Introduction
Atopic dermatitis (AD) is a common dermatological
disease with prevalence rates of 15-30% in children and 2-10% in
adults (1). The combination of
genetic, immune and environmental stimuli appears to contribute to
the manifestation of AD. Hereditary susceptibility is considered a
primary factor. To elucidate the genetic factors of AD, multiple
genetic studies have been performed and a series of susceptibility
genes/loci have been identified, which indicate two major
biological pathways responsible for the etiology of AD, including
skin epithelial dysfunction and innate/adaptive immune response
dysregulation, which affect each other (2). However, its exact pathogenesis has
remained largely elusive.
Exosomes are thought to have important roles in
intercellular communication and have been proven to be potential
markers for numerous diseases, such as tumours (3) and renal disease (4). Along with the identification of RNA in
exosomes in 2007 and the development of high-throughput techniques
for nucleic acid analyses, there have been a growing number of
studies on RNA sequences in exosomes (5). One class of small noncoding (snc)RNAs,
transfer RNA-derived fragments (tRFs), which are generated from
transfer RNA (tRNA), have been suggested to have roles in the
regulation of gene expression, cell proliferation, RNA processing,
modulation of the DNA damage response, priming of viral reverse
transcriptase, neurodegeneration and tumour suppression (6). tRFs have been implicated in various
infections, such as human T-cell leukaemia virus type 1(7) and HIV-1-infected cells (8). Different tRFs have been identified in
cellular proliferation and tRF signatures have been described in
myelodysplastic syndromes (9) and
various cancer types, such as prostate cancer (10) and squamous cell carcinoma (11). These studies highlighted the
potential role of tRFs in disease progression. A database of tRFs
has been developed where each unique tRF has been given a name
following the progress of high-throughput sequencing and analyses
of small tRNA fragments (12).
According to their mapped positions on the primary or mature tRNA
transcript, tRFs may be classified into various types, such as tRNA
halves (tiRNA/tiR) (13), tRF-1,
tRF-3, tRF-5(14), and endogenous
tRFs (i-tRFs) (15).
However, to the best of our knowledge, no previous
study has characterized tRFs in AD. In the present study, the
possible roles of tRFs in AD were explored, which may provide novel
information regarding the pathogenesis of AD.
Materials and methods
Ethics statement and human
specimens
This study was approved by the ethics committee of
Shanghai Skin Disease Hospital (Shanghai, China) and was conducted
according to the principles of the Declaration of Helsinki. A total
of 23 pediatric patients with AD meeting the Hanifin-Rajka
diagnostic criteria (16) and 23
healthy controls were enrolled from Shanghai Skin Disease Hospital
(Shanghai, China) between December 1, 2017 and October 1, 2018 in
the present study. All controls were evaluated by experienced
doctors and were required to have no atopic disease history in
individuals and their immediate family members over three
generations. All patients and controls were required to have no
history of malignancy or other systemic autoimmune diseases. None
of the patients were treated with systemic drugs in the two weeks
prior to examination. The disease severity in patients was
evaluated using the objective SCORing Atopic Dermatitis (SCORAD)
index (17), according to which
patients were divided into the mild (0-24 points), moderate (25-50
points) and severe (51-103 points) groups. Written informed consent
forms were signed by all of the subjects and their guardians. There
was no significant difference between the two groups in terms of
sex or age. The clinical features of the AD and control groups are
presented in Table I.
 | Table IClinical features of subjects in the
AD and control groups. |
Table I
Clinical features of subjects in the
AD and control groups.
| Item | AD group (n=23) | Control group
(n=23) |
|---|
| Age (years) | 9.5 (3-13) | 9.0 (6-12) |
| Sex
(female/male) | 12/11 | 10/13 |
| SCORAD | | - |
|
Mild | 7 | - |
|
Moderate | 10 | - |
|
Severe | 6 | - |
Plasma from patients with AD and
healthy controls
Peripheral venous blood was collected into EDTA
anticoagulant tubes, stored at 4˚C and further processed within 1
h. The blood was centrifuged at 2,500 x g at 4˚C for 10 min and the
supernatant was collected and stored at -80˚C.
Exosome isolation and transmission
electron microscopy (TEM) examination
Using the ExoQuick Plasma Prep and Exosome
Precipitation kit (cat. no. EXOQ5TM-1, System Biosciences LLC)
according to the manufacturer's protocol, exosomes were isolated
from plasma. The following protocol were followed: i) Purified
thrombin was diluted to a concentration of [611 U/ml] in PBS; ii) 4
µl of [611 U/ml] thrombin per 0.5 ml plasma was added to a final
concentration of 5 U/ml; iii) the mixture was then incubated at
room temperature for 5 min while mixing; iv) centrifugation was
performed in a standard microfuge at 4,500 x g for 5 min at room
temperature; v) the visible fibrin pellet at the bottom of the
tubes was transferred to a new clean tube; vi) the serum-like
supernatant was then treated with ExoQuick to precipitate exosomes
for 30-60 min at 5˚C; vii) 5-10 µl of the exosome supernatant was
absorbed and added to a copper net, precipitated for 1 min, and the
floating liquid was removed from the edge with a filter paper at
room temperature; viii) the sample was rinsed with PBS at room
temperature; ix) 10 µl phosphotungstic acid was dropped on a copper
net, precipitated for 1 min, before the floating liquid was
absorbed with filter paper at room temperature; and x) this was
fried at room temperature for 2 min, before TEM was performed
(JEM-1200EX, Japan Electronics Corporation).
