Introduction
Eosinophils are a type of white blood cell formed
from stem cells in the bone marrow. Raised eosinophil count is
associated with a wide variety of allergic, rheumatologic,
infectious, neoplastic and rare idiopathic disorders (1). Several drugs, including antimalarials,
non-steroid anti-inflammatories and anticonvulsants, are associated
with eosinophilia (2).
Eosinophilia has been documented as a
well-established side effect of the antipsychotic drug clozapine,
as it is seen in ~1% of clozapine-treated patients (3). However, limited data are available
regarding the potential effect of other antipsychotic medications
on eosinophil count (4,5).
Despite the lack of systematic studies, eosinophilia
related to antipsychotic use has been described in a number of case
reports; olanzapine, most notably, has been associated with
increased eosinophil count (6-8)
whilst it has also been found to cause eosinophilic pleural
effusion (6), drug reaction with
eosinophilia and systemic symptoms (DRESS) syndrome (9), eosinophilic myocarditis (10), as well as hypersensitivity syndrome
(11). Aripiprazole has also been
associated with DRESS syndrome (12) and eosinophilic myocarditis (13), Quetiapine has been associated with
eosinophilia (14,15), refractory cardiac myocarditis and
DRESS syndrome (14), and one case
report described risperidone-induced acute eosinophilic pneumonia
(16).
The purpose of this study was to generate a larger
case series on the possible association between raised eosinophil
count and any of the following six -first and second-generation
antipsychotic drugs: Olanzapine, Aripiprazole, Quetiapine,
Risperidone, Haloperidol and Amisulpride. The present study did not
include any patients on Clozapine, since its relation to increased
eosinophil count is well documented in the literature (3,17).
Materials and methods
Ethics approval
The study was conducted at the Department of
Psychiatry at the University General Hospital ‘Attikon’, a tertiary
care multispecialty hospital in Athens, Greece. Ethics approval for
this study and approval for use of patient data were obtained from
the Scientific Committee of ‘Attikon’ Hospital (Athens, Greece)
with the approval number (of the research project of which this
present study is a part) being 14/210109. All patients gave
informed consent on admission with regards to the use of their
data, which they could withdraw at any point. All procedures
performed in the study were in accordance with the ethical
standards of the Helsinki Declaration (1964) and its later
amendments.
Data collection and inclusion
criteria
Data were obtained from the database of electronic
and paper patient records of the Department of Psychiatry of
Attikon Hospital. Patients admitted to the Department of Psychiatry
between April 4, 2016 and November 1, 2019 who received standard
clinical treatment at the time of the study were included if they
met the following criteria: i) Sufficient duration of
hospitalisation (t>2 weeks), so that the fluctuation in the
eosinophil count could be monitored; ii) monotherapy treatment with
one antipsychotic medication in-hospital; iii) no administration of
other medication that could contribute to eosinophil count
fluctuations (e.g., antidepressants or antibiotics) during hospital
stay; iv) access to a reliable past medical history, including
electronic medical records; v) either no history of receiving
antipsychotics for a 6-month period prior to hospitalization or
being antipsychotic naïve; vi) if they had at least two blood tests
measuring eosinophil count during their hospital stay; and vii) no
history of any serious organic illness (such as respiratory
problems, including pneumonia, intestinal problems, such as
colitis, skin conditions, recent or chronic infections, allergies,
cancer, autoimmune disorders and haematological disorders) that
could in any way affect eosinophil count. Also, no history of any
specific eosinophilic disorders, such as eosinophilic
granulomatosis with polyangiitis (EGPA, formerly known as
Churg-Strauss), eosinophilic fasciitis, eosinophilic lung
disorders, hypereosinophilic syndrome and eosinophilic leukemia.
