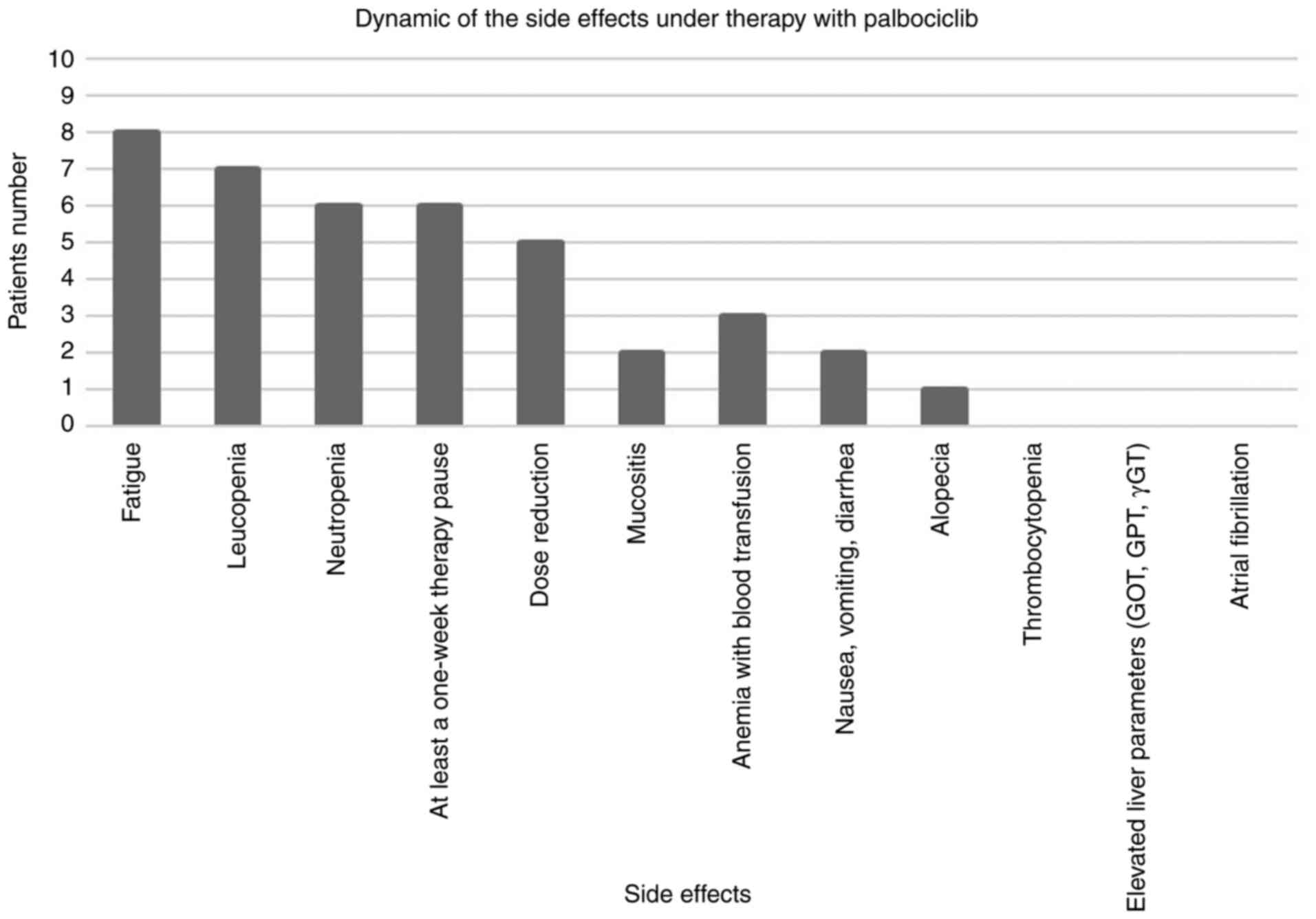
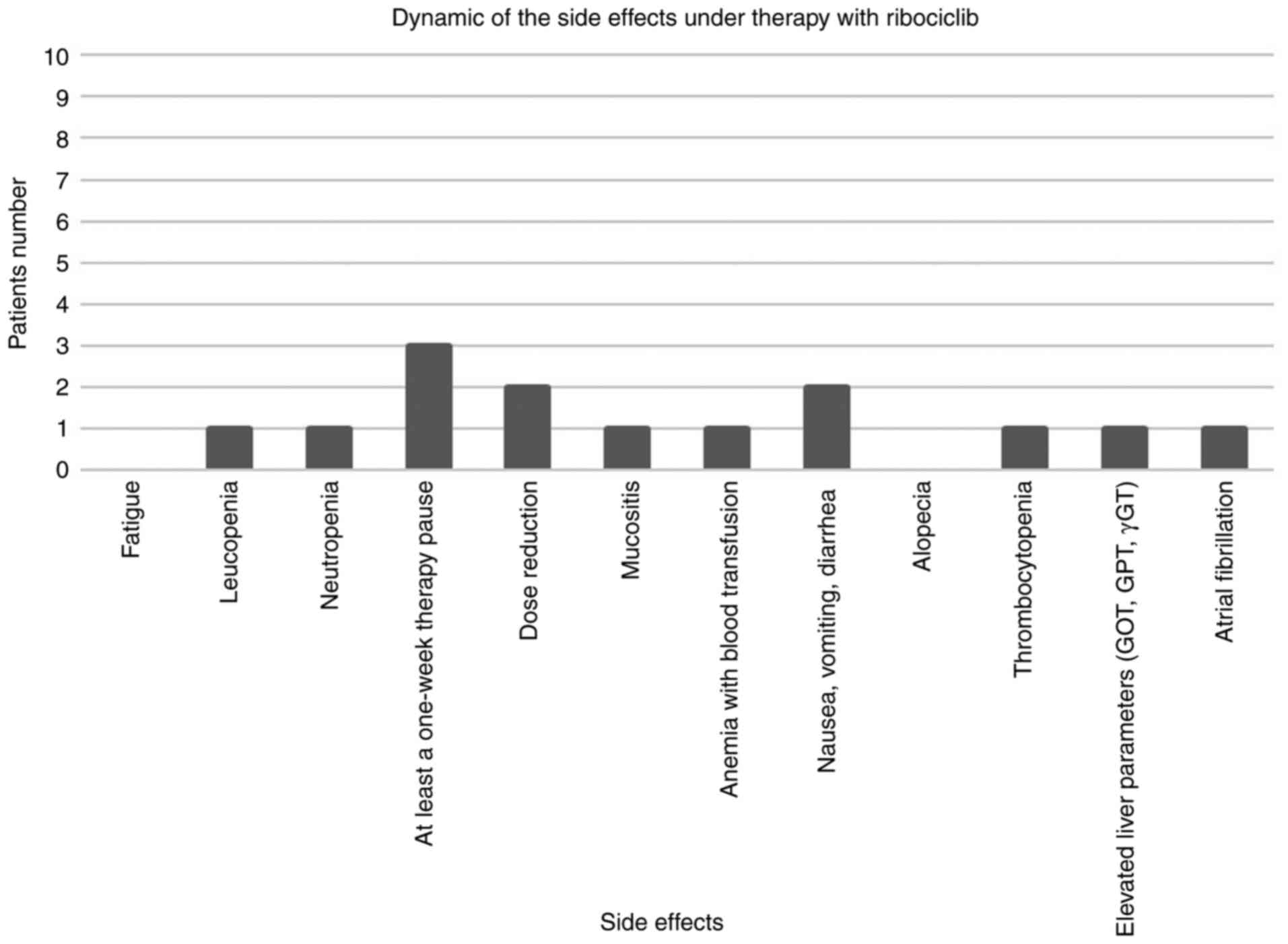
|
1
|
World Health Organisation: Breast cancer.
http://www.who.int/cancer/prevention/diagnosis-screening/breast-cancer/en/.
Accessed September 15, 2020.
|
|
2
|
Ji F, Yang CQ, Li XL, Zhang LL, Yang M, Li
JQ, Gao HF, Zhu T, Cheng MY, Li WP, et al: Risk of breast
cancer-related death in women with a prior cancer. Aging (Albany
NY). 12:5894–5906. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Nadji M, Gomez-Fernandez C, Ganjei-Azar P
and Morales AR: Immunohistochemistry of estrogen and progesterone
receptors reconsidered: Experience with 5,993 breast cancers. Am J
Clin Pathol. 123:21–27. 2005.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Cancer Genome Atlas Network. Comprehensive
molecular portraits of human breast tumours. Nature. 490:61–70.
2012.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Howell A, Cuzick J, Baum M, Buzdar A,
Dowsett M, Forbes JF, Hoctin-Boes G, Houghton J, Locker GY and
Tobias JS: ATAC Trialists' Group. Results of the ATAC (arimidex,
tamoxifen, alone or in combination) trial after completion of 5
years' adjuvant treatment for breast cancer. Lancet. 365:60–62.
2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Finn RS, Martin M, Rugo HS, Jones S, Im
SA, Gelmon K, Harbeck N, Lipatov ON, Walshe JM, Moulder S, et al:
Palbociclib and letrozole in advanced breast cancer. N Engl J Med.
375:1925–1936. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lundberg AS and Weinberg RA: Control of
the cell cycle and apoptosis. Eur J Cancer. 35:531–539.
1999.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Caldon CE, Daly RJ, Sutherland RL and
Musgrove EA: Cell cycle control in breast cancer cells. J Cell
Biochem. 97:261–274. 2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sherr CJ, Beach D and Shapiro GI:
Targeting CDK4 and CDK6: From discovery to therapy. Cancer Discov.
6:353–367. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Shapiro GI: Cyclin-dependent kinase
pathways as targets for cancer treatment. J Clin Oncol.
24:1770–1783. 2006.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ortega S, Malumbres M and Barbacid M:
Cyclin D-dependent kinases, INK4 inhibitors and cancer. Biochim
Biophys Acta. 1602:73–87. 2002.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gul A, Leyland-Jones B, Dey N and Pe P: A
combination of the PI3K pathway inhibitor plus cell cycle pathway
inhibitor to combat endocrine resistance in hormone
receptor-positive breast cancer: A genomic algorithm-based
treatment approach. Am J Cancer Res. 8:2359–2376. 2018.PubMed/NCBI
|
|
13
|
Miller TW, Balko JM, Fox EM, Ghazoui Z,
Dunbier A, Anderson H, Dowsett M, Jiang A, Smith RA, Maira SM, et
al: ERα-dependent E2F transcription can mediate resistance to
estrogen deprivation in human breast cancer. Cancer Discov.
1:338–351. 2011.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Finn RS, Dering J, Conklin D, Kalous O,
Cohen DJ, Desai AJ, Ginther C, Atefi M, Chen I, Fowst C, et al: PD
0332991, a selective cyclin D kinase 4/6 inhibitor, preferentially
inhibits proliferation of luminal estrogen receptor-positive human
breast cancer cell lines in vitro. Breast Cancer Res.
11(R77)2009.PubMed/NCBI View
Article : Google Scholar
|
|
15
|
Finn RS, Crown JP, Lang I, Boer K,
Bondarenko IM, Kulyk SO, Ettl J, Patel R, Pinter T, Schmidt M, et
al: The cyclin-dependent kinase 4/6 inhibitor palbociclib in
combination with letrozole versus letrozole alone as first-line
treatment of oestrogen receptor-positive, HER2-negative, advanced
breast cancer (PALOMA-1/TRIO-18): A randomised phase 2 study.
Lancet Oncol. 16:25–35. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Hortobagyi GN, Stemmer SM, Burris HA, Yap
YS, Sonke GS, Paluch-Shimon S, Campone M, Blackwell KL, André F,
Winer EP, et al: Ribociclib as first-line therapy for HR-positive,
advanced breast cancer. N Engl J Med. 375:1738–1748.
2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Cersosimo RJ: Cyclin-dependent kinase 4/6
inhibitors for the management of advanced or metastatic breast
cancer in women. Am J Health Syst Pharm. 76:1183–1202.
2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Guo L, Hu Y, Chen X, Li Q, Wei B and Ma X:
Safety and efficacy profile of cyclin-dependent kinases 4/6
inhibitor palbociclib in cancer therapy: A meta-analysis of
clinical trials. Cancer Med. 8:1389–1400. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pfizer Inc: IBRANCE 75/100/125 mg full
prescribing information. http://labeling.pfizer.com/ShowLabeling.aspx?id=2191.
Accessed September 15, 2020.
|
|
20
|
Novartis Pharmaceuticals Corporation:
Kisqali 200 mg prescribing information. https://www.novartis.us/sites/www.novartis.us/files/kisqali.pdf.
Accessed September 15, 2020.
|
|
21
|
Sonke GS, Hart LL, Campone M, Erdkamp F,
Janni W, Verma S, Villanueva C, Jakobsen E, Alba E, Wist E, et al:
Ribociclib with letrozole vs. letrozole alone in elderly patients
with hormone receptor-positive, HER2-negative breast cancer in the
randomized MONALEESA-2 trial. Breast Cancer Res Treat. 167:659–669.
2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Diéras V, Rugo HS, Schnell P, Gelmon K,
Cristofanilli M, Loi S, Colleoni M, Lu DR, Mori A, Gauthier E, et
al: Long-term pooled safety analysis of palbociclib in combination
with endocrine therapy for HR+/HER2-advanced breast cancer. J Natl
Cancer Inst. 111:419–430. 2019.PubMed/NCBI View Article : Google Scholar
|