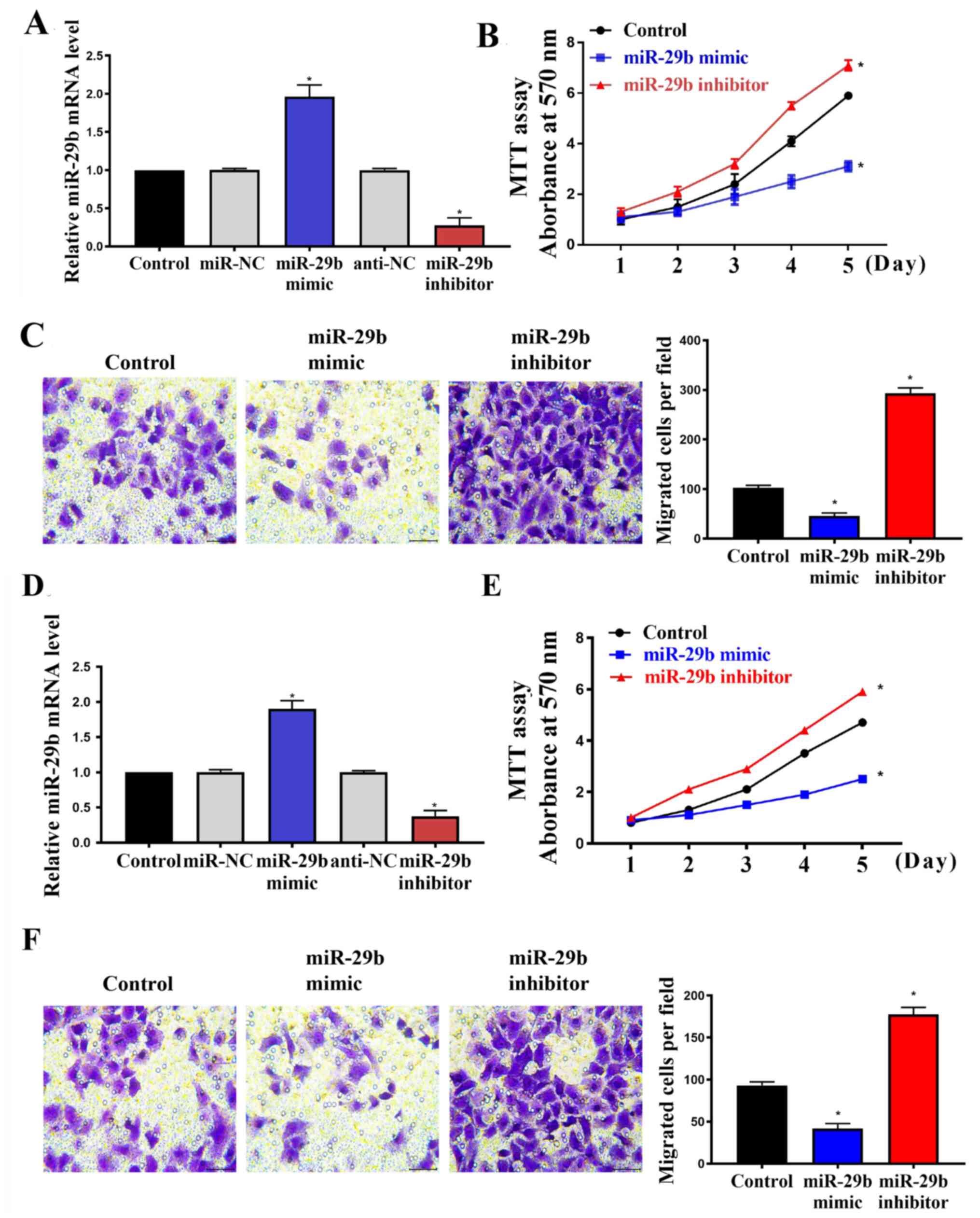
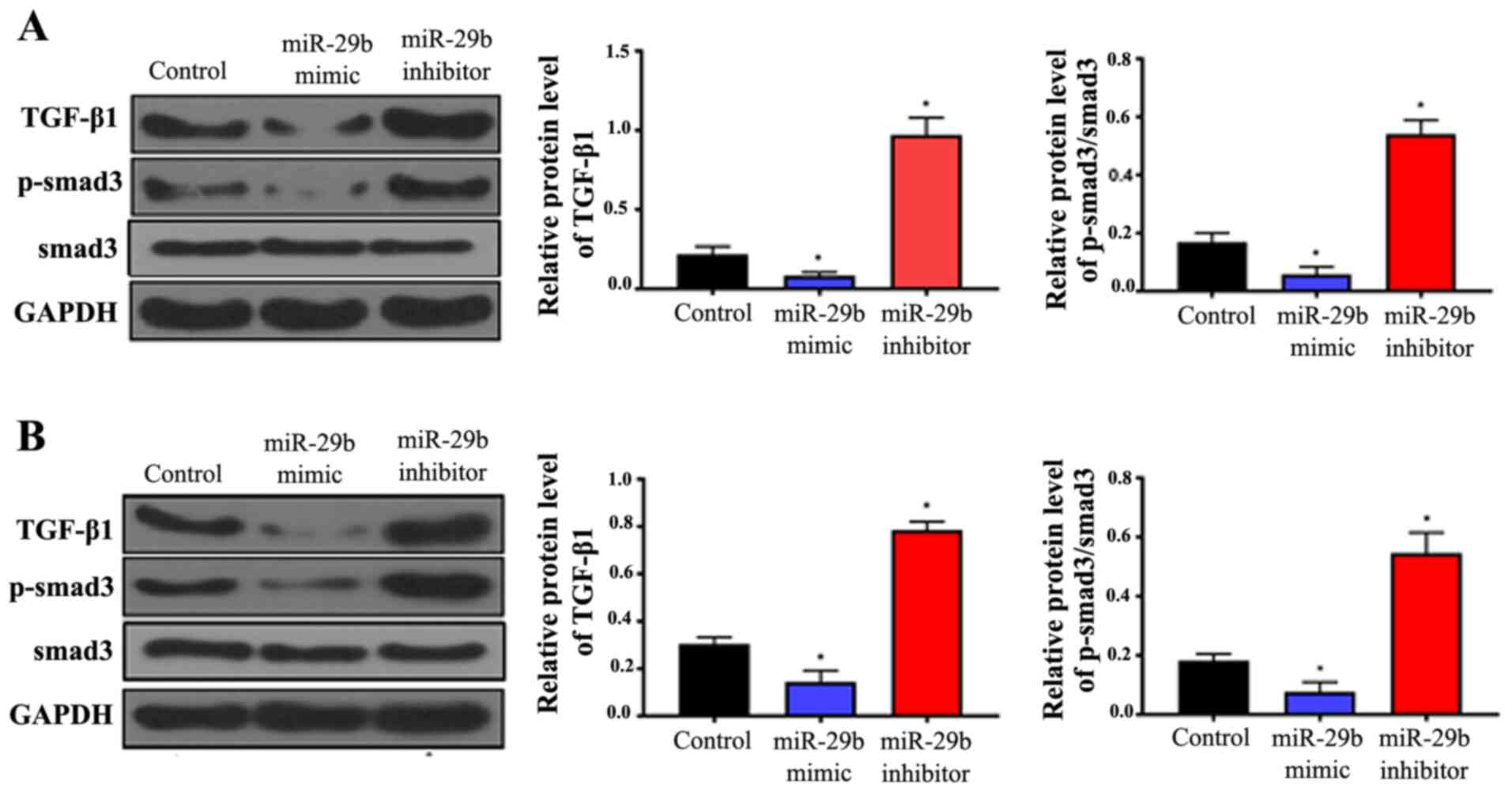
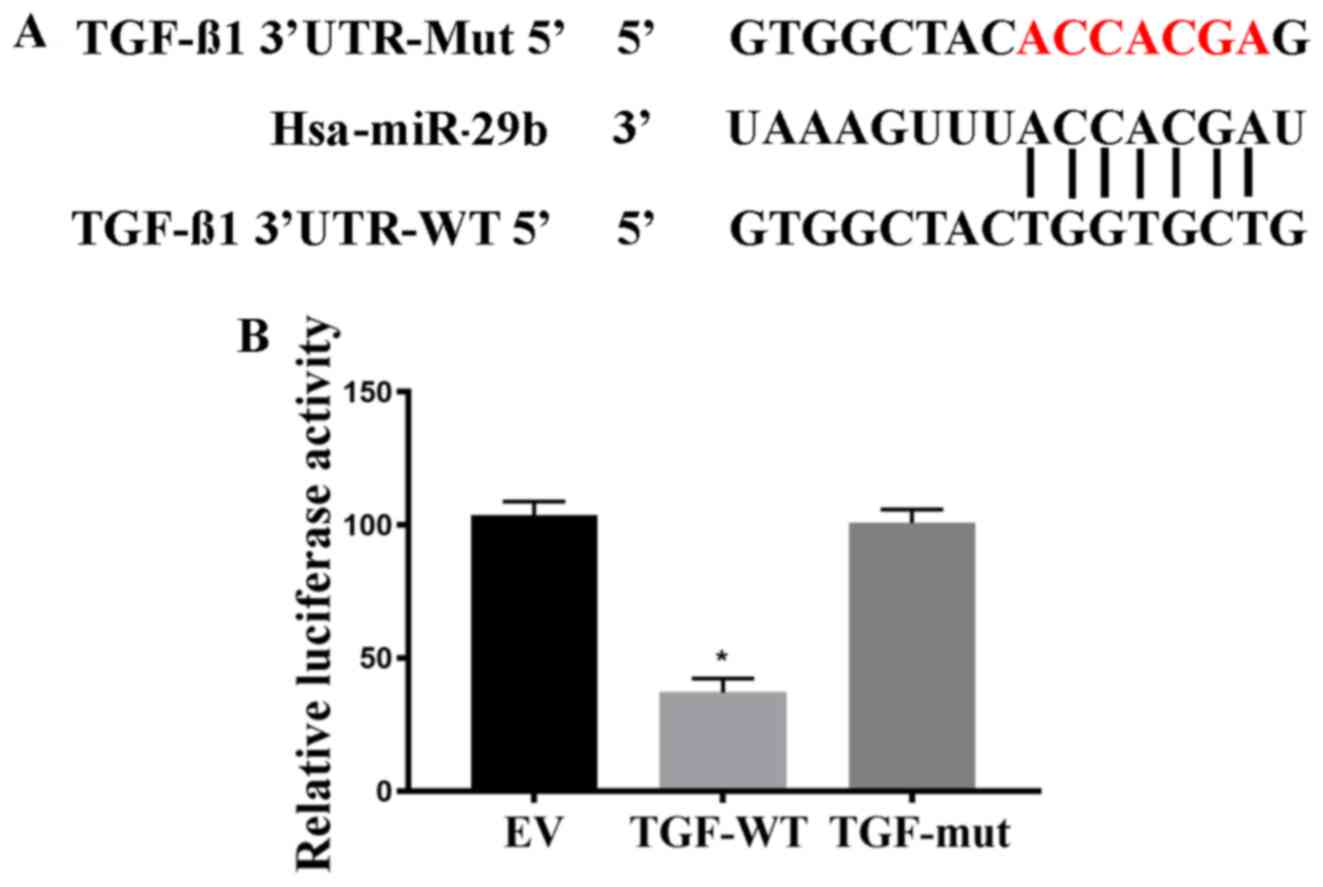
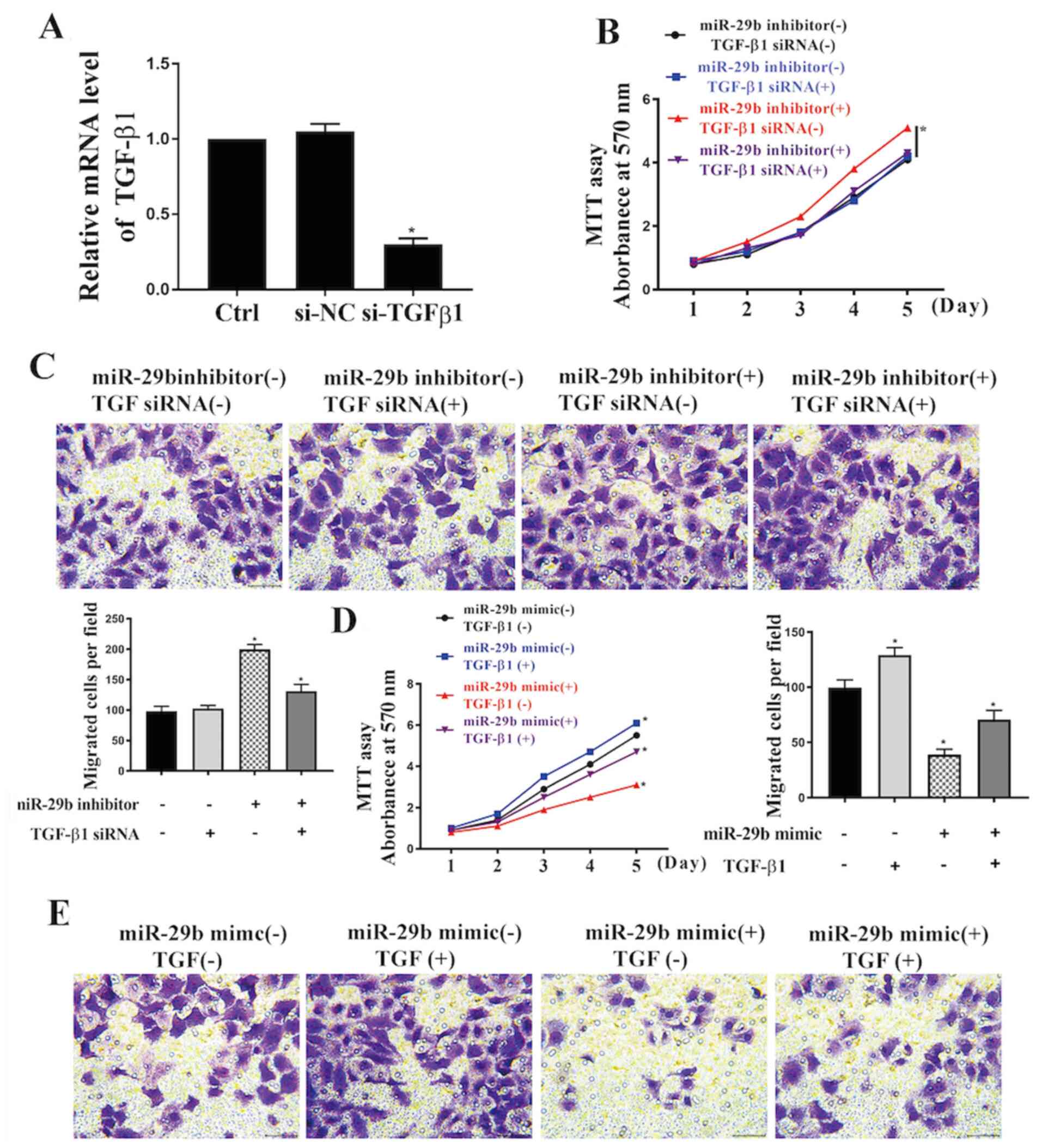
|
1
|
Frösen J, Tulamo R, Paetau A, Laaksamo E,
Korja M, Laakso A, Niemelä M and Hernesniemi J: Saccular
intracranial aneurysm: Pathology and mechanisms. Acta Neuropathol.
123:773–786. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Bor AS, Rinkel GJ, Adami J, Koffijberg H,
Ekbom A, Buskens E, Blomqvist P and Granath F: Risk of subarachnoid
haemorrhage according to number of affected relatives: A population
based case-control study. Brain. 131:2662–2665. 2008.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Louw DF, Asfora WT and Sutherland GR: A
brief history of aneurysm clips. Neurosurg Focus.
11(E4)2001.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ailawadi G, Moehle CW, Pei H, Walton SP,
Yang Z, Kron IL, Lau CL and Owens GK: Smooth muscle phenotypic
modulation is an early event in aortic aneurysms. J Thorac
Cardiovasc Surg. 138:1392–1399. 2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Nakajima N, Nagahiro S, Sano T, Satomi J
and Satoh K: Phenotypic modulation of smooth muscle cells in human
cerebral aneurysmal walls. Acta Neuropathol. 100:475–480.
2000.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ali MS, Starke RM, Jabbour PM, Tjoumakaris
SI, Gonzalez LF, Rosenwasser RH, Owens GK, Koch WJ, Greig NH and
Dumont AS: TNF-α induces phenotypic modulation in cerebral vascular
smooth muscle cells: Implications for cerebral aneurysm pathology.
J Cereb Blood Flow Metab. 33:1564–1573. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kawai-Kowase K and Owens GK: Multiple
repressor pathways contribute to phenotypic switching of vascular
smooth muscle cells. Am J Physiol Cell Physiol. 292:C59–C69.
2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Owens GK, Kumar MS and Wamhoff BR:
Molecular regulation of vascular smooth muscle cell differentiation
in development and disease. Physiol Rev. 84:767–801.
2004.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Lacolley P, Regnault V, Segers P and
Laurent S: Vascular smooth muscle cells and arterial stiffening:
Relevance in development, aging, and disease. Physiol Rev.
97:1555–1617. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mao N, Gu T, Shi E, Zhang G, Yu L and Wang
C: Phenotypic switching of vascular smooth muscle cells in animal
model of rat thoracic aortic aneurysm. Interact Cardiovasc Thorac
Surg. 21:62–70. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
van Rooij E, Marshall WS and Olson EN:
Toward microRNA-based therapeutics for heart disease: The sense in
antisense. Circ Res. 103:919–928. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Small EM and Olson EN: Pervasive roles of
microRNAs in cardiovascular biology. Nature. 469:336–342.
2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
van Rooij E and Olson EN: MicroRNA
therapeutics for cardiovascular disease: Opportunities and
obstacles. Nat Rev Drug Discov. 11:860–872. 2012.PubMed/NCBI View
Article : Google Scholar
|
|
14
|
Liu D, Han L, Wu X, Yang X, Zhang Q and
Jiang F: Genome-wide microRNA changes in human intracranial
aneurysms. BMC Neurol. 14(188)2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yan B, Guo Q, Fu FJ, Wang Z, Yin Z, Wei YB
and Yang JR: The role of miR-29b in cancer: Regulation, function,
and signaling. Onco Targets Ther. 8:539–548. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zhang Y, Huang XR, Wei LH, Chung AC, Yu CM
and Lan HY: miR-29b as a therapeutic agent for angiotensin
II-induced cardiac fibrosis by targeting TGF-β/Smad3 signaling. Mol
Ther. 22:974–985. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
McMullen JR and Bernardo BC: Inhibition of
miR-29 protects against cardiac hypertrophy and fibrosis: New
insight for the role of miR-29 in the heart. Non-coding RNA
Investig. 2(14)2018.
|
|
18
|
Wang G, Matsuura I, He D and Liu F:
Transforming growth factor-{beta}-inducible phosphorylation of
Smad3. J Biol Chem. 284:9663–9673. 2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Sun L, Zhao M, Zhang J, Lv M, Li Y, Yang
X, Liu A and Wu Z: MirR-29B downregulation induces phenotypic
modulation of vascular smooth muscle cells: Implication for
intracranial aneurysm formation and progression to rupture. Cell
Physiol Biochem. 41:510–518. 2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Fang J, Xu H, Yang C, Morsalin S,
Kayarthodi S, Rungsrisuriyachai K, Gunnal U, Mckenzie B, Rao VN and
Reddy ES: Ets related gene and Smad3 proteins collaborate to
activate transforming growth factor-beta mediated signaling pathway
in ETS related gene-positive prostate cancer cells. J Pharm Sci
Pharmacol. 1:175–181. 2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kole AJ, Swahari V, Hammond SM and
Deshmukh M: miR-29b is activated during neuronal maturation and
targets BH3-only genes to restrict apoptosis. Genes Dev.
25:125–130. 2011.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Eyholzer M, Schmid S, Wilkens L, Mueller
BU and Pabst T: The tumour-suppressive miR-29a/b1 cluster is
regulated by CEBPA and blocked in human AML. Br J Cancer.
103:275–284. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Tsai S, Hollenbeck ST, Ryer EJ, Edlin R,
Yamanouchi D, Kundi R, Wang C, Liu B and Kent KC: TGF-beta through
Smad3 signaling stimulates vascular smooth muscle cell
proliferation and neointimal formation. Am J Physiol Heart Circ
Physiol. 297:H540–H549. 2009.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Flanders KC, Sullivan CD, Fujii M, Sowers
A, Anzano MA, Arabshahi A, Major C, Deng C, Russo A, Mitchell JB,
et al: Mice lacking Smad3 are protected against cutaneous injury
induced by ionizing radiation. Am J Pathol. 160:1057–1068.
2002.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Shi Y and Massagué J: Mechanisms of
TGF-beta signaling from cell membrane to the nucleus. Cell.
113:685–700. 2003.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Ma J, Wang Q, Fei T, Han JD and Chen YG:
MCP-1 mediates TGF-beta-induced angiogenesis by stimulating
vascular smooth muscle cell migration. Blood. 109:987–994.
2007.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Salcedo R, Ponce ML, Young HA, Wasserman
K, Ward JM, Kleinman HK, Oppenheim JJ and Murphy WJ: Human
endothelial cells express CCR2 and respond to MCP-1: Direct role of
MCP-1 in angiogenesis and tumor progression. Blood. 96:34–40.
2000.PubMed/NCBI
|
|
29
|
Ma F, Li Y, Jia L, Han Y, Cheng J, Li H,
Qi Y and Du J: Macrophage-stimulated cardiac fibroblast production
of IL-6 is essential for TGF β/Smad activation and cardiac fibrosis
induced by angiotensin II. PLoS One. 7(e35144)2012.PubMed/NCBI View Article : Google Scholar
|
|
30
|
van Rooij E, Sutherland LB, Thatcher JE,
DiMaio JM, Naseem RH, Marshall WS, Hill JA and Olson EN:
Dysregulation of microRNAs after myocardial infarction reveals a
role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci USA.
105:13027–13032. 2008.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Davis-Dusenbery BN, Wu C and Hata A:
Micromanaging vascular smooth muscle cell differentiation and
phenotypic modulation. Arterioscler Thromb Vasc Biol. 31:2370–2377.
2011.PubMed/NCBI View Article : Google Scholar
|
|
32
|
ten Dijke P and Arthur HM: Extracellular
control of TGFbeta signalling in vascular development and disease.
Nat Rev Mol Cell Biol. 8:857–869. 2007.PubMed/NCBI View
Article : Google Scholar
|
|
33
|
Petsophonsakul P, Furmanik M, Forsythe R,
Dweck M, Schurink GW, Natour E, Reutelingsperger C, Jacobs M, Mees
B and Schurgers L: Role of vascular smooth muscle cell phenotypic
switching and calcification in aortic aneurysm formation.
Arterioscler Thromb Vasc Biol. 39:1351–1368. 2019.PubMed/NCBI View Article : Google Scholar
|