RNA extraction, small RNA library
preparation and sequencing
Total RNA from plasma exosomes was extracted with
TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.).
Subsequently, purified RNAs were sent to Yingbio for constructing
small RNA libraries and performing small RNA sequencing analysis.
In brief, the small RNA was bound with 3'- and 5'-adapters and
complementary (c)DNA constructs were created by reverse
transcription (RT) followed by PCR. The small RNA fragments (15-40
nt) were excised and purified and the purified libraries were
quantified and validated. Small RNA sequencing was performed on an
Illumina HiSeq 2500 (Illumina, Inc.) (18).
Data analysis
Low-quality reads and short reads (<15 nt) of the
raw sequencing data were filtered out. All small RNA clean reads
were aligned to the piwi-interacting RNA (piRNA) database
(http://pirnabank.ibab.ac.in/), miRbase
database (http://www.mirbase.org/), National
Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov/), tRF database (tRFdb;
http://genome.bioch.virginia.edu/trfdb/), Genomic tRNA
database (http://gtrnadb.ucsc.edu/) and
MintBase (https://mintbase.io/) to identify known
sncRNAs. The clean data were analysed to detect the expression of
sncRNAs between patients and controls, the criteria for
differentially expressed small RNAs were as follows: Log2FC, >1
or <-1; P<0.05. MiRanda (http://www.microrna.org/microrna/home.do; selection
criteria, score ≥150 and energy <-20) and RNAhybrid (https://omictools.com/rnahybrid-tool;
selection criteria, energy <-25) were used to predict the target
mRNAs of the screened small RNAs with significant differences. The
overlapping genes of these two databases were taken as the final
result of the target gene prediction. Gene Ontology (GO) assignment
(http://www.geneontology.org/) and the
Kyoto Encyclopedia of Genes and Genomes (KEGG) database (http://www.genome.jp/kegg) were used to analyse the
functions and pathways of all target genes.
RT-quantitative (q)PCR
A total of 100 ng of RNA was reverse-transcribed to
cDNA using the NEBNext® Multiplex Small RNA Library Prep
Kit (NEB; cat. no. E7560S) according to the manufacturer's
protocol. First, 1 µl of RT primer and 100 ng RNA was mixed, and
then added to RNase-Free H2O to make up 12 µl. The
mixture was incubated at 65˚C for 5 min. Subsequently, 4 µl of
buffer (5X), 2 µl of dNTP Mix, 1 µl of protector RNase Inhibitor, 1
µl of transcriptase, and 12 µl of the mixture from the previous
step, were added, followed by incubation for 1 h at 42˚C, then at
70˚C for 5 min. The obtained cDNA was cryopreserved. Next, 5 µl of
2X Master Mix (Roche Diagnostics, 0.3 µl of PCR specific primer
forward, and 0.3 µl of PCR specific primer reverse was mixed, and
added to ddH2O to make up 9 µl. Then, 9 µl mixture was
added to each well of the 384-PCR plate, along with 1 µl of the
corresponding cDNA. qPCR was performed on an ABI Q6 detection
system (Applied Biosystems; Thermo Fisher Scientific, Inc.). The
thermocycling conditions were as follows: 95˚C for 10 min; followed
by 95˚C for 15 sec, then to 60˚C for 60 sec, for 45 cycles. U6 was
selected as the internal control. The PCR specific primer sequences
were as follows: tRF-33-Q99P9P9NH57SD3 forward
5'-GCAGGCTTCTGTAGTGTAGTGGTTA-3' and reverse,
5'-AGTGCGTGTCGTGGAGTCG-3'; tRF-28-QSZ34KRQ590K forward,
5'-AGGCTCGTTGGTCTAGGGGTA-3' and reverse, 5'-AGTGCGTGTCGTGGAGTCG-3';
and U6 forward, 5'-CGATACAGAGAAGATTAGCATGGC-3' and reverse,
5'-AACGCTTCACGAATTTGCGT-3'. The 2-ΔΔCq method was used
to quantify the tRFs (19).
Statistical analysis
Spearman's nonparametric correlation analysis was
used to analyse the correlation between the expression of tRFs and
disease severity using SPSS 23 (IBM Corp.). The predictive value of
biomarkers regarding the probability of AD was determined by
receiver operating characteristic (ROC) curve analysis, which was
performed using MedCalc software (version 10.4.7.0; MedCalc). The
area under the ROC curve (AUC) was calculated to evaluate the
diagnostic potential of tRFs for AD. The optimal diagnostic score
of the signature of each tRF was evaluated at the cut-off value
with the largest Youden index. P<0.05 was considered to indicate
statistical significance. All the experimental data were from at
least three independent experiments.
Results
Characterization of the isolated
plasma exosomes by TEM
There was no significant difference between the two
groups in terms of sex or age. The clinical features of the AD and
control groups are presented in Table
I. To obtain the purified exosomes, plasma samples from
pediatric patients with AD and individuals from the control group
were subjected to centrifugation. TEM revealed that the plasma
exosomes were spherical particles with a diameter of 100-150 nm and
a complete membrane structure (Fig.
S1), which indicated the successful isolation of exosomes from
human plasma.
Sequencing of tRFs from plasma
exosomes
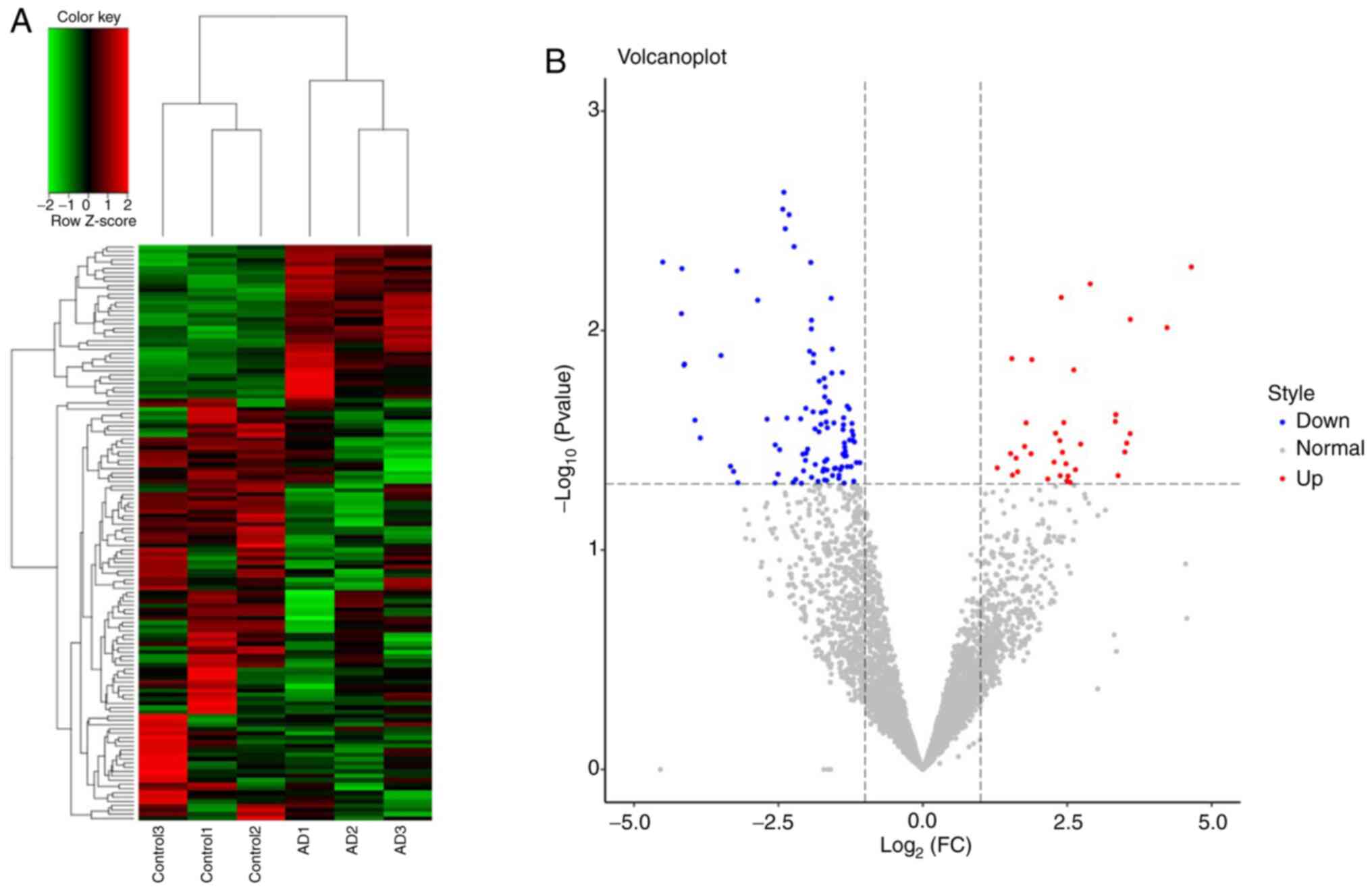
A total of 4,637 tRFs were identified between 3
patients with AD and 3 controls. Of these tRFs, 135 were
significantly differentially expressed, including 36 upregulated
and 99 downregulated tRFs (Fig. 1);
the details are provided in Table
SI. A total of 6 types of tRFs were identified, namely tRF-1,
tRF-3, tRF-5, 3'-halves, 5'-halves and i-tRF (Table SII).
Target gene prediction and functional
analysis of differentially expressed tRFs
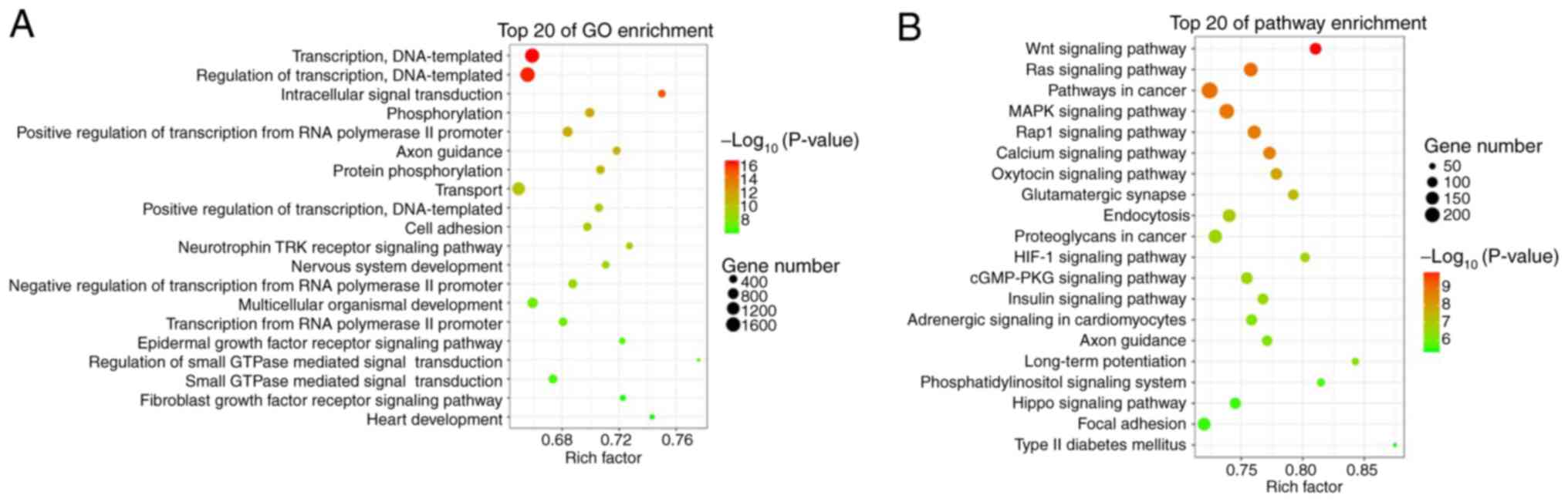
Analyses with the miRanda and RNAhybrid databases
identified 58,227 target genes associated with 135 differentially
expressed tRFs. GO analysis revealed that these target genes were
functionally enriched in transcription, DNA templating, regulation
of transcription, intracellular signal transduction, transport and
epidermal growth factor receptor signalling pathway (Fig. 2A). KEGG pathway analysis revealed
that these target genes were enriched in the Wnt signalling
pathway, Ras signalling pathway, MARK signalling pathway, Rap1
signalling pathway, calcium signalling pathway, endocytosis and
hypoxia-inducible factor (HIF)-1 signalling pathway (Fig. 2B).
Validation of differentially expressed
tRFs
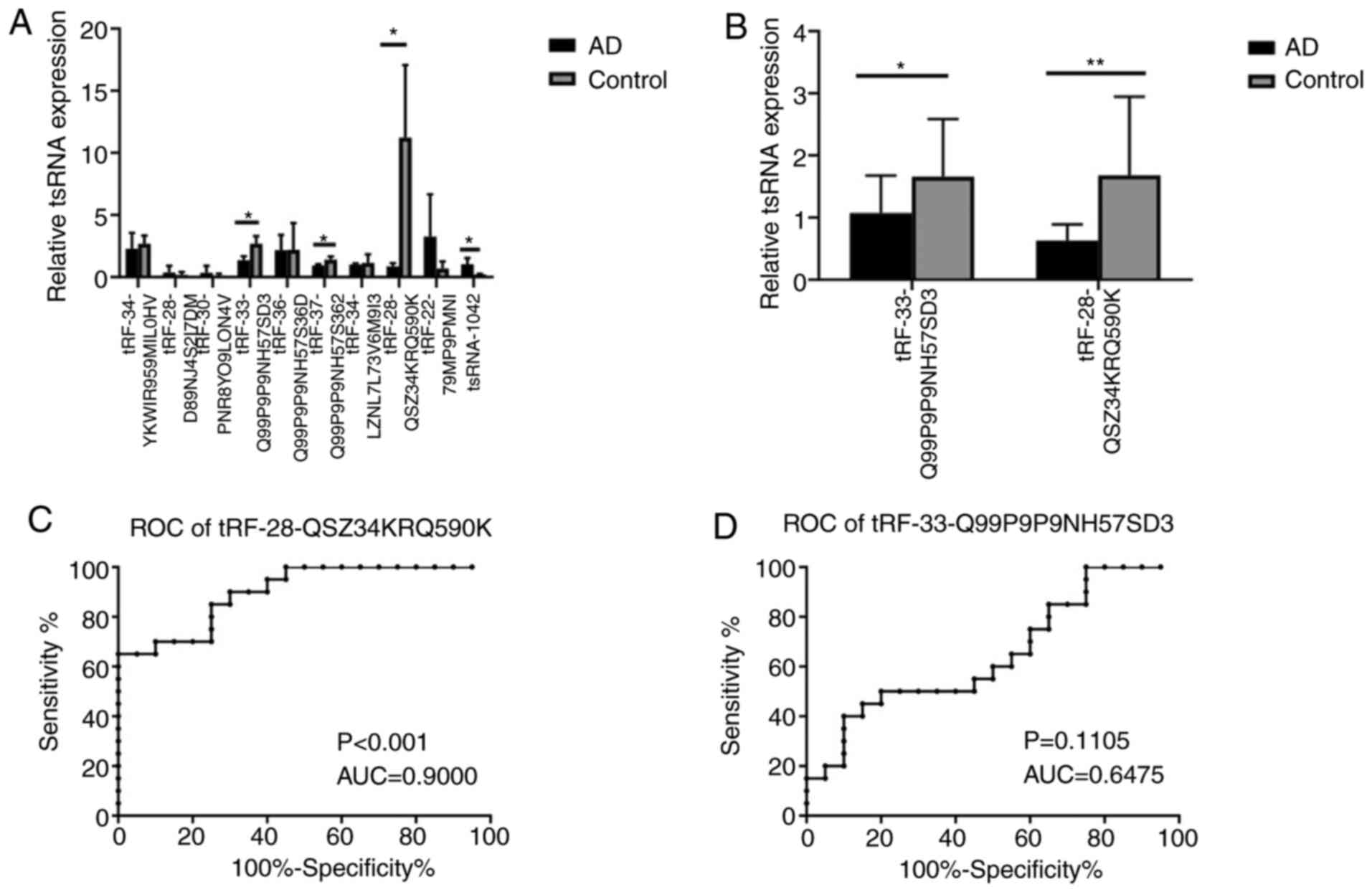
To further confirm the tRFs, 10 differentially
expressed tRFs, including 6 downregulated tRFs
(tRF-34-YKWIR959MIL0HV, tRF-34-LZNL7L73V6M9I3, tRF-28-QSZ34KRQ590K,
tRF-33-Q99P9P9NH57SD3, tRF-36-Q99P9P9NH57S36D and
tRF-37-Q99P9P9NH57S362) and 4 upregulated tRFs
(tRF-28-D89NJ4S2I7DM, tRF-30-PNR8YO9LON4V, tRF-22-79MP9PMNI and
tsRNA-1042), were subjected to RT-qPCR analysis in plasma exosomes
from 3 patients with AD and 3 controls. A total of four tRFs,
namely tRF-28-QSZ34KRQ590K, tRF-33-Q99P9P9NH57SD3,
tRF-37-Q99P9P9NH57S362 and tsRNA-1042, exhibited differential
expression (P=0.037, 0.030, 0.047 and 0.041 respectively; Fig. 3A). Of these, tRF-28-QSZ34KRQ590K and
tRF-33-Q99P9P9NH57SD3 were further selected to be validated by
RT-qPCR in 20 patients with AD and 20 controls. The results
indicated that the expression of both of the tRFs exhibited
significant differences between the AD and control groups
(P<0.001 and 0.048, respectively; Fig. 3B).
ROC curve analyses for
tRF-28-QSZ34KRQ590K and tRF-33-Q99P9P9NH57SD3
ROC curve analysis was used to determine the
diagnostic accuracy of plasma exosomal tRF-28-QSZ34KRQ590K and
tRF-33-Q99P9P9NH57SD3. The ΔCq value of each sample was used for
ROC curve analysis. and the AUCs of the two tRFs were 0.900 and
0.6475, respectively (P<0.001 and P=0.1105; Fig. 3C and D, respectively), which revealed that
plasma exosomal tRF-28-QSZ34KRQ590K may serve as a biomarker for
AD.
Correlation of tRF-28-QSZ34KRQ590K
with AD severity
According to the SCORAD, the patients with AD were
divided into three groups (Table
I). There was no correlation between the expression of tRFs and
disease severity (P=0.842, data not shown).
Discussion
Exosomes are thought to have important roles in
intercellular communication and have been proven to be potential
markers for numerous diseases. The discovery of tRFs in exosomes
and their importance in gene regulation have substantially promoted
an increase in their study in recent years. They are closely
related to the occurrence of numerous human diseases and have the
potential to become novel biomarkers for diseases (20). In the present study, the first
analysis of tRFs in plasma exosomes from patients with AD was
performed. Exosomes were isolated successfully from plasma with
combined centrifugation and were then further identified by TEM. A
total of 135 differentially expressed plasma exosomal tRFs were
identified in 3 subjects with AD and 3 controls. GO and KEGG
pathway enrichment analyses revealed a series of the possible
functions and pathways of the target genes of these 135
differentially expressed tRFs. The downregulation of plasma
exosomal tRF-28-QSZ34KRQ590K and tRF-33-Q99P9P9NH57SD3 was further
verified by RT-qPCR in 20 subjects with AD and 20 controls. The ROC
curve analysis indicated that only tRF-28-QSZ34KRQ590K may be a
potential biomarker for AD.
tRFs have multiple biological functions, including
acting as signalling molecules in cell proliferation and stress
responses and as regulators of gene expression, and have been
considered to be involved in RNA processing, suppression of
translation, neurodegeneration and modulation of the DNA damage
response (20). Identifying the
entire repertoire of the roles of tRFs is particularly important to
better understand their functions. In the present study, a total of
135 differentially expressed plasma exosomal tRFs were identified
in a small sample, including 36 upregulated and 99 downregulated
tRFs, which contained 6 types of tRFs, namely tRF-1, tRF-3, tRF-5,
3'-halves, 5'-halves and i-tRF. tRFs represent a heterogeneous
class of sncRNAs and different sncRNAs are still being identified.
In addition, there are limitations concerning tRF identification by
current methods; in particular, certain tRNA modifications may
block reverse transcriptase and modified tRFs may escape detection
(21). Thus, it is possible that
additional classes of tRFs are uncovered in the future. The present
study was the first to identify differentially expressed plasma
exosomal tRFs in AD, which provided a preliminary outline on tRFs
in AD, and additional functional studies are required to further
understand their mechanisms of action.
The different subclasses of tRFs have been indicated
to be involved in several cellular functions that are also
implicated in cancer, infection and neurodegenerative disorders
(21). In the present study, two
downregulated differentially expressed plasma exosomal tRFs,
tRF-28-QSZ34KRQ590K and tRF-33-Q99P9P9NH57SD3, were verified in a
larger sample by RT-qPCR. Growing evidence has indicated that tRFs
regulate gene expression, epigenetic inheritance, apoptosis and RNA
degradation and stability, although their detailed biological
functions have not been fully elucidated. Downregulated
tRF-28-QSZ34KRQ590K was demonstrated to be a significant marker for
AD in the ROC curve analysis. Studies have revealed that tRFs may
serve as noninvasive diagnostic biomarkers for various diseases
(20). Therefore,
tRF-28-QSZ34KRQ590K may also be a diagnostic biomarker for AD in
pediatric patients, which may provide a novel source for the early
diagnosis and targeted treatment of AD in the future. However, no
correlation between tRF-28-QSZ34KRQ590K and the severity of AD was
obtained (P>0.05). Thus, further studies are required to clarify
the potential role of tRF-28-QSZ34KRQ590K in AD.
Analysis with the miRanda and RNAhybrid databases
retrieved 58,227 target genes associated with the 135
differentially expressed tRFs. GO analysis indicated functional
enrichment of these target genes in transcription, DNA templating,
regulation of transcription, intracellular signal transduction and
epidermal growth factor receptor signalling pathways. Epidermal
growth factor receptor signalling has been previously reported to
have a protective role in AD and to modulate IL-17 responses in the
skin (22). KEGG pathway analysis
suggested that these target genes were enriched in pathways
including the Wnt, Ras, MAPK, Rap1, calcium and HIF-1 signalling
pathways. The Rap1 signalling pathway (23) and MAPK signalling pathway (24) have been reported to be associated
with inflammation in previous studies and the inflammatory response
has also been suggested to have roles in the pathogenesis of AD
(25). Therefore, the present study
implied that plasma exosome tRFs may participate in the
pathogenesis of AD by being involved in the inflammatory response
and the epidermal growth factor receptor signalling pathway.
However, further functional studies are required to better
understand the pathogenesis of AD.
tRF-28-QSZ34KRQ590K demonstrated a significant
diagnostic value for AD in the ROC curve analysis. There were 28
target genes of tRF-28-QSZ34KRQ590K, including tachykinin receptor
1, RANBP2-like and GRIP domain-containing 1, non-SMC condensin I
complex subunit H (NCAPH), ectodysplasin-A receptor,
contactin-associated protein family member 5, cytochrome P450
family (CYP)27 subfamily C member 1, WD repeat domain 33, POTE
ankyrin domain family member F, insulin receptor substrate 1,
thyroid hormone receptor interactor 12, S-antigen visual arrestin,
ATPase plasma membrane Ca2+ transporting 2,
neurexophilin and PC-esterase domain family member 3, Rho GTPase
activating protein 31, p21 (RAC1)-activated kinase 2, midnolin,
unc-13 homolog A, potassium channel tetramerization domain
containing 15, endogenous retrovirus group V member 1 envelope,
zinc finger and SCAN domain containing 18, KIAA0930 (also known as
C22orf9, LSC3), SET binding factor 1, forkhead box K1, AVL9 cell
migration-associated, FKBP prolyl isomerase family member 6, X-ray
repair cross complementing 2, phosphatase and actin regulator 1 and
mitochondrial calcium uniporter regulator 1. A previous study
reported on a pediatric patient with AD, mental retardation,
autistic features, epilepsy, developmental delay and abnormal
immunological results, who carried a 7.9 Mb de novo deletion
of chromosome 22q13.2/qter, a region containing the NCAPH2, SH3 and
multiple ankyrin repeat domains 3 and CYP2D6 genes (26). These genes are associated with the
T-cell immune response and the inflammatory response has also been
suggested to have roles in the pathogenesis of AD. Thus,
tRF-28-QSZ34KRQ590K may be involved in the pathogenesis of AD by
affecting the inflammatory response. Further studies are required
to uncover the role of tRF-28-QSZ34KRQ590K in the pathogenesis of
AD. At present, it remains undetermined whether tRF-28-QSZ34KRQ590K
is specifically expressed in AD, although it has not been reported
to be expressed in any other diseases, to the best of our
knowledge. However, tRF-28-QSZ34KRQ590K exhibited a significant
diagnostic value in the ROC curve analysis, which suggested that
tRF-28-QSZ34KRQ590K may be a potential biomarker for pediatric
patients with AD, although further studies are required to confirm
the role of this tRF in AD.
In conclusion, in the present study, 135
differentially expressed tRFs in the plasma exosomes of pediatric
patients with AD were identified, and the significantly
downregulated tRF-28-QSZ34KRQ590K and tRF-33-Q99P9P9NH57SD3 were
further verified. tRF-28-QSZ34KRQ590K exhibited significance in the
ROC curve analysis. Thus, tRF-28-QSZ34KRQ590K may be a potential
biomarker for AD in pediatric patients. To the best of our
knowledge, the present study was the first to report on the roles
of tRFs in AD. It provided novel biological information for
uncovering the pathogenesis of AD and a novel potential source for
the early diagnosis and targeted treatment of AD in the future.
However, further studies are required to better understand the
roles of tRFs in AD.
Supplementary Material
Verification of exosomes isolated from
plasma of patients with atopic dermatitis and controls. The
morphology of exosomes was analyzed by transmission electron
microscopy. (A) Magnified window displaying an exosome (scale bar,
100 nm) and (B) original microscopy image (scale bar, 200 nm).
Differentially expressed 135 tRFs in 3
subjects with AD compared with 3 controls.
Six types of differentially expressed
tRFs identified in 3 patients with atopic dermatitis and 3
controls.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural Science
Foundation of China (grant nos. 81602744 and 81573063).
Availability of data and materials
The raw data of small RNA sequencing of plasma
exosomes has been uploaded to the sequence read archive (http://www.ncbi.nlm.nih.gov/bioproject/609458;
submission ID: SUB7071247; BioProject accession no. PRJNA609458).
The raw data will be publicly available on March 1, 2021.
Authors' contributions
LM and LJ involved in drafting the manuscript and
revising it critically for important intellectual content, and have
made substantial contributions in the conception and design of the
current study. JC, HR, ZG, YW and FW were involved in the
acquisition of data, analysis and interpretation of data. LY was
responsible for the conception and design of the current study and
manuscript revision. LM and LY confirm the authenticity of all the
raw data. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
This study was approved by the ethics committee of
Shanghai Skin Disease Hospital (Shanghai, China) and all subjects
and their guardians provided written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Abrams EM and Sicherer S: Cutaneous
sensitization to peanut in children with atopic dermatitis: A
window to prevention of peanut allergy. JAMA Dermatol. 155:13–14.
2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Bin L and Leung DY: Genetic and epigenetic
studies of atopic dermatitis. Allergy Asthma Clin Immunol.
12(52)2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Guescini M, Genedani S, Stocchi V and
Agnati LF: Astrocytes and Glioblastoma cells release exosomes
carrying mtDNA. J Neural Transm (Vienna). 117:1–4. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Miranda KC, Bond DT, McKee M, Skog J,
Paunescu TG, Da Silva N, Brown D and Russo LM: Nucleic acids within
urinary exosomes/microvesicles are potential biomarkers for renal
disease. Kidney Int. 78:191–199. 2010.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Valadi H, Ekstrom K, Bossios A, Sjostrand
M, Lee JJ and Lotvall JO: Exosome-mediated transfer of mRNAs and
microRNAs is a novel mechanism of genetic exchange between cells.
Nat Cell Biol. 9:654–659. 2007.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Kumar P, Kuscu C and Dutta A: Biogenesis
and function of transfer RNA-related fragments (tRFs). Trends
Biochem Sci. 41:679–689. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ruggero K, Guffanti A, Corradin A, Sharma
VK, De Bellis G, Corti G, Grassi A, Zanovello P, Bronte V, Ciminale
V and D'Agostino DM: Small noncoding RNAs in cells transformed by
human T-cell leukemia virus type 1: A role for a tRNA fragment as a
primer for reverse transcriptase. J Virol. 88:3612–3622.
2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yeung ML, Bennasser Y, Watashi K, Le SY,
Houzet L and Jeang KT: Pyrosequencing of small non-coding RNAs in
HIV-1 infected cells: Evidence for the processing of a
viral-cellular double-stranded RNA hybrid. Nucleic Acids Res.
37:6575–6586. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Guo Y, Bosompem A, Mohan S, Erdogan B, Ye
F, Vickers KC, Sheng Q, Zhao S, Li CI, Su PF, et al: Transfer RNA
detection by small RNA deep sequencing and disease association with
myelodysplastic syndromes. BMC Genomics. 16(727)2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Martens-Uzunova ES, Jalava SE, Dits NF,
van Leenders GJ, Moller S, Trapman J, Bangma CH, Litman T,
Visakorpi T and Jenster G: Diagnostic and prognostic signatures
from the small non-coding RNA transcriptome in prostate cancer.
Oncogene. 31:978–991. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Victoria Martinez B, Dhahbi JM, Nunez
Lopez YO, Lamperska K, Golusinski P, Luczewski L, Kolenda T, Atamna
H, Spindler SR, Golusinski W and Masternak MM: Circulating small
non-coding RNA signature in head and neck squamous cell carcinoma.
Oncotarget. 6:19246–19263. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kumar P, Mudunuri SB, Anaya J and Dutta A:
tRFdb: A database for transfer RNA fragments. Nucleic Acids Res.
43:D141–D145. 2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Thompson DM and Parker R: Stressing out
over tRNA cleavage. Cell. 138:215–219. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Lee YS, Shibata Y, Malhotra A and Dutta A:
A novel class of small RNAs: tRNA-derived RNA fragments (tRFs).
Genes Dev. 23:2639–2649. 2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Goodarzi H, Liu X, Nguyen HC, Zhang S,
Fish L and Tavazoie SF: Endogenous tRNA-derived fragments suppress
breast cancer progression via YBX1 displacement. Cell. 161:790–802.
2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Rajka G and Hanifin JM: Diagnostic
features of atopic dermatitis. Acta Dermato-venereologica.
60:44–47. 1980.
|
|
17
|
Severity scoring of atopic dermatitis. The
SCORAD index. Consensus report of the European task force on atopic
dermatitis. Dermatology. 186:23–31. 1993.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zhang L, Liu S, Wang JH, Zou J, Zeng H,
Zhao H, Zhang B, He Y, Shi J, Yoshida S and Zhou Y: Differential
expressions of microRNAs and transfer RNA-derived Small RNAs:
Potential targets of choroidal neovascularization. Curr Eye Res.
44:1226–1235. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Mazzei M, Vascellari M, Zanardello C,
Melchiotti E, Vannini S, Forzan M, Marchetti V, Albanese F and
Abramo F: Quantitative real time polymerase chain reaction
(qRT-PCR) and RNAscope in situ hybridization (RNA-ISH) as effective
tools to diagnose feline herpesvirus-1-associated dermatitis. Vet
Dermatol. 30:e491–e147. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shen Y, Yu X, Zhu L, Li T, Yan Z and Guo
J: Transfer RNA-derived fragments and tRNA halves: Biogenesis,
biological functions and their roles in diseases. J Mol Med.
96:1167–1176. 2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Soares AR and Santos M: Discovery and
function of transfer RNA-derived fragments and their role in
disease. Wiley Interdiscip Rev RNA: 8, 2017 doi:
10.1002/wrna.1423.
|
|
22
|
Zhang Z, Xiao C, Gibson AM, Bass SA and
Khurana Hershey GK: EGFR signaling blunts allergen-induced IL-6
production and Th17 responses in the skin and attenuates
development and relapse of atopic dermatitis. J Immunol.
192:859–866. 2014.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Wu A, Chen H, Xu C, Zhou J, Chen S, Shi Y,
Xu J, Gan J and Zhang J: miR-203a is involved in HBx-induced
inflammation by targeting Rap1a. Exp Cell Res. 349:191–197.
2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kyriakis JM and Avruch J: Mammalian MAPK
signal transduction pathways activated by stress and inflammation:
A 10-year update. Physiol Rev. 92:689–737. 2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Hirota T, Takahashi A, Kubo M, Tsunoda T,
Tomita K, Sakashita M, Yamada T, Fujieda S, Tanaka S, Doi S, et al:
Genome-wide association study identifies eight new susceptibility
loci for atopic dermatitis in the Japanese population. Nat Genet.
44:1222–1226. 2012.PubMed/NCBI View
Article : Google Scholar
|
|
26
|
Chen CP, Lin SP, Chern SR, Tsai FJ, Wu PC,
Lee CC, Chen YT, Chen WL and Wang W: A de novo 7.9 Mb deletion in
22q13.2→qter in a boy with autistic features, epilepsy,
developmental delay, atopic dermatitis and abnormal immunological
findings. Eur J Med Genet. 53:329–332. 2010.PubMed/NCBI View Article : Google Scholar
|