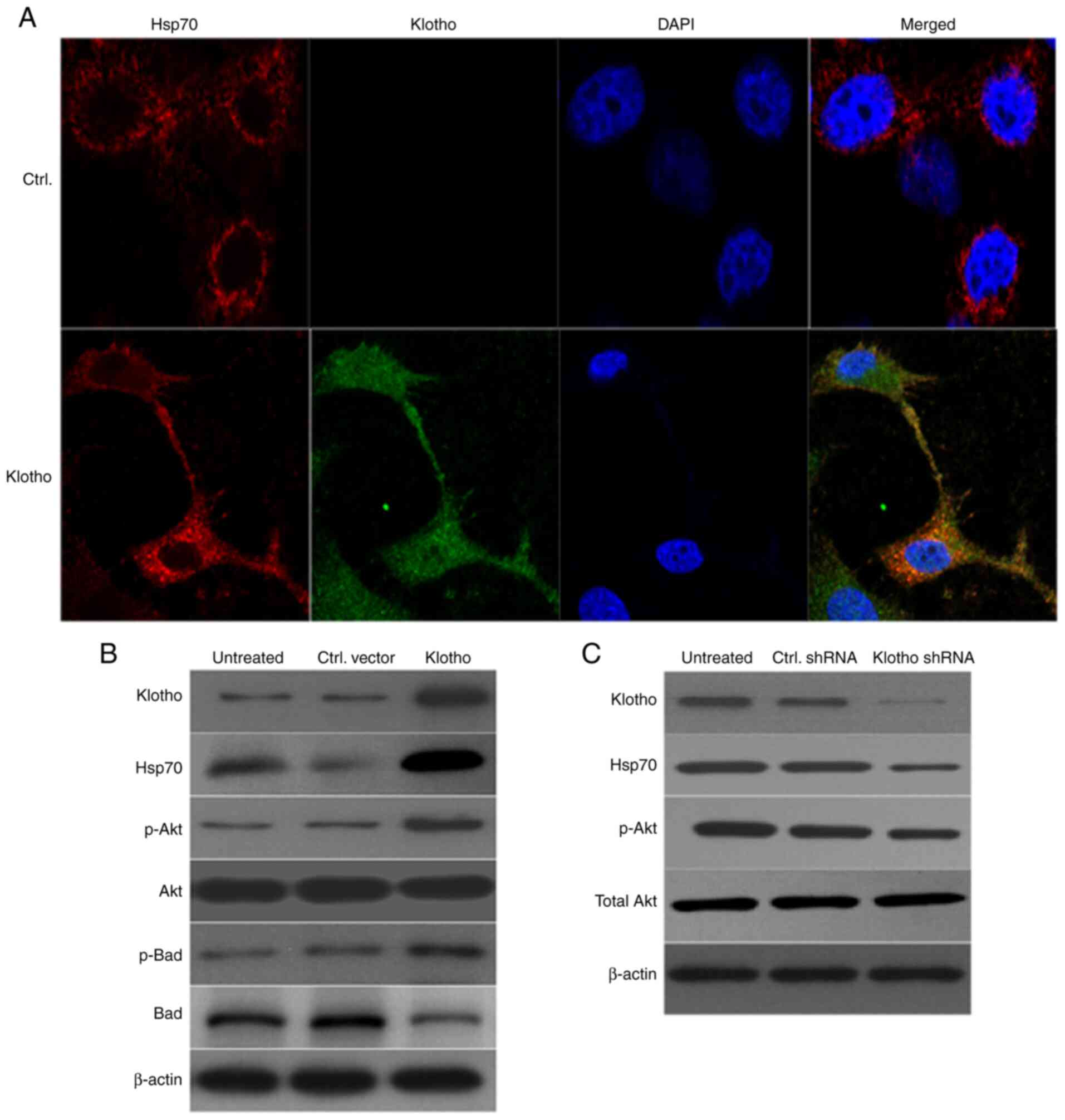
|
1
|
Alabas OA, Jernberg T, Pujades-Rodriguez
M, Rutherford MJ, West RM, Hall M, Timmis A, Lindahl B, Fox KA,
Hemingway H, et al: Statistics on mortality following acute
myocardial infarction in 842 897 Europeans. Cardiovasc Res.