The medical history was obtained through the patients' own
self-report and also through the available electronic medical
records of the patients, which were accessible once they were
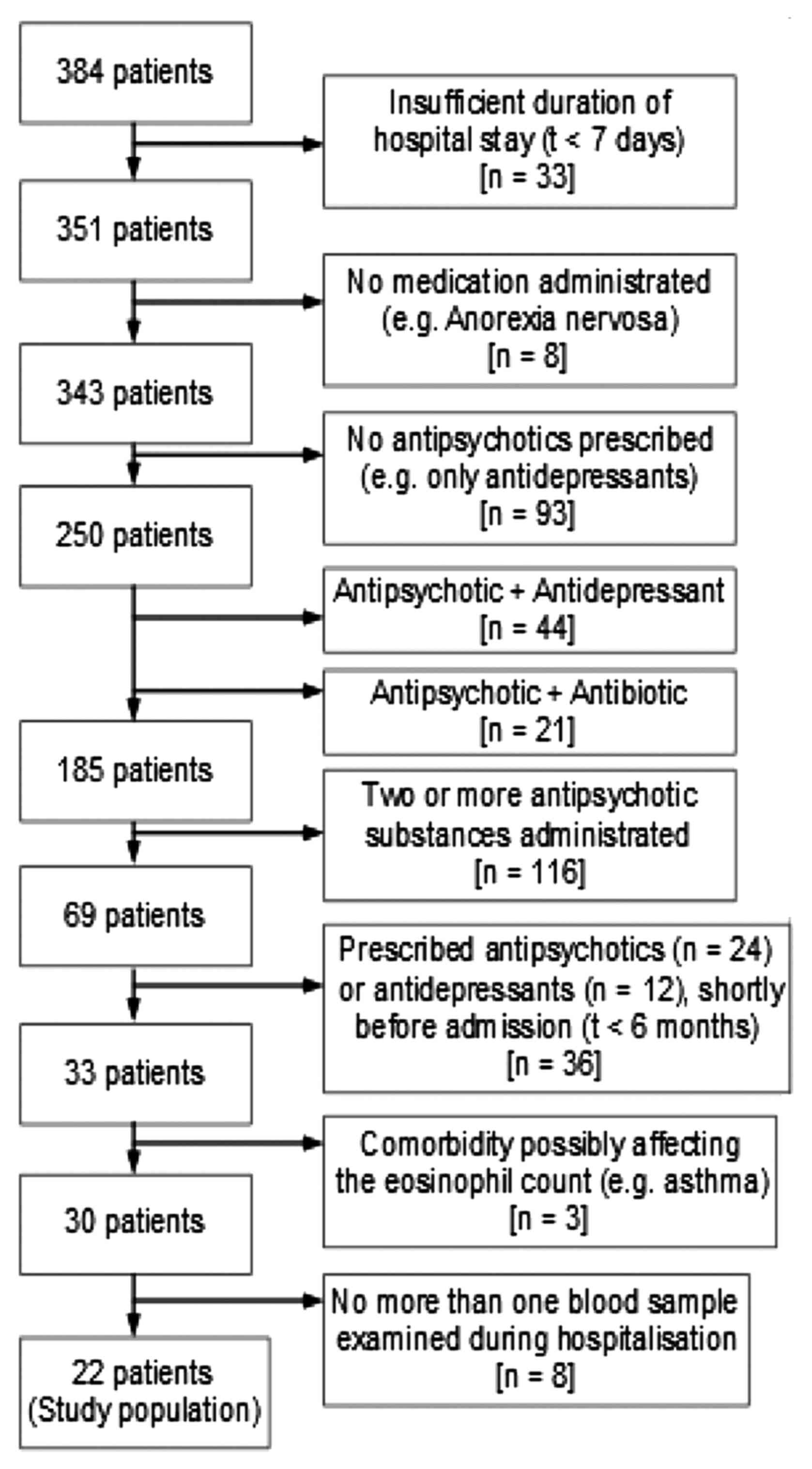
admitted to hospital (Fig. 1, for
details and flow-chart).
Statistical analysis
Patients included were tested every week for a
period of 2-6 weeks and the levels of eosinophils (absolute
eosinophil count in K/µl and eosinophil WBC percentage) were
extracted. The first measurement of the eosinophils represents the
baseline eosinophil count before the patients started treatment
with antipsychotic medication.
To study the effect of time and drug type on the
concentration (or relative %) the slopes of the least square
regression lines for each patient are presented and were analysed
with single factor ANOVA.
Summary measures analysis and single factor (or one
way) ANOVA were applied to test the difference in the means of the
eosinophil concentrations or relative % when there were sufficient
(3 or more) patients in a group by patient and by time (18,19).
Results
Sample characteristics
A total of 384 patients admitted in the 3 ½-year
period and receiving antipsychotic treatment were identified. Of
these, 22 patients met the inclusion criteria of the present study
(Fig. 1)
The mean age of the 22 patients included was 45.2
years, 36% were female, and the average duration of the hospital
stay was 22 days. Of those, 9 (40.9%) received monotherapy with
Olanzapine, 6 (27.3%) Haloperidol, 3 (13.6%) Aripiprazole, 2 (9.1%)
Amisulpride, 1 (4.5%) Quetiapine and 1 (4.5%) Risperidone.
Changes in eosinophil count associated
with antipsychotic use
Different patients responded differently to each
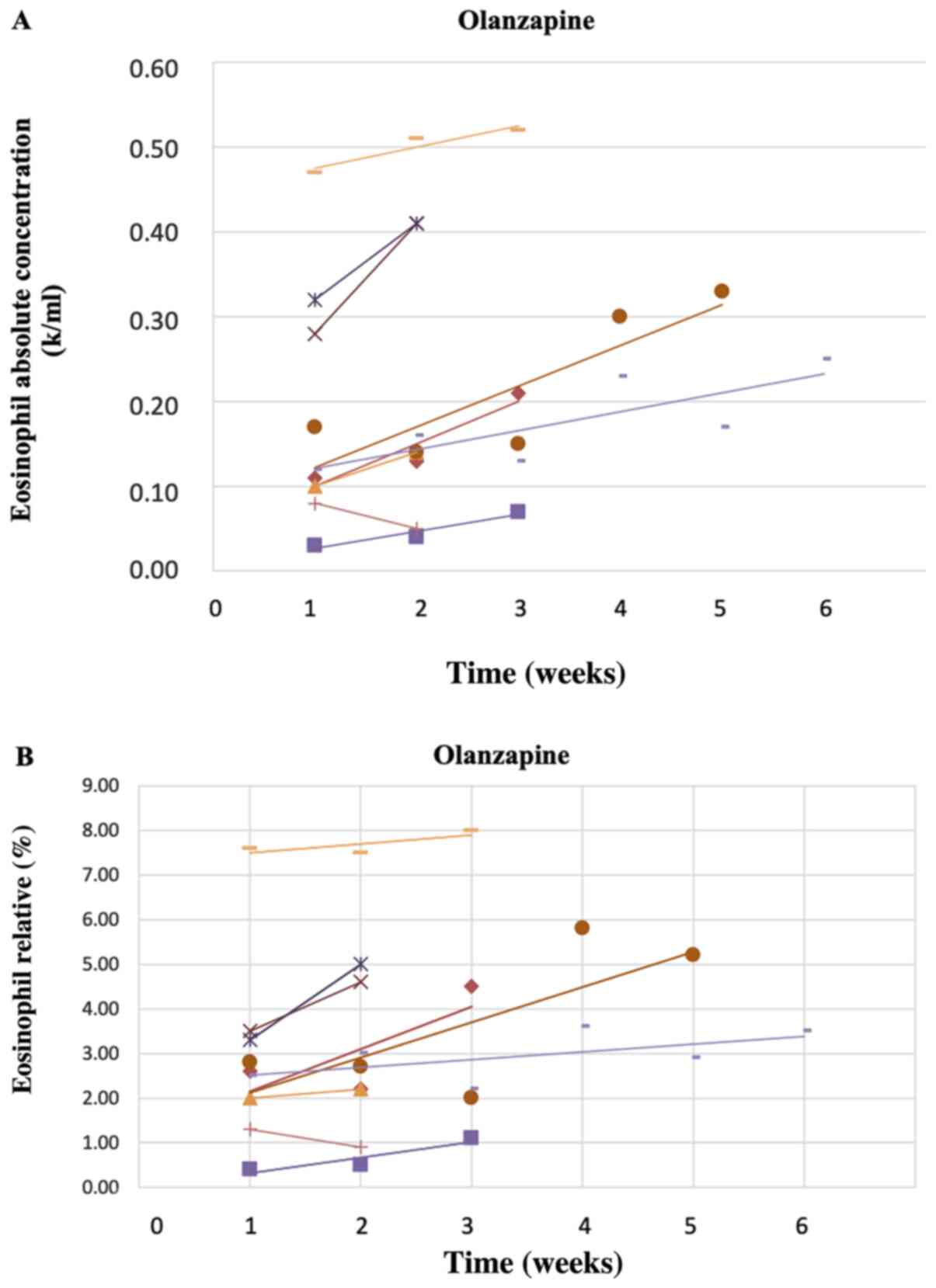
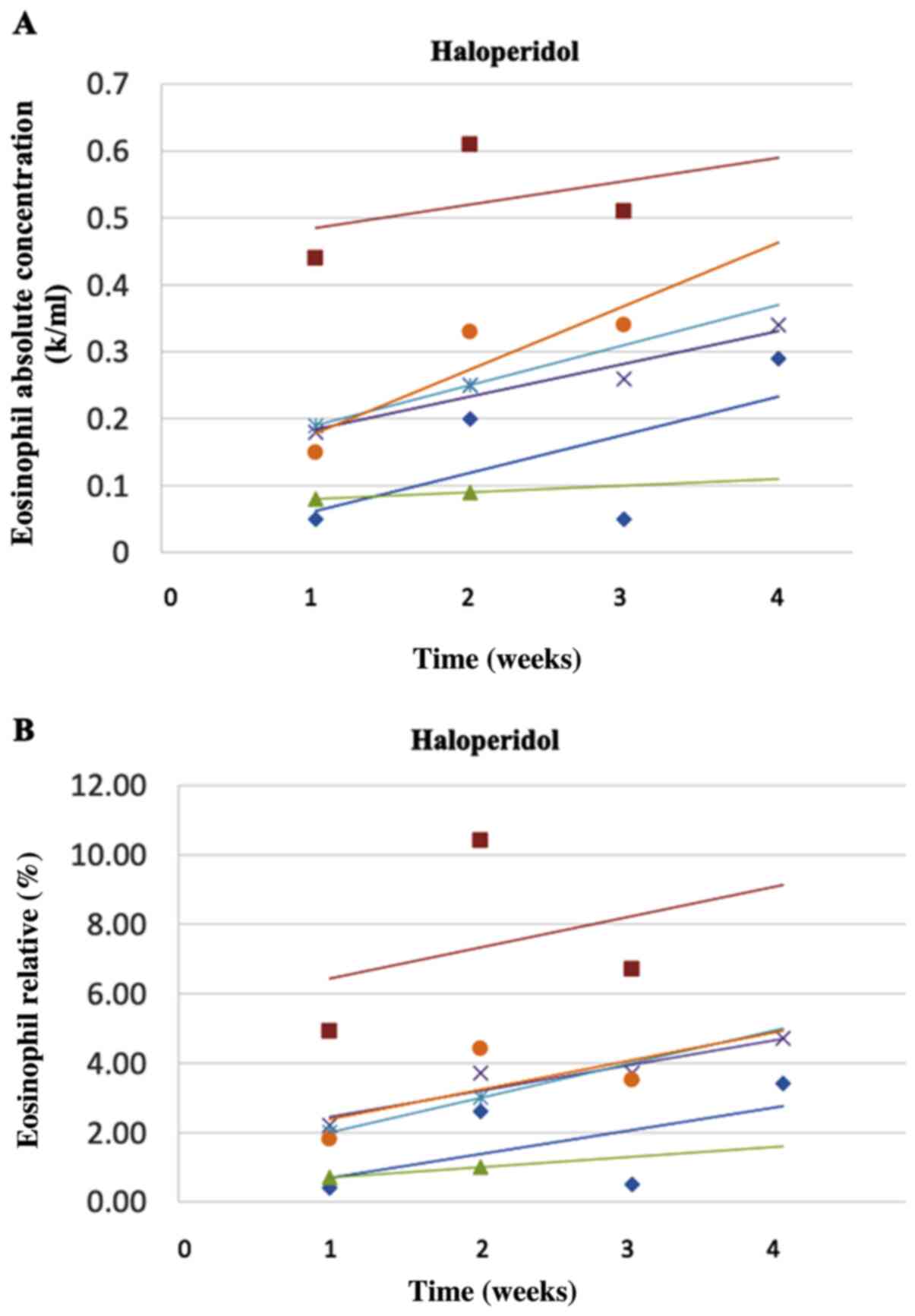
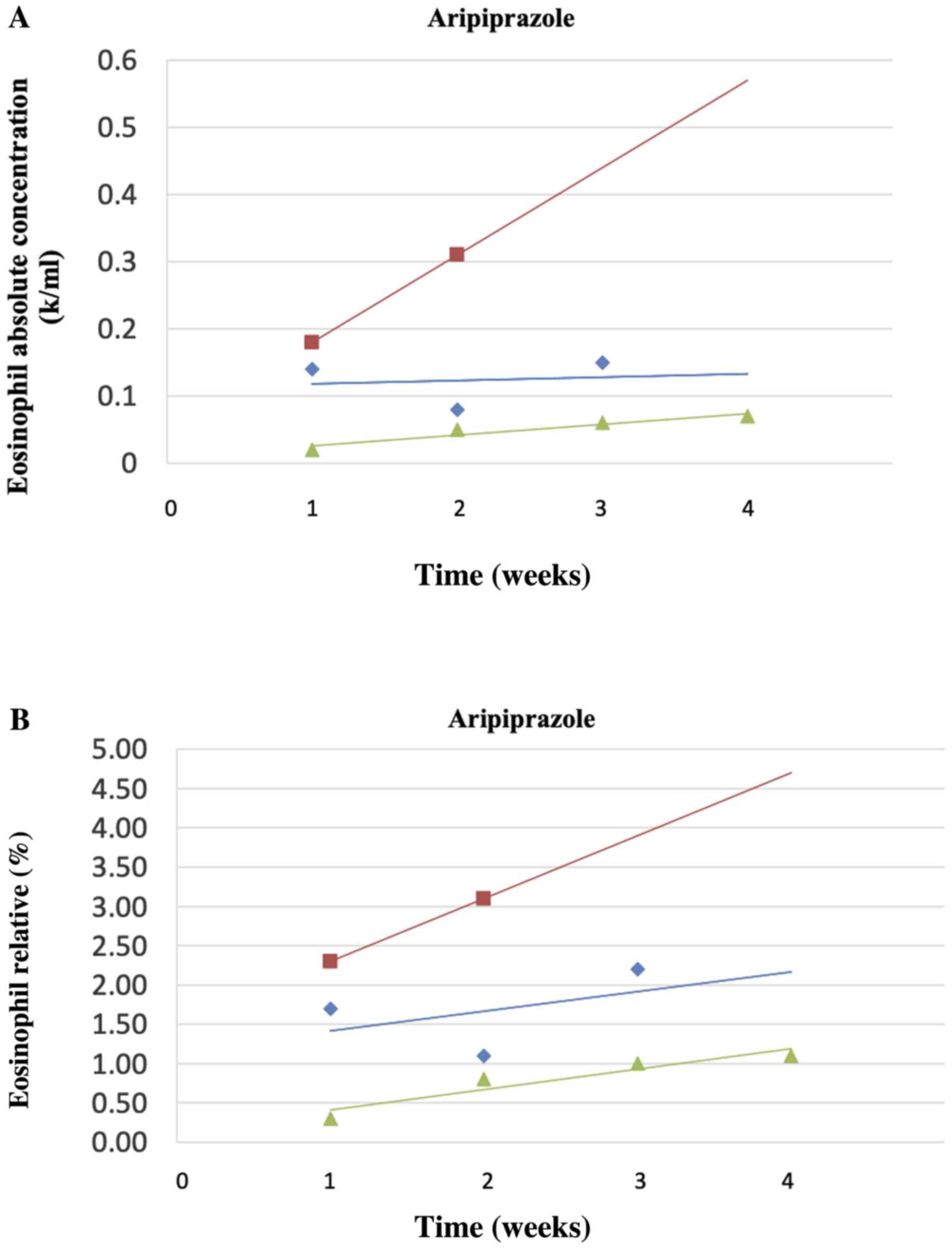
drug in terms of eosinophil count. Figs. 2A and B, 3A and
B, and 4A and B
show the corresponding changes in the absolute count (Figs. 2A, 3A and 4A)
and relative % (Figs. 2B, 3B and 4B)
for Olanzapine, Haloperidol and Aripiprazole, respectively (where
three or more patients were included for each drug) over time. A
trendline was fitted to the data of each patient and is shown in
the figures. Increases in the mean absolute count and relative %
were observed for all antipsychotic medications. Haloperidol showed
the greatest increase overall, ~100% for both the absolute count
and the %. Of note, the antipsychotic doses used were the regular
-within therapeutic range- doses that are used in clinical
practice. More specifically, for Olanzapine the doses used were
between 5 and 20 mg daily, for Aripiprazole they were between 10
and 30 mg daily and for Haloperidol they were between 5 and 20 mg
daily.
Significant differences for eosinophils' relative %
over time were detected for 9 patients in the Olanzapine group
(F=12.3>F-critical; P<0.001), the 6 patients in the
Haloperidol group (F=5.8>F-critical; P=0.006), and the 3
patients in the Aripiprazole group (F=11.3>F-critical; P=0.009),
while significant differences were also seen in the absolute
eosinophil values in patients on Olanzapine (F=17.2>F-critical;
P<0.001), Haloperidol (F=8.1>F-critical, P=0.002) and
Aripiprazole (F=12.0>F-critical; P=0.008).
No statistically significant differences in the
means among drugs over time and no differences in the absolute and
relative % eosinophil count from one week to the next were detected
for the three antipsychotics Olanzapine (F=0.3; P=0.91),
Haloperidol (F=0.7; P=0.59) and Aripiprazole (F=0.1; P=0.97).
Discussion
The present study investigated the impact of
antipsychotic monotherapy on the eosinophil count of a case series
of 22 psychiatric inpatients over time. The present findings
suggest a linear increase in the absolute concentration and the
relative % of eosinophils with time for at least 3 of the
antipsychotic drugs, Olanzapine, Haloperidol and Aripiprazole (for
which there were at least 3 patients on each). An increase between
the first and last measurement of the eosinophil count was also
seen for the other three antipsychotics (Amisulpride, Risperidone
and Quetiapine) which were taken by groups of two patients or a
single patient (data not shown).
Although there were differences in the increase of
the eosinophil count among patients taking the same medication as a
function of time, there was no difference in the rate of increase
between medications, which means each medication caused a similar
effect on the eosinophils (increases with similar variance) of the
patients with time.
To the best of our knowledge, this is the first case
series investigating the association between antipsychotics and
eosinophilic count. The study's strengths include that patients
were on antipsychotic monotherapy and that both first and second
generation antipsychotics were investigated. In addition, strict
exclusion criteria were applied to minimize the influence of other
factors that could cause eosinophilia. This strict approach has
generated a more homogenous, but small case series lacking
statistical power to detect significant findings between
medications. Hence, our findings indicate that antipsychotic
medications have an effect on the eosinophil count, but more
patients need to be studied for a longer period of time. Also,
since the primary aim of the study was to explore the effect of
antipsychotic use on eosinophil count, data on neutrophils were not
collected and analysed; however, this matter will be dealt with in
a future study.
Although there are no confirmed mechanisms by which
antipsychotics may cause eosinophilia, two possible explanations
can be found in the literature. The first mechanism is called
pharmacologic interaction with immune receptors (p-I concept) and
suggests that medications may be associated with eosinophilia by
activating T-cells. This would lead to the release of cytokines
stimulating eosinophils directly and/or eosinophil precursors
(20). The second possible
mechanism concerns the molecule of serotonin. According to animal
studies, serotonin and eotaxin have been demonstrated to have an
eosinophil chemoattractant profile. This most likely occurs via the
5HT2A receptor (21), which has
been reported to be a mediator of inflammation outside of the
central nervous system (21,22).
The serotoninergic action of antipsychotic drugs is mediated via
their affinity for 5-HT2A receptors, especially at low dosage. It
is therefore arguable that the serotonergic action of antipsychotic
drugs through antagonism of 5-HT2A receptors could result in a rise
in eosinophil count. Serotonin eosinophilic-specific
chemo-attracting action induced by a low dose antipsychotic
(risperidone) has been previously implicated as a mechanism leading
to eosinophilic pneumonia (16).
In conclusion, the present case series shows a
possible signal in the association between antipsychotic use and
raised eosinophil count. The small sample size however precludes
inferring causal associations. However, it adds to the literature
in an under-researched area, and suggests that potential serious
consequences of raised eosinophils, such as DRESS syndrome
(9), eosinophilic myocarditis
(13) and pneumonia (16), warrant further investigations. Our
estimation is that a sufficiently powered future study should have
at least 8-10 patients per drug, and 5-8 consecutive measurements
(i.e., follow-up for 5-8 weeks) per patient for all patients. Such
a study is being constructed, since data collection regarding
eosinophils and antipsychotic use remains ongoing in our
department, and the results will be presented in the near future
once a sufficiently powered sample is reached.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
KT and CM wrote the original draft, edited and
critically revised the manuscript. KT and IH confirmed the
authenticity of all the raw data. KT, CM, MK, IT, NSi, SK, AO, EA,
ER, DT, DAS and NSm made substantial contributions to the
conception and design of the current study and interpreted the
data. IH, IT, EA and ER acquired the data. MK, CM, IH and KT made
substantial contributions to data handling, figure preparation and
analysis of the data. All authors critically revised and edited the
manuscript. All authors substantially contributed to the
conception, writing and revision of the work and read and approved
the final content of the manuscript.
Ethics approval and consent to
participate
The authors assert that all procedures contributing
to this work comply with the ethical standards of the relevant
national and institutional committees on human experimentation and
with the Helsinki Declaration of 1975, as revised in 2008. The
study was conducted at the Department of Psychiatry at the
University General Hospital ‘Attikon’, a tertiary care
multispecialty hospital in Athens, Greece. Ethics Approval for this
study and approval for use of patient data were obtained by the
Scientific Committee of ‘Attikon’ Hospital (Athens, Greece)
(approval no. 14/210109). All patients provided informed consent on
admission with regards to the use of their data, which they could
withdraw at any time.
Patient consent for publication
Not applicable.
Competing interests
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
The other authors declare that they have no competing
interests.
References
|
1
|
Klion AD: Eosinophilia: A pragmatic
approach to diagnosis and treatment. Hematology Am Soc Hematol Educ
Program, 92-97: 2015.
|
|
2
|
Rauscher C and Freeman A: Drug-induced
eosinophilia. Allergy Asthma Proc. 39:252–256. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kadiyala PK, Ahmed MA, Pinto DA and Mathai
JP: Clozapine induced eosinophilia: An often neglected important
adverse effect. Indian J Psychiatry. 57:429–430. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gentile S: Safety concerns associated with
second-generation antipsychotic long-acting injection treatment. A
systematic update. Horm Mol Biol Clin Investig 36, 2018.
|
|
5
|
Rettenbacher MA, Hofer A, Kemmler G and
Fleischhacker WW: Neutropenia induced by second generation
antipsychotics: A prospective investigation. Pharmacopsychiatry.
43:41–44. 2010.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Huang J, Yu Y, Lin W, Zhang D, Deng Z and
Ding Q: Olanzapine-induced peripheral eosinophilia and eosinophilic
pleural effusion: A case report. Medicine (Baltimore).
97(e9996)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tournikioti K, Douzenis A, Antoniadou A,
Papazahos K, Moschos C, Papageorgiou C and Rizos EN: Eosinophilia
Associated With Olanzapine. J Clin Psychopharmacol. 36:180–181.
2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mathias S, Schaaf LWN and Sonntag A:
Eosinophilia associated with olanzapine. J Clin Psychiatry.
63:246–247. 2002.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Penchilaiya V, Kuppili PP, Preeti K and
Bharadwaj B: DRESS syndrome: Addressing the drug hypersensitivity
syndrome on combination of Sodium Valproate and Olanzapine. Asian J
Psychiatr. 28:175–176. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Vang T, Rosenzweig M, Bruhn CH,
Polcwiartek C, Kanters JK and Nielsen J: Eosinophilic myocarditis
during treatment with olanzapine-report of two possible cases. BMC
Psychiatry. 16(70)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Raz A, Eilam O, Hayek T, Bergman R and
Yungerman T: A Case report of olanzapine-induced hypersensitivity
syndrome. Am J Med Sci. 321:156–158. 2001.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Taleb S, Zgueb Y, Ouali U, Jomli R, Kort Y
and Nacef F: Drug reaction with eosinophilia and systemic symptoms
syndrome related to aripiprazole therapy. J Clin Psychopharmacol.
39:691–693. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Christoffersen RK, Vestergård LD, Høimark
L and Vesterby A: Eosinophilic myocarditis and sudden unexpected
death in a younger patient treated with antipsychotics. Ugeskr
Laeger. 173:2799–2800. 2011.PubMed/NCBI(In Danish).
|
|
14
|
Hagiwara H, Fukushima A, Iwano H and Anzai
T: Refractory cardiac myocarditis associated with drug rash with
eosinophilia and systemic symptoms syndrome due to anti-bipolar
disorder drugs: A case report. Eur Hear J Case Rep.
2(yty100)2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chen L, Tan P and Tan X: Case report of
eosinophilia induced by quetiapine. Shanghai Arch psychiatry.
27:374–377. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Rizos E, Tsigkaropoulou E, Lambrou P,
Kanakaki M, Chaniotou A, Alevyzakis E and Liappas I:
Risperidone-induced acute eosinophilic pneumonia. In Vivo.
27:651–653. 2013.PubMed/NCBI
|
|
17
|
Lee J, Takeuchi H, Fervaha G, Powell V,
Bhaloo A, Bies R and Remington G: The effect of clozapine on
hematological indices: A 1-Year Follow-Up Study. J Clin
Psychopharmacol. 35:510–516. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Matthews JN, Altman DG, Campbell MJ and
Royston P: Analysis of serial measurements in medical research.
BMJ. 300:230–235. 1990.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Matthews JNS: A refinement to the analysis
of serial data using summary measures. Stat Med. 12:27–37.
1993.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Pichler WJ, Beeler A, Keller M, Lerch M,
Posadas S, Schmid D, Spanou Z, Zawodniak A and Gerber B:
Pharmacological interaction of drugs with immune receptors: The p-i
concept. Allergol Int. 55:17–25. 2006.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Boehme SA, Lio FM, Sikora L, Pandit TS,
Lavrador K, Rao SP and Sriramarao P: Cutting edge: Serotonin is a
chemotactic factor for eosinophils and functions additively with
eotaxin. J Immunol. 173:3599–3603. 2004.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Baganz NL and Blakely RD: A Dialogue
between the immune system and brain, spoken in the language of
serotonin. ACS Chem Neurosci. 4:48–63. 2013.PubMed/NCBI View Article : Google Scholar
|