116:149–157. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Ioacara S, Popescu AC, Tenenbaum J,
Dimulescu DR, Popescu MR, Sirbu A and Fica S: Acute Myocardial
Infarction Mortality Rates and Trends in Romania between 1994 and
2017. Int J Environ Res Public Health. 17(285)2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Neri M, Riezzo I, Pascale N, Pomara C and
Turillazzi E: Ischemia/reperfusion injury following acute
myocardial infarction: A critical issue for clinicians and forensic
pathologists. Mediators Inflamm. 2017(7018393)2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wang M and Wan J: Soluble regulators of
Interleukin-1 signaling: Novel biomarkers for early acute
myocardial infarction diagnosis and to predict ischemia/reperfusion
injury? Int J Cardiol. 274(357)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Feng M, Wang Q, Wang H and Guan W: Tumor
necrosis factor-alpha preconditioning attenuates liver
ischemia/reperfusion injury through preserving sarco/endoplasmic
reticulum calcium-ATPase function. J Surg Res. 184:1109–1113.
2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Li X, Dai Y, Yan S, Shi Y, Han B, Li J,
Cha L and Mu J: Down-regulation of lncRNA KCNQ1OT1 protects against
myocardial ischemia/reperfusion injury following acute myocardial
infarction. Biochem Biophys Res Commun. 491:1026–1033.
2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tu CC, Wan BY and Zeng Y: STIM2 knockdown
protects against ischemia/reperfusion injury through reducing
mitochondrial calcium overload and preserving mitochondrial
function. Life Sci. 247(116560)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yue R, Xia X, Jiang J, Yang D, Han Y, Chen
X, Cai Y, Li L, Wang WE and Zeng C: Mitochondrial DNA oxidative
damage contributes to cardiomyocyte ischemia/reperfusion-injury in
rats: Cardioprotective role of lycopene. J Cell Physiol.
230:2128–2141. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Wang Z, Sun R, Wang G, Chen Z, Li Y, Zhao
Y, Liu D, Zhao H, Zhang F, Yao J, et al: SIRT3-mediated
deacetylation of PRDX3 alleviates mitochondrial oxidative damage
and apoptosis induced by intestinal ischemia/reperfusion injury.
Redox Biol. 28(101343)2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wang X, He F, Liao Y, Song X, Zhang M, Qu
L, Luo T, Zhou S, Ling Y, Guo J, et al: Baicalin pretreatment
protects against myocardial ischemia/reperfusion injury by
inhibiting mitochondrial damage-mediated apoptosis. Int J Cardiol.
168:4343–4345. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yang Y, Duan W, Jin Z, Yi W, Yan J, Zhang
S, Wang N, Liang Z, Li Y, Chen W, et al: JAK2/STAT3 activation by
melatonin attenuates the mitochondrial oxidative damage induced by
myocardial ischemia/reperfusion injury. J Pineal Res. 55:275–286.
2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Xie Y, Jiang D, Xiao J, Fu C, Zhang Z, Ye
Z and Zhang X: Ischemic preconditioning attenuates
ischemia/reperfusion-induced kidney injury by activating autophagy
via the SGK1 signaling pathway. Cell Death Dis.
9(338)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhou LY, Zhai M, Huang Y, Xu S, An T, Wang
YH, Zhang RC, Liu CY, Dong YH, Wang M, et al: The circular RNA ACR
attenuates myocardial ischemia/reperfusion injury by suppressing
autophagy via modulation of the Pink1/ FAM65B pathway. Cell Death
Differ. 26:1299–1315. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Di Bona D, Accardi G, Virruso C, Candore G
and Caruso C: Association of Klotho polymorphisms with healthy
aging: A systematic review and meta-analysis. Rejuvenation Res.
17:212–216. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wang Q, Su W, Shen Z and Wang R:
Correlation between soluble alpha-Klotho and renal function in
patients with chronic kidney disease: A Review and meta-analysis.
Biomed Res Int. 2018(9481475)2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Mosa O, Skitek M and Jerin A: Validity of
Klotho, CYR61 and YKL-40 as ideal predictive biomarkers for acute
kidney injury: Review study. Sao Paulo Med J. 135:57–65.
2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kuwahara N, Sasaki S, Kobara M, Nakata T,
Tatsumi T, Irie H, Narumiya H, Hatta T, Takeda K, Matsubara H, et
al: HMG-CoA reductase inhibition improves anti-aging klotho protein
expression and arteriosclerosis in rats with chronic inhibition of
nitric oxide synthesis. Int J Cardiol. 123:84–90. 2008.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yang K, Wang C, Nie L, Zhao X, Gu J, Guan
X, Wang S, Xiao T, Xu X, He T, et al: Klotho protects against
indoxyl sulphate-induced myocardial hypertrophy. J Am Soc Nephrol.
26:2434–2446. 2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ramez M, Rajabi H, Ramezani F, Naderi N,
Darbandi-Azar A and Nasirinezhad F: The greater effect of
high-intensity interval training versus moderate-intensity
continuous training on cardioprotection against
ischemia-reperfusion injury through Klotho levels and attenuate of
myocardial TRPC6 expression. BMC Cardiovasc Disord.
19(118)2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Takenaka T, Kobori H, Miyazaki T, Suzuki
H, Nishiyama A, Ishii N, Yamashita M and Hayashi M: Klotho protein
supplementation reduces blood pressure and renal hypertrophy in
db/db mice, a model of type 2 diabetes. Acta Physiol (Oxf).
225(e13190)2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Donate-Correa J, Martín-Núñez E, Delgado
NP, de Fuentes MM, Arduan AO, Mora-Fernández C and Navarro González
JF: Implications of Fibroblast growth factor/Klotho system in
glucose metabolism and diabetes. Cytokine Growth Factor Rev.
28:71–77. 2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Fountoulakis N, Maltese G, Gnudi L and
Karalliedde J: Reduced levels of anti-ageing hormone Klotho predict
renal function decline in type 2 diabetes. J Clin Endocrinol Metab.
103:2026–2032. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhou X and Wang X: Klotho: A novel
biomarker for cancer. J Cancer Res Clin Oncol. 141:961–969.
2015.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Fakhar M, Najumuddin Gul M and Rashid S:
Antagonistic role of Klotho-derived peptides dynamics in the
pancreatic cancer treatment through obstructing WNT-1 and Frizzled
binding. Biophys Chem. 240:107–117. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Li Q, Li Y, Liang L, Li J, Luo D, Liu Q,
Cai S and Li X: Klotho negatively regulated aerobic glycolysis in
colorectal cancer via ERK/HIF1alpha axis. Cell Commun Signal.
16(26)2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Pako J, Bikov A, Barta I, Matsueda H,
Puskas R, Galffy G, Kerpel-Fronius A, Antus B and Horvath I:
Assessment of the circulating klotho protein in lung cancer
patients. Pathol Oncol Res. 26:233–238. 2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wang N, Ma J, Ren Y, Xiang S and Jia R:
Secreted klotho from exosomes alleviates inflammation and apoptosis
in acute pancreatitis. Am J Transl Res. 11:3375–3383.
2019.PubMed/NCBI
|
|
28
|
Sugiura H, Yoshida T, Mitobe M, Yoshida S,
Shiohira S, Nitta K and Tsuchiya K: Klotho reduces apoptosis in
experimental ischaemic acute kidney injury via HSP-70. Nephrol Dial
Transplant. 25:60–68. 2010.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kim SJ, Cheresh P, Eren M, Jablonski RP,
Yeldandi A, Ridge KM, Budinger GR, Kim DH, Wolf M, Vaughan DE, et
al: Klotho, an antiaging molecule, attenuates oxidant-induced
alveolar epithelial cell mtDNA damage and apoptosis. Am J Physiol
Lung Cell Mol Physiol. 313:L16–L26. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zhu H, Gao Y, Zhu S, Cui Q and Du J:
Klotho Improves cardiac function by suppressing reactive oxygen
species (ROS) mediated apoptosis by modulating Mapks/Nrf2 signaling
in doxorubicin-induced cardiotoxicity. Med Sci Monit. 23:5283–5293.
2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Song S, Gao P, Xiao H, Xu Y and Si LY:
Klotho suppresses cardiomyocyte apoptosis in mice with
stress-induced cardiac injury via downregulation of endoplasmic
reticulum stress. PLoS One. 8(e82968)2013.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Liang X, Li B, Huang Q, Liu D and Ma H:
Klotho prevents DEX-induced apoptosis in MC3T3-E1 osteoblasts
through the NF-κB signaling pathway. Biochem Biophys Res Commun.
507:355–361. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Cui W, Leng B, Liu W and Wang G:
Suppression of apoptosis in human umbilical vein endothelial cells
(HUVECs) by Klotho protein is associated with reduced endoplasmic
reticulum oxidative stress and activation of the PI3K/AKT pathway.
Med Sci Monit. 24:8489–8499. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Aijaz S, Ahmed N, Akhter Z, Sattar S,
Lakhani S, Malik R and Pathan A: Clinical characteristics and
in-hospital outcome in percutaneous coronary interventions with ST
elevation myocardial infarction patients developing acute kidney
injury. J Pak Med Assoc. 69:1827–1833. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wang C, Pei YY, Ma YH, Ma XL, Liu ZW and
Zhu JH: Risk factors for acute kidney injury in patients with acute
myocardial infarction. Chin Med J (Engl). 132:1660–1665.
2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Wang DX, Huang Z, Li QJ, Zhong GQ, He Y,
Huang WQ, Cao XL, Tu RH and Meng JJ: Involvement of HSP90 in
ischemic postconditioning-induced cardioprotection by inhibition of
the complement system, JNK and inflammation. Acta Cir Bras.
35(e202000105)2020.PubMed/NCBI View Article : Google Scholar